Abstract
Introduction
The use of total shoulder arthroplasty (TSA) to treat primary glenohumeral osteoarthritis (GHOA) is increasing. Factors influencing patient satisfaction after surgery have not been well documented. The aim of this study was to determine demographic, radiologic, and surgical, factors predictive for satisfaction after TSA for GHOA.
Materials and Methods
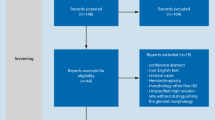
Between 2005 and 2012, 95 shoulders undergoing TSA for GHOA by a single surgeon were eligible for inclusion in the study. Age, gender, previous surgeries, American Society of Anesthesiologists (ASA) score, and Walch glenoid morphology were analyzed as satisfaction predictors. Patients with Walch glenoid type C were excluded.
Results
Data on 80/92(87 %) shoulders were available at a mean of 3 years (range 2–9). Three complications (3 %) and 2 failures (2 %) occurred. The outcome scores collected significantly improved from preoperative values (p < 0.05). Median patient satisfaction was 10/10. Gender, age, previous surgery, ASA score, and Walch morphology were not associated with patient satisfaction.
Conclusions
TSA provided excellent results for patients with idiopathic GHOA with low complication and failure rates. Outcomes after TSA for type B glenoid morphology with posterior subluxation were similar to outcomes after TSA for centered type A morphology. Overall patient satisfaction was high and was not influenced by the demographic, anatomic, and surgical variables investigated.
Level of Evidence: III, Therapeutic study, Retrospective Cohort Study
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The use of total shoulder arthroplasty (TSA) to treat symptomatic glenohumeral osteoarthritis (GHOA) is increasing [1]. In 2008, approximately 27,000 TSAs were performed in the United States, 77 % of which were done to manage GHOA [1]. Several studies have revealed better results and a lower revision rate for TSA compared to hemiarthroplasty for treatment of primary GHOA [2–4]. The most common causes for failure or complications following TSA are prosthetic loosening, glenohumeral instability, periprosthetic fracture, rotator cuff tears, infection, neural injury, and deltoid muscle dysfunction [5, 6]. Overall reported complication rates after TSA vary between 3 and 22.6 % [3, 6–12]. Secondary surgery rates have been reported ranging from 6.5 to 11.2 % [9, 10, 13, 14]. Even though long-term revision-free survival rates of primary TSA are reportedly high, 95 % at 10 years [15] and 83.2 % at 20 years [16], the identification of preoperative factors affecting outcomes is lacking in the literature.
Chen et al. reported a significant correlation between patients’ age and satisfaction after TSA as well as a significant correlation between satisfaction and the ASES score [17]. In addition to female gender, the abnormal glenoid morphology as defined by the Walch classification has been associated with inferior TSA outcomes. It has been reported that patients with glenoid type B according to the Walch classification involving posterior subluxation of the humeral head are at increased risk for complications and failure due to recurrent instability and early glenoid loosening [18–20]. Johnson et al. recently found that the overall patient’s preoperative health as measured by the American Society of Anesthesiologists (ASA, scale 1–6) score greater than 2 significantly increased the risk for surgical complications in TSAs, reverse TSAs, and revision arthroplasties, with a three times increased risk for prosthesis failure [21]. Harreld et al. demonstrated that patient satisfaction after TSA correlated more with subjective than objective measures such as the ASES score, Simple Shoulder Test, and SF-36 summary scores [13]. They concluded that subjective measures had a relatively low correlation with objective measures.
The purpose of this study was to analyze predictors for satisfaction after anatomic TSA for primary idiopathic GHOA. According to previously published variables [21–23], the hypothesis was that men, older age, no prior surgeries, ASA score of 1 or 2 (compared to 3 or more), and Walch glenoids A1 and A2 with centered humeral head (compared to B1 and B2 glenoids with posterior subluxation of the humeral head) would be predictors of patient satisfaction after TSA.
Materials and Methods
Study design
Institutional Review Board (IRB) approval was obtained prior to the initiation of this study. Between December 2005 and January 2013, all patients who underwent anatomic TSA for idiopathic GHOA performed by the senior surgeon (PJM) were assessed for eligibility. Patients with concomitant pathologies such as biceps tendinitis or small rotator cuff tears were included as minor changes within the rotator cuff have not been shown to significantly affect the functional outcome following TSA [24]. Patients with non-idiopathic osteoarthritis, e.g., resulting from rheumatoid arthritis, previous glenohumeral dislocations, or fractures of either the proximal humerus or glenoid, were excluded.
Prospective data collection
All data were prospectively collected and retrospectively analyzed. Demographic data (age, gender, and dominant shoulder), previous surgeries on the index shoulder, surgical techniques, concomitant pathologies, adjuvant treatments, and perioperative complications were collected for analysis. Minimum 2 years postoperative clinical scores were obtained.
Shoulder-specific clinical outcome scores were collected both pre- and postoperatively and included the American Shoulder and Elbow Surgeons’ Score (ASES; range 0 = 100, 100 = best), Quick Disabilities of the Arm Shoulder and Hand Score (QuickDASH; range 0 = 100, 0 = best), Single Assessment Numeric Evaluation (SANE; range 0 = 100, 100 = best), and the overall general health Short Form-12 Physical Component Summary (SF-12 PCS) scores. In addition, since the ASES score is composed of 50 % pain and 50 % function, both subscales were individually analyzed.
The ASA score (range 1–6, 1 = best) was retrospectively determined from a chart review. Glenoid morphology was categorized retrospectively according to the Walch classification [25] for analysis by means of preoperative MRI or CT scans on axial cuts. Type A1 glenoids are concentric with minor central erosion, and A2 glenoids are concentric with major central erosion. B1 glenoids have posterior humeral head subluxation with joint space narrowing and retroversion. B2 glenoids are biconcave with posterior subluxation of the humeral head (Fig. 1). Type C glenoids (dysplastic with retroversion greater than 25°) were excluded from the study.
All TSA were implanted via a deltopectoral approach and a lesser tuberosity osteotomy (Fig. 2). In patients with a B2 glenoid, the glenoid was eccentrically reamed to achieve a flat plane and essentially convert the B2 situation to a B1 situation (subluxation without biconcave glenoid). Bone grafting was not used in any case.
When analyzing factors associated with patient satisfaction, patients with an ASA score of 1 or 2 representing generally healthy patients were grouped together and compared to those with an ASA score of 3. In addition, patients with a Walch glenoid types A1 and A2 (centered)were grouped and compared to Walch glenoid types B 1 and B2 (posterior subluxation).
According to the general definition by Clavien et al. [26], failure was defined as revision surgery on the index shoulder for component loosening or dislocation, while complications included infection/wound dehiscence, nerve damage, periprosthetic fracture, arthrofibrosis/stiffness, rotator cuff tendon rupture, and increased local pain [26].
Statistical analysis
The primary goal of this analysis was to test whether gender, age, previous surgery, ASA grade, and Walch type were predictors of satisfaction following TSA. First, comparisons were made between preoperative and postoperative outcome scores using the paired t test. Next, potential predictors for patient satisfaction were assessed with nonparametric tests including the Mann–Whitney U-test and Spearman’s rho. Independent t tests were used to test for associations between potential predictors and the other outcome scales (ASES, DASH, SANE, SF12). Equal group variances were not assumed. Statistical significance was declared for p values less than 0.05, and all statistical analyses were performed using IBM SPSS Statistics, version 20 (Armonk, NY, USA).
Source of funding
There was no source of external funding.
Results
Between December 2005 and January 2013, a total of 132 shoulders underwent TSA by a single sports medicine fellowship-trained orthopedic surgeon. Among these, 83 patients (56 men, 27 women) with 95 shoulders (12 bilaterals) underwent a TSA for primary idiopathic GHOA at a median age of 66 years (range 44–80). Three patients with type C glenoid according to Walch were excluded from this study. Two patients with failure of TSA and secondary conversion to reverse TSA were excluded from the statistical outcome analysis: One patient with ASA grade 3 (1 %) sustained a periprosthetic humeral shaft fracture that was treated with ORIF 20 months after TSA implantation. Six years later, the shoulder function deteriorated due to loosening of the glenoid component (initially glenoid type A1). One year after conversion to reverse TSA, the ASES score was 78. Another patient (ASA grade 2) experienced loosening of the glenoid component and had conversion to reverse TSA 2 years after primary TSA implantation (glenoid type B2). The ASES score 1.5 years after implantation of the reverse TSA was 83.3.
Ninety shoulders were included in the final data analysis. Three (3 %) shoulders had a complication treated surgically (1 wound infection treated with prosthesis-retaining irrigation and debridement, 1 arthrofibrosis treated with arthrolysis and capsular releases, and 1 rupture of the subscapularis (SSC) tendon treated with SSC repair). The patients who had complications were included in the data analysis. Minimum 2-year follow-up was available for 78/90 (87 %) shoulders at a mean of 3 years (range 2–9) postoperatively. There were no significant differences in demographic or baseline patient-reported outcome variables between the patients with and without 2-year follow-up.
Median patient satisfaction was 10/10 (range 2–10, 1st quartile = 7). As shown in Table 1, patients demonstrated significant improvement in the ASES, SF-12 PCS, QuickDASH, and SANE scores from preoperative levels. There were substantial ceiling effects in patient satisfaction (56 %) and postoperative versions of ASES pain subscale (70 %), ASES function subscale (27 %), and ASES total (27 %).
Information on sex, age, previous surgery, and ASA score was available for all 78 patients with outcome data. As to be expected at an orthopedic referral practice, all patients in this study were either ASA 1, 2, or 3. MRI or CT imaging was available for 57/78 (73 %) patients for retrospective review of the glenoid morphology. According to Walch, 24/57 (42.1 %) of the patients had a glenoid type A1, 13/57 (22.8 %) had a glenoid type A2, 10/57 (17.5 %) had a glenoid type B1, and 14/57 (24.6 %) had a glenoid type B2. Table 2 presents group comparisons of patient satisfaction and patient-reported outcome scales for sex, previous surgery, ASA score, and Walch classification type. These four candidate predictors were not found to be significantly associated with any of the 5 postoperative outcomes measures that were investigated. Spearman correlations between age at surgery and patient outcomes are shown in Table 3. Age was not correlated with any of the five outcomes measures.
Time to follow-up was negatively correlated with preoperative SF-12 PCS score (ρ = −0.263; p = 0.046), indicating that the patient population may be generally healthier as the study period progressed. Correspondingly, both postoperative SF-12 PCS (ρ = −0.392; p < 0.001) and patient satisfaction (ρ = −0.264; p = 0.019) were also negatively correlated with time to follow-up. Two factor models showed that our five hypothesized predictors were not significantly associated with postoperative SF-12 PCS even when adjusting for length of follow-up.
Discussion
The most important finding of the current study was that anatomic TSA for the treatment of primary idiopathic GHOA provided excellent results with low complication and failure rates. Outcomes after TSA for type B glenoid morphology with posterior subluxation were similar to outcomes after TSA for centered type A morphology. In addition, demographic, anatomic, and surgical variables did not influence patient satisfaction after anatomic TSA. This is contrary to previously published literature [21–23], despite our study consisting of a similar age and gender distribution to previously reported studies [3, 4, 8, 12, 13, 17, 27, 28].
According to the previously reported finding that an ASA score greater than 2 increases the risk of medical and surgical complications for hip and knee arthroplasties, Johnson et al. recently analyzed TSAs, reverse TSAs, and revision arthroplasties [21]. They did not find a correlation to medical complications, but found that an ASA score greater than 2 significantly increased the risk for surgical complications, with a three times increased risk for prosthesis failure. In the current study, 1/6 (17 %) of the patients with ASA score of 3 (n = 6) had a TSA failure whereas only 1/86 (1.2 %) of patients with ASA score of 1 or 2 sustained a TSA failure. The ASES and SANE scores of patients with an ASA score of 3 were inferior and more variable than those of patients with ASA scores of 1 or 2. However, the low sample size of ASA three patients (n = 6) and high variability likely resulted in a non-significant difference. Of interest, the patient population analyzed by Johnson et al. had a larger percentage of ASA class 3 or above (37 %) compared to the current studies cohort (6.5 %), indicating that the patient population analyzed in this study were healthier [21].
The most common causes for failure or complications following TSA are prosthetic loosening, glenohumeral instability, periprosthetic fracture, rotator cuff tears, infection, neural injury, and deltoid muscle dysfunction [5, 6] Compared to reported overall complication rates after TSA ranging from 3 to 22.6 % [3, 6–12], our rate of 5 % (2 % failure and 3 % complications) is on the lower end of the spectrum. The same applies for our secondary surgery rate of 5 % (2 failures and 3 complications), which is lower than what other studies have found as previous studies reported a secondary surgery rate ranging from 6.5 to 11.2 % [9, 10, 13, 14]. Our low failure and complication rates may be explained by a variety of factors: a rather healthy patient population in terms of ASA score (91.7 % of patients having an ASA score of 1 or 2), a single-surgeon (PJM) high-output practice, the use of a lesser tuberosity osteotomy for subscapularis repair [29], the use of an anatomic TSA design [30, 31], as well as standardized surgical technique and postoperative rehabilitation. Two recent studies revealed significantly less blood loss, lower surgical time, shorter length of stay, and better outcomes for TSA by high-volume surgeons performing more than 15 cases annually compared with lower volume practitioners [28, 32]. Surgical skills and experiences likely contributed to the decreased complication and failure rates seen in the current patient cohort.
Failure of the glenoid component is considered to be the most important long-term complication [5, 11, 33]. Of interest, the risk for both loosening of the glenoid component and surgical revision after TSA has not changed in reported literature between 1976 and 2007. The reported yearly rate of symptomatic glenoid loosening is 1.2 %, and surgical revision is 0.8 % [23]. In addition to female gender, the glenoid-shape/Walch classification has been associated with a poorer outcome after TSA due to recurrent instability and early glenoid loosening [18–20]. Papadonikolakis et al. found the Walch classification to be a significant risk factor when grouping all distorted glenoid morphologies (A2, B1, B2, C) together and comparing to A1 glenoids. However, there were no significant differences when comparing the distorted glenoid morphologies individually to A1 glenoids [23]. The current study combined glenoid types B1 and B2 given the clinical challenges associated with this morphology due to posterior subluxtion of the humeral head and compared outcomes to patients with glenoid types A1 and A2 with centered humeral head. Comparable results were found between the two groups. This is an important finding as a recent study advocated that elderly patients (mean age of 74.1) with a B2 glenoid should undergo a reverse TSA due to concerns of poor outcomes and instability with TSA [22]. Within a cohort of 128 osteoarthritic shoulder treated with TSA, the humeral head was preoperatively found to be posteriorly subluxated in 22 shoulders (18 %) [19]. These shoulders with preoperative posterior subluxation of the humeral head had a lower ASES score, more pain, and decreased active external rotation than patients with a preoperatively centered humeral head following either TSA or hemiarthroplasty. These findings are contrary to ours as the outcomes after TSA for patients with preoperatively centered humeral head and patients with preoperative posterior subluxation of the humeral were found to be pretty similar in this study. Similar to the results of Walch et al. [20], however, the rate of glenoid component loosening was 7 %. Despite good functional outcomes and high satisfaction, implantation of TSA for primary idiopathic GHOA had the highest risk of glenoid loosening in patients with type B2 glenoid.
Ten-year revision-free survival rates of primary TSA are reported between 89 and 95 % [15, 27, 34]. Twenty-year revision-free survival rates range between 70 and 83.2 % [16, 27, 34]. However, Raiss et al. [27] also reported that all clinical parameters first substantially improved postoperatively compared to the preoperative baseline, reaching a plateau at 1 year of follow-up. Their results then remained stable for 8 years and then started to slightly deteriorate until 15 years postoperatively. After an average of 15 years, the clinical results were inferior to those at the 6-month follow-up [27]. This is somewhat consistent with the findings of our study, demonstrating that longer follow-up was significantly correlated with lower both pre- and postoperative SF-12 PCS scores and patient satisfaction. This may indicate that the baseline health of the cohort increased during the study period, or it may also represent a potential reporting bias of patients.
Although TSA has been shown by the current study and others to be a reliable treatment modality for patients with GHOA, several studies have shown unacceptable outcomes in younger patients due to increased rates of component loosening [35], decreased component survival [18], and significantly higher risk of revision [36]. Therefore, alternative treatments including arthroscopic management of GHOA have been investigated [37–40]. A recent Markov decision analysis demonstrated that the arthroscopic management was preferable in patients less than 47 years of age, while TSA was the better option in patients over 66 years. For patients between 47 and 66 years of age, the decision mainly depends on individual factors and both options may be reasonable [41].
Several limitations apply to this study. First, our sample size may have been too small to detect other potential predictors for satisfaction. Second, our patient population was medically healthier in terms of ASA score (93.5 % of patients having an ASA score of 1 or 2 than previously published ones, which may be due to the selection of our referral practice and may have influenced outcomes. Lastly, the ceiling effects observed in the patient satisfaction and ASES scales may inhibit the ability of statistical analysis to identify relevant predictors for these outcome scores.
Conclusion
Anatomic TSA provided excellent results for patients with idiopathic GHOA with low complication and failure rates. Outcomes after TSA for posteriorly subluxed type B glenoid morphology were similar to outcomes after TSA for centered type A morphology. Overall, patient satisfaction was high and was not influenced by the demographic, anatomic, and surgical variables investigated.
References
Kim SH, Wise BL, Zhang Y, Szabo RM (2011) Increasing incidence of shoulder arthroplasty in the United States. J Bone Jt Surg Am 93:2249–2254. doi:10.2106/JBJS.J.01994
Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A (2005) A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Jt Surg Am 87:1947–1956. doi:10.2106/JBJS.D.02854
Edwards TB, Kadakia NR, Boulahia A, Kempf JF, Boileau P, Nemoz C, Walch G (2003) A comparison of hemiarthroplasty and total shoulder arthroplasty in the treatment of primary glenohumeral osteoarthritis: results of a multicenter study. J Shoulder Elb Surg 12:207–213. doi:10.1016/S1058-2746(02)86804-5
Orfaly RM, Rockwood CA Jr, Esenyel CZ, Wirth MA (2003) A prospective functional outcome study of shoulder arthroplasty for osteoarthritis with an intact rotator cuff. J Shoulder Elb Surg 12:214–221
Bohsali KI, Wirth MA, Rockwood CA Jr (2006) Complications of total shoulder arthroplasty. J Bone Jt Surg Am 88:2279–2292
Wirth MA, Rockwood CA Jr (1994) Complications of shoulder arthroplasty. Clin Orthop Relat Res 307:47–69
Chalmers PN, Gupta AK, Rahman Z, Bruce B, Romeo AA, Nicholson GP (2014) Predictors of early complications of total shoulder arthroplasty. J Arthroplast 29:856–860. doi:10.1016/j.arth.2013.07.002
Farng E, Zingmond D, Krenek L, SooHoo NF (2011) Factors predicting complication rates after primary shoulder arthroplasty. J Shoulder Elb Surg 20:557–563. doi:10.1016/j.jse.2010.11.005
Gonzalez JF, Alami GB, Baque F, Walch G, Boileau P (2011) Complications of unconstrained shoulder prostheses. J Shoulder Elbow Surg 20:666–682. doi:10.1016/j.jse.2010.11.017
Kiet TK, Feeley BT, Naimark M, Gajiu T, Hall SL, Chung TT, Ma CB (2015) Outcomes after shoulder replacement: comparison between reverse and anatomic total shoulder arthroplasty. J Shoulder Elb Surg 24:179–185. doi:10.1016/j.jse.2014.06.039
Norris TR, Iannotti JP (2002) Functional outcome after shoulder arthroplasty for primary osteoarthritis: a multicenter study. J Shoulder Elb Surg 11:130–135
Puskas B, Harreld K, Clark R, Downes K, Virani NA, Frankle M (2013) Isometric strength, range of motion, and impairment before and after total and reverse shoulder arthroplasty. J Shoulder Elb Surg 22:869–876. doi:10.1016/j.jse.2012.09.004
Harreld K, Clark R, Downes K, Virani N, Frankle M (2013) Correlation of subjective and objective measures before and after shoulder arthroplasty. Orthopedics 36:808–814. doi:10.3928/01477447-20130523-29
van de Sande MA, Brand R, Rozing PM (2006) Indications, complications, and results of shoulder arthroplasty. Scand J Rheumatol 35:426–434
Young A, Walch G, Boileau P, Favard L, Gohlke F, Loew M, Molé D (2011) A multicentre study of the long-term results of using a flat-back polyethylene glenoid component in shoulder replacement for primary osteoarthritis. J Bone Jt Surg Br 93:210–216. doi:10.1302/0301-620X.93B2.25086
Schoch B, Schleck C, Cofield RH, Sperling JW (2015) Shoulder arthroplasty in patients younger than 50 years: minimum 20-year follow-up. J Shoulder Elb Surg 24:705–710. doi:10.1016/j.jse.2014.07.016
Chen AL, Bain EB, Horan MP, Hawkins RJ (2007) Determinants of patient satisfaction with outcome after shoulder arthroplasty. J Shoulder Elb Surg 16:25–30
Denard PJ, Raiss P, Sowa B, Walch G (2013) Mid- to long-term follow-up of total shoulder arthroplasty using a keeled glenoid in young adults with primary glenohumeral arthritis. J Shoulder Elb Surg 22:894–900. doi:10.1016/j.jse.2012.09.016
Iannotti JP, Norris TR (2003) Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Jt Surg Am 85-A:251–258 PMID: 12571302
Walch G, Moraga C, Young A, Castellanos-Rosas J (2012) Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elb Surg 21:1526–1533. doi:10.1016/j.jse.2011.11.030
Johnson CC, Sodha S, Garzon-Muvdi J, Petersen SA, McFarland EG (2014) Does preoperative American Society of Anesthesiologists score relate to complications after total shoulder arthroplasty? Clin Orthop Relat Res 472:1589–1596. doi:10.1007/s11999-013-3400-1
Mizuno N, Denard PJ, Raiss P, Walch G (2013) Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Jt Surg Am 95:1297–1304. doi:10.2106/JBJS.L.00820
Papadonikolakis A, Neradilek MB, Matsen FA 3rd (2013) Failure of the glenoid component in anatomic total shoulder arthroplasty. J Bone Jt Surg Am 95:2205–2212. doi:10.2106/JBJS.L.00552
Ahearn N, McCAnn PA, Tasker A, Sarangi PP (2013) The influence of rotator cuff pathology on functional outcome in total shoulder replacement. Int J Shoulder Surg 7:127–131. doi:10.4103/0973-6042.123509
Walch G, Badet R, Boulahia A, Khoury A (1999) Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty 14:756–760
Clavien PA, Sanabria JR, Strasberg SM (1992) Proposed classification of complications of surgery with examples of utility in cholecystectomy. Surgery 111:518–526
Raiss P, Bruckner T, Rickert M, Walch G (2014) Longitudinal observational study of total shoulder replacements with cement: fifteen to 20-year follow-up. J Bone Jt Surg Am 96:198–205. doi:10.2106/JBJS.M.00079
Singh A, Yian EH, Dillon MT, Takayanagi M, Burke MF, Navarro RA (2014) The effect of surgeon and hospital volume on shoulder arthroplasty perioperative quality metrics. J Shoulder Elb Surg 23:1187–1194. doi:10.1016/j.jse.2013.11.017
Ponce BA, Ahluwalia RS, Mazzocca AD, Gobezie RG, Warner JJ, Millett PJ (2005) Biomechanical and clinical evaluation of a novel lesser tuberosity repair technique in total shoulder arthroplasty. J Bone Jt Surg Am 87(Suppl 2):1–8
Jeong J, Bryan J, Iannotti JP (2009) Effect of a variable prosthetic neck-shaft angle and the surgical technique on replication of normal humeral anatomy. J Bone Jt Surg Am 91:1932–1941. doi:10.2106/JBJS.H.00729
Sassoon A, Schoch B, Rhee P, Schleck CD, Harmsen WS, Sperling JW, Cofield RH (2013) The role of eccentric and offset humeral head variations in total shoulder arthroplasty. J Shoulder Elb Surg 22:886–893. doi:10.1016/j.jse.2012.09.008
Singh JA, Ramachandran R (2015) Does hospital volume predicts outcomes and complications after total shoulder arthroplasty in the United States? Arthritis Care Res (Hoboken) 67(6):885–890. doi:10.1002/acr.22507
Matsen FA 3rd, Clinton J, Lynch J, Bertelsen A, Richardson ML (2008) Glenoid component failure in total shoulder arthroplasty. J Bone Jt Surg Am 90:885–896. doi:10.2106/JBJS.G.01263
Singh JA, Sperling JW, Cofield RH (2011) Revision surgery following total shoulder arthroplasty: analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Jt Surg Br 93:1513–1517. doi:10.1302/0301-620X.93B11.26938
Sperling JW, Cofield RH, Rowland CM (2004) Minimum 15-year follow-up of Neer hemiarthroplasty and total shoulder arthroplasty in patients aged 50 years or younger. J Shoulder Elb Surg 13:604–613
Dillon MT, Inacio MC, Burke MF, Navarro RA, Yian EH (2013) Shoulder arthroplasty in patients 59 years of age and younger. J Shoulder Elb Surg 22:1338–1344. doi:10.1016/j.jse.2013.01.029
Millett PJ, Horan MP, Pennock AT, Rios D (2013) Comprehensive Arthroscopic Management (CAM) procedure: clinical results of a joint-preserving arthroscopic treatment for young, active patients with advanced shoulder osteoarthritis. Arthroscopy 29:440–448. doi:10.1016/j.arthro.2012.10.028
Millett PJ, Schoenahl JY, Allen MJ, Motta T, Gaskill TR (2013) An association between the inferior humeral head osteophyte and teres minor fatty infiltration: evidence for axillary nerve entrapment in glenohumeral osteoarthritis. J Shoulder Elb Surg 22:215–221. doi:10.1016/j.jse.2012.05.030
Van Thiel GS, Sheehan S, Frank RM, Slabaugh M, Cole BJ, Nicholson GP, Romeo AA, Verma NN (2010) Retrospective analysis of arthroscopic management of glenohumeral degenerative disease. Arthroscopy 26:1451–1455. doi:10.1016/j.arthro.2010.02.026
Weinstein DM, Bucchieri JS, Pollock RG, Flatow EL, Bigliani LU (2000) Arthroscopic debridement of the shoulder for osteoarthritis. Arthroscopy 16:471–476
Spiegl UJ, Faucett SC, Horan MP, Warth RJ, Millett PJ (2014) The role of arthroscopy in the management of glenohumeral osteoarthritis: a markov decision model. Arthroscopy 30:1392–1399. doi:10.1016/j.arthro.2014.06.011
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This research was supported by the Steadman Philippon Research Institute. The Institute receives research support from the following entities: Smith & Nephew Endoscopy, Inc.; Arthrex, Inc.; Siemens Medical Solutions USA, Inc.; Ossur Americas, Inc.; Opedix, Inc. This work was not supported directly by outside funding or grants. Dr. Peter J. Millett has received from Arthrex something of value (exceeding the equivalent of US$500) not related to this manuscript or research. He is a consultant and receives payments from Arthrex and has stock options in GameReady and Vumedi. Dr. Maximilian Petri, Dr. Simon Euler and Dr. J. Christoph Katthagen have had their research position at the Steadman Philippon Research institute funded by Arthrex. The other authors receive support from the Steadman Philippon Research Institute.
Additional information
M. Petri and S. A. Euler contributed equally to this study.
Institutional Review Board Approval: Vail Valley Medical Center.
Rights and permissions
About this article
Cite this article
Petri, M., Euler, S.A., Dornan, G.J. et al. Predictors for satisfaction after anatomic total shoulder arthroplasty for idiopathic glenohumeral osteoarthritis. Arch Orthop Trauma Surg 136, 755–762 (2016). https://doi.org/10.1007/s00402-016-2452-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-016-2452-6