Abstract
Purpose
Breast-conserving therapy (BCT) is the standard procedure for most patients with primary breast cancer (BC). By contrast, axillary management is still under transition to find the right balance between avoiding of morbidity, maintaining oncological safety, and performing a staging procedure. The rising rate of primary systemic therapy creates further challenges for surgical management.
Methods
Patients with primary, non-metastatic BC treated between 01.01.2003 and 31.12.2016 under guideline-adherent conditions were included in this study. For this prospectively followed cohort, breast and axillary surgery patterns are presented in a time-trend analysis as annual rate data (%) for several subgroups.
Results
Overall, 6700 patients were included in the analysis. While BCT rates remained high (mean 2003–2016: 70.4%), the proportion of axillary lymph node dissection has declined considerably from 80.1% in 2003 to 16.0% in 2016, while the proportion for sentinel lymph node biopsy (SLND) has increased correspondingly from 10.3 to 76.4%. Among patients with cT1-2, cN0 breast cancer receiving BCT with positive SLND, the rate of axillary completion has decreased from 100% in 2008 to 24.4% in 2016.
Conclusions
In the past decade, SLNB has been established as the standard procedure for axillary staging of clinically node-negative patients. Surgical morbidity has been further reduced by the rapid implementation of new evidence from the ACOSOG Z0011 trial into clinical routine. The results reflect the transition towards more individually tailored, less invasive treatment for selected patient subgroups, especially in regards to axillary lymph node management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC) is the most frequently diagnosed malignancy in women worldwide [1]. Mortality due to BC has declined over the last decade in most developed countries because of new developments in screening, diagnostics, and (neo)adjuvant systemic therapy in interdependency with structural (multidisciplinarity, specialized breast units) and quality improvement measures (evidence-based guidelines and certification processes) [2, 3]. These changes have been accompanied by a growing understanding of molecular genetics in the past decade [4, 5]. The increased knowledge has provided a new therapeutic view of BC as a heterogeneous disease that can be classified into different biological subtypes with various clinical and pathological features and different patterns of therapeutic response [6, 7]. This leads to the necessity of individually tailored treatment, which is implemented today for every single case within specialized breast units, through a multidisciplinary tumor board.
Besides the progress on outcome due to the advantages of (primary) systemic therapy [8], surgery is still an essential and obligatory part of treatment for the early stage BC. Since the important studies of Fisher et al. [9] and Veronesi et al. [10] 25 years ago, breast-conserving therapy (BCT) has become established as the standard treatment for most patients with the early BC, with oncological outcomes comparable to a more radical surgical approach. Surgical morbidity has been further reduced during the past 2 decades through the implementation of axillary staging via sentinel lymph node biopsy (SLND). It has been shown that the selective removal of the sentinel lymph node(s) has lower morbidity [11] with oncological outcomes equivalent to those of complete axillary lymph node dissection (ALND) [12]. Thus, SLND is the current standard of care for a clinically unsuspicious axilla (cN0) [13, 14].
In recent years, the paradigm of compulsory completion with ALND upon finding positive sentinel lymph node(s) (SLN) has been called into question by several trials. The IBCSG 23-01 study showed that ALND could be avoided in patients with micrometastasis in SLN(s) [15]. The American College of Surgeons Oncology Group (ACOSOG) Z0011 trial was a further step in decreasing the morbidity of the surgical procedure and thus tailoring the operation of the axilla more individually. This randomized trial investigated the impact of omitting an ALND in clinically node-negative patients with cT1-2 tumors and 1 or 2 positive sentinel nodes who had undergone BCT followed by radiation of the affected breast. In the ACOSOG Z011 trial, SLND alone resulted in equivalent local control, disease-free survival, and overall survival rates compared to those seen after complete ALND. [16]. The AMAROS trial was able to show similar loco-regional control rates for patients with cT1-2 cN0 tumors with a positive SNLD that were treated with radiotherapy of the axillary region as an alternative to ALND [17]. In the ACOSOG Z0011 trial, only patients with BCT were included, whereas, in AMAROSS, 17% of the patients received a mastectomy as surgical treatment of the breast.
To reconstruct the transitions of surgical management in routine management over the past decade, breast and axillary surgery patterns are shown in a time-trend analysis. Therefore, the prospectively collected data of 6700 female cases with primary BC treated at a breast unit between 01.01.2003 and 31.12.2016 under routine clinical management were analyzed.
Methods
Patient selection
In 2002, a patient registry was set up for the purpose of quality monitoring and improvement. Since 1st January 2003, all patients treated at the Heidelberg University Breast Center have had data collected in a standardized way and entered into the registry at the time of their consultations and treatment. Here, we performed a post hoc analysis of that registry data. Our ethics commission provided approval to perform and report this analysis of data. All cases included in this cohort study are managed under certified conditions which were monitored frequently by a yearly re-certification process by the German certification board of the German Cancer Society (DKG) and the German Society of Senology (DGS).
Patients from the registry were included in the present analysis if they had invasive or carcinoma-in situ cancer of the breast and were newly diagnosed or treated between 1st January 2003 and 31st December 2016, including a total of n = 7904 cases. Any one of the following characteristics led to exclusion from this analysis (in this order, in total n = 1204): male sex (n = 54), no primary diagnosis (n = 29), distant metastasis at the time of diagnosis (n = 429), and patients who did not receive surgery for whatever reason (n = 692), most of whom were reports to third parties in combination with external surgery and/or adjuvant treatment. Thus, the final cohort for this study comprised n = 6700 cases that had breast surgery between 2003 and 2016.
Statistical analysis
Breast and axillary surgical treatment patterns at the Heidelberg Breast Unit are presented here in a longitudinal time-trend analysis. The information was analyzed descriptively and is presented as the annual percentage rate. Descriptive statistics were used to characterize the patient sample and the epidemiology of their tumor characteristics. Data were analyzed using SPSS software version 22 (IBM; Armonk, NY, USA). For visualization of the annual rates, the corresponding graphs were generated in Excel 2016 (Microsoft; Redmond, WA, USA).
Results
Patient and tumor characteristics
The final study sample comprised n = 6700 cases who underwent breast surgery. Among them, 5942 (88.7%) had invasive carcinoma, and 758 cases (11.3%) had carcinoma-in situ (CIS). Most invasive BC cases were hormone-receptor positive (estrogen: 76.9%, progesterone: 70.8%) and HER-2 negative (82.2%), and had a maximum tumor size of 2 cm (pT1: 61.3%, ypT1: 36.4%) and did not have axillary lymph node involvement (69.9%). The median age of the whole cohort was 57. Table 1 presents detailed patient and tumor characteristics.
Most of all cases underwent BCT (n = 4719 patients, 70.4%). Axillary lymph node management for invasive carcinoma was provided for a total of 5942 patients. Among them, 2928 patients (49.3%) had SLND alone, 2178 (36.7%) had ALND alone, 742 (12.5%) had SLND with subsequent ALND (i.e., completion of the axillary dissection), and 94 (1.6%) had no axillary intervention. Among the 5942 patients with invasive breast cancer, 2659 (44.8%) had chemotherapy, of whom 1660 (62.4%) underwent adjuvant chemotherapy (ACT), and 999 (37.6%) underwent neoadjuvant chemotherapy (NACT).
Changes in breast and axillary surgery between 2003 and 2016
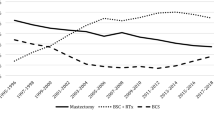
BCT rates were analyzed for the whole cohort as well as separately for invasive vs. CIS tumors and NACT vs. ACT between 2003 and 2016. The proportion of all patients who underwent BCT has declined from 83.5% in 2003 to 61.4% in 2006 (the lowest annual rate) and then remained stable in the past decade until 2016 on a somewhat higher level (2016: 72.3%, mean 2003–2016: 70.4%). There are almost parallel developments separately for invasive vs. in situ tumors (Fig. 1). Concerning invasive cases with adjuvant vs. neoadjuvant therapy, there were almost similar rates of BCT in the recent years, although the BCT rates for NACT cases were significantly lower than for ACT cases in 2005–2008 (2003: 78.2% vs. 84.9%; 2016: 68.7% vs. 62.6%); (Fig. 2).
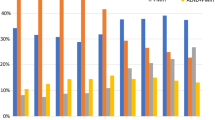
Axillary management patterns were analyzed for the whole cohort and also separately for the management after BCT vs. after mastectomy, and for invasive cases with NACT vs. ACT for 2003–2016. A distinct development is visible here: the proportion of ALND alone has declined from 80.1% in 2003 to 16.0% in 2016, while the proportion for SLND (alone) has increased correspondingly from 10.3 to 76.4% (Fig. 3a). Separately concerning the performed breast surgery procedure, there was a higher rate of SLND as axillary staging for patients with BCT (2016: 84.1%) in comparison to patients with mastectomy (2016: 58.5%) (Fig. 3b, c).
The overall lymph node findings after axillary staging (including SLND + ALND) were analyzed as well, in total and separately for the ACT vs. NACT group (Fig. 4). A similar trend is visible here: the average number of lymph nodes removed has declined over the study period, both in the ACT group (Fig. 4a) and in the group after NACT (Fig. 4b). However, in the group after NACT, the average number of lymph nodes affected has declined in comparison to the ACT group. Thus, the ratio of how many lymph nodes had to be removed for every affected lymph node is still higher in the NACT group, although, over the time period, the ratio remained stable in both groups, in favor of the ACT group.
Adoption of the ACOSOG Z0011 and AMAROS trials into clinical practice
We have analyzed the rates of completion ALND over time along the ACOSOG Z0011 trial results [16], as an example of the adoption of new findings from clinical trials into routine management. Since the presentation of those trial results in 2010 at Annual Meeting of the American Society of Clinical Oncology (ASCO), a rising percentage of patients have been treated accordingly to those results (2008: 0.0% vs. 2016: 75.6%) (Fig. 5a). On the other hand, another relevant trial with equivalent intention has not been implemented in a comparable matter. Applying the AMAROS study criteria, our results suggest that only a low proportion of patients with positive SLNs have been treated by axillary radiotherapy only in recent years as recommended by that clinical trial (2016: 8.6%) (Fig. 5b).
Discussion
Specialized breast units are in front to bring changing recommendations based on new evidence from clinical trials into routine practice. Although periodic evaluations are required as part of the implementation of re-certification audits and national benchmarking processes, it can be useful to analyze the outcome of these patterns of care over a longer period as already done for this cohort [18].
The general time-trend for our cohort during the past decade follows a development towards more individually tailored and less invasive treatment concepts for selected patient subgroups. In particular, this development involves: first, systemic treatment with a rising portion of primary systemic therapy [8]; second, surgery, as it was the focus in this study.
Concerning surgery, a noteworthy trend was the rising rate for SLNB from 10% in 2003 to almost 80% in 2016, which was introduced first into clinical practice in the late 1990s [19, 20]. Thus, ALND as a primary staging procedure was replaced completely by SLNB in clinically node-negative patients, which has reduced the associated morbidity for most BC patients. Concerning the surgical procedure in the breast, the proportion BCT (ca. 70%) and mastectomy (ca. 30%) stayed more or less unchanged during the past decade. In other words, BCT with subsequent radiotherapy has remained the standard treatment. Its equivalency to mastectomy regarding survival has been well established since the main trials from Veronesi et al. 25 years ago [21]. Nonetheless, mastectomy was performed on average in about 25–30% of our registry sample each year during the study period, comparable to epidemiologic rates [22].
For patients who underwent mastectomy, the rate of ALND declined from almost 90% in 2003 to less than 40% in 2016. The rising rate for primary systemic therapy and the increase of a pathologic complete response in recent years might be related to a decreasing rate of patients who have a positive SLND result after neoadjuvant chemotherapy. For these patients, subsequent axillary completion could be spared. The overall rate of affected lymph nodes after NACT is also lower than in the ACT patients, supporting the effectiveness of primary systemic therapy.
The rates of BCT and mastectomy during the past decade in our registry have been comparable to results on the European level presented by the European Society of Breast Cancer Specialists (EUSOMA) [22]. A rising rate of mastectomies, as reported in the US (due to prophylactic procedures, extended genetic testing, and rising assumed individual safety awareness), was not observed in our cohort [23, 24]. Despite this recent rise of mastectomy rates, the general trend toward less invasive treatment options remains indisputable [25], and further impacts on guideline recommendations can be expected. The latest important controversy was generated by the ACOSOG Z0011 trial. The results (which were controversial due to study design reasons) implied the waiving of ALND even for subgroups of the early breast cancer patients with 1–2 positive lymph nodes and breast conservation treatment with guideline-adherent adjuvant therapy [15, 16]. Moreover, further results from a study by Dengel et al. imply that a subsequent ALND could be avoided according to the ACOSOG Z0011 inclusion criteria for approximately 85% of patients with clinically node-negative disease who underwent BCT in routine clinical practice and were found to have positive sentinel nodes. The remaining patients with either gross extracapsular extension or ≥ 3 positive nodes are at high risk for extensive residual axillary disease and a completion axillary dissection is still warranted [26]. Due to the high impact and controversy that was created by the ACOSOG Z0011 study, we also applied those study criteria to our study sample. This was done to show how new research results were integrated into routine care over time. In our study sample, a rising portion of patients were treated according to the ACOSOG Z0011 criteria (up to 75% in 2016), which shows the rapid uptake, since the results were presented in 2010 at ASCO annual meeting, but not yet considered by official guideline recommendations. Furthermore, for a monocentric U.S. cohort, this rate of 75% for the implementation of the ACOSOG Z0011 results was reported already for the year 2012 [27], i.e., 2–3 years earlier than in our cohort. Nonetheless, omitting ALND according to ACOSOG Z0011 also implies a relevant impact on adjuvant treatment decisions due to the loss of the prognostic value of the number of affected lymph nodes in the axilla, which should be considered in this context, as reported earlier [28]. Although the AMAROS study had similar results suggesting axillary radiotherapy instead of ALND, it did not have a relevant impact on treatment decision for patients with positive SLN(s) in our study sample. Most of the patients enrolled in the AMAROS trial were treated without axillary lymph node dissection or axillary radiotherapy according to the results of the ACOSOG Z0011 study [29]. Moreover, neither study considered the potential effect of primary systemic therapy on affected axillary lymph nodes by excluding patients after treatment with NACT [30]. In clinical routine, about two-thirds of patients receiving chemotherapy today are treated in a neoadjuvant setting [8]. Despite the discrepancy between the implementation of ACOSOG Z0011 vs. AMAROS, the results presented here in a time-trend analysis from a single breast care unit show a remarkable uptake of results from recent clinical research into routine practice. Future study results might specify cohorts with clinically nodal-negative axilla who do not need any surgical axillary staging with SLND at all, e.g., results from the running INSEMA trial [31].
Conclusions
Our results reflect the fundamental developments in surgical management of early BC patients in the past decade in a specialized breast unit. BCT is still the standard procedure for most patients. A rising number of patients have been spared the unnecessary morbidity of ALND in the past decade, mainly due to implementation of the SLND technique. Future trials will define more precise subgroups that might be spared from any unnecessary axillary intervention. Furthermore, the impact of NACT for breast and axillary surgical management should be answered in future trials.
References
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63(1):11–30
Desantis C, Ma J, Bryan L, Jemal A (2014) Breast cancer statistics, 2013. CA Cancer J Clin 64(1):52–62
Early Breast Cancer Trialists’ Collaborative Group (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365(9472):1687–1717
Cancer Genome Atlas Network (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70
Ellis MJ, Perou CM (2013) The genomic landscape of breast cancer as a therapeutic roadmap. Cancer Discov 3(1):27–34
Prat A, Perou CM (2011) Deconstructing the molecular portraits of breast cancer. Mol Oncol 5(1):5–23
Weigelt B, Baehner FL, Reis-Filho JS (2010) The contribution of gene expression profiling to breast cancer classification, prognostication and prediction: a retrospective of the last decade. J Pathol 220(2):263–280
Hennigs A, Riedel F, Marme F, Sinn P, Lindel K, Gondos A et al (2016) Changes in chemotherapy usage and outcome of early breast cancer patients in the last decade. Breast Cancer Res Treat 160(3):491–499
Fisher B, Redmond C, Poisson R, Margolese R, Wolmark N, Wickerham L et al (1989) Eight-year results of a randomized clinical trial comparing total mastectomy and lumpectomy with or without irradiation in the treatment of breast cancer. N Engl J Med 320(13):822–828
Veronesi U, Banfi A, Salvadori B, Luini A, Saccozzi R, Zucali R et al (1990) Breast conservation is the treatment of choice in small breast cancer: long-term results of a randomized trial. Eur J Cancer 26(6):668–670
Mansel RE, Fallowfield L, Kissin M, Goyal A, Newcombe RG, Dixon JM et al (2006) Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC Trial. J Natl Cancer Inst 98(9):599–609
Veronesi U, Paganelli G, Viale G, Luini A, Zurrida S, Galimberti V et al (2006) Sentinel-lymph-node biopsy as a staging procedure in breast cancer: update of a randomised controlled study. Lancet Oncol 7(12):983–990
Kreienberg R, Albert US, Follmann M, Kopp IB, Kuhn T, Wockel A (2013) Interdisciplinary GoR level III guidelines for the diagnosis, therapy and follow-up care of breast cancer: short version-AWMF Registry No.: 032-045OL AWMF-Register-Nummer: 032-045OL-Kurzversion 3.0, Juli 2012. Geburtshilfe und Frauenheilkunde 73(6):556–583
D’Angelo-Donovan DD, Dickson-Witmer D, Petrelli NJ (2012) Sentinel lymph node biopsy in breast cancer: a history and current clinical recommendations. Surg Oncol 21(3):196–200
Galimberti V, Cole BF, Zurrida S, Viale G, Luini A, Veronesi P et al (2013) Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23-01): a phase 3 randomised controlled trial. Lancet Oncol 14(4):297–305
Giuliano AE, Hunt KK, Ballman KV et al (2011) Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 305(6):569–575
Donker M, van Tienhoven G, Straver ME, Meijnen P, van de Velde CJ, Mansel RE et al (2014) Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol 15(12):1303–1310
Heil J, Gondos A, Rauch G, Marme F, Rom J, Golatta M et al (2012) Outcome analysis of patients with primary breast cancer initially treated at a certified academic breast unit. Breast (Edinburgh, Scotland) 21(3):303–308
Edge SB, Niland JC, Bookman MA, Theriault RL, Ottesen R, Lepisto E et al (2003) Emergence of sentinel node biopsy in breast cancer as standard-of-care in academic comprehensive cancer centers. J Natl Cancer Inst 95(20):1514–1521
Rescigno J, Zampell JC, Axelrod D (2009) Patterns of axillary surgical care for breast cancer in the era of sentinel lymph node biopsy. Ann Surg Oncol 16(3):687–696
Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A et al (2002) Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med 347(16):1227–1232
Garcia-Etienne CA, Tomatis M, Heil J, Friedrichs K, Kreienberg R, Denk A et al (2012) Mastectomy trends for early-stage breast cancer: a report from the EUSOMA multi-institutional European database. Eur J Cancer 48(13):1947–1956
Mamtani A, Morrow M (2017) Why are there so many mastectomies in the United States? Ann Rev Med 68:229–241
Pesce CE, Liederbach E, Czechura T, Winchester DJ, Yao K (2014) Changing surgical trends in young patients with early stage breast cancer, 2003 to 2010: a report from the National Cancer Data Base. J Am Coll Surg 219(1):19–28
Garcia-Fernandez A, Chabrera C, Garcia-Font M, Fraile M, Barco I, Gonzalez S et al (2015) A study comparing two consecutive historical periods in breast cancer with a focus on surgical treatment, loco-regional recurrence, distant metastases and mortality. Clin Transl Oncol 17(4):296–305
Dengel LT, Van Zee KJ, King TA, Stempel M, Cody HS, El-Tamer M et al (2014) Axillary dissection can be avoided in the majority of clinically node-negative patients undergoing breast-conserving therapy. Ann Surg Oncol 21(1):22–27
Caudle AS, Hunt KK, Tucker SL, Hoffman K, Gainer SM, Lucci A et al (2012) American College of Surgeons Oncology Group (ACOSOG) Z0011: impact on surgeon practice patterns. Ann Surg Oncol 19(10):3144–3151
Aigner J, Smetanay K, Hof H, Sinn HP, Sohn C, Schneeweiss A et al (2013) Omission of axillary dissection according to ACOSOG Z0011: impact on adjuvant treatment recommendations. Ann Surg Oncol 20(5):1538–1544
Boughey JC (2014) How do the AMAROS trial results change practice? Lancet Oncol 15(12):1280–1281
Kantor O, Pesce C, Liederbach E, Wang CH, Winchester DJ, Yao K (2017) Are the ACOSOG Z0011 trial findings being applied to breast cancer patients undergoing neoadjuvant chemotherapy? Breast J 23(5):554–562
Reimer T, Stachs A, Nekljudova V, Loibl S, Hartmann S, Wolter K, Hildebrandt G, Gerber B (2017) Restricted axillary staging in clinically and sonographically node-negative early invasive breast cancer (c/iT1-2) in the context of breast conserving therapy: first results following commencement of the intergroup-sentinel-mamma (INSEMA) trial. Geburtshilfe Frauenheilkd 77(2):149–157
Acknowledgements
We would like to thank Christian Lange, Brigitte Wiegand, and Ibrahim Kilic for the medical documentation and data management. We would also like to thank Michael Hanna, Ph.D., (Mercury Medical Research & Writing) for proof-reading the manuscript.
Funding
Funding for this research was provided by the German Cancer Aid (Grant No. 70112082).
Author information
Authors and Affiliations
Contributions
Protocol/project development: AH, FR, and JH. Data collection or management: JH, MG, FS, JR, and CS. Data analysis: FR and AH. Manuscript writing/editing: FR, AH, and JH. Manuscript review: FR, JH, MG, BS, SH, SS, JR, FS, CS, and AH.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interests (e.g., employment, consultancies, stock ownership, honoraria, paid expert testimony, patent applications/registrations, or grants, or other funding with regard to this study) for any of the authors.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the University of Heidelberg and was conducted in accordance with the Declaration of Helsinki. Because the study was deemed as without risk and used only anonymized routinely collected data, the Ethics Committee of the University of Heidelberg did not require approval for patient consent.
Rights and permissions
About this article
Cite this article
Riedel, F., Heil, J., Golatta, M. et al. Changes of breast and axillary surgery patterns in patients with primary breast cancer during the past decade. Arch Gynecol Obstet 299, 1043–1053 (2019). https://doi.org/10.1007/s00404-018-4982-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4982-3