Abstract
The objective of this study was to investigate in more detail the structure of multilamellar beta-carotene-loaded liposome dispersions produced by proliposome hydration. Such dispersions were stabilized using xanthan gum as a thickener in different concentrations, and their stabilities were monitored for 90 days. The vesicles exhibited an average diameter in the range of 700 to 3000 nm, and the liposomes were capable of protecting β-carotene from degradation for a period of 90 days. The dispersions were also characterized by transmission electron microscopy, differential scanning calorimetry, rheology, and small-angle X-ray scattering (SAXS). The thermal analyses showed that neither the β-carotene nor the xanthan gum affected the liposome bilayer structure. The presence of the xanthan gum, which affects the scattering intensity, was not an obstacle to obtain the structural parameters by SAXS modeling, as a modified modeling strategy (Gaussian deconvolution) was applied. This modeling resulted in 40 symmetric layers, and the results obtained with the variation of temperature were in agreement with the gel-liquid crystalline transition temperature obtained by calorimetric measurements. Additionally, the rheological data showed that xanthan gum was not as effective as a mixture of xanthan gum and guar gum at stabilizing the liposomes, most likely due to the higher stiffness of the gum alone compared with that of its combination with guar gum.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The potential use of liposomes in food products has been more extensively explored in recent years, and several studies of the production of liposomes for encapsulating food bioactives can be found in the literature [25–37].
Because food formulations are mostly water based, liposomes are of great interest for their potential to overcome the technological drawbacks of incorporating hydrophobic bioactives, e.g., vitamin E [21–43], essential oils [11, 12, 39, 42], ferrous sulfate [5, 6], polyphenols [10, 14, 18, 30, 44], l-carnosine [19], and carotenoids [3, 24, 35, 36, 38, 41].
However, the effective application of liposomes in food systems is quite limited because of the lack of studies focusing on processes that are suitable for scaling up and increasing capacity to produce the phospholipid vesicles in large quantities, which are typical of the food industry. Furthermore, many processes that have been implemented in the pharmaceutical industry use organic solvents that are completely forbidden for food. To establish parameters for an effective implementation of liposome production for food applications, it is necessary to investigate organic solvent-free methods that are capable of producing massive amounts of vesicles. Another important consideration is the cost of the microencapsulation system; multilamellar liposomes (MLVs) would be cheaper to produce than small unilamellar vesicles (SUVs) because they would not require the use of high-pressure homogenizers or microfluidization systems.
The type of liposome employed determines its stability in a complex medium such as a food matrix. The formulation of the phospholipid vesicle must be engineered with consideration for the conditions to which it would be subjected in a food matrix, such as the presence of salt, sugars, proteins, and other colloidal structures, as well as the possibility of thermal treatments during food processing, which could also destabilize the liposomal system. Under such conditions, SUV could be more stable than MLV because of their reduced diameter. Therefore, it is important to design a liposomal system that can be scaled up at a cost suitable for food applications, which is certainly much lower than that for the pharmaceutical industry.
Some efforts have been made to develop innovative scale-up solutions. Laouini et al. [17] refer to liposome production techniques, such as the heating method, freeze-drying, supercritical reverse phase evaporation (SCRPE), modified ethanol injection, and hydration of proliposomes. Patil and Jadhav [28], in a recent review, emphasize the need for the development of novel processes for liposome production, particularly techniques that do not require detergents or organic solvents. In this context, a notable method for producing liposomes in massive amounts is the hydration of proliposomes. Proliposomes are dry phospholipid particles that, when hydrated at controlled temperatures and stirring conditions, can result in the formation of multilamellar liposomes [1, 15, 40]. The use of proliposomes has the advantage of inserting the bioactive in a matrix that provides high structural integrity, stability, and functionality [1]. There are various known methods to produce proliposomes, and one of the well-known processes in the literature is the spray-drying method [15, 24, 41]. This method of liposome production could be easily scaled up because spray drying is a unit operation that is extensively used in the food industry.
Beta-carotene is the carotenoid with the highest pro-vitamin A activity [8]. Furthermore, beta-carotene is an important antioxidant that may have a protective function against degenerative conditions such as cancer and cardiovascular diseases [8–31]. However, the use of beta-carotene in food can be limited for two reasons: (1) high hydrophobicity (log P = 14.7), which limits its dispersion in water-based media, and (2) sensitivity to chemical degradation, which could decrease its effectiveness as a pigment as well as its physiological benefits [27, 36]. Therefore, the use of colloidal dispersions, such as emulsions and liposomes, to microencapsulate beta-carotene could provide a physico-chemical barrier against oxidants and highly increase its dispersibility in aqueous environments. The multilamellar liposomes studied in the present research work have already been incorporated in yogurt in a previous study published by Toniazzo et al. [38], with promising technological and sensorial results. In such a study, liposomes were used to incorporate beta-carotene as a natural dye in the dairy product and the results showed that the phospholipid vesicles can be considered as a possible alternative to be used as vehicles in food matrices. The present study is the characterization of these beta-carotene-loaded multilamellar liposomes produced by spray-dried proliposome hydration, a scalable production method applicable to the food industry. The liposomes were produced by hydrating the proliposomes obtained by spray drying, and to stabilize the liposome dispersions, it was necessary to use xanthan gum as a thickening agent. The data obtained were in the form of measurements of vesicle size distribution, zeta potential, protection capacity of the encapsulated carotenoid, rheological characterization, thermal behavior by differential scanning calorimetry (DSC), transmission electron microscopy (TEM), and small-angle X-ray scattering (SAXS).
Material and methods
Materials
The phospholipid used was a purified hydrogenated soy phosphatidylcholine (Phospholipon 90H, average molecular weight of 800 g) obtained from Lipoid GmbH (Ludwigshafen, Germany) (including at a minimum of 90 % phosphatidylcholine and a maximum of 4 % of lysophosphatidylcholine, with a minimum of 98 % palmitic acid plus stearic acid and a maximum of 2 % C18 unsaturated aliphatic chains).
Beta-carotene was obtained from Sigma (St. Louis, MO, USA) in its crystalline powder form. Xanthan gum (Grindsted® Xanthan 80) was donated by Danisco (Cotia, Brazil). All the reagents used in the study were reagent grade. Deionized water (from a Millipore system) was used throughout the experiments.
Production of liposomes
Liposome dispersions were produced via hydration of proliposomes, which were produced using a spray-drying method described by Moraes et al. [24] and Toniazzo et al. [38]. The proliposomes were prepared with 3.2 g of phospholipid, 0.270 g of sucrose (Synth, Diadema, Brazil), and 0.120 g of beta-carotene and were solubilized in anhydrous ethanol (Synth, Diadema, Brazil). The proliposomes (7.5 g proliposome L−1) were hydrated with deionized water by ultra-agitation (IKA T25, IKA, Germany) at 12,000 rpm for 15 min at 60 °C. The thickening agent (xanthan gum (XG), 0.20, 0.25, or 0.30 % in mass) was slowly added to the liposome dispersion under magnetic stirring (3600 rpm) at room temperature. The samples were prepared in triplicate and stored under refrigeration. Sodium benzoate (Synth, Brazil; 0.02 % in mass) was added to avoid microbiological contamination.
Determination of the particle size distribution and zeta potential of liposomes
The size distribution of the liposomes was measured by photon correlation spectroscopy using a ZetaPlus analyzer (Brookhaven Instruments Company, USA) at 25 °C and a He-Ne laser at 627 nm with an incidence angle of 90°. The samples were diluted with ultra-purified water to weaken the opalescence before measuring the particle mean diameter. The zeta potential was obtained using the same equipment used for electrophoretic mobility measurements, in water, adjusting the conductivity to 50 μS/cm with 1 M KCl. Data analyses were performed using the software included with the system.
Quantification of beta-carotene encapsulated in the liposomes
For these determinations, a liquid-liquid extraction with 6 mL of hexane and 9 mL of anhydrous ethanol added to 1 mL of liposome dispersion was carried out. The mixture was then vigorously shaken. Ethanol was used to rupture the liposomes before beta-carotene extraction into the hexane phase. After 2 h at a controlled temperature of 25 °C, the hexane phase was removed and the absorbance was read at 450 nm (Libra S22; Biochrom, UK). The beta-carotene levels were determined with respect to a calibration curve of pure beta-carotene in hexane, with an R 2 value of 0.996 [24–38].
Differential scanning calorimetry
The thermal behavior of the liposomes was assessed by DSC, using a TA2010 differential scanning calorimeter controlled by a TA5000 system (TA Instruments, New Castle, USA). The samples (approximately 10 mg) were placed in hermetically sealed aluminum TA pans and heated from 10 to 100 °C using a heating ramp of 10 °C/min. An empty pan was used as a reference. The heating was carried out under an inert atmosphere (45 mL/min N2). The results were analyzed using the Universal Analysis V1.7F software (TA Instruments, New Castle, USA).
Transmission electron microscopy
The morphology of the liposomes was observed by TEM using a CEM-902 (Carl Zeiss, Oberkochen, Germany) microscope coupled with a Castaing-Henry-Ottensmeyer filter spectrometer. The samples were diluted to 0.25 mM (total phospholipid concentration). The colorant used was uranyl acetate (1 % w/v) (Electron Microscopy Sciences, Hatfield, USA). The reagent was filtered through a plastic syringe coupled with a 0.22-μm pore size filter. On the surface of a grid covered with a parlodium plastic film and evaporated carbon, a drop of the liposome dispersion was added, and after 5 min, the excess sample was removed with a filter paper. A drop of uranyl acetate was added to stain the liposomes, and the excess colorant was removed with a filter paper. The images were recorded using a Proscan high-speed slow-scan CCD camera and processed in the iTEM universal TEM imaging platform.
Rheological measurements
A rheometer (AR2000 advanced rheometer; TA Instruments, New Castle, USA) fitted with concentric cylinders (internal radius = 14 mm, external radius = 15 mm, height = 42 mm, and gap = 5920 μm) was used to carry out the rheological tests with the liposome dispersions. All the samples were analyzed at a controlled temperature of 10 ± 1 °C. The relaxation period of the samples before the beginning of the process was 2 min. The flow curves were obtained by measuring shear stress, while the shear rate was increased from 0.01 to 100 s−1 and decreased from 100 to 0.01 s−1. The experimental data were fitted to the power law model according to Eq. (1)
where σ is the shear stress (Pa), k is the consistency index (Pa sn), n is the flow index, and ẏ is the shear rate (s−1). The results were analyzed using the Rheology Advantage Data Analysis V.5.3.1 software (TA Instruments, New Castle, USA).
Small-angle X-ray scattering
SAXS measurements were performed at the Brazilian Synchrotron Light Laboratory (LNLS) at the SAXS2 beamline. The sample holder [2], a cell measuring 1 × 4 × 8 mm3, was placed in a device coupled to a water-circulating thermal bath, thus enabling measurements at controlled temperature. The wavelength of the incoming monochromatic X-ray beam was 1.48 Å, and the distance between the sample and the detector was set to 700 mm, providing a q (scattering vector modulus) interval from 0.013 to 0.33 Å−1, where q = 4π(sin θ)/λ and 2θ is the scattering angle. The 2D scattering data were collected on a MAR detector, and the integration of the SAXS patterns and data treatment was performed using the FIT2D software [13]. Error estimation and normalization to absolute scale were performed using the SuperSAXS package. A rectangular beam with 300 × 100 μm dimensions was used. This beam size was further convoluted with a detector resolution of 180 μm. The experimental setup provided a resolution in q values of Δq = 0.00129 Å−1.
Results and discussion
Stability of the liposome dispersions: size distribution, zeta potential, and encapsulated beta-carotene
The liposomes produced with different concentrations of stabilizing xanthan gum presented an average hydrodynamic diameter in the range of 700 to 3000 nm, which is a characteristic of multilamellar dispersions. The particle size distribution was monitored over a storage period of 90 days, and the curves are shown in Fig. 1.
In the absence of xanthan gum, the liposome dispersions destabilized completely in less than 24 h of storage under refrigeration (7–10 °C), resulting in visible phase separation. This was the reason it was necessary to add thickeners (gums) to the system. The minimum gum concentration needed to avoid phase separation was 0.20 %; with lower concentrations of thickener, the system destabilized in less than 48 h under refrigeration. However, concentrations greater than 0.30 % XG made the system highly viscous, which was not a desirable characteristic for incorporation in food systems. Therefore, the range tested was from 0.20 to 0.30 % XG, and Fig. 1 shows that the liposome dispersions stabilized with 0.30 % XG were more capable of maintaining the size distribution over the storage period, as practically no changes were observed in the corresponding size distribution curve. Therefore, the liposome dispersions stabilized with 0.30 % XG were more stable in relation to the size distributions during 90 days of storage.
Table 1 shows that the values of the zeta potential of the samples, which is an indirect measurement of the stability of colloidal dispersions, significantly changed after 90 days but remained below −30 mV in all cases. Zeta potential values below −30 mV are typical of stable dispersions, and the zeta potential never rose above this number for the liposomes encapsulating the beta-carotene. These results are similar to the data obtained by Toniazzo et al. [38], who produced MLV by proliposome hydration but used a mixture of xanthan gum and guar gum as a thickening agent used.
The stabilization of the liposome dispersions using the polysaccharides was certainly due to the polysaccharides’ capacity to increase the viscosity of the continuous phase via the formation of a polymeric network, a mechanism that has been widely explored in the stabilization of food emulsions [4]. This rheological control mechanism is most effective at relatively low-particle volume fractions, where individual droplets are separately immobilized in a polysaccharide network [4].
The network formed by xanthan gum is shown in the micrograph obtained by transmission electron microscopy of the liposome dispersions stabilized with 0.20 % xanthan gum (Fig. 2). The biopolymeric network can be clearly visualized in the background of the micrograph, and it was responsible for delaying aggregation of the liposomes during storage. This micrograph also shows the spherical shape of the liposomes, and it substantiates the average hydrodynamic sizes obtained by quasi-elastic light scattering.
The encapsulation of beta-carotene was complete, as it was 100 % incorporated in the liposomes, as previously described in studies by Moraes et al. [24] and Toniazzo et al. [38], in which the proliposomes were also hydrated at controlled conditions. The fresh samples had, on average, a molar percentage of approximately 3.30 % of beta-carotene in the phospholipid vesicles. According to the literature, such a value is slightly above that obtained in studies of the incorporation of beta-carotene in multilamellar liposomes [29, 33, 34], but in those cases, the vesicles were produced using the film hydration method. The production of liposomes via the hydration of proliposomes has the advantage of incorporating the bioactive in the phospholipid matrix prior to the procedure, compared with the majority of the methods of liposome production, which incorporate the bioactive only in the bilayer formation step; this advantage allows a higher quantity of beta-carotene to be incorporated into the liposomes [24].
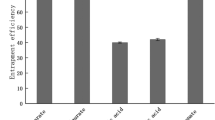
The temporal profile of beta-carotene concentration (Fig. 3), however, indicates that the carotenoid suffered extensive decomposition in the first 10 days of storage. At the end of this initial period, the samples contained, on average, 2.10 % molar beta-carotene, a value that remained stable up to the 90th day of storage. The similar amounts of remaining beta-carotene in the liposomes, despite the differences in the initial quantity, indicates that most of the molecules of the carotenoid were deeply inserted in the more internal phospholipid bilayers of the multilamellar liposomes, which is more difficult for the oxidant to reach. As noted below, the SAXS analyses indicated that the multilamellar liposomes presented more than 40 phospholipid bilayers. Such a value of approximately 2.5 % molar agrees with the results obtained by Moraes et al. [24] and Toniazzo et al. [38], who studied similar liposome systems, and with those of Pintea et al. [29] and Socaciu et al. [33], who also studied multilamellar liposomes incorporating beta-carotene. It seems to be the superior limit of the beta-carotene/phospholipid molar ratio to keep the carotenoid deep inside enough the various bilayers of the liposomes to be protected from oxidative damage.
The data presented in Table 1 also indicate that the quantity of xanthan gum added to the dispersions to stabilize them did not affect the decomposition rate of the encapsulated beta-carotene.
Rheological characterization of liposome dispersions
As for the rheological behavior of liposomes stabilized with different xanthan gum concentrations, the results were fit to the power law model. The determination coefficient for this model was 0.99 for all the samples. It is the model that fits the rheological data obtained in flow assays of colloidal diluted dispersions (e.g., emulsions) and slightly flocculated dispersions [22]. Therefore, the liposomes stabilized with a xanthan gum showed a non-Newtonian behavior of the pseudoplastic type, as expected, due to the presence of xanthan gum as a thickener. The flow curves showed that the rheological behavior of liposomes was not changed over the storage period, as observed in Fig. 4, where the two curves practically overlapped.
Table 2 shows the mean values of apparent viscosity of the liposome dispersions stabilized with xanthan gum in different concentrations. As expected, increasing the concentration of xanthan gum led to an increase in the viscosity of the system; in this case, the increase in viscosity of the aqueous phase was able to prevent decomposition after 90 days only in the system containing 0.30 % of xanthan gum. Some considerations can be made when comparing the data obtained here with the data obtained by Toniazzo et al. [38]. In the latter study, the liposomes were in a concentration two times higher than that in the present study, and a concentration of 0.10 % total gum (being 0.09 % guar gum and 0.01 % xanthan gum) was sufficient to prevent decomposition for 90 days. In the present study, compared with the study of Toniazzo et al. [38], three times more thickening agent was required to stabilize half the liposome amount. The viscosity of the most stable system in Toniazzo et al. [38] was, on average, 0.069 Pa s−1, whereas the system in this study had a value of 0.235 Pa s−1. Therefore, the explanation for such a result is related not only to an increase in the continuous phase viscosity but also to the fact that only xanthan gum was used in the present study. Xanthan gum is a polysaccharide of high stiffness, much higher than that of galactomannan (guar gum), and this rigidity imparts a much more limited degree of mobility to the chains and, hence, much longer relaxation times [7]. In a liposomal system, destabilization in the presence of a thickener is likely to be limited by the lowered diffusion capacity of liposomes. However, the polysaccharide molecules can work as obstacles for the diffusion of the phospholipid vesicles, and more flexible chains, such as the guar gum molecules, would present a higher number of configurations that could more efficiently prevent contact among the phospholipid vesicles. Therefore, a smaller amount of thickener could be used to stabilize a higher concentration of liposomes.
Thermal behavior and small-angle X-ray scattering
The thermogram of the liposomes is shown in Fig. 5, and it exhibits one endothermic transition represented by a single narrow peak that is attributed to the gel-liquid crystalline phase transition of the phospholipid vesicles. Calorimetric analyses were carried out only with the most stable liposome dispersion (0.30 % XG). The transition temperature (T m) was at approximately 54 °C, identical to that reported by Toniazzo et al. [38], who studied the same type of liposome dispersion, but thickened with a mixture of guar gum and xanthan gum. The type of thickening agent, therefore, did not seem to influence the gel-liquid crystalline transition. This is another indication that there are no any modifications in the bilayer structure due to possible interactions among the phospholipids and the polysaccharide chains. In fact, the polysaccharides only built a polymer network in the continuous phase, as shown in Fig. 2, and they were not adsorbed or anchored on the surfaces of the liposomes. The structure of the liposome bilayer was not modified by the presence of beta-carotene because the T m of empty multilamellar liposomes produced with the same phospholipid was 51.7 °C [42].
The SAXS data were also collected only for the most stable formulation of liposome dispersion over the storage period (0.30 % XG). The sample was subjected to temperatures ranging from 30 to 57 °C. As observed, a characteristic profile of multilamellar systems was obtained (Fig. 6a). The data were modeled using the Gaussian deconvolution method [26], which permits modeling the form factor and structure factor simultaneously; as a result, it is possible to obtain information regarding the electron density profile of the lipid bilayer (Fig. 6b) and several other parameters, which are summarized in Table 3. For this modeling method, it was necessary to introduce the contribution from the polymer network (xanthan gum). This contribution was obtained in two steps. First, the scattering intensity of the xanthan gum in solution was measured alone at three different temperatures. From this result (data not shown), one can demonstrate that the scattering contribution from the xanthan gum can be described using a Gaussian chain
where u = 〈R 2 G 〉q 2 and 〈R 2 G 〉 are the average radius of gyration of the polymer chain. This expression was incorporated into the Gaussian deconvolution procedure to obtain the correct description of the polymer contribution and, consequently, the correct form factor and structure factor from the bilayer.
Scattering results for the multilamellar liposome dispersions with 0.30 % XG as a thickener. a SAXS data (open circles) for the various temperatures. The theoretical model (solid lines) is also shown in the plot. b Obtained electron density profile for each temperature. As expected, the structure becomes less ordered with an increase in temperature
With the increase of temperature, several interesting features are observed both on the bilayer shape and on the structure factor parameters. As shown in Fig. 6(b), the electron density profile indicates a symmetric bilayer, which is typical of zwitterionic phospholipids [16], and that the bilayer loses ordering with an increase in temperature, which is most likely due to the increase in thermal energy in the system and the subsequent approximation of the gel-liquid crystalline transition point at 54 °C. In addition, the vesicles decrease the number of layers (N), starting at approximately 40 for 30 °C and decreasing to approximately 20 for 50 °C.
Notably, the membrane shows an increase in flexibility only after 54 °C, where one can observe a substantial increase in the Caillè parameter (η), which is associated with the bilayer bending rigidity [16]. A higher Caillè parameter indicates that the membrane is more flexible, which is consistent with the differential scanning calorimetry data showing that the gel-liquid crystalline transition of the liposomes is also approximately 54 °C. The interplanar spacing also varied, with a maximum value of approximately 50 °C (74.86 Å). In all the cases, it was necessary to include the contribution from a Gaussian chain, which indicates that the xanthan gum made an important contribution to the scattering intensity. The abrupt variation on the radius of gyration between 40 °C (∼16 Å) and 45 °C (∼11 Å) and above can indicate a change in the polysaccharide conformation. Such a change is related to the shifts from an ordered state to a disordered state of the xanthan gum chain [9], corresponding to a helix-coil transition of the polysaccharide backbone and simultaneous release of the lateral chains, followed by a progressive decrease in the rigidity of the (1–4)-β-d-glucan chain as the temperature rises from 40 to 60 °C [23].
Conclusions
The experimental data obtained in this investigation demonstrated the ability to produce beta-carotene-loaded multilamellar liposomes via proliposome hydration and to stabilize the dispersions with xanthan gum. Additionally, it was possible to clearly visualize the biopolymeric network stabilizing the dispersions via transmission electron microscopy. The multilamellar character of the liposomes was shown by the SAXS data, which, despite the presence of xanthan gum, could be modeled to obtain important parameters such as the bilayer electron density, the Caillè parameter (and, indirectly, the rigidity of the membrane), and the number of layers. The Gaussian deconvolution method was capable of being used to decouple the effect of the xanthan gum in the scattering intensity to obtain the cited parameters, with good correlation. In addition, the data obtained by SAXS agreed with the calorimetric behavior of the system.
In addition, the rheological measurements showed that xanthan gum alone was less effective as a thickening agent in the liposome dispersions than a mixture of guar gum and xanthan gum. Such a characteristic was most likely due to the stiffness of the xanthan gum, which limits the number of configurations it can assume and, consequently, allows a higher degree of movement of the liposomes in the aqueous medium, which could lead to faster destabilization.
References
Alves GP, Santana MHA (2004) Phospholipid dry powder produced by spray drying processing: structural, thermodynamic and physical properties. Powder Technol 145:139–148
Cavalcanti LP, Torriani IL, Plivelic TS, Oliveira CLP, Kellermann G, Neuenschwander R (2004) Two new sealed sample cells for small angle x-ray scattering from macromolecules in solution and complex fluids using synchrotron radiation. Rev Sci Instrum 75:4541–4546
De Paz E, Martín A, Cocero MJ (2012) Formulation of β-carotene with soybean lecithin by PGSS (particles from gas saturated solutions)-drying. J Supercrit Fluids 72:125–133
Dickinson E (2009) Hydrocolloids as emulsifiers and emulsion stabilizers. Food Hydrocoll 23:1473–1482
Ding B, Xia S, Hayat K, Zhang X (2009) Preparation and stability of ferrous glycinate liposomes. J Agric Food Chem 57:2938–2944
Ding B, Zhang X, Hayat K, Xia S, Jia C, Xie M, Liu C (2011) Preparation, characterization and the stability of ferrous glycinate liposomes. J Food Eng 102:202–208
Doublier JL, Cuvelier G (2006) Gums and hydrocolloids: functional aspects. In: Eliasson AC (ed) Carbohydrates in foods, 2nd edn. Taylor and Francis, Boca Raton, pp 233–271
Fraser PD, Bramley PM (2004) The biosynthesis and nutritional uses of carotenoids. Prog Lipid Res 43:228–265
Garcia-Ochoa F, Santos VE, Casas JA, Gómez E (2000) Xanthan gum: production, recovery and properties. Biotechnol Adv 18:549–579
Gibis M, Zeeb B, Weiss J (2014) Formation, characterization, and stability of encapsulated hibiscus extract in multilayered liposomes. Food Hydrocoll 38:28–39
Gortzi O, Lalas S, Chinou I, Tsaknis J (2007) Evaluation of the antimicrobial and antioxidant activities of Origanum dictamnus extracts before and after encapsulation in liposomes. Molecules 12:932–945
Gortzi O, Lalas S, Chinou I, Tsaknis J (2008) Reevaluation of bioactivity and antioxidant activity of Myrtus communis extract before and after encapsulation in liposomes. Eur Food Res Technol 226:583–590
Hammersley AP, Svensson SO, Hanfland M, Fitch AN, Hausermann D (1996) Two-dimensional detector software: from real detector to idealised image or two-theta scan. High Pressure Res 14:235–248
Hasan M, Belhaj N, Benachour H, Barberi-heyob M, Kahn CJF, Jabbari E, Linder M, Arab-Tehrany E (2014) Liposome encapsulation of curcumin: physico-chemical characterizations and effects on MCF7 cancer cell proliferation. Int J Pharm 461:519–528
Kikuchi H, Yamauchi H, Hirota S (1991) A spray drying method for the mass production of liposomes. Chem Pharm Bull 39:1522–1527
Kucerka N, Nieh MP, Katsaras J (2010) Small-angle scattering from homogeneous and heterogeneous lipid bilayers. In: Iglic A, Tien HT (eds) Advances in planar lipid bilayers and liposomes, vol 12. Academic, Burlington, pp 201–236
Laouini A, Jaafar-Maalej C, Limayen-Blouza I, Sfar S, Charcosset C, Fessi H (2012) Preparation, characterization and applications of liposomes: the state of art. J Colloid Sci Biotechnol 1:147–168
Lu Q, Li D-C, Jiang J-G (2011) Preparation of a tea polyphenol nanoliposome system and its physicochemical properties. J Agric Food Chem 59:13004–13011
Maherani B, Aab-Tehrany E, Kheirolomoom A, Cleymand F, Linder M (2012) Influence of lipid composition on physicochemical properties of nanoliposomes encapsulating natural dipeptide antioxidant l-carnosine. Food Chem 134:632–640
Maherani B, Arab-Tehrany E, Mozafari MR, Gaiani C, Linder M (2011) Liposomes: a review of manufacturing techniques and targeting strategies. Curr Nanosci 7:436–452
Marsanasco M, Márquez AL, Wagner JR, Alonso SV, Chiaramoni NS (2011) Liposomes as vehicles for vitamins E and C: an alternative to fortify orange juice and offer vitamin C protection against heat treatment. Food Res Int 44:3039–3046
McClements DJ (2005) Food emulsions: principles, practice and techniques, 2nd edn. CRC, Washington
Milas M, Rinaudo M (1979) Conformational investigation on the bacterial polysaccharide xanthan. Carbohydr Res 76:189–196
Moraes M, Carvalho JMP, Silva CR, Cho S, Sola MR, Pinho SC (2013) Liposomes encapsulating β-carotene produced by proliposomes method: characterization and shelf life of powders and phospholipid vesicles. Intl J Food Sci Technol 48:274–282
Mozafari MR, Johnson C, Hatziantoniou S, Demetzos C (2008) Nanoliposomes and their applications in food nanotechnology. J Liposome Res 18:309–327
Oliveira CLP, Gerbelli BB, Silva ERT, Nallet F, Navailles L, Oliveira EA, Pedersen JS (2012) Gaussian deconvolution: a useful method for a form-free modeling of scattering data from mono- and multilayered planar systems. J Appl Crystallogr 45:1278–1286
Parada J, Aguilera JM (2007) Food microstructure affects the bioavailability of several nutrients. J Food Sci 72:21–32
Patil YP, Jadhav S (2014) Novel methods for liposome preparation. Chem Phys Lipids 177:8–18
Pintea A, Diehl HA, Momeu C, Aberle L, Socaciu C (2005) Incorporation of carotenoid esters into liposomes. Biophys Chem 118:7–14
Rashidinejad A, Birch EJ, Sun-Waterhouse D, Everett DW (2014) Delivery of green tea catechin and epigallocatechin gallate in liposomes incorporated into low-fat hard cheese. Food Chem 156:176–183
Ratnam DV, Ankola DD, Bhardwaj V, Sahana DK, Kumar MNVR (2006) Role of antioxidants in prophylaxis and therapy: a pharmaceutical perspective. J Control Release 11:189–207
Rocha FM, Pinho SC, Santana MHA, Zollner RL (2001) Preparation and characterization of affinity magnetoliposomes useful for detection of antiphospholipid antibodies. J Magn Magn Mater 225:101–108
Socaciu C, Bojarski P, Aberle L, Diehl HA (2002) Different ways to insert carotenoids into liposomes affect structure and dynamics of the bilayer differently. Biophys Chem 99:1–15
Socaciu C, Jessel R, Diehl HA (2000) Competitive carotenoid and cholesterol incorporation into liposomes: effects of membrane phase transition, fluidity, polarity and anisotropy. Chem Phys Lipids 106:79–88
Tan C, Xia S, Xue J, Xie J, Feng B, Zhang X (2013) Liposomes as vehicles for lutein: preparation, stability, liposomal membrane dynamics, and structure. J Agric Food Chem 61:8175–8184
Tan C, Xue J, Lou X, Abbas S, Guan Y, Feng B, Zhanga X, Xia S (2014) Liposomes as delivery systems for carotenoids: comparative studies of loading ability, storage stability and in vitro release. Food Funct 5:1232–1240
Taylor TM, Weiss J, Davidson PM, Bruce BD (2005) Liposomal nanocapsules in food science and agriculture. Food Sci Nutr 45:587–605
Toniazzo T, Berbel IF, Cho S, Fávaro-Trindade CS, Moraes ICF, Pinho SC (2014) β-carotene-loaded liposome dispersions stabilized with xanthan and guar gums: physicochemical stability and feasibility of application in yogurt. LWT Food Sci Technol 59:1265–1273
Van Vuuren SF, du Toit LC, Parry A, Pillay V, Choonara YE (2010) Encapsulation of essential oils within a polymeric liposomal formulation for enhancement of antimicrobial efficacy. Nat Prod Commun 5:1401–1409
Wagner A, Vorauer-Uhl K (2011) Liposome technology for industrial purposes. J Drug Deliv. doi:10.1155/2011/591325
Xia F, Hu D, Jin H, Zhao Y, Liang J (2012) Preparation of lutein proliposomes by supercritical anti-solvent technique. Food Hydrocoll 26:456–463
Yoshida PA, Yokota D, Foglio MA, Rodrigues RAF, Pinho SC (2010) Liposomes incorporating essential oil of Brazilian cherry (Eugenia uniflora L.): characterization of aqueous dispersions and lyophilized formulations. J Microencapsul 27:416–425
Zhao P, Xiong H, Peng H, Wang Q, Han D, Bai C, Li Y, Shi S, Deng B (2011) PEG-coated lyophilized proliposomes: preparation, characterization and in vitro release evaluation of vitamin E. Eur Food Res Technol 232:467–654
Zou L-Q, Liu W, Liu W-L, Liang R-H, Li T, Liu C-M, Cao Y-L, Niu J, Liu Z (2014) Characterization and bioavailability of tea polyphenol nanoliposome prepared by combining an ethanol injection method with dynamic high-pressure microfluidization. J Agric Food Chem 62:934–941
Acknowledgments
The authors are grateful to FAPESP for the financial support (project 2009/01087-7) and the scholarships awarded (projects 2009/14868-7, 2011/01277-0, and 2011/03901-3). The authors also thank Danisco for the donation of xanthan gum and Carlos Alberto Paula Leite for the micrographs of the liposomes.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carvalho, J.M.P., Toniazzo, T., Cavalcanti, L.P. et al. Physico-chemical stability and structural characterization of thickened multilamellar beta-carotene-loaded liposome dispersions produced using a proliposome method. Colloid Polym Sci 293, 2171–2179 (2015). https://doi.org/10.1007/s00396-015-3594-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3594-8