Abstract
Purpose
Labor induction is performed in 20 % of pregnancies. However, the impact of the indications for induction on its failure rate has hardly been investigated. We aimed to evaluate the association of indications for labor induction with failure rate.
Methods
Background and delivery-related data were retrospectively collected for all women with a viable term singleton pregnancy, who underwent labor induction with a PGE2 vaginal insert in 2013–2014. Reasons for induction were categorized as maternal indications, hypertensive disorders, premature rupture of membranes, and fetal indications. Induction failure was defined as Bishop score ≤7 at 24 h after PGE2 administration, cesarean delivery due to latent phase dystocia or removal of the insert due to non-reassuring fetal heart rate followed by emergency cesarean delivery. Outcome measures were rate of induction failure (primary) and rate of cesarean delivery (secondary).
Results
The cohort included 1066 women. Those who failed induction (n = 213, 20 %) were more likely to be nulliparous (69.5 vs. 45.7 %, p < 0.0001), older (31 vs. 30 years, p = 0.047), and at an earlier gestational age (39.4 vs. 40.0 weeks, p < 0.0001). Among nulliparous women, maternal indications were significantly associated with induction failure (aOR 2.52, 95 % CI 1.28–4.95, p = 0.007) and cesarean delivery (aOR 2.36, 95 % CI 0.40–2.29, p = 0.019). Among multiparous women, hypertensive disorders (aOR 7.26, 95 % CI 1.89–27.87, p = 0.004) and maternal indications (aOR 4.22, 95 %CI 1.14–15.58, p = 0.031) were significantly associated with induction failure but not cesarean delivery.
Conclusions
The indication for induction of labor may impact its failure rate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Induction of labor (IoL) by ripening the cervix is a common obstetrical intervention, performed in approximately 20 % of pregnancies [1]. It is routinely advised for gravid women with an unfavorable cervix in whom maternal or fetal health risks outweigh the anticipated risk of ongoing gestation [2]. As such, specific indications for IoL may include, among others, premature rupture of membranes (PROM), diabetes, hypertensive disorders, maternal chronic diseases, fetal growth restriction, postdate pregnancy, oligohydramnios, and cholestasis of pregnancy. At the same time, it is important to consider that IoL itself may be associated with some complications, namely, uterine tachysystole with or without corresponding fetal heart rate changes [3–6], inadequate labor progression [7, 8], and cesarean delivery [9–16]. Factors increasing the risk of failed IoL are unfavorable cervix, low parity, older maternal age, early gestational age, high neonatal birth weight, maternal short stature, and increased body mass index [17–19].
Most studies of IoL to date have included highly varied populations in terms of gestational age and method of cervical ripening and indications. Whether the specific indications for IoL have different effects on the likelihood of its success or failure has hardly been investigated [20, 21]. The studies were also limited by the bias of defining cesarean delivery as a primary outcome for IoL, as cesarean delivery may also be performed for reasons unrelated to the induction procedure, long after active labor has been achieved.
Therefore, the aim of the present study was to evaluate the impact of indications for labor induction on the rate of induction failure and the risk of cesarean delivery.
Materials and methods
The database of a tertiary university-affiliated medical center was retrospectively reviewed for all women with a viable singleton pregnancy who underwent cervical ripening between January 2013 and December 2014. The study was approved by our local institutional review board (Approval No. 0351-13).
Study population
Inclusion criteria were vertex presentation, Bishop Score ≤7 and gestational age 34–41 completed weeks. Exclusion criteria were multiple gestation, non-vertex presentation, prior cesarean delivery, stillbirth, and chromosomal or major structural fetal anomalies. Women who had elective IoL for nonmedical reasons and women for whom data were insufficient to properly ascertain the indication for IoL were excluded as well.
Data collection
The following data were collected for all eligible patients from the departmental health records: demographic and obstetric characteristics, physical characteristics, pregnancy-related complications, indication for labor induction, findings on cervical examination at admission, gestational age at delivery, mode of delivery, indications for cesarean delivery, and neonatal birth weight.
Induction of labor protocol
According to our departmental protocol, a prostaglandin E2 (PGE2) slow-release vaginal insert placed in the posterior fornix (Propess 10 mg, Ferring Pharmaceuticals AB, Kiel, Germany) was used for labor induction in all the cases. Physician assessment and fetal heart rate monitoring were performed immediately before and 1 h after insertion and again at 12 and 24 h or in the event of painful uterine contractions, vaginal bleeding, or suspected PROM. The insert was removed on findings of cervical ripening (Bishop Score >7), uterine hyperstimulation (>5 contractions in 10 min), non-reassuring fetal heart rate, or if none of these has occurred at 24 h after placement. Women in whom cervical ripening was achieved were transferred to the delivery room, and if necessary, labor was augmented with oxytocin and/or amniotomy under continuous electronic fetal heart rate monitoring. If cervical ripening was not achieved, a subsequent trial was undertaken using either oxytocin or an extra-amniotic balloon, depending on the Bishop score, pattern of uterine contractions and physician discretion.
Definitions
Reasons for inducing labor were categorized into four groups on the basis of previous medical literature [22]: (1) maternal indication: diabetes mellitus, renal disease, pulmonary disease, cardiovascular disease, or other chronic illnesses (2) any hypertensive disorder of pregnancy; (3) PROM; and (4) fetal indication: suspected fetal growth restriction (sonographic estimated fetal weight <10th percentile according to local reference values [23]), oligohydramnios (amniotic fluid index ≤5 cm), suspected macrosomia (sonographic estimated fetal weight ≥4000 g), cholestasis of pregnancy, or postdate pregnancy (gestational age ≥41 + 0 weeks). IoL was considered failed under the following conditions: (1) Bishop score <7 at 24 h after PGE2 administration (lack of cervical ripening); (2) Cesarean delivery due to prolonged latent phase. This was defined as labor dystocia 24 h after PGE2 administration and dilatation of <4 cm after at least 12 h of oxytocin infusion with adequate contractions in the presence of ruptured membranes; or (3) removal of the PGE2 insert before 24 h due to hyperstimulation and non-reassuring fetal heart rate, leading to an emergency cesarean delivery.
Outcome measures
The primary outcome measure was the IoL failure rate and the secondary outcome measure—cesarean delivery rate, both stratified by the preliminary indication for IoL. As pre-labor PROM seems to be the most natural indication for IoL with the lowest IoL failure rates, we evaluated the other indications with reference to it. In addition, as parity is considered a major determinant for successful IoL, outcomes were analyzed separately for nulliparous and multiparous women.
Statistical analysis
Data analysis was performed with the SPSS package, v21.0 (Chicago, IL). Student’s t test and Mann–Whitney U test were used to compare continuous variables with and without normal distribution, respectively, between groups. Chi-square and Fisher’s exact tests were used for categorical variables, as appropriate. Logistic regression analysis was used to adjust IoL failure rates to known confounders, such as maternal age, gestational age at delivery and neonatal birth weight. Differences were considered significant at p < 0.05.
Results
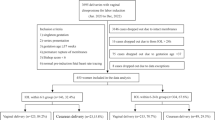
Of the 15,564 women who gave birth at our center during the study period, 1066 (6.8 %) met the inclusion criteria. IoL failed in 213 (20 %), due to lack of cervical ripening (n = 162, 76 %), hyperstimulation and non-reassuring fetal heart rate (n = 40, 18.8 %), and labor dystocia (n = 11, 5.2 %) (Fig. 1).
About half the cohort was nulliparous (n = 538, 50.5 %) and half multiparous (n = 526, 49.5 %). The nulliparous group accounted for a considerably higher percentage of the induction failures (148/538, 27.5 %) than the multiparous group (65/528, 12.3 %). In both the groups, the most common reason for performing IoL was fetal indication, followed by maternal indication. The relative proportion of IoL procedures for maternal indication was higher in the multiparous group (27.7 vs. 17.7 %) (Table 1).
The demographic and obstetrical characteristics of the study cohort, stratified by IoL failure or success, are presented in Table 2. Women who failed IoL were significantly more likely to be nulliparous (69.5 vs. 45.7 %, p < 0.0001), older (median age 31 vs. 30 years, p = 0.047), and at an earlier median gestational age at delivery (39.4 vs. 40.0 weeks, p < 0.0001). There were no differences between the groups in pre-gestational body mass index or neonatal birth weight.
Failed IoL and cesarean delivery rate by indication: nulliparous women
Analysis of the nulliparous group revealed a significant association of the indication for induction with induction failure. The risk of failure was higher when IoL was performed because of maternal indication (41.1 %) than hypertensive disorders (33.3 %), fetal indication (23.7 %), or PROM (20.7 %) (Fig. 2). On logistic regression analysis controlling for maternal age, gestational age at delivery, and neonatal birth weight, only maternal indication remained significantly associated with the failure of IoL (aOR 2.52, 95 % CI 1.28–4.95, p = 0.007) and with cesarean delivery (aOR 2.36, 95 % CI 0.40–2.29, p = 0.019) (Table 3).
Failure of IoL and cesarean delivery rate by indication: multiparous women
Analysis of the multiparous group revealed a significant association of the indication for induction with induction failure. The risk of failure was higher when IoL was performed because of hypertensive disorders (26.7 %) than maternal indication (15.8 %), fetal indication (8.5 %), or PROM (5.9 %) (Fig. 2). On logistic regression analysis controlling for maternal age, gestational age at delivery, and neonatal birth weight, hypertensive disorders (aOR 7.26, 95 % CI 1.89–27.87, p = 0.004), and maternal indication (aOR 4.22, 95 %CI 1.14–15.58, p = 0.031) remained significantly associated with the failure of IoL but not with cesarean delivery (Table 4).
Discussion
This retrospective study of women undergoing cervical ripening with PGE2 sought to evaluate the association between the indication for IoL and rates of IoL failure and cesarean delivery. There were several important findings: (1) the overall IoL failure rate was 20 % and the overall rate of cesarean delivery, 14.2 %; (2) women who failed IoL were significantly more likely to be nulliparous, older and at an earlier gestational age; and (3) among nulliparous women, a maternal indication for IoL was associated with a higher risk of failed IoL and cesarean delivery, while among multiparous women, a maternal indication and hypertensive disorders were associated with a higher risk of unsuccessful IoL but not cesarean delivery.
The literature reports IoL failure rates ranging from 20 to 30 % [24, 25] and cesarean delivery rates of approximately 20 % [26, 27]; both of these are compatible with our findings. As in the present study, others have identified nulliparity [10], advanced maternal age [19], and earlier gestational age [28] as risk factors for IoL failure. There are also reports of a significant association of gestational age with cesarean delivery [19–21, 28–30]. Interestingly, in the only two studies focusing on the effect of specific indications for induction on the risk of post-induction cesarean delivery, Parkes et al. [20] found a significant association of IoL failure with older gestational age (≥40 weeks; aOR = 2.028, 95 % CI 1.143–3.594, p = 0.0016), and Gerli et al. [21] found that postdate pregnancy was the only significant risk factor. Thus, gestational age may have a bimodal effect in the setting of IoL. Several studies have suggested that the optimal timing for labor induction is 36–40 weeks of gestation and that the success rate is lower before and after this period [29, 30].
In terms of indications for IoL, Parkes et al. [20] reported that among the 343 nulliparous women out of the 796 women evaluated, a fetal indication for IoL was carried with a significantly higher risk of cesarean delivery (aOR = 3.76, 95 % CI 1.153–12.265) than the other indication categories. However, in the multiparous group, no association was found for any of the indication categories. They suggested that these results represent another aspect of the well-established risk of perinatal morbidity and mortality in compromised fetuses. By contrast, in our nulliparous group, only maternal indication was significantly associated with failed IoL and with cesarean delivery among the indication categories. We speculate that this discrepancy is attributable to the different outcome measures of our study, or alternatively to the specific indications within the fetal indication group, which may have a different impact on the success of induction (growth restriction, macrosomia or oligohydramnios). We limited the definition of failed IoL to a failure to achieve cervical ripening or active labor. This was done to avoid introducing a bias, as the decision to perform intrapartum cesarean delivery may also be influenced by other factors stemming from the active labor itself, long after induction has been successfully achieved. The assumption that IoL is a better outcome measure than cesarean delivery was further strengthened by our detection of a significant effect of both maternal indication and hypertensive disorders on IoL failure in the multiparous group. As such, our results probably represent the true association of IoL indications with the process of induction itself, without confounding factors. The study of Gerli et al. [21] is also subject to the same bias. In addition, it was limited by the categorization of the cohort of 324 women by nine individual indications for IoL, which led to relatively small numbers for each.
The strengths of our study include the inclusion of a large uniform cohort of women all of whom underwent the same management protocol in terms of IoL and delivery as well as and the case-by-case review of the data by specialists. The indications were grouped by category to ensure an adequate sample. Furthermore, we used more accurate outcome measures: earlier studies evaluated rates of cesarean delivery regardless of the success or failure of induction, whereas we evaluated the success or failure of IoL it terms of the achievement of active labor, regardless of the ultimate mode of delivery. The main study limitation is its retrospective design.
In conclusion, the indications for IoL apparently have an important impact on its success or failure. Our data suggest that maternal indications for IoL pose a significant risk of failure in both nulliparous and multiparous women.
References
Chauhan SP, Ananth CV (2012) Induction of in the United States: a critical appraisal of appropriateness and reducibility. Semin Perinatol 36:336–343
American College of Obstetricians and Gynecologists (2009) Induction of labor. ACOG practice bulletin no. 107. Obstet Gynecol 114:386–397
Agency for Healthcare Research and Quality (2008) Maternal and neonatal outcomes of elective induction of labor. AHRQ, Rockville (MD) (AHRQ Evidence Report/Technology Assessment No.176)
Witter FR, Rocco LE, Johnson TR (1992) A randomized trial of prostaglandin E2 in a controlled-release vaginal pessary for cervical ripening at term. Am J Obstet Gynecol 166:830–834
Gelber S, Sciscione A (2006) Mechanical methods of cervical ripening and labor induction. Clin Obstet Gynecol 49:642–657
Hofmeyr GJ, Gülmezoglu AM (2003) Vaginal misoprostol for cervical ripening and induction of labor. Cochrane Database Syst Rev 1:CD000941
Vahratian A, Zhang J, Troendle JF, Sciscione AC, Hoffman MK (2005) Labor progression and risk of cesarean delivery in electively induced nulliparas. Obstet Gynecol 105:698–704
Rouse DJ, Owen J, Hauth JC (2000) Criteria for failed labor induction: prospective evaluation of a standardized protocol. Obstet Gynecol 96:671–677
Isono W, Nagamatsu T, Uemura Y, Fujii T, Hyodo H, Yamashita T, Kamei Y, Kozuma S, Taketani Y (2011) Prediction model for the incidence of emergent cesarean section during induction of labor specialized in nulliparous low-risk women. J Obstet Gynaecol Res 2011(37):1784–1791
Ennen CS, Bofill JA, Magann EF, Bass JD, Chauhan SP, Morrison JC (2009) Risk factors for cesarean delivery in preterm, term and post-term patients undergoing induction of labor with an unfavorable cervix. Gynecol Obstet Invest 67:113–117
Cnattingius R, Hoglund B, Kieler H (2005) Emergency cesarean delivery in induction of labor: an evaluation of risk factors. Acta Obstet Gynecol Scand 84:456–462
Roos N, Sahlin L, Ekman-Ordeberg G, Kieler H, Stephansson O (2010) Maternal risk factors for postterm pregnancy and cesarean delivery following labor induction. Acta Obstet Gynecol Scand 89:1003–1010
Moore LE, Rayburn WF (2006) Elective induction of labor. Clin Obstet Gynecol 49:698–704
Luthy DA, Malmgren JA, Zingheim RW (2004) Cesarean delivery after elective induction in nulliparous women: the physician effect. Am J Obstet Gynecol 191:1511–1515
Cammu H, Martens G, Ruyssinck G, Amy JJ (2002) Outcome after elective labor induction in nulliparous women: a matched cohort study. Am J Obstet Gynecol 186:240–244
Bailit JL, Gregory KD, Reddy UM, Gonzalez-Quintero VH, Hibbard JU, Ramirez MM, Branch DW, Burkman R, Haberman S, Hatjis CG, Hoffman MK, Kominiarek M, Landy HJ, Learman LA, Troendle J, Van Veldhuisen P, Wilkins I, Sun L, Zhang J (2010) Maternal and neonatal outcomes by labor onset type and gestational age. Am J Obstet Gynecol 202:245.e1–245.e12
Pevzner L, Rayburn WF, Rumney P, Wing DA (2009) Factors predicting successful labor induction with dinoprostone and misoprostol vaginal inserts. Obstet Gynecol 114:261–267
Beckmann M (2007) Predicting a failed induction. Aust N Z J Obstet Gynaecol 47:394–398
Tolcher MC, Holbert MR, Weaver AL, McGree ME, Olson JE, El-Nashar SA, Famuyide AO, Brost BC (2015) Predicting cesarean delivery after induction of labor among nulliparous women at term. Obstet Gynecol 126:1059–1068
Parkes I, Kabiri D, Hants Y, Ezra Y (2016) The indication for induction of labor impacts the risk of cesarean delivery. J Matern Fetal Neonatal Med 29:224–228
Gerli S, Favilli A, Giordano C, Bini V, Di Renzo GC (2013) Single indications of induction of labor with prostaglandins and risk of cesarean delivery: a retrospective cohort study. J Obstet Gynaecol Res 39:926–931
Simpson JL, Richards DS, Otano L (2012) Induction of labor. In: Gabbe SG, Niebyl JR, Simpson JL et al (eds) Obstetrics: normal and problem pregnancies, 6th edn. Saunders Elsevier, Philadelphia
Dollberg S, Haklai Z, Mimouni FB, Gorfein I, Gordon ES (2005) Birth weight standards in the live-born population in Israel. Isr Med Assoc J 7:11–314
Vrouenraets FP, Roumen FJ, Dehing CJ, van den Akker ES, Aarts MJ, Scheve EJ (2005) Bishop score and risk of cesarean delivery after induction of labor in nulliparous women. Obstet Gynecol 105:690–697
Johnson DP, Davis NR, Brown AJ (2003) Risk of cesarean delivery after induction at term in nulliparous women with an unfavorable cervix. Am J Obstet Gynecol 188:1565–1570
Ehrenthal DB, Jianh X, Strobino DM (2010) Labor induction and the risk of a caesarean delivery among nulliparous women at term. Obstet Gynecol 116:35–42
Crowley P (2000). Interventions for preventing or improving outcome delivery at or beyond term. Cochrane Database Syst Rev 1(2):CD000170
Melamed N, Yogev Y, Hadar E, Hod M, Ben-Haroush A (2008) Preinduction cervical ripening with prostaglandin E2 at preterm. Acta Obstet Gynecol Scand 87:63–67
Heffner LJ, Elkin E, Fretts RC (2003) Impact of labor induction, gestational age, and maternal age on cesarean delivery rates. Obstet Gynecol 102:287–293
Seyb ST, Berka RJ, Socol ML, Dooley SL (1999) Risk of caesarean delivery with elective induction of labor at term in nulliparas. Obstet Gynecol 94:600–607
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to report.
Research involving human participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
This study was approved by the local institutional review board (IRB) of the Rabin medical center.
Obtaining informed consent was waived by the IRB.
Rights and permissions
About this article
Cite this article
Gabbay-Benziv, R., Hadar, E., Ashwal, E. et al. Induction of labor: does indication matter?. Arch Gynecol Obstet 294, 1195–1201 (2016). https://doi.org/10.1007/s00404-016-4171-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-016-4171-1