Abstract
Purpose
Several experimental studies showed that magnesium intake improved insulin resistance and glucose uptake in diabetes patients. However, epidemiological studies on the association between magnesium intake and diabetes risk have yielded inconsistent results. We investigated whether magnesium intake is related to the risk of developing diabetes in a population-based cohort study in Japan.
Methods
Study subjects were participants in the Takayama study. A total of 13,525 residents in Takayama City, Japan, responded to a self-administered questionnaire in 1992 and to a follow-up questionnaire seeking information about diabetes in 2002. Magnesium and other nutrient intakes were estimated from a validated food frequency questionnaire administered at the baseline.
Results
During a follow-up of 10 years, 438 subjects reported diabetes newly diagnosed by physician. Compared with women in the low quartile of magnesium intake, women in the high quartile were at a significantly reduced risk of diabetes (HR 0.50; 95 % CI 0.30–0.84; P-trend 0.005) after adjustments for covariates. In men, there was no association between magnesium intake and the risk of diabetes.
Conclusion
These results suggest that diets with a high intake of magnesium may decrease the risk of diabetes in women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Magnesium is one of the most abundant ions present in cells. It serves as a necessary cofactor in over 300 enzymatic reactions that require adenosine triphosphate and as a key component in various reactions that require kinases [1, 2]. Magnesium is believed to play a role in insulin activity and glucose homeostasis. In contrast, insulin regulates the magnesium homeostasis. Several animal studies demonstrated a significant reduction in the risk of diabetes after feeding with a magnesium-rich diet [3]. Hypomagnesemia in diabetic patients has been widely recognized [4]. Dietary magnesium intake was inversely associated with fasting serum insulin in diabetes patients [5, 6]. Magnesium depletion has a negative impact on glucose homeostasis and insulin sensitivity in diabetic patients, as well as on the evolution of complications such as retinopathy, neuropathy, hypertension, and cardiovascular disease [5, 7–11]. Magnesium supplementation improved insulin action and glycemic control among patients with diabetes [12, 13]. Although these studies suggest that dietary magnesium may have a beneficial effect for diabetic subjects, the role of magnesium intake on insulin and glucose homeostasis in non-diabetic subjects is unknown. Magnesium intake might have a protective effect against the development of diabetes.
Several prospective epidemiologic studies have explored the associations of dietary magnesium with diabetes in Europe and the USA [6, 14–18]. Some studies showed a significantly inverse association between magnesium intake and the risk of diabetes [15–18], but others showed no association [6, 14]. It has been reported that insulin sensitivity is lower among Japanese subjects than among Caucasians [19, 20]. It is of interest to examine the association between magnesium intake and diabetes risk in the Japanese population. However, to our knowledge, only three studies prospectively examined this association in the Japanese population, and the results have been inconsistent [21–23]. Therefore, in this study we investigated whether magnesium intake from food is related to the risk of developing diabetes among Japanese men and women in a cohort study.
Methods
Study group
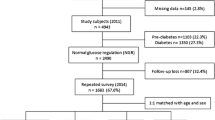
Subjects in this study were cohort members from a population-based cohort study conducted in Takayama City, Gifu Prefecture, Japan (the Takayama study). The Takayama study, in which 36,990 men and women aged 35 years and over were registered, was established in September 1992. The study was initiated to identify dietary and lifestyle factors in relation to morbidity from chronic diseases. At the baseline, subjects filled out a self-administered questionnaire on demographic characteristics, smoking, diet, physical activity, and reproductive and medical histories, including diabetes. A total of 14,427 men and 17,125 women, 85.3 % of the target population, completed the questionnaire. Details of the study design have been described elsewhere [24].
The 169-item FFQ including alcoholic beverages was used to collect dietary information over the previous year. For each food item or dish, subjects were asked their consumption frequency and their usual unit or portion size. Nine response options were available for eating frequency, from “never” to “two or more times per day.” Slightly different options were used for beverage intake: from “never” to “four or more times per day.” A standard portion size was specified for each food item, and the respondents were asked to choose their usual portion from among three categories. We calculated the average daily intake of nutrients including magnesium by multiplying the frequency of the consumption of each food by its nutrient content per serving and totaling the nutrient intake for all food items. We obtained information on use of supplements of multivitamin, vitamin A, β-carotene, vitamin C, calcium, and vitamin E, but not of magnesium. Therefore, dietary intake of magnesium did not include supplemental magnesium. We also could not provide the results after controlling for any supplemental magnesium ingested by participants.
A detailed description of the FFQ and the method used for calculating nutrient intake has been published previously [25]. The FFQ was validated in this population by comparing 3-day diet records, four 24-h recalls, and 12 daily diet records kept over a 1-year period [25]. Correlation coefficients between the FFQ and the 12 daily diet records for the intakes of magnesium, total energy, and total fat were 0.56, 0.44, and 0.31, respectively, in men and 0.59, 0.53, and 0.52, respectively, in women. Body mass index was calculated as weight in kilograms divided by the square of height in meters. To assess the amount of regular physical activity, the average number of hours per week spent on listed physical activities, such as strenuous sports, vigorous work, and moderate exercise, was sought. The amount of time per week spent at each intensity of activity was multiplied by its corresponding energy expenditure requirements, expressed in metabolic equivalents (METs), and summed to yield a physical activity score (MET-h/week). Further details and the validity information of the physical activity questionnaire have been previously reported [26, 27].
Follow-up and endpoints
For the present study, participants who were younger than 70 years at baseline (n = 26,546) were the target population. In July 2002, a follow-up survey seeking information on diabetes was conducted. By the time this follow-up survey began, 1505 participants were deceased, 51 participants were physically unable to complete the questionnaire, and 2598 participants had relocated. After these exclusions, the follow-up questionnaire was distributed to 22,392 persons, of whom 14,975 (66.9 %) responded to the questionnaire. Compared with non-respondents to the follow-up questionnaire, respondents were more likely to be older, better educated (≥15 years), and never smokers and had lower intakes of alcohol and coffee, as described elsewhere [28, 29]. Magnesium intake did not differ between respondents and non-respondents.
The specific endpoint of this study was incidence of diabetes. The participants who developed diabetes between the baseline survey and the follow-up survey were identified. The participants were asked whether they had been diagnosed with diabetes by a physician and, if so, at what age. Because 30 men and 10 women who developed diabetes during the follow-up period did not provide information regarding the time of diagnosis, we assigned median values for time to diagnosis among persons who developed diabetes for men and women separately. We previously reported that the sensitivity and specificity of self-reported diabetes as compared with the reference standard, defined by hemoglobin A1c level ≥6.1 % (JDS) [30], were 57.4 and 96.5 %, respectively, in a subgroup of participants (n = 214) [31]. The sensitivity and specificity did not differ greatly between subjects with low (below the median) and high (above the median) intake of magnesium; the values were 95.9 and 60.0 % in the low intake group and 97.1 and 50.0 % in the high intake group, respectively. The sensitivity and specificity were not dependent on the covariates in the model except for BMI; the values were 97.8 and 80.0 % in the low BMI group and 94.6 and 44.4 % in the high BMI group, respectively.
For the present analysis, participants who were identified as having had diabetes at the baseline from the follow-up questionnaire (n = 100) were excluded. Additionally, participants who reported a diagnosis of coronary heart disease, stroke, cancer, and diabetes at the baseline were excluded (n = 1350). After these exclusions, a total of 13,525 subjects (5885 men and 7640 women) were included in the present analysis.
Statistical analysis
Analyses were performed on men and women separately. Dietary intake of magnesium was log-transformed to better approximate a normal distribution and then adjusted for total energy intake using the residual method by Willett [32]. Subjects were divided into four equal groups on the basis of quartile of magnesium intake. Baseline characteristics were presented according to the quartile of dietary magnesium intake. We used the Cox proportional hazards model to estimate the hazard ratios (HRs) and 95 % confidence intervals (95 % CIs) of incident diabetes in each category of magnesium intake compared with the low quartile. The trends of associations were assessed using the median values of magnesium intake in each quartile. Age was adjusted in the initial model. In multivariable models, we also adjusted for potential confounders of diet–diabetes associations (15, 17, 21, 23), including body mass index, level of education (≤11, 12–14, or ≥15 years), physical activity (MET-h/week), smoking status (men: never smoker, former smoker, current smoker with ≤30 years of smoking, or current smoker with >30 years of smoking; women: never smoker, former smoker, or current smoker), history of hypertension (yes or no), menopausal status (yes or no; women only), and dietary factors, including glycemic load and daily intakes (continuous) of alcohol and coffee, total energy, and total fat. The glycemic load is the calculated product of the carbohydrate content and glycemic index (GI) of a food. The GI is a tool that allows for quantification of the postprandial blood glucose response to dietary carbohydrates from foods [33]. Statistical analyses were performed using SAS statistical software, version 9.3 (SAS Institute). Significance was defined as a two-sided P value <0.05.
Results
Characteristics of the study population, by sex and quartile of magnesium intake without magnesium supplement, are shown in Table 1. In the present study, the means of magnesium intake were 406 (SD 162) mg/day for men and 374 (SD 152) mg/day for women. Men and women with a higher intake of magnesium were older and less likely to be current smokers. They also had higher intakes of fat, vegetable, and fruit and a lower glycemic load. Women who had a greater intake of magnesium were more likely to be postmenopausal. The data presented for magnesium intake may have been overestimated by the questionnaire, since in the validity study, magnesium estimates were about 40 percent higher based on the questionnaire than were estimates based on diet records. Using data from the validation study (81 men and 101 women), we examined the association between the reporting accuracy and the variables given in Table 1. In men, the greatest difference was observed between men with high (above the median) and low (below the median) intake of alcohol; the correlation coefficients between the FFQ and the 3-day diet records for magnesium intake were 0.43 and 0.13, respectively, although the difference was not significant. In women, the greatest but nonsignificant difference was observed between women with high (above the median) and low (below the median) intake of coffee; the corresponding correlation coefficients were 0.17 and 0.61, respectively. The BMI did not affect the correlation coefficients for magnesium intake in both men and women; the values were 0.40 and 0.32 in men with high and low BMI, respectively, and 0.30 and 0.40 in women with high and low BMI, respectively.
During a follow-up of 10 years, we documented 438 incident cases of diabetes. The HR and 95 % CI for diabetes according to quartile of magnesium intake are given in Table 2. In men, there was no significant association between magnesium intake and the risk of diabetes and the association was not altered after adjusted for confounders. In women, unadjusted HR (95 % CI) was 0.95 (0.62–1.45), 0.84 (0.54–1.30), and 1.09 (0.72–1.64) for the second to the highest quartiles as compared with the lowest quartile, respectively. After adjustment for covariates, the highest and the second highest quartiles of magnesium intake were associated with a decreased risk of diabetes. The trend was also statistically significant (P-trend: 0.005). Confounding effect due to glycemic load, alcohol, and fat intake was great; the HR (95 % CI) for the highest as compared with the lowest quartile of magnesium intake was 0.89 (0.59–1.36) without controlling for these three variables. BMI did not appear to be a strong confounder nor mediator; the HR for the highest as compared with the lowest quartile of magnesium intake was 0.61 (0.39–0.94) after excluding BMI from the covariates. Additional adjustments for vitamin supplement use (yes or no) or intakes of calcium, potassium, phosphorous, vitamin C, folate, fish, vegetables, and fruits did not alter the results substantially; for example, the HR (95 % CI) for the highest as compared with the lowest quartile of magnesium intake was 0.29 (0.16–0.55) after controlling for vitamin supplement use and intakes of vegetables and fruits.
Discussion
In this prospective study, we found an inverse association between magnesium intake and the risk of developing diabetes among women after multivariable adjustment for several risk factors for diabetes. The high quartile of magnesium intake had a 50 % lower risk of developing diabetes than the low quartile among women. Two large prospective cohort studies among Western women, the Iowa Women’s Health Study [18] and the Nurses’ Health Study [15], observed similar results; the declines in the risk of diabetes were 24 and 38 %, respectively. On the other hand, the Women’s Health Study reported no significant association in the US women [6]. Among Japanese women, Kirii et al. [23] observed a 32 % reduction in the risk of diabetes associated with the highest quintile of magnesium intake, although this did not obtain statistical significance. A similar reduction in diabetes risk (37 %) in the highest quartile of magnesium intake was reported by Hata et al. [22] after controlling for sex. However, another study conducted among Japanese women observed no association [21]. The estimates of magnesium intake were similar among subjects in the USA, the UK, and Japan [34].
Studies on the effect of magnesium on insulin or glucose levels among non-diabetic subjects are few. One study reported that daily magnesium supplements (4.5 g/day for 4 weeks) improved insulin secretion in elderly non-obese subjects [13]. Some mechanisms have been proposed to explain the potential effects of magnesium on insulin and glucose actions. Diminished intracellular levels of magnesium may impair tyrosine kinase activity at insulin receptors and reduce glucose-stimulated insulin secretion at the pancreatic β-cell [6, 35]. The beneficial effect of magnesium on insulin and glucose control may lead to protection against diabetes.
We failed to find an inverse association between magnesium intake and the risk of diabetes in men. The observed discrepancy in our results between men and women might be explained by gender differences in the metabolism of magnesium. Some studies have reported a higher incidence of hypomagnesemia in women compared with men [36]. Women had significantly lower levels of serum magnesium than men [0.76 ± 0.10 vs. 0.81 ± 0.08 mmol/L (mean ± SD), P < 0.05] [37]. A positive association between magnesium intake and blood magnesium concentration has been reported [7]. Low blood magnesium concentrations indicate low magnesium stores [38]. Women’s magnesium body stores may be lower, and depletion would be more likely to occur in women than in men. The role of dietary magnesium in insulin regulation may be more important in women than in men, which would explain the observed risk reduction in diabetes in women. A large cohort of men in the USA, the Health Professionals Follow-up Study [16, 17], reported a significant inverse association between magnesium intake and diabetes risk. In Japan, Kirii et al. [23] reported a risk reduction (36 %) among men in the highest quartile of magnesium intake, but this association was not statistically significant. Nanri et al. [21] observed no association.
The major strengths of the present study include a population-based prospective design and the use of a validated FFQ. Despite these strengths, the present study has several limitations that warrant mention. First, the response rate to the follow-up questionnaire was not high (66.9 %). The relatively low response rate may increase the potential for selection bias, such as differential participation among the case groups by magnesium intake. However, based on dietary data obtained on both respondents and non-respondents, we do not expect a large amount of bias. The second limitation is that the incidence of diabetes was ascertained based on self-reported information. However, a previous study in Japan confirmed that the self-reported history of diabetes in the questionnaire was consistent with the diagnosis by physicians (sensitivity 80.8 %, specificity 99.3 %) [39]. Nevertheless, no screening for undiagnosed diabetes was done. The sensitivity of self-reported diabetes as compared with the criterion defined by hemoglobin A1c level was not high in our subsample, although similar sensitivity of self-reported diabetes in comparison with biomarkers has been reported in other studies [40, 41]. Therefore, there is a concern that women who were healthier were more likely diagnosed because of more visits to the doctor. It is also possible that women who were less healthy were more likely diagnosed because they have risk factors like high BMI. The third limitation is that magnesium intake assessed at only one time point may not reflect the changes in magnesium intake during the follow-up period. The fourth, despite the use of a validated FFQ, some degree of misclassification of dietary intake is to be expected, just as in other nutritional epidemiologic studies. However, it is unlikely that magnesium intake on our FFQ would be systematically underestimated among the incident diabetes cases. Finally, we could not obtain information on family history of diabetes as well as genetic traits. Genetic factors such as KCNQ1 [42], KCNJ15 gene [43], and ANK1 locus [44] have been implicated in the diabetes risk among Asians.
In summary, magnesium intake was inversely associated with incidence of diabetes in women. Potential favorable effects of magnesium on insulin and glucose control may explain our findings.
Abbreviations
- FFQ:
-
Food frequency questionnaire
References
Pham PC, Pham PM, Pham SV, Miller JM, Pham PT (2007) Hypomagnesemia in patients with type 2 diabetes. Clin J Am Soc Nephrol 2:366–373. doi:10.2215/CJN.02960906
Barbagallo M, Dominguez LJ (2007) Magnesium metabolism in type 2 diabetes mellitus, metabolic syndrome and insulin resistance. Arch Biochem Biophys 458:40–47. doi:10.1016/j.abb.2006.05.007
Balon TW, Gu JL, Tokuyama Y, Jasman AP, Nadler JL (1995) Magnesium supplementation reduces development of diabetes in a rat model of spontaneous NIDDM. Am J Physiol 269:E745–E752
Mather HM, Nisbet JA, Burton GH, Poston GJ, Bland JM, Bailey PA, Pilkington TR (1979) Hypomagnesaemia in diabetes. Clin Chim Acta 95:235–242. doi:10.1016/0009-8981(79)90364-4
Ma J, Folsom AR, Melnick SL, Eckfeldt JH, Sharrett AR, Nabulsi AA, Hutchinson RG, Metcalf PA (1995) Associations of serum and dietary magnesium with cardiovascular disease, hypertension, diabetes, insulin, and carotid arterial wall thickness: the ARIC study. Atherosclerosis Risk in Communities Study. J Clin Epidemiol 48:927–940. doi:10.1016/0895-4356(94)00200-A
Song Y, Manson JE, Buring JE, Liu S (2004) Dietary magnesium intake in relation to plasma insulin levels and risk of type 2 diabetes in women. Diabetes Care 27:59–65. doi:10.2337/diacare.27.1.59
Paolisso G, Barbagallo M (1997) Hypertension, diabetes mellitus, and insulin resistance: the role of intracellular magnesium. Am J Hypertens 10:346–355. doi:10.1016/S0895-7061(96)00342-1
Lordes Lima M, Cruz T, Pousada JC, Rodrigues LE, Barbosa K, Canguçu V (1998) The effect of magnesium supplementation in increasing doses on the control of type 2 diabetes. Diabetes Care 21:682–686. doi:10.2337/diacare.21.5.682
The Diabetes Control and Complications Trial Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329:977–986. doi:10.1056/NEJM199309303291401
Nadler JL, Buchanan T, Natarajan R, Antonipillai I, Bergman R, Rude R (1993) Magnesium deficiency produces insulin resistance and increased thromboxane synthesis. Hypertension 21:1024–1029. doi:10.1161/01.HYP.21.6.1024
Altura BM, Altura BT, Gebrewold A, Ising H, Günther T (1984) Magnesium deficiency and hypertension: correlation between magnesium-deficient diets and microcirculatory changes in situ. Science 223:1315–1317
Saris NE, Mervaala E, Karppanen H, Khawaja JA, Lewenstam A (2000) Magnesium. An update on physiological, clinical and analytical aspects. Clin Chim Acta 294:1–26. doi:10.1016/S0009-8981(99)00258-2
Paolisso G, Sgambato S, Gambardella A, Pizza G, Tesauro P, Varricchio M, D’Onofrio F (1992) Daily magnesium supplements improve glucose handling in elderly subjects. Am J Clin Nutr 55:1161–1167
Kao WH, Folsom AR, Nieto FJ, Mo JP, Watson RL, Brancati FL (1999) Serum and dietary magnesium and the risk for type 2 diabetes mellitus: the Atherosclerosis Risk in Communities Study. Arch Intern Med 159:2151–2159. doi:10.1001/archinte.159.18.2151
Salmerón J, Manson JE, Stampfer MJ, Colditz GA, Wing AL, Willett WC (1997) Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. JAMA 277:472–477. doi:10.1001/jama.1997.03540300040031
Lopez-Ridaura R, Willett WC, Rimm EB, Liu S, Stampfer MJ, Manson JE, Hu FB (2004) Magnesium intake and risk of type 2 diabetes in men and women. Diabetes Care 27:134–140. doi:10.2337/diacare.27.1.134
Salmerón J, Ascherio A, Rimm EB, Colditz GA, Spiegelman D, Jenkins DJ, Stampfer MJ, Wing AL, Willett WC (1997) Dietary fiber, glycemic load, and risk of NIDDM in men. Diabetes Care 20:545–550. doi:10.2337/diacare.20.4.545
Meyer KA, Kushi LH, Jacobs DR Jr, Slavin J, Sellers TA, Folsom AR (2000) Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am J Clin Nutr 71:921–930
Møller JB, Dalla Man C, Overgaard RV, Ingwersen SH, Tornøe CW, Pedersen M, Tanaka H, Ohsugi M, Ueki K, Lynge J, Vasconcelos NM, Pedersen BK et al (2014) Ethnic differences in insulin sensitivity, β-cell function, and hepatic extraction between Japanese and Caucasians: a minimal model analysis. J Clin Endocrinol Metab 99:4273–4280. doi:10.1210/jc.2014-1724
Fukushima M, Suzuki H, Seino Y (2004) Insulin secretion capacity in the development from normal glucose tolerance to type 2 diabetes. Diabetes Res Clin Pract 66:S37–S43. doi:10.1016/j.diabres.2003.11.024
Nanri A, Mizoue T, Noda M, Takahashi Y, Kirii K, Inoue M, Tsugane S (2010) Magnesium intake and type II diabetes in Japanese men and women: the Japan Public Health Center-based Prospective Study. Eur J Clin Nutr 64:1244–1247. doi:10.1038/ejcn.2010.138
Hata A, Doi Y, Ninomiya T, Mukai N, Hirakawa Y, Hata J, Ozawa M, Uchida K, Shirota T, Kitazono T et al (2013) Magnesium intake decreases type 2 diabetes risk through the improvement of insulin resistance and inflammation: the Hisayama Study. Diabet Med 30:1487–1494. doi:10.1111/dme.12250
Kirii K, Iso H, Date C, Fukui M, Tamakoshi A (2010) Magnesium intake and risk of self-reported type 2 diabetes among Japanese. J Am Coll Nutr 29:99–106. doi:10.1080/07315724.2010.10719822
Shimizu H (1996) A basic report on Takayama Study. Department of Public Health, Gifu University School of Medicine, Gifu
Shimizu H, Ohwaki A, Kurisu Y, Takatsuka N, Ido M, Kawakami N, Nagata C, Inaba S (1999) Validity and reproducibility of a quantitative food frequency questionnaire for a cohort study in Japan. Jpn J Clin Oncol 29:38–44. doi:10.1093/jjco/29.1.38
Shimizu H (2002) A supplementary comment on “reliability and validity of a questionnaire for assessment of physical activity in epidemiological studies” published in journal of epidemiology, 1998. J Epidemiol 12:54
Suzuki I, Kawakami N, Shimizu H (1998) Reliability and validity of a questionnaire for assessment of energy expenditure and physical activity in epidemiological studies. J Epidemiol 8:152–159
Nagata C, Nakamura K, Fujii K, Kawachi T, Takatsuka N, Oba S, Shimizu H (2008) Smoking and risk of cedar pollinosis in Japanese men and women. Int Arch Allergy Immunol 147:117–124. doi:10.1159/000135698
Oba S, Nagata C, Nakamura K, Fujii K, Kawachi T, Takatsuka N, Shimizu H (2010) Consumption of coffee, green tea, oolong tea, black tea, chocolate snacks and the caffeine content in relation to risk of diabetes in Japanese men and women. Br J Nutr 103:453–459. doi:10.1017/S0007114509991966
Seino Y, Nanjo K, Tajima N, Kadowaki T, Kashiwagi A, Araki E, Ito C, Inagaki N, Iwamoto Y, Kasuga M et al (2010) Report of the committee on the classification and diagnostic criteria of diabetes mellitus. J Diabetes Investig 1:212–228. doi:10.1111/j.2040-1124.2010.00074.x
Nagata C, Nakamura K, Wada K, Tsuji M, Tamai Y, Kawachi T (2013) Branched-chain amino acid intake and the risk of diabetes in a Japanese community: the Takayama study. Am J Epidemiol 178:1226–1232. doi:10.1093/aje/kwt112
Willett WC (2013) Implication of total energy intake for epidemiological analyses. In: Willett WC (ed) Nutritional epidemiology, 3rd edn. Oxford University Press, New York, pp 260–286
Jenkins DJ, Kendall CW, Augustin LS, Franceschi S, Hamidi M, Marchie A, Jenkins AL, Axelsen M (2002) Glycemic index: overview of implications in health and disease. Am J Clin Nutr 76:266S–273S
Zhou BF, Stamler J, Dennis B, Moag-Stahlberg A, Okuda N, Robertson C, Zhao L, Chan Q, Elliott P (2003) Nutrient intakes of middle-aged men and women in China, Japan, United Kingdom, and United States in the late 1990s: the INTERMAP study. J Hum Hypertens 17:623–630. doi:10.1038/sj.jhh.1001605
Suárez A, Pulido N, Casla A, Casanova B, Arrieta FJ, Rovira A (1995) Impaired tyrosine-kinase activity of muscle insulin receptors from hypomagnesaemic rats. Diabetologia 38:1262–1270
Sheehan JP (1991–1992) Magnesium deficiency and diabetes mellitus. Magnes Trace Elem 10:215–219
Ryschon TW, Rosenstein DL, Rubinow DR, Niemela JE, Elin RJ, Balaban RS (1996) Relationship between skeletal muscle intracellular ionized magnesium and measurements of blood magnesium. J Lab Clin Med 127:207–213. doi:10.1016/S0022-2143(96)90080-3
Takaya J, Higashino H, Kobayashi Y (2004) Intracellular magnesium and insulin resistance. Magnes Res 17:126–136
Wada K, Yatsuya H, Ouyang P, Otsuka R, Mitsuhashi H, Takefuji S, Matsushita K, Sugiura K, Hotta Y, Toyoshima H et al (2009) Self-reported medical history was generally accurate among Japanese workplace population. J Clin Epidemiol 62:306–313. doi:10.1016/j.jclinepi.2008.04.006
Waki K, Noda M, Sasaki S, Matsumura Y, Takahashi Y, Isogawa A, Ohashi Y, Kadowaki T, Tsugane S (2005) Alcohol consumption and other risk factors for self-reported diabetes among middle-aged Japanese: a population-based prospective study in the JPHC study cohort I. Diabet Med 22:323–331. doi:10.1111/j.1464-5491.2004.01403.x
Schneider AL, Pankow JS, Heiss G, Selvin E (2012) Validity and reliability of self-reported diabetes in the atherosclerosis risk in communities study. Am J Epidemiol 176:738–743. doi:10.1093/aje/kws156
Unoki H, Takahashi A, Kawaguchi T, Hara K, Horikoshi M, Andersen G, Ng DP, Holmkvist J, Borch-Johnsen K, Jørgensen T et al (2008) SNPs in KCNQ1 are associated with susceptibility to type 2 diabetes in East Asian and European populations. Nat Genet 40:1098–1102. doi:10.1038/ng.208
Okamoto K, Iwasaki N, Nishimura C, Doi K, Noiri E, Nakamura S, Takizawa M, Ogata M, Fujimaki R, Grarup N et al (2010) Identification of KCNJ15 as a susceptibility gene in Asian patients with type 2 diabetes mellitus. Am J Hum Genet 86:54–64. doi:10.1016/j.ajhg.2009.12.009
Imamura M, Maeda S, Yamauchi T, Hara K, Yasuda K, Morizono T, Takahashi A, Horikoshi M, Nakamura M, Fujita H et al (2012) A single-nucleotide polymorphism in ANK1 is associated with susceptibility to type 2 diabetes in Japanese populations. Hum Mol Genet 21:3042–3049. doi:10.1093/hmg/dds113
Acknowledgments
Our study has been financially supported by the National Cancer Center Research and Development Fund and the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The manuscript does not contain clinical studies or patient data.
Rights and permissions
About this article
Cite this article
Konishi, K., Wada, K., Tamura, T. et al. Dietary magnesium intake and the risk of diabetes in the Japanese community: results from the Takayama study. Eur J Nutr 56, 767–774 (2017). https://doi.org/10.1007/s00394-015-1122-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-015-1122-8