Abstract
Purpose
This study aims to evaluate the efficacy of hysteroscopic curettage combined with progestin therapy in young patients with early-stage endometrial cancer (EC) and endometrial atypical hyperplasia (EAH) who wished to preserve their fertility.
Methods
This prospective cohort study included 16 patients with early-stage EC and 25 patients with EAH in Dalian Maternal and Child Health Hospital from August 2014 to October 2018. All patients received fertility-sparing therapy with hysteroscopic evaluation every 3 months until achieving complete response (CR). Demographic, clinical, and pathological data follow-up information as well as fertility outcomes was analyzed.
Results
There were 92.6% (37/41) patients who achieved CR. The mean treatment duration to CR was 7.47 ± 2.91 months. BMI ≤ 30 kg/m2 was associated with shorter treatment duration to achieve CR (P = 0.003). Among the patients who attempted to conceive, 30.3% (10/33) had successful pregnancy, and 18.2% (6/33) delivered live births. The implementation of assisted reproductive technology (ART) is closely associated with pregnancy (P = 0.001).
Conclusion
The fertility-sparing therapy, hysteroscopic curettage combined with progestin therapy, of early young EC and EAH patients is safe and effective. BMI is the main factor affecting the duration of CR. After achieving CR, ART can significantly improve the pregnancy rate of these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endometrial cancer (EC) is one of the three major malignant tumors of the female reproductive system. In recent years, the incidence of endometrial cancer has gradually increased, and the age of onset has become lower. About 25% of patients are premenopausal women, of which 5% are patients younger than 40 years old [1, 2]. Endometrial atypical hyperplasia (EAH) is a precancerous lesion of endometrial cancer, which has a significant probability of progressing to endometrial cancer within 5 years. Due to its earlier onset, an increasingly larger number of young women who have not yet given birth suffer from this disease [3,4,5]. This highlights the need for fertility-sparing therapy.
Hysteroscopic curettage, combined with progestin therapy, to preserve fertility is controversial [6,7,8,9]. It has several advantages because it can accurately observe the size, location, and range of endometrial lesions in the uterine cavity; improve the positive rate of uterine curettage; reduce leakage of aspiration; and accurately evaluate the degree of myometrial invasion. Hysteroscopic endometrial lesion resection can also be performed simultaneously to achieve a clear diagnosis, identify the precise degree of the lesion, and reduce the tumor load. It has been a highly recognized treatment in recent years [10], with a clinical–pathological complete response rate of over 90% [10,11,12]. However, the number of cases reported in literature is still small, and the factors affecting clinical–pathological response and pregnancy are currently unclear.
The primary purpose of this study is to evaluate the effectiveness of hysteroscopic surgery combined with high-dose progestin as a fertility-sparing therapy for patients with early EC and EAH. The secondary aim is to evaluate various factors that may affect fertility-sparing therapy and pregnancy outcomes to provide a basis for the standardized treatment of early EC and EAH with fertility preservation.
Methods
Study design and patients
This study was a prospective observational study. We included patients diagnosed and pathologically confirmed with highly differentiated EC or EAH in Dalian Maternity and Child Health Hospital from August 2014 to October 2018. All patients were initially diagnosed and treated. The study was approved by the ethics committee of our institution. All study participants signed written informed consent.
Our inclusion criteria for fertility-sparing therapy followed 2014 NCCN guideline [13], which included patients who: (1) were 40 years old or younger with a strong desire and requirement to preserve fertility; (2) diagnosed with endometrioid adenocarcinoma or EAH as the pathological type; (3) had well-differentiated (G1) carcinoma; (4) was progestin receptor-positive immunohistochemically; (5) had lesions confined to the uterine mucosa confirmed using transvaginal color Doppler sonography (TVS) and magnetic resonance imaging (MRI) examination that excluded invasion of the myometrium and cervical stroma without extrauterine metastasis; (6) no increase in serum cancer antigen 125 (CA125); (7) no contraindication for drug treatment or equipment use; (8) had good compliance to therapy and close follow-up; and (9) understood the advantages and disadvantages of fertility-sparing therapy, and voluntarily chose the treatment with fertility preservation.
The exclusion criteria included patients who had: (1) non-endometrioid adenocarcinoma, such as clear cell carcinoma or serous papillary carcinoma; (2) moderate or poor grades of differentiation (G2, G3); (3) myometrial infiltration or cervical stromal involvement; (4) evidence of extrauterine metastasis, such as peritoneal, ovarian, and lymph node metastasis; (5) contraindications to progestin therapy; (6) poor compliance and follow-up; (7) desire to withdraw from the clinical study; and (8) thrombosis.
All patients’ age, marriage status, childbearing history, gravidity and parity, menstrual history, past medical history, family history of cancer, and body mass index (BMI), as well comorbidities such as hypertension, diabetes, heart disease, liver and kidney disease, breast disease, and thrombotic disease were recorded.
All patients underwent hysteroscopy and comprehensive diagnostic curettage before fertility-sparing therapy. All the endometrial specimens were reviewed by at least two experienced pathologists to confirm the pathological type and grade of differentiation of the endometrial lesions. Progestin receptors were detected by immunohistochemistry.
All patients underwent TVS and/or MRI before treatment to exclude myometrial invasion, cervical stromal involvement, ovarian tumor, and extrauterine metastasis. CA125 was likewise evaluated.
Progestin therapy regimen
Hysteroscopy and curettage were performed for all patients before treatment. After the operation, oral megestrol acetate or levonorgestrel intrauterine system (LNG-IUS) insertion regimen was used for the fertility-sparing treatment.
The oral MA regimen was as follows: patients took oral MA 160 mg/d for 21 days (starting from the 5th day after hysteroscopic curettage or the 5th day of menstruation). After stopping the medication, the patient was observed for menstrual cramps then started on the next treatment cycle 5 days after menstruation. If the patient still had no menstruation after 2 weeks of drug withdrawal, the next cycle of treatment was started to avoid the influence of a prolonged non-treatment interval. This converted the therapy to a continuous regimen, oral MA 160 mg/day for 2 months, followed by evaluation. The other patients were treated with LNG-IUS insertion. All patients with diabetes mellitus or insulin resistance before beginning the treatment were given additional oral metformin 250 mg/dose, three times daily, with meals.
Evaluation of the therapeutic effect
The first efficacy evaluation was conducted after 12 weeks of treatment and every 3 months thereafter. Evaluation included TVS, MRI, hysteroscopy, or diagnostic curettage to obtain endometrial tissue for pathological examination. The efficacy criteria were as follows: complete response (CR), the pathological examination of the endometrium after treatment exhibits the proliferative phase, secretory phase, or without any abnormal endometrial changes; partial response (PR), endometrial lesions with a reduced grade, residual cancer, or atypical hyperplasia; no response or stable disease (SD), the pathological examination results are the same as before treatment, and there is no change in the endometrium; and progressive disease (PD), when there are clear signs of myometrial infiltration, pathological upgrade to high-grade endometrial cancer, or extrauterine metastasis during treatment.
Based on the results, we decided whether to continue or terminate the conservative treatment. During fertility-preservation therapy, if there was a clear evidence of disease progression, conservative treatment was terminated immediately, and surgical treatment was performed. Patients with CR were transferred to an assisted reproductive center for assisted pregnancy treatment. For unmarried patients or those not practicing family planning for any reason, maintenance treatment was continued, consisting of oral short-acting contraceptives, oral progestin on the second half of the menstrual cycle, implantation of LNG-IUS, or only close monitoring of menstruation patterns.
The initial treatment period can last for two to three cycles (6–9 months). If there is no documented response to high-dose progestin after more than 6 months, disease progression or metastasis needed to be ruled out. If the above conditions were excluded, treatment was continued for another 3–6 months, with close follow-up. For patients who failed to achieve complete pathological response after four cycles (12 months) of fertility-sparing treatment, patient underwent thorough evaluation and the treatment plan was modified.
During fertility-sparing therapy, adverse reactions were regularly monitored, including liver and kidney function injury, drug allergy, weight changes, gastrointestinal reaction, lower limb swelling and pain, cardiovascular system disease, or thromboembolic disease. Fertility-sparing therapy was terminated in the event of a serious adverse reaction.
Statistical analysis
SPSS 19.0 (SPSS, Inc., Chicago, IL, USA) statistical software was used to analyze the data. Categorical variables were expressed as the frequency (percentage) and were analyzed using Chi-square test or Fisher’s exact test. Continuous data were shown as mean ± standard deviation, and normal distribution data were analyzed using Student’s t test, while nonnormal distribution data were analyzed using the Mann–Whitney U test. Kaplan–Meier method was used for univariate analysis. P < 0.05 was defined as statistically significant.
Results
Study cohort
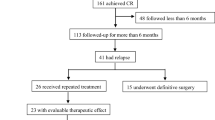
A total of 41 patients were included in this study, including 16 cases of early EC and 25 cases of EAH. The median age was 34 years (25–40 years). The median BMI was 30.04 (24.54–42.86). We treated 27 cases with MA, 10 cases with MA and metformin, and 4 cases with LNG-IUD. The median follow-up time was 32 months (8–65 months). Patient characteristics are shown in Table 1.
Of the 41 conservatively treated patients, 4 patients showed menorrhagia, of which 1 patient showed heavy vaginal bleeding with blood clots and severe anemia (hemoglobin 48 g/L). Other complications include eight cases with oligomenorrhea and four cases of abnormal urinary bleeding (> 20 days). Normal menstrual cycles were documented in 24 cases (Table 1).
Outcomes of treatment
After hysteroscopic diagnostic curettage and high-dose progestin therapy, CR was observed in 37 patients, PR in 1, SD in 2, and PD in 1 patient (Table 1). The overall response rate (CR + PR) was 92.6%. The patient with PD was 28 years old with a BMI of 28.84 (obesity in China). She was treated in the emergency department due to heavy vaginal bleeding and underwent hysteroscopy and curettage. Pathological examination showed EAH. The patient was treated with MA 160 mg/d for 6 months, then underwent repeat hysteroscopic diagnostic curettage. Endometrial biopsy showed a well-differentiated endometrioid adenocarcinoma with myometrial invasion, and was classified as PD. The patient underwent laparoscopic total hysterectomy, double adnexectomy, and pelvic lymph node dissection with cytologic examination of the abdominal washing fluid. The postoperative pathological results were well-differentiated endometrioid adenocarcinoma with local invasion of the deep myometrium, without intravascular tumor thrombus or lymph node metastasis. On regular postoperative follow-up, the patient remains tumor free.
During progestin treatment, the most common adverse drug reaction was weight gain in 53.6% (22/41), followed by irregular vaginal bleeding in 17% (7/41). There were three patients with mild liver dysfunction, simultaneously treated with liver protection treatment, which did not affect the continuity of fertility-sparing therapy. No serious adverse events related to drug treatment and hysteroscopy were found, such as thromboembolism, severe renal or liver dysfunction, severe infection, or uterine perforation.
A total of 14 patients in the EC group obtained CR, and the average treatment time was 8.33 ± 2.49 months. CR was achieved in a total of 23 patients in the EAH group, and the average treatment time was 6.74 ± 3.00 months. There was no statistical difference between the two groups (P = 1.000). We also evaluated other factors that may affect the CR rate and therapeutic duration to achieve CR (Table 2). Univariate Kaplan–Meier analysis indicated that BMI ≤ 30 (P = 0.001), PCOS (P = 0.022), and diabetes mellitus (P = 0.009) were related to therapeutic duration.
In the EAH group, 17.39% (4/23) of patients with CR had disease recurrence, wherein three had atypical hyperplasia, and one had highly differentiated endometrioid adenocarcinoma. The recurrence time was 15, 33, 10, and 28 months, respectively. All four patients refused surgical treatment and met the criteria for fertility-sparing therapy after evaluation. All patients were given a second cycle oral progestin. At present, two cases obtained CR, while two cases are still undergoing treatment.
In the EC group, 14.3% (2/14) of the patients with CR had a recurrence. The recurrence time was 24 and 30 months. All patients with EC had well-differentiated endometrioid adenocarcinoma, and underwent surgical treatment (total hysterectomy, double adnexectomy, and pelvic lymphadenectomy). Currently, they remain tumor free.
Pregnancy outcomes
In this study, a total of 33 women were desirous of pregnancy. Among them, 10 women became pregnant for a total of 12 events during the study period, with 6 live births. The overall pregnancy rate was 30.0%, and the fertility rate was 18.1%. The fertility rate of the two groups is shown in Table 1. We evaluated the factors that may affect the pregnancy rate (Table 3). Univariate Kaplan–Meier analysis showed that assisted reproductive technology (ART) was associated with successful pregnancy (P = 0.001).
Discussion
We prospectively studied the pregnancy outcomes of hysteroscopic surgery combined with progestin therapy in patients with EAH and early EC who met the criteria for fertility-preserving therapy. About 90% of patients obtained CR. Obesity was closely related to longer treatment duration before achieving CR. Approximately 33% of the patients had successful pregnancy, and the implementation of ART increased chances to a successful pregnancy.
Previous studies showed that the CR rate of fertility-sparing therapy varies greatly, ranging from 49 to 100% [14,15,16,17,18]. Many studies have explored the factors affecting CR, but the results are unclear. The goal of fertility-sparing therapy is not the complete reversal of endometrial lesions, but the ability to achieve pregnancy and deliver live births. The reported pregnancy rate varies considerably, mostly between 40 and 50% [11]. It is difficult for clinicians to discern suitable candidates for fertility-sparing therapy. Therefore, it is necessary to study the factors influencing CR rate and pregnancy rate.
The CR rate is directly related to the inclusion criteria. All patients included in this study satisfied the NCCN guidelines [13]. Notably, the NCCN guidelines do not limit the age of patients with endometrial cancer for fertility-sparing treatment. Most literature reported that patients with endometrial cancer undergoing fertility-sparing treatment were ≤ 40 years old, but there are also a small number of patients between 41 and 45. These patients obtained CR after treatment, but few ultimately achieved fertility [19]. We only included patients under 40 years old. This is because there is a significant correlation between ovarian function and age. Serum anti-Müllerian hormone (AMH) in Chinese women dropped significantly after 35 years old, with an average value of only 1.29 ng/ml after 40 years old, and is exhausted after 45 years old [20]. Similarly, patients with endometrial cancer often have infertility, and AMH in infertile patients is significantly lower than that in normal women [21, 22]. In this study, we theorized that patients undergoing fertility preservation therapy will take longer to become pregnant after hysteroscopic surgery and MA treatment. By the time the regimen was completed, these patients were older, thus a lower pregnancy rate, affecting the pregnancy rate calculation.
The use of hysteroscopic surgery to treat fertility-sparing endometrial cancer patients was first published by Mazzon et al. [6]. Since then, many researchers evaluated its efficacy [1, 12, 23]. A meta-analysis indicated that hysteroscopy plays a crucial role in the early diagnosis, fertility-sparing treatment, and evaluation of endometrial cancer [10]. However, repeated hysteroscopy pose complications, including intrauterine adhesions, and positive ascites cytology. In fertility-sparing therapy, progestin is supplemented after hysteroscopic resection of the lesion. Progestin can inhibit the growth of the endometrium, delaying wound healing. Theoretically, this increases the risk of intrauterine adhesions, thereby reducing the pregnancy success rate, which is contrary to the original intention of preserving fertility. In this study, two patients had failed pregnancies due to intrauterine adhesions. Therefore, it is very important to balance the scope and the effect of surgery. It was reported that diagnostic laparoscopy could also be performed for each patient to evaluate whether the patient has intra-abdominal lesions during condition evaluation, which is the most accurate method [24]. However, since diagnostic laparoscopy is an invasive operation and considering economic reasons, patients were not asked to have such an examination.
We analyzed the factors influencing the CR of EAH and EC from fertility-sparing therapy. Our results showed that higher BMI was the main influencing factor. Park et al. considered that a BMI ≥ 25 kg/m2 before treatment was significantly correlated with low CR rate and high recurrence rate for progestin treatment; after treatment, it was also an important factor for high recurrence rate [25]. Yang et al. reported that a BMI ≥ 25 kg/m2 and intrauterine lesions larger than 2 cm were the influencing factors of prolonged CR time [11]. In this study, BMI affected CR time, but not CR rate. Patients with BMI ≤ 30 needed a shorter time to achieve CR. Since BMI is a modifiable factor, future studies should investigate whether it can increase the CR rate, shorten the time to reach CR, or increase the pregnancy rate through weight modification.
Due to the large differences in the criteria and treatment schemes of patients with fertility-sparing therapy included in different literatures, it is challenging to compare the outcomes of different literatures. Park et al. reported that the pregnancy rate and live birth rate of patients in the ART group were significantly higher than those in the natural pregnancy group. In contrast, the ectopic pregnancy rate and spontaneous abortion rate of the two groups were not significantly different [26]. Tong et al. found that the preterm birth rate and multiple pregnancy rate of the ART group were significantly higher than those of natural pregnancy group [27]. Signorelli et al. showed that intermittent treatment with progesterone can achieve better therapeutic effect than continuous medication. High dose progesterone can induce apoptosis of endometrial cancer cells, but it will lead to drug resistance of endometrial cells if used continuously for a long time [28]. In this study, ART greatly improved the pregnancy rate of patients, wherein only one patient became pregnant naturally. We believe that the further development of ART, combined with the complete resolution of endometrial lesions and correcting other infertility factors, can help patients achieve the goal of pregnancy and childbirth.
There are some limitations to our study. First, this was a single-center study with a small sample size due to the strict inclusion criteria for patients with fertility-sparing therapy. Future studies should include a multi-center research to increase the sample size. Second, ART is expensive, thus many patients are prevented from using this modality to achieve successful pregnancy. Further, the short follow-up time may have resulted in the low recurrence rate and pregnancy rate in this study.
In conclusion, hysteroscopic surgery combined with progestin therapy as a fertility-sparing management for patients with early EC and EAH patients is safe and effective. This complies with the individualized, humanized, and minimally invasive treatment concept of modern tumor treatment. BMI is the main factor affecting the length of time required to obtain CR. After treatment, ART can significantly improve pregnancy outcomes. With further research, a higher pregnancy rate and fertility rate can be achieved.
Availability of data and material
The datasets used and analyzed during the current study are included in this article.
References
Falcone F, Laurelli G, Losito S, Di Napoli M, Granata V, Greggi S (2017) Fertility preserving treatment with hysteroscopic resection followed by progestin therapy in young women with early endometrial cancer. J Gynecol Oncol 28:e2
Contreras N, Sabadell J, Verdaguer P, Julià C, Fernández-Montolí M (2022) Fertility-sparing approaches in atypical endometrial hyperplasia and endometrial cancer patients: current evidence and future directions. Int J Mol Sci 23:2531
Tomao F, Peccatori F, Pup LD, Franchi D, Zanagnolo V, Panici PB, Colombo N (2015) Special issues in fertility preservation for gynecologic malignancies. Crit Rev Oncol Hematol 97:206–219
Falcone F, Balbi G, Di Martino L, Grauso F, Salzillo ME, Messalli EM (2014) Surgical management of early endometrial cancer: an update and proposal of a therapeutic algorithm. Med Sci Monit 20:1298–1313
La Rosa VL, Garzon S, Gullo G, Fichera M, Sisti G, Gallo P, Riemma G, Schiattarella A (2020) Fertility preservation in women affected by gynaecological cancer: the importance of an integrated gynaecological and psychological approach. Ecancermedicalscience 14:1035
Mazzon I, Corrado G, Masciullo V, Morricone D, Ferrandina G, Scambia G (2010) Conservative surgical management of stage IA endometrial carcinoma for fertility preservation. Fertil Steril 93:1286–1289
Lee CL, Huang KG, Chen HL, Yen CF (2008) The roles of endoscopy in endometrial cancer. Taiwan J Obstet Gynecol 47:379–383
Bese T, Demirkiran F, Guralp O, Sanioglu C, Arvas M (2009) Transtubal transport of carcinoma cells into the peritoneal cavity after saline infusion via transcervical route in patients with endometrial carcinoma. Int J Gynecol Cancer 19:682–685
Garzon S, Uccella S, Zorzato PC, Bosco M, Franchi MP, Student V, Mariani A (2021) Fertility-sparing management for endometrial cancer: review of the literature. Minerva Med 112:55
Guillon S, Popescu N, Phelippeau J, Koskas M (2019) A systematic review and meta-analysis of prognostic factors for remission in fertility-sparing management of endometrial atypical hyperplasia and adenocarcinoma. Int J Gynaecol Obstet 146:277–288
Yang B, Xu Y, Zhu Q, Xie L, Shan W, Ning C, Xie B, Shi Y, Luo X, Zhang H, Chen X (2019) Treatment efficiency of comprehensive hysteroscopic evaluation and lesion resection combined with progestin therapy in young women with endometrial atypical hyperplasia and endometrial cancer. Gynecol Oncol 153:55–62
Wang Q, Guo Q, Gao S, Xie F, Du M, Dong J, Sui L, Xie K (2015) Fertility-conservation combined therapy with hysteroscopic resection and oral progesterone for local early stage endometrial carcinoma in young women. Int J Clin Exp Med 8:13804
Koh WJ, Greer BE, Abu-Rustum NR, Apte SM, Campos SM, Chan J, Cho KR, Cohn D, Crispens MA, Dupont N, Eifel PJ, Fader AN, Fisher CM, Gaffney DK, George S, Han E, Huh WK, Lurain JR, Martin L, Mutch D, Remmenga SW, Reynolds RK, Small WJ, Teng N, Tillmanns T, Valea FA, McMillian N, Hughes M (2014) Uterine neoplasms, version 1.2014. J Natl Compr Canc Netw 12:248–280
Ushijima K, Yahata H, Yoshikawa H, Konishi I, Yasugi T, Saito T, Nakanishi T, Sasaki H, Saji F, Iwasaka T, Hatae M, Kodama S, Saito T, Terakawa N, Yaegashi N, Hiura M, Sakamoto A, Tsuda H, Fukunaga M, Kamura T (2007) Multicenter phase II study of fertility-sparing treatment with medroxyprogesterone acetate for endometrial carcinoma and atypical hyperplasia in young women. J Clin Oncol 25:2798–2803
Pal N, Broaddus RR, Urbauer DL, Balakrishnan N, Milbourne A, Schmeler KM, Meyer LA, Soliman PT, Lu KH, Ramirez PT, Ramondetta L, Bodurka DC, Westin SN (2018) Treatment of low-risk endometrial cancer and complex atypical hyperplasia with the levonorgestrel-releasing intrauterine device. Obstet Gynecol 131:109–116
Gotlieb WH, Beiner ME, Shalmon B, Korach Y, Segal Y, Zmira N, Koupolovic J, Ben-Baruch G (2003) Outcome of fertility-sparing treatment with progestins in young patients with endometrial cancer. Obstet Gynecol 102:718–725
Ota T, Yoshida M, Kimura M, Kinoshita K (2005) Clinicopathologic study of uterine endometrial carcinoma in young women aged 40 years and younger. Int J Gynecol Cancer 15:657–662
Andress J, Pasternak J, Walter C, Kommoss S, Krämer B, Hartkopf A, Brucker SY, Schönfisch B, Steinmacher S (2021) Fertility preserving management of early endometrial cancer in a patient cohort at the department of women’s health at the university of Tuebingen. Arch Gynecol Obstet 304:215–221
Cade TJ, Quinn MA, Rome RM, Neesham D (2013) Long-term outcomes after progestogen treatment for early endometrial cancer. Aust N Z J Obstet Gynaecol 53:566–570
Zhu J, Li T, Xing W, Lin H, Ou J (2018) Chronological age vs biological age: a retrospective analysis on age-specific serum anti-Mullerian hormone levels for 3280 females in reproductive center clinic. Gynecol Endocrinol 34:890–894
Okunola T, Olusegun AK, Morebise LO, Salawu A, Omitinde SO (2017) Follicle stimulating hormone and anti-Mullerian hormone among fertile and infertile women in Ile-Ife, Nigeria: is there a difference? Int J Fertil Steril 11:33–39
Bedenk J, Vrtacnik-Bokal E, Virant-Klun I (2020) The role of anti-Mullerian hormone (AMH) in ovarian disease and infertility. J Assist Reprod Genet 37:89–100
Shan B, Ren Y, Sun J, Tu X, Jiang Z, Ju X, Zang R, Wang H (2013) A prospective study of fertility-sparing treatment with megestrol acetate following hysteroscopic curettage for well-differentiated endometrioid carcinoma and atypical hyperplasia in young women. Arch Gynecol Obstet 288:1115–1123
Tock S, Jadoul P, Squifflet JL, Marbaix E, Baurain JF, Luyckx M (2018) Fertility sparing treatment in patients with early stage endometrial cancer, using a combination of surgery and GnRH agonist: a monocentric retrospective study and review of the literature. Front Med (Lausanne) 5:240
Park JY, Seong SJ, Kim TJ, Kim JW, Bae DS, Nam JH (2017) Significance of body weight change during fertility-sparing progestin therapy in young women with early endometrial cancer. Gynecol Oncol 146:39–43
Park J, Seong SJ, Kim T, Kim JW, Kim SM, Bae D, Nam J (2013) Pregnancy outcomes after fertility-sparing management in young women with early endometrial cancer. Obstet Gynecol 121:136–142
Xlxj TONG (2013) Fertility-preserving treatment and pregnancy outcomes in the early stage of endometrial carcinoma. Chin Med J (Engl) 126:2965–2971
Signorelli M, Caspani G, Bonazzi C, Chiappa V, Perego P, Mangioni C (2009) Fertility-sparing treatment in young women with endometrial cancer or atypical complex hyperplasia: a prospective single-institution experience of 21 cases. BJOG 116:114–118
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
None.
Author information
Authors and Affiliations
Contributions
YX: project development, data collection, manuscript editing and submission; GL: data analysis; DDL: data collection or management; JYJ: data collection; RQG: design and supervise the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
All procedures performed in studies were in accordance with the ethical standards of the institutional and/or national research committee (the Dalian Municipal Maternal and Child Health Care Hospital) and with the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xi, Y., Liu, G., Liu, D. et al. Efficacy and pregnancy outcomes of hysteroscopic surgery combined with progestin as fertility-sparing therapy in patients with early stage endometrial cancer and atypical hyperplasia. Arch Gynecol Obstet 307, 583–590 (2023). https://doi.org/10.1007/s00404-022-06626-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06626-w