Abstract
Purpose
To evaluate maternal and neonatal outcomes in patients with intrahepatic cholestasis of pregnancy (ICP).
Methods
Patients who gave birth in our hospital between January 2018 and March 2022 were retrospectively reviewed from the hospital database and patient file records. The study comprised 1686 patients, 54 in the ICP group and 1632 controls. Patients who had ICP after 20 weeks of gestation and were monitored and delivered at our facility were enrolled. Maternal demographic and obstetric characteristics data were examined. Perinatal outcomes were also assessed. Logistic regression analysis was used to determine adverse maternal outcomes.
Results
The mean age was 29 years. ART, GDM, and preeclampsia were significantly higher in the ICP group. The mean serum bile acid level was 19.3 ± 3 μmol/L in the ICP group. There was a higher risk of GDM and pre-eclampsia in women with ICP compared with those without and a significant association between ICP and adverse perinatal outcomes. There was a statistically significant relation between the presence of ICP and spontaneous preterm delivery, iatrogenic preterm delivery, 5th-minute Apgar scores < 7, and NICU requirement. No significant relationship was found between the presence of ICP and SGA and meconium. There was a significant relationship between the presence of ICP, mode of delivery, and PPH (p < 0.05). Those with ICP had a lower gestational week and birth weight, and higher rates of cesarean delivery and PPH.
Conclusion
ICP should prompt close monitoring and management to mitigate the potential exacerbation of adverse outcomes, including preeclampsia, GDM, and preterm birth.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Our study suggested that patients with ICP had increased risk of development of GDM and preeclampsia. Accordingly, we showed that it was associated with preterm birth and adverse perinatal outcomes. |
Introduction
Intrahepatic cholestasis of pregnancy (ICP) is the most common pregnancy-specific liver disease. It is observed in an average of 1/1000 to 1/10,000 of pregnancies and has a benign character that usually resolves in the postpartum period without causing any hepatic sequelae or mortality in the mother [1]. ICP is a pregnancy-specific condition that frequently occurs in the third trimester or late second trimester. It is a condition complicated by pruritus in the body, increased liver enzymes, and fasting bile acid levels (≥ 10 µmol/L) [2, 3]. In contrast to the maternal condition, fetal morbidity and mortality are increased in cases of intrahepatic cholestasis in which severe maternal morbidity and mortality are not usually observed. Intrahepatic cholestasis may cause preterm labor, abnormal intrapartum fetal heart tracing and sudden intrapartum fetal death [4, 5]. The most common reason for admission to the hospital in these patients is widespread itching in the extremities and skin lesions as a result of itching [6]. Although clearly not identified some of the following factors including genetic predisposition, environmental and hormonal factors also play a role [7, 8]. Ursodeoxycholic acid is the first choice in reducing maternal symptoms [9].
Along with gestational cholestasis, both preeclampsia and gestational diabetes mellitus (GDM) affect the mother and fetus. The first study to systematically describe highly significant associations between ICP and GDM and preeclampsia was conducted in 2013 [10]. Although the relationship of ICP with GDM and preeclampsia has been evaluated in studies, there are few studies examining the relationship of ICP with GDM and preeclampsia [10,11,12,13,14,15,16]. A meta-analysis found that the risk of GDM and preeclampsia increased approximately twofold in women with cholestasis [10]. Another study revealed an increased risk of GDM in pregnant women with cholestasis [11]. Another two studies revealed an increased risk of preeclampsia in pregnant women with cholestasis, and especially in twin pregnant women [12, 13]. In a retrospective cohort study, compared with the general population, a greater proportion of pregnant women with cholestasis were also affected by GDM, preeclampsia and/or spontaneous preterm birth [14]. In a prospective study, ICP was associated with impaired glucose tolerance, dyslipidemia, and increased fetal growth. These findings may have implications for the future health of affected offspring [15]. A significant difference was found in patients with ICP compared with controls terms of the incidence of hypothyroidism, thrombophilia, gestational diabetes, gestational hypertension, postpartum hemorrhage, and preterm birth in patients with ICP [16]. In addition, there is a high degree of heterogeneity and relative lack of reliable data in studies of ICP and GDM. In this study, we aimed to review the adverse maternal and perinatal outcomes of intrahepatic cholestasis in our hospital and to investigate the relationship between intrahepatic cholestasis, and the risk of GDM and preeclampsia.
Materials and methods
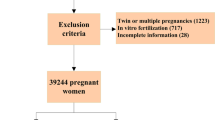
Patients who delivered in our hospital between January 2018 and March 2022 were retrospectively reviewed from the hospital database and patient file records. In our study, 1686 patients were evaluated, 54 patients in the ICP group, and 1632 patients in the control group. Patients who had ICP after 20 weeks of gestation and were followed and delivered at our institution were included in the study group. The diagnosis of ICP was based on elevated liver enzymes and elevated maternal serum total bile acid levels (≥ 10 μmol/L). Women who delivered at our institution during the study period were included to the control group. Ultrasonographic evaluations of all patients were performed by an obstetrician and gynecologist. All pregnancies complicated with severe congenital malformations such as chromosomal abnormalities and/or multiple congenital anomalies, all twin and higher multiple pregnancies, and patients with missing data were excluded. Data including maternal demographic and obstetrics characteristics, maternal age, gravida, parity, pre-pregnancy body mass index (BMI), assisted reproductive technique (ART), and presence of GDM and preeclampsia were analyzed. Perinatal outcomes, gestational age at birth, birth weight, induction of labor, delivery method, postpartum hemorrhage, preterm delivery (iatrogenic and spontaneous), small for gestational age (SGA), Apgar score, stillbirth, and neonatal death data were evaluated. Preeclampsia was defined according to the American College of Obstetricians and Gynecologist guidelines [17]. GDM was defined according to World Health Organization criteria [18]. Preterm labor was defined as delivery after 24 weeks and before 37 completed gestational weeks. SGA was defined as a birth weight < 10th percentile and low Apgar score was defined as a score < 7 at 5 min after birth. Stillbirth was used to define fetal death at or above 20 weeks of gestation, and neonatal death was defined as the death of an infant between 0 and 28 days after birth. The grading criteria for BMI were based on the recommendations of the Obesity Working Groups. Postpartum hemorrhage (PPH) was defined as blood loss of ≥ 500 mL within 24 h after vaginal delivery or ≥ 1000 mL after cesarean delivery. Severe PPH was defined as blood loss of ≥ 1000 mL within 24 h after delivery [19]. The primary outcome of this study was to determine the incidence of GDM and preeclampsia among women with ICP. Secondary outcomes were to determine the relationship among ICP, stillbirth, gestational age at delivery, birth weight, induction of labor, mode of delivery, presence of PPH, preterm delivery (iatrogenic and spontaneous), SGA, 5 min Apgar score, neonatal intensive care unit (NICU) hospitalization, and neonatal death.
The study was approved by the Buca Seyfi Demirsoy Training and Research Hospital on August 23, 2023, with the protocol number 2023/8–156. The study was written in accordance with the principles of the Declaration of Helsinki. In our study, an informed consent form was obtained from the patients. First, the data obtained from the participants were summarized using descriptive statistics. Then, the independent samples t-test was used for the difference of means between binary categories and the Chi-square test was used to analyze the relationships between categorical data. Statistical significance was accepted as p < 0.05. Binary logistic regression analysis was performed to analyze risk factors for preeclampsia and GDM. The research data were statistically analyzed using the SPSS version 20 software package (IBM SPSS Statistics, IBM Corporation, Armonk, NY, USA).
Results
The mean age of the participants was 29.71 ± 5.13 years. ICP was present in 3.2% of the participants. Preeclampsia and GDM was encountered at rates of 1.7% and 7.2%, respectively. Cesarean section was performed in 49.3% of the participants. The mean serum bile acid level was 19.3 ± 3 μmol/L in the ICP group. Table 1 shows the distribution of demographic and pregnancy characteristics between the groups.
There was a statistically significant difference between the mean gestational week (36.2 vs. 37.6, Cohen’s d: 1, r: −0.4; p < 0.001) and birth weight (2847 vs. 3127 g, Cohen’s d: −0.7, r: −0.35; p < 0.001) according to the presence or absence of ICP (p < 0.001). The rate of cesarean delivery and PPH were higher in those with ICP (p < 0.001 and p = 0.044, respectively). There was no significant relationship between the presence of ICP and SGA and meconium (p = 0.055 and p = 0.151, respectively). Spontaneous preterm delivery, iatrogenic preterm delivery, 5th minute Apgar score < 7, and NICU need were found to be proportionally higher in those with ICP (Table 2).
Table 3 shows the comparison of the patients with ICP and controls. ICP increased the risk of preeclampsia. Furthermore, ICP increased the risk of GDM. Age, BMI, gravida, parity, and ART were not associated with a risk of preeclampsia and GDM. Table 3 shows risk factors for development of preeclampsia and GDM (Table 3).
Discussion
This retrospective study had two main findings. ICP showed a strong relationship with GDM and pre-eclampsia. Women with ICP had a significantly increased risk for adverse perinatal outcomes. In our study, consistent with previous studies, we demonstrated a higher risk of GDM and preeclampsia in women with ICP compared with those without ICP [10, 14].
ICP was reported to be a rare condition that occurs with a prevalence of 1/1000 to 1/10,000 [1]. Although the literature reports the incidence of ICP as 1 in 1000, the higher rate observed in our study may be attributed to its cross-sectional design and the referral of some patients to our clinic due to ICP. Although rarely observed, ICP was reported to be associated with unfovarable maternal outcomes. Martineau et al. reported that ICP was characterized by glucose intolerance and dyslipidemia, consistent with the metabolic syndrome [15]. The mechanisms underlying the direct association of ICP with GDM and preeclampsia remain unclear. Women with ICP exhibited higher triglyceride, total cholesterol, low-density lipoprotein and very-low-density lipoprotein levels and these indices were also related with an increased risk of preeclampsia and GDM [16, 17]. In our study, cholesterol levels were not evaluated. It has been shown that women with higher BMI and impaired metabolic pathways may also be at increased risk for adverse outcomes of pregnancy [18, 19]. We found no significant relationship between GDM and preeclampsia and obesity.
In a meta-analysis conducted by Qin et al., which included a large population, a positive correlation was found between ART and pregnancy-related complications such as preeclampsia and GDM [20]. In current the study, there were only 50 ART pregnancies, and we showed that they were not a risk factor for either GDM or preeclampsia. We think that this contrast can only be explained by the low number of ART pregnancies included in the study. On the other hand, our study revealed that there was a correlation between ART and the presence of ICP. In parallel, in the study conducted by Jie et al., it was shown that the incidence of ICP was higher in ART pregnancies [21].
We observed a significant relationship between ICP and adverse perinatal outcomes. The relationship between the presence of ICP and spontaneous preterm delivery, iatrogenic preterm delivery, 5th minute Apgar scores < 7, and NICU need showed adverse effects of ICP on perinatal outcomes. On the other hand, there was no significant relationship between the presence of ICP and SGA and meconium. In the study published by Geenes et al., which included a very large population, it was found that the risk of meconium-stained amniotic fluid was higher in severe ICP. However, no neonatal meconium aspiration syndrome was shown in the study, and it was claimed that the NICU need was generally associated with preterm birth and the negative consequences it caused. In addition, in the same study, fewer babies with SGA were found in the cholestasis group than in the control group [22], similar to our study. Another recent study by Obiegbusi et al. showed that SGA rates were not different between patients and controls [23]. As a result, it is not possible to talk about a clear relationship between SGA and ICP. Williamson et al. previously reported that ICP was associated with an increased risk of stillbirth [24]. ICP is characterized by fetal death without any clear cause, accordingly, it is advised to consider making delivery decisions based on bile acid levels to reduce the risks of ICP on stillbirth [24]. With effective management during pregnancy, there was no increased incidence of stillbirth in our study, in agreement with previously published studies. In patients with ICP, the mean gestational age and birth weight were found to be low. The Royal College of Obstetrics and Gynecology does not recommend active management, although optimal timing of delivery in ICP is difficult given the paucity of high-quality studies [25].
A recent meta-analysis showed an increased risk of stillbirth in women with ICP when serum bile acid concentrations were 100 µmol/L or more [26], but bile acid is not always a reliable monitoring index [5]. Relatedly, stillbirth may not have been detected because the highest serum acid level in our study group was 29.9 µmol/L. We encourage physicians to monitor bile acid levels and recommend that they make an individualized decision about whether the risks of preterm birth outweigh the risks of disease.
In our study, both spontaneous and iatrogenic preterm birth were found higher in the ICP group. The high rate of iatrogenic preterm labor may reflect concerns regarding the risk of fetal death or adverse outcomes in pregnancies complicated by high maternal serum bile acids. Physicians managing these pregnancies may prefer a management strategy of induction of labor prior to 37 weeks despite emerging concerns about special education needs [27] and poorer school performance [28] in babies born late preterm. Spontaneous preterm labor may be explained by a dose-dependent bile acid effect on myometrial contractility, as has been demonstrated in rodents [29]. Furthermore, myometrial cells from women with ICP are more responsive to oxytocin, and cells from normal women demonstrate an increased response to oxytocin in the presence of bile acids [30, 31].
PPH is defined as blood loss of more than 500 mL within 24 h after delivery. In our study, PPH was more frequent in patients with ICP. The increased incidence of PPH in patients with ICP may be attributed to a possible association between ICP and PPH. In the ICP group, the mean gestational week and birth weight were found to be lower, and the rate of cesarean delivery was found to be higher. However, in these patients, PPH was treated using conservative methods without the need for any surgical or additional procedures such as postpartum hysterectomy, uterine artery ligation, hypogastric artery ligation, balloon tamponade, and b-lynch suturing. Additionally, no patients required massive blood product transfusion and no exitus occurred due to PPH. This study was conducted as a single-center, retrospective cohort study with large samples of patients with ICP demonstrating the prevalence and characteristics of ICP in our hospital.
In the present study, cesarean section rates were found to be higher in pregnancies in the ICP group. This rate was determined as 72.2% in the ICP group. The rate of birth by cesarean section is increasing worldwide [32]. According to the 2022 data of the Ministry of Health in Turkey, the cesarean section birth rate among live births was determined as 60.1% [33]. We think that this difference is related to the perinatal risks brought by ICP. In a recent study, Kong et al. demonstrated that women with severe ICP also had a significantly higher incidence of planned and unplanned CS compared those with mild ICP because the indications for CS showed that fetal intolerance was higher in severe ICP compared with mild ICP [34]. Shemer et al. reported that the risk of emergency cesarean section in ICP with spontaneous onset of labor did not differ from that in women without ICP with spontaneous onset of labor [35]. Based on the studies of Brouwers et al. and Stenhiles et al., it is known that ICP increases the risk of preterm labor, abnormal intrapartum fetal heart tracing, and sudden intrapartum fetal death [4, 5]. These known risks are the most likely reasons why the patients with ICP have a higher frequency of cesarean section compared with spontaneous delivery.
There are some limitations of our study. This study can be considered as a retrospective study, which may raise some concerns about data quality. These risks are thought to be related to patient selection risks and information bias. Furthermore, we did not evaluate the relationship between ICP and GDM or ICP and pre-eclampsia for time of onset. The relationship between bile acid concentrations and the risk of GDM or pre-eclampsia was not evaluated. Therefore, it is necessary to further analyze the time frame for any relationship between ICP and GDM or pre-eclampsia and to better understand the potential mechanisms of any correlation.
In conclusion, the presence of ICP should prompt close monitoring and management to mitigate the potential exacerbation of adverse outcomes, including preeclampsia, GDM, and preterm birth.
Data availability
The data that support the findings of this study are available upon reasonable request. The raw data and materials used in this research can be obtained from the corresponding author upon request.
References
Pathak B, Sheibani L, Lee RH (2010) Cholestasis of pregnancy. Obstet Gynecol Clin North Am 37(2):269–282. https://doi.org/10.1016/j.ogc.2010.02.011
Diken Z, Usta IM, Nassar AH (2014) A clinical approach to intrahepatic cholestasis of pregnancy. Am J Perinatol 31(1):1–8. https://doi.org/10.1055/s-0033-1333673
Estiú MC, Monte MJ, Rivas L, Moirón M, Gomez-Rodriguez L, Rodriguez-Bravo T, Marin JJ, Macias RI (2015) Effect of ursodeoxycholic acid treatment on the altered progesterone and bile acid homeostasis in the mother-placenta-foetus trio during cholestasis of pregnancy. Br J Clin Pharmacol 79(2):316–329. https://doi.org/10.1111/bcp.12480
Brouwers L, Koster MP, Page-Christiaens GC, Kemperman H, Boon J, Evers IM, Bogte A, Oudijk MA (2015) Intrahepatic cholestasis of pregnancy: maternal and fetal outcomes associated with elevated bile acid levels. Am J Obstet Gynecol 212(1):100.e1–7. https://doi.org/10.1016/j.ajog.2014.07.026
Sentilhes L, Verspyck E, Pia P, Marpeau L (2006) Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol 107(2 Pt 2):458–460. https://doi.org/10.1097/01.AOG.0000187951.98401.f7
Williamson C, Mackillop L (2009) Diseases of the liver, biliary system, and pancreas. In: Creasy RK, Resnik R (eds) Creasy & Resnik’s maternal-fetal medicine, 6th edn. Saunders Elsevier, Philadelphia, pp 1059–1077
Than NN, Neuberger J (2013) Liver abnormalities in pregnancy. Best Pract Res Clin Gastroenterol 27(4):565–575. https://doi.org/10.1016/j.bpg.2013.06.015
Pařízek A, Hill M, Dušková M, Vítek L, Velíková M, Kancheva R, Šimják P, Koucký M, Kokrdová Z, Adamcová K, Černý A, Hájek Z, Stárka L (2016) A comprehensive evaluation of steroid metabolism in women with intrahepatic cholestasis of pregnancy. PLoS ONE 11(8):e0159203. https://doi.org/10.1371/journal.pone.0159203
Biberoglu E, Kirbas A, Daglar K, Kara O, Karabulut E, Yakut HI, Danisman N (2016) Role of inflammation in intrahepatic cholestasis of pregnancy. J Obstet Gynaecol Res 42(3):252–257. https://doi.org/10.1111/jog.12902
Arafa A, Dong JY (2020) Association between intrahepatic cholestasis of pregnancy and risk of gestational diabetes and preeclampsia: a systematic review and meta-analysis. Hypertens Pregnancy 39(3):354–360. https://doi.org/10.1080/10641955.2020.1758939
Martineau M, Raker C, Powrie R, Williamson C (2014) Intrahepatic cholestasis of pregnancy is associated with an increased risk of gestational diabetes. Eur J Obstet Gynecol Reprod Biol 176:80–85. https://doi.org/10.1016/j.ejogrb.2013.12.037
Mor M, Shmueli A, Krispin E, Bardin R, Sneh-Arbib O, Braun M, Arbib N, Hadar E (2020) Intrahepatic cholestasis of pregnancy as a risk factor for preeclampsia. Arch Gynecol Obstet 301(3):655–664. https://doi.org/10.1007/s00404-020-05456-y
Raz Y, Lavie A, Vered Y, Goldiner I, Skornick-Rapaport A, Landsberg Asher Y, Maslovitz S, Levin I, Lessing JB, Kuperminc MJ, Rimon E (2015) Severe intrahepatic cholestasis of pregnancy is a risk factor for preeclampsia in singleton and twin pregnancies. Am J Obstet Gynecol 213(3):395.e1–8. https://doi.org/10.1016/j.ajog.2015.05.011
Marathe JA, Lim WH, Metz MP, Scheil W, Dekker GA, Hague WM (2017) A retrospective cohort review of intrahepatic cholestasis of pregnancy in a South Australian population. Eur J Obstet Gynecol Reprod Biol 218:33–38. https://doi.org/10.1016/j.ejogrb.2017.09.012
Martineau MG, Raker C, Dixon PH, Chambers J, Machirori M, King NM, Hooks ML, Manoharan R, Chen K, Powrie R, Williamson C (2015) The metabolic profile of intrahepatic cholestasis of pregnancy is associated with impaired glucose tolerance, dyslipidemia, and increased fetal growth. Diabetes Care 38(2):243–248. https://doi.org/10.2337/dc14-2143
Granese R, Calagna G, Alibrandi A, Martinelli C, Romeo P, Filomia R, Ferraro MI, Piccione E, Ercoli A, Saitta C (2023) Maternal and neonatal outcomes in intrahepatic cholestasis of pregnancy. J Clin Med 12(13):4407. https://doi.org/10.3390/jcm12134407
Jin WY, Lin SL, Hou RL, Chen XY, Han T, Jin Y, Tang L, Zhu ZW, Zhao ZY (2016) Associations between maternal lipid profile and pregnancy complications and perinatal outcomes: a population-based study from China. BMC Pregnancy Childbirth 21(16):60. https://doi.org/10.1186/s12884-016-0852-9
Somprasit C, Tanprasertkul C, Rattanasiri T, Saksiriwutth P, Wongkum J, Kovavisarach E, Jongfueangparinya K, Panichakul P, Wuthiwong J (2015) High pre-pregnancy body mass index and the risk of poor obstetrics outcomes among Asian women using BMI criteria for Asians by World Health Organization Western Pacific Region (WPRO): a large cohort study. J Med Assoc Thai 98(Suppl 2):S101–S107
Cnattingius S, Bergström R, Lipworth L, Kramer MS (1998) Prepregnancy weight and the risk of adverse pregnancy outcomes. N Engl J Med 338(3):147–152. https://doi.org/10.1056/NEJM199801153380302
Qin J, Liu X, Sheng X, Wang H, Gao S (2016) Assisted reproductive technology and the risk of pregnancy-related complications and adverse pregnancy outcomes in singleton pregnancies: a meta-analysis of cohort studies. Fertil Steril 105(1):73-85.e1–6. https://doi.org/10.1016/j.fertnstert.2015.09.007
Jie Z, Yiling D, Ling Y (2015) Association of assisted reproductive technology with adverse pregnancy outcomes. Iran J Reprod Med 13(3):169–180
Geenes V, Chappell LC, Seed PT, Steer PJ, Knight M, Williamson C (2014) Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology 59(4):1482–1491. https://doi.org/10.1002/hep.26617
Obiegbusi CN, Dong XJ, Obiegbusi SC (2023) Pregnancy outcomes following antenatal screening for intrahepatic cholestasis of pregnancy (ICP). Taiwan J Obstet Gynecol 62(6):809–816. https://doi.org/10.1016/j.tjog.2023.09.001
Williamson C, Hems LM, Goulis DG, Walker I, Chambers J, Donaldson O, Swiet M, Johnston DG (2004) Clinical outcome in a series of cases of obstetric cholestasis identified via a patient support group. BJOG 111(7):676–681. https://doi.org/10.1111/j.1471-0528.2004.00167.x
Bicocca MJ, Sperling JD, Chauhan SP (2018) Intrahepatic cholestasis of pregnancy: review of six national and regional guidelines. Eur J Obstet Gynecol Reprod Biol 231:180–187. https://doi.org/10.1016/j.ejogrb.2018.10.041
Ovadia C, Seed PT, Sklavounos A, Geenes V, Di Ilio C, Chambers J, Kohari K, Bacq Y, Bozkurt N, Brun-Furrer R, Bull L, Estiú MC, Grymowicz M, Gunaydin B, Hague WM, Haslinger C, Hu Y, Kawakita T, Kebapcilar AG, Kebapcilar L, Kondrackienė J, Koster MPH, Kowalska-Kańka A, Kupčinskas L, Lee RH, Locatelli A, Macias RIR, Marschall HU, Oudijk MA, Raz Y, Rimon E, Shan D, Shao Y, Tribe R, Tripodi V, Yayla Abide C, Yenidede I, Thornton JG, Chappell LC, Williamson C (2019) Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet 393(10174):899–909. https://doi.org/10.1016/S0140-6736(18)31877-4. (Erratum in: Lancet. 2019 Mar 16;393(10176):1100)
MacKay DF, Smith GC, Dobbie R, Pell JP (2010) Gestational age at delivery and special educational need: retrospective cohort study of 407,503 schoolchildren. PLoS Med 7(6):e1000289. https://doi.org/10.1371/journal.pmed.1000289.PMID:20543995;PMCID:PMC2882432
Quigley MA, Poulsen G, Boyle E, Wolke D, Field D, Alfirevic Z, Kurinczuk JJ (2012) Early term and late preterm birth are associated with poorer school performance at age 5 years: a cohort study. Arch Dis Child Fetal Neonatal Ed 97(3):F167–F173. https://doi.org/10.1136/archdischild-2011-300888. (Epub 2012 Jan 3 PMID: 22215800)
Campos GA, Castillo RJ, Toro FG (1988) Efecto de los ácidos biliares sobre la contractibilidad miometral en útero gestante aislado [Effect of bile acids on the myometral contractility of the isolated pregnant uterus]. Rev Chil Obstet Ginecol 53(4):229–233
Germain AM, Kato S, Carvajal JA, Valenzuela GJ, Valdes GL, Glasinovic JC (2003) Bile acids increase response and expression of human myometrial oxytocin receptor. Am J Obstet Gynecol 189(2):577–582. https://doi.org/10.1067/s0002-9378(03)00545-3
Israel EJ, Guzman ML, Campos GA (1986) Maximal response to oxytocin of the isolated myometrium from pregnant patients with intrahepatic cholestasis. Acta Obstet Gynecol Scand 65(6):581–582. https://doi.org/10.3109/00016348609158391
Demirbaş M, Karabel MP, İnci MB (2018) Türkiye’de ve dünya’da değişen sezaryen sıklığı ve olası nedenleri. Sakarya Tıp Dergisi Ocak 7(4):158–163. https://doi.org/10.31832/smj.368600
Kong C, Zhu Z, Mei F (2023) Risk factors associated with cesarean section and adverse fetal outcomes in intrahepatic cholestasis of pregnancy. Front Pediatr 30(11):1136244. https://doi.org/10.3389/fped.2023.1136244
Wikström Shemer EA, Thorsell M, Marschall HU, Kaijser M (2013) Risks of emergency cesarean section and fetal asphyxia after induction of labor in intrahepatic cholestasis of pregnancy: a hospital-based retrospective cohort study. Sex Reprod Healthc 4(1):17–22. https://doi.org/10.1016/j.srhc.2012.11.005
Funding
The authors declare no funding was received.
Author information
Authors and Affiliations
Contributions
Ata Can: Data collection or management, Data analysis, Manuscript writing/editing. Atlihan Ufuk: Data collection or management, Data analysis, Manuscript writing/editing. Avsar Huseyin Aytug: Protocol/project development, Data collection or management, Data analysis, Manuscript writing/editing. Erkilinc Selcuk: Protocol/project development, Manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no confict of interest.
Ethical approval
Ethical approval for this study was obtained from the ‘Buca Seyfi Demirsoy Training and Research Hospital’ on August 23, 2023, with the protocol number 2023/8–156.
Informed consent
A separate declaration of informed consent of the patients in this retrospective hospital data collection was waived. There were no minors included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Avsar, H.A., Atlıhan, U., Ata, C. et al. Intrahepatic cholestasis of pregnancy and its association with preeclampsia and gestational diabetes: a retrospective analysis. Arch Gynecol Obstet 310, 221–227 (2024). https://doi.org/10.1007/s00404-024-07507-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-024-07507-0