Abstract
Rheological properties of concentrated oil-in-water emulsions containing dissolving polymers in both phases, partially playing a role of surfactants, were studied. Additionally, nanoparticles were added to the aqueous phase, and they had an influence on rheological behavior and emulsion stability. The main peculiarity of the objects is the superposition of viscoelastic properties related to the presence of polymers and to interface interactions. Emulsion viscoelasticity were characterized by three separate relaxation modes with very different relaxation times. They reflect relaxation processes of polymeric origin inside both phases, which are dilute polymer solutions, and elasticity of interface layers. Presence of nanoparticles strongly affects the rheological properties leading to the increase in the apparent viscosity, elastic modulus, and yield stress of emulsions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
So-named Pickering emulsions stabilized by solid micro- or nanoparticles were studied in numerous publications [1–8]. Usually the subject of these studies was classical oil-in-water or reverse emulsions. We have chosen a rather peculiar emulsion, which can be called a “polymeric emulsion” because different polymers are present in both phases. Naturally, polymers give their own input into viscoelastic properties of emulsions in addition to viscoelasticity provided by surface effects. Besides, one expects that polymer components can have an influence on stability of emulsions [9].
One of the components in emulsions was an aqueous solution of hydroxypropyl cellulose (HPC) and the second one was an oil solution of polyisoprene. These polymers are not real surfactants, but there are some indications on surface activity of HPC [10–12]. Additionally, different nanoparticles were added to the aqueous solution.
Perspective field for application of such a complex system composed of two polymers is using these emulsions as precursor for pressure-sensitive adhesives. In particular, polyisoprene [13], block copolymers [14, 15], and HPC [16]-based adhesive of such a kind are known. Using polymer blends is useful for controlling morphology [17, 18] and increasing tackifier of adhesives [19]. Besides, such pressure-sensitive adhesives can be used for transdermal drug delivery systems [20, 21].
The goal of this work was to understand the role of solid particles for emulsions in which both phases consist in polymer solutions, and one can suspect that a superposition of polymer and solid particles as co-surfactant takes place.
Materials and methods
The object of this work is the rheology of direct emulsions of the oil-in-water type. The following phases were used: 5 wt.% solution of HPC (Klucel JF, M w = 1.4 · 105) in distilled water and 5 wt.% solution of cis-1,4-polyisoprene (Isolene 400, M w = 9.0 · 104) in n-pentadecane.
Nanoparticles—0.5 wt.% of Na-montmorillonite Cloisite Na+ (MMT) or ultradispersed nanodiamonds of the detonation synthesis (NaD) nanoparticles—were added to the aqueous phase. The characteristic size of the NaD elementary unit is 5 nm, but they are inclined to agglomerate forming structures with size of 80–140 nm. Particles of MMT form aggregated of the similar size.
The imposition of solid additives and emulsification were performed using the disperser IKA Ultra-Turrax T18. The rate of the agitator rotation was 24,000 rpm, and processing time was 15 min. As a result, homogeneous (on the macroscopic level) emulsions were obtained. No foaming took place. The droplet diameter lies in the interval of 2–6 μm. The droplet size of the disperse phase in a system without solid particles was close to the upper boundary of the specified range, while addition of MMT shifted the characteristic size to the lower boundary. The emulsions under study without nanoparticles were stable during at least a week. Stability was understood as the absence of evident phase separation of emulsion into two layers. The introduction of NaD increased stability up to 2 weeks, and nothing happened in the systems with MMT during at least a year.
Weight content of an oil phase in emulsions was 50 wt.% (56.6 vol.%). So, it was a concentrated emulsion but below the limit of the closest packing. Rheological properties of less concentrated emulsions were also measured. The content of the disperse phase was 12.6, 24.5, 35.8, 46.4, 56.6, and 66.1 vol.% (consequently, 10, 20, 30, 40, 50, and 60 wt.%).
Rheological measurements were carried out mainly on a Physica MCR 301 (Anton Paar) rheometer. The cone-and-plate pair was used. The diameter of the cone was 50 mm, and the angle between plate and cone surface was 1°. Additionally, rheological properties of samples were measured using a RheoStress 600 (Thermo Scientific HAAKE) rheometer with a 20-mm parallel plate cell. The gap between plates was 1 mm and the surface of plates was either smooth or grooved.
The interfacial properties were studied using the Physica MCR 301 rheometer and a bicone arrangement (d = 68.28 mm, α = 2 × 5°) placed in a cylindrical cell (d = 80 mm) at the boundary between oil and water phases [22, 23].
All experiments at steady shear flow were carried out in two regimes. The viscosity, η, were measured either at rate-controlled or at stress-controlled shearing. The complex modulus and its elastic G' and loss G" components were measured at harmonic oscillations.
Measurements of the properties were carried out in the following modes of deformation:
-
frequency dependencies of the components of the complex modulus (G' and G") at low strains corresponding to the linear region of viscoelasticity at frequencies ranging from 0.0628 to 628 s−1;
-
amplitude dependence of the large-strain elastic modulus by amplitude scanning at a fixed angular frequency of 6.28 s−1 [24];
-
dependencies of shear stress on shear rate (flow curves) by scanning along the shear rate in the range between 0.001 and 1000 s−1 at duration of every step of 3 min and the rate interval between steps equal to one fifth of the decimal order.
Results and discussion
Viscous flow
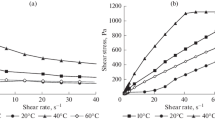
Flow curves of emulsions are shown in Fig. 1. Emulsions additionally stabilized by nanoparticles possess higher viscosity in comparison with emulsions stabilized only by polymers solved on both phases. However, it is worth mentioning that the change of Na-MMT for hydrophobic organo-modified MMT (Cloisite 20A or Cloisite 30B) or replacement of nanodiamonds for carbon nanotubes in equivalent content did not allow us to obtain stable emulsions. They were disintegrated into separate phase in several minutes. It is the same regardless whether these components are placed either into aqueous or oil phase.
According the used protocol, the flow curves in Fig. 1 were obtained in the rate-controlled scanning mode. Then the maximal Newtonian branches should be discussed to answer whether they respond to the steady flow regimes. This problem became actual in light of the publication [25], where it was stated that the Newtonian branches in visco-plastic media are artifacts obliged to the fact that transient states of deformations were practically measured. An analogous result was earlier obtained in [26], where a systematic transformation from the Newtonian flow to the yield stress behavior was observed and that was treated as rheopexy in the behavior of concentrated emulsions.
To answer this concern, an independent experiment has been performed. Shear stress evolution (and consequently, the apparent viscosity) was observed at prolong fixed rate shearing. Figure 2 demonstrates the results of the “apparent viscosity” measurements (defined as the ratio of current stresses to a specified shear rate). As is seen, a pronounced transient process happens reflecting the unlimited increase of apparent viscosity with the tendency to the yield stress, just as was described in [25–27].
So, the “observed” Newtonian plateau in Fig. 1 seems not to have any physical meaning and is an artifact, indeed. Its apparent values are related to the method of measuring based on scanning with a rather short step. Actually, we have to consider these dispersions as a visco-plastic medium with flow curves which should look as shown in Fig. 1 by thin lines corresponding to the yield stress.
Meanwhile, the picture is somewhat more complicated. If the experiment is carried out at a very low fixed stress, evolution of the apparent viscosity looks as shown in Fig. 3. These data demonstrate the existence of a clear limit, i.e., the Newtonian plateau. So, this dispersion demonstrates a dualism of the rheological behavior (or viscosity bifurcation) at the low stress/rate limit depending on the mode of deformation [28–30].
Not quite “traditional” shape of flow curves with the bend in the middle part also draws attention. The same shape of flow curves was observed in [26] for highly concentrated water-in-oil emulsions. Bends on the flow curves were explained (and experimentally proven by direct observations) by the change in the mechanism of flow—from the rolling of larger droplets over smaller ones to bulk shear deformation of droplets. Possibly, an analogue effect may be proposed in this case. However, the droplet size in our case is essentially smaller than used in [26], and it makes hardy possible carrying out optical observations.
The above presented and discussed results on measuring viscous properties of emulsion were obtained using the cone-and-plate instrument with a small angle between cone and plate surfaces. So, the stress field was homogeneous throughout a sample.
Meanwhile, in many publications of the last decade (e.g., [29, 31–36]), it was shown that layer stratification (“shear banding”) can take place in the flow of numerous multi-component systems and emulsions, in particular. So, it seems reasonable to compare the above presented results with data obtained in different flow conditions bearing in mind to clear up whether we are dealing with the homogeneous flow or not.
Independent flow characteristics of dispersions were obtained by measuring their rheological properties in rotational flow between two parallel disks on the “plate-and-plate” unit. The flat disks as well as grooved surfaces were used. The latter allowed us to believe that slip at the boundary surfaces of a measuring cell was excluded.
The results of the comparative study are shown in Fig. 4. One can see that the data obtained with different geometry of flow at two rheometers coincide up to the definite limit. However, shear stresses and consequently the apparent viscosity values obtained with smooth surfaces sharply decrease in comparison with the other experimental points starting from some shear rate (or stress), marked by the arrow in Fig. 4. The comparison of data obtained for smooth and grooved surfaces should be treated as the transition to the wall sliding. Meanwhile, data obtained on the “cone-and-plate” and “plate-and-plate” with smooth surface units coincide. It can be understood as proof that these data correspond to the homogeneous bulk flow without shear banding formation.
Figure 1 shows that the type of stabilization noticeably affects the quantitative measurements of theological properties: solid particles strongly increase apparent viscosity and the strength of the emulsion structure (characterized by the yield stress).
Viscoelasticity of dispersions
The emulsions under study as well as nanoparticle containing dispersions are viscoelastic media. In Fig. 5, the frequency dependencies of dynamic modulus components are presented. They were obtained for the linear region of viscoelasticity. Addition of nanoparticles leads to the increase of the elastic modulus which can exceed the loss modulus in some cases. This effect is in line with the increase in the apparent viscosity and the yield stress
The increase of the moduli values along the increase of frequency is observed for the whole frequency range. The transition to the plateau can be seen in the low frequency limit only. This is not typical for concentrated emulsions [37].
The model [38, 39] of highly concentrated emulsion elasticity predicts the existence of plateau as a consequence of the droplet compression. However, quite different mechanisms dominate for emulsions under consideration at high frequencies. They can be related, first, with the presence of polymers in both phases and, second, with the elasticity of an interfacial layer [40].
The tendency to the constant values of moduli at low frequencies can be related to the existence of the yield stress intrinsic for different multi-component materials. The MMT containing dispersion is the most noteworthy in this relation. The elastic modulus is higher than loss modulus for this system. It might be explained by the well-developed structure formed by lamellar nanoparticles of anisometric shape. So, the G 0 values for emulsions stabilized with solid particles are by an order higher than without them. It can be a consequence of the own percolation structure formed by these particles.
Figure 5 demonstrates that viscoelasticity of emulsions comes out in a rather wide frequency range. Therefore, the presentation of viscoelastic functions via a relaxation spectrum should cover a rather wide range of relaxation times. Then, bearing in mind main relaxation mechanism (macromolecular motion in both phases and viscoelasticity of an interfacial layer), it seems reasonable to limit a relaxation spectrum by three discrete relaxation time values, which are spaced far from each other. This approach can be presented by the following equation
where G 0 is the equilibrium rubbery modulus corresponding to low frequencies, τ i are three relaxation times, and G i are their weights.
The presentation of the relaxation spectrum via three relaxation modes is quite arbitrary but useful for understanding the obtained experimental results.
Since G 0 does not depend on frequency, it can be related to the physics of droplet elasticity presented by the Princen-Mason model [38, 39]. The three relaxation times, τ i , characterize the viscoelastic behavior of emulsions. The values of all these parameters found by the least-square method are shown in Fig. 6.
As is seen, the addition of nanoparticles leads to the acceleration of relaxation processes, possibly due to emulsifying—the break-up of dispersed droplets at the processing stage. The difference in the nature of nanoparticles is not very large in this respect. So, just the nanoparticle presence influences the rate of relaxation. At the same time, the G i values increase. This is the reflection of the increase in the emulsion stability due to the presence of nanoparticles. As was said above, these particles lead to the increase of the elasticity of the interface, i.e., they modify its mobility and increase long-term kinetic stability of emulsions. The impact of MMT is more noticeable than NaD. This is analogous to the role of nanoparticles in viscous properties of emulsions (Fig. 1).
It is also interesting to find the limit of linearity at periodic oscillation because it means the start of the destruction of the structure of emulsions. Figure 7 presents the amplitude dependence of the elastic modulus of the systems under study. It is seen that we can measure viscoelastic properties of emulsions confidently in the linear range and the deformation limit of linearity is approximately 1 %. The growth of the elasticity with addition of nanoparticles providing better stability of emulsions is also well established from these data.
Interfacial rheological properties
Investigation of the interfacial (2D) rheological properties of emulsions is an important method for the estimation of their stability [22]. There are a lot of experimental data based on various methods devoted to the rheology of interfacial layers [41]. However, this theme could not be treated as exhausted because there are so many different emulsion systems and the correlation between interfacial and bulk rheological properties of emulsions is a rather rare subject of investigation.
Figure 8a shows the shear rate dependencies of the equilibrium values (limited values after prolonged exposition) of the interfacial viscosity for aqueous phase/oil phase interfaces with different nanoparticles. The introduction of NaD does not change the interface viscosity. The addition of MMT into the aqueous phase results in the significant increase in viscosity and smoothening the non-Newtonian character of flow curves.
Rheological properties of the aqueous phase/oil phase interfacial boundary: a flow curves for systems without nanoparticles (1), with 1 wt.% NaD (2) or MMT (3); b frequency dependencies of the nterface moduli of elasticity (1) and losses (2) for system with 1 wt.% MMT nanoparticles in the aqueous phase
Frequency dependencies of the components of the interface dynamic modulus were measured for interface with MMT (Fig. 8b). It is worth stressing that there is no noticeable interface elasticity for pure emulsions and only addition of nanoparticles (MMT) creates elasticity of the interfacial layers.
The elasticity of the interfacial boundary is higher than its dissipative component (G s ′ > G s ″). So, the interfacial layer can be treated as an elastic film stabilizing an emulsion. This situation is assumed as one of the criteria determining the stability of an emulsion [42]. One can presume that introduction of nanoparticles acts as an additional stabilizing factor, i.e., a synergetic effect of the emulsion stabilization by polymer and nanoparticles takes place like described for the other systems [3].
Concentration dependence of the rheological properties
Flow curves of the emulsions with different content of a disperse phase stabilized by MMT are presented in Fig. 9. Generally speaking, the flow curves are similar though their shape for relatively dilute (12.6 vol.%) emulsions has some differences at low shear rates. As was said above, the existence of the Newtonian plateau is likely artificial and caused due to the method of measurements because the steady flow is not reached in the rate scanning mode of shearing. In the transition to the low concentration (12.6 vol.%) emulsion, the steady state, possibly, is reached and therefore we see the increase of apparent viscosity reflecting the approach to the yield stress. The physical origin of the yield stress can be related to the formation of the percolation network created by the MMT nanoparticles. These data say about the necessity of caution in treating the apparent viscosity in the low shear rate range as the “Newtonian” viscosity that is in line with a conception developed in [22].
Frequency dependencies of the complex dynamic modulus components for the same set of emulsions are shown in Fig. 10. It is seen that the elastic modulus is higher than the loss modulus for all emulsions with concentration of the oil phase ≥56.6 vol.% in the whole frequency range.
As was said above, viscoelastic properties of emulsions manifest themselves in a rather wide frequency range (Figs. 5 and 10). So, the model with three far distinct relaxation times can be used for visual presentation of viscoelastic properties of emulsions in this case too, as is written by Eq. 1.
The results of relaxation modes calculation are shown in Fig. 11 and the reliability of predictions of the dynamic modulus is shown in Fig. 10 (solid lines) in comparison with the experimental data (points). As is seen, the spectrum of relaxation phenomena is rather wide and the concentration dependencies of various modes are different.
Relatively low concentrations of dissolved polymers allow us to suppose that the corresponding viscoelastic effects are related to the short relaxation times τ 2 and τ 3. So, as well as in the case of a concentrated emulsion, it is reasonable to think that the high-frequency relaxation modes reflect the viscoelasticity of polymer solutions, and this is out of special interest for this discussion. The main interest, evidently, is connected with the “equilibrium” modulus value G 0 and the first mode of the slowest relaxation process with time τ 1 and its weight G 1.
The values of G 0 and G 1 are close to each other and can be presented by the same concentration dependence (Fig. 11a). It is essential that the noticeable elasticity becomes quite evident at the disperse phase concentration of 12.6 vol.%. It is rather obvious that this effect cannot be explained by the deformation (compression) of droplets as the reason of elasticity. The long-term component of relaxation (τ 1 and its partial modulus G 1) can be explained by the elasticity of interfacial layers reinforced by nanoparticles. In the increase of concentration, the interparticle interaction becomes stronger and this results in the sharp increase in the modulus G 1 and the relaxation time τ 1 (Fig. 11b).
Conclusion
Systematic studies of rheological properties of concentrated direct oil-in-water emulsions formed by polymer solutions and modified by solid nanoparticles (in aqueous phase) showed that these systems demonstrate non-linear viscoelastic behavior characteristic for the heterogeneous multi-component systems.
In the range of low shear rate (stresses), bifurcation of the viscous properties is observed. The prolonged deformation shows the unlimited growth of stress with the transition to the yield stress. So, emulsions with dissolved polymeric components as well as emulsion-based dispersions have to be treated as visco-plastic media. Meanwhile, at low fixed stresses, the maximal Newtonian viscosity is confidently observed. The unusual bend on the flow curves is explained by the change in the mechanism of deformation (flow) of concentrated emulsions. Addition of nanoparticles results in the increase of the elasticity of emulsions. That correlated with the increase of their stability.
Relaxation processes proceed in different and far separated time scales. It is connected with short-term relaxation inside both phases (which are semi-dilute polymer solutions) as well as long-term relaxation on heterogeneous interface layers containing solid nanoparticles.
References
Binks BP (2002) Particles as surfactants—similarities and differences. Curr Opin Colloid Interface Sci 7:21–41
Binks BP, Whytby CP (2005) Nanoparticle silica-stabilised oil-in-water emulsions: improving emulsion stability. Colloid Surf A 253:105–115
Binks BP, Desforges A, Duff DG (2007) Synergistic stabilization of emulsions by a mixture of surface-active nanoparticles and surfactant. Langmuir 23:1098–1106
Wolf B, Lam S, Kirkland M, Frith WJ (2007) Shear thickening of an emulsion stabilized with hydrophilic silica particles. J Rheol 51:465–478
Ashbya NP, Binks BP (2000) Pickering emulsions stabilised by Laponite clay particles. Phys Chem Chem Phys 2:5640–5646
Melle S, Lask M, Fuller GG (2005) Pickering emulsions with controllable stability. Langmuir 21:2158–2162
Wang J, Yang F, Tan J, Liu G, Xu J, Sun D (2010) Pickering emulsions stabilized by a lipophilic surfactant and hydrophilic platelike particles. Langmuir 26:5397–5404
Aveyard R, Binks BP, Clint JH (2003) Emulsions stabilised solely by colloidal particles. Adv Colloid Interface Sci 100–102:503–546
Okada M, Maeda H, Fujii S, Nakamura Y, Furuzono T (2012) Formation of Pickering emulsions stabilized via interaction between nanoparticles dispersed in aqueous phase and polymer end groups dissolved in oil phase. Langmuir 28:9405–9412
Chang SA, Gray DG (1978) The surface tension of aqueous hydroxypropyl cellulose solutions. J Colloid Interface Sci 67:255–265
Mezdoura S, Cuveliera G, Cashb MJ, Michon C (2007) Surface rheological properties of hydroxypropyl cellulose at air–water interface. Food Hydrocoll 21:776–781
McNally EJ, Zografi G (1990) Spread and adsorbed monolayers of hydroxypropyl cellulose and hydroxyethyl cellulose at the air–water interface. J Colloid Interface Sci 138:61–68
Yarusso DJ, Rivard RJ, Ma J (1999) Properties of polyisoprene-based pressure sensitive adhesives crosslinked by electron beam irradiation. J Adhesion 69:201–215
Gibert FX, Marin G, Derail C, Allal A, Lechat J (2003) Rheological properties of hot melt pressure-sensitive adhesives based on styrene–isoprene copolymers. Part 1: a rheological model for [sis-si] formulations. J Adhes 79:825–852
Sasaki M, Fujita K, Adachi M, Fujii S, Nakamura Y, Urahama Y (2008) The effect of tackifier on phase structure and peel adhesion of a triblock copolymer pressure-sensitive adhesive. Int J Adhes Adhes 28:372–381
Borodulina T, Bermesheva E, Smirnova N, Ilyin S, Brantseva T, Antonov S (2014) Adhesive properties of liquid crystalline hydroxypropyl cellulose–propylene glycol blends. J Adhes Sci Technol 28:1629–1643
Class JB, Chu SG (1985) The viscoelastic properties of rubber–resin blends. I. The effect of resin structure. J Appl Polym Sci 30:805–814
Tobing S, Klein A, Sperling LH, Petrasko B (2001) Effect of network morphology on adhesive performance in emulsion blends of acrylic pressure sensitive adhesives. J Appl Polym Sci 81:2109–2117
Leong YC, Lee LMS, Gan SN (2003) The viscoelastic properties of natural rubber pressure‐sensitive adhesive using acrylic resin as a tackifier. J Appl Polym Sci 88:2118–2123
Tan HS, Pfister WR (1999) Pressure-sensitive adhesives for transdermal drug delivery systems. Pharm Sci Technol Today 2:60–69
Valenta C, Auner BG (2004) The use of polymers for dermal and transdermal delivery. Eur J Pharm Biopharm 58:279–289
Krägel J, Derkatch SR (2010) Interfacial shear rheology. Curr Opin Colloid Interface Sci 15:246–255
Erni P, Fischer P, Windhab EJ, Kuznezov V, Stettin H, Läuger J (2003) Stress- and strain-controlled measurements of interfacial shear viscosity and viscoelasticity at liquid/liquid and gas/liquid interfaces. Rev Sci Instr 74:4916–4924
Ilyin S, Kulichikhin V, Malkin A (2014) Characterization of material viscoelasticity at large deformations. Appl Rheol 24:13653
Møller PCF, Fall A, Bonn D (2009) Origin of apparent viscosity in yield stress fluids below yielding. EPL 87:38004
Masalova I, Taylor M, Kharatiyan E, Malkin AY (2005) Rheopexy in highly concentrated emulsions. J Rheol 49:839–849
Malkin A, Ilyin S, Roumyantseva T, Kulichikhin V (2013) Rheological evidence of gel formation in dilute poly (acrylonitrile) solutions. Macromolecules 46:257–266
Coussot P, Nguyen QD, Huynh HT, Bonn D (2002) Viscosity bifurcation in thixotropic, yielding fluids. J Rheol 46:573–589
Malkin A, Ilyin S, Semakov A, Kulichikhin V (2012) Viscoplasticity and stratified flow of colloid suspensions. Soft Matter 8:2607–2617
Ilyin SO, Pupchenkov GS, Krasheninnikov AI, Kulichikhin VG, Malkin AY (2013) Rheology of aqueous poly (ethylene oxide) solutions reinforced with bentonite clay. Colloid J 75:267–273
Berret JF, Porte G, Decruppe JP (1997) Inhomogeneous shear flows of wormlike micelles: mA master dynamic phase diagram. Phys Rev E 55:1668
Britten MM, Callaghan TP (1997) Two-phase shear band structures at uniform stress. Phys Rev Lett 78:4930
Salmon JB, Manneville S, Colin A (2003) Shear banding in a lyotropic lamellar phase. I. Time-averaged velocity profiles. Phys Rev E 68:051503
Olmsted PD (2008) Perspectives on shear banding in complex fluids. Rheol Acta 47:283–300
Dhont JKG, Briels WJ (2008) Gradient and vorticity banding. Rheol Acta 47:257–281
Ilyin S, Roumyantseva T, Spiridonova V, Semakov A, Frenkin E, Malkin A, Kulichikhin V (2011) Gels of cysteine/Ag-based dilute colloid systems and their rheological properties. Soft Matter 7:9090–9103
Derkach SR (2009) Rheology of emulsions. Adv Colloid Interface Sci 151:1–23
Princen HM (1986) Osmotic pressure of foams and highly concentrated emulsions. I. Theoretical considerations. Langmuir 2:519–524
Mason TG, Lacasse MD, Grest GS, Levine D, Bibette J, Weitz DA (1997) Osmotic pressure and viscoelastic shear moduli of concentrated emulsions. Phys Rev E 56:2150–3166
Foudazi R, Masalova I, Malkin AY (2010) The role of interdroplet interaction in the physics of highly concentrated emulsions. Colloid J 72:74–92
Kragel J, Derkatch SR (2009) Interfacial shear rheology—an overview of measuring techniques and their applications. In: Miller R, Liggieri L (eds) Interfacial rheology. Brill Publ, Leiden, pp 372–428
Fainerman VB, Möbius D, Miller R (2001) Surfactants: chemistry, interfacial properties, applications. Elsevier, Amsterdam
Acknowledgment
The authors are grateful to the Russian Scientific Foundation (agreement #14-23-00003 of August 7, 2014) for financial support of the work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilyin, S.O., Kulichikhin, V.G. & Malkin, A.Y. Rheological properties of emulsions formed by polymer solutions and modified by nanoparticles. Colloid Polym Sci 293, 1647–1654 (2015). https://doi.org/10.1007/s00396-015-3543-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3543-6