Abstract
Objective
In this study, we aimed to assess the safety and efficacy of Janus kinase (JAK) inhibitors in patients with ankylosing spondylitis (AS).
Methods
We conducted a Bayesian network meta-analysis using direct and indirect data from randomized controlled trials (RCTs), and examined the safety and efficacy of JAK inhibitors in active AS patients exhibiting inadequate response or intolerance to two or more non-steroidal anti-inflammatory drugs (NSAIDs).
Results
RCTs included a total of 406 patients (203 experimental subjects and 203 controls) from three studies on upadacitinib, filgotinib, and tofacitinib. Assessment of SpondyloArthritis International Society 20% improvement (ASAS20), ASAS40, and ASAS5/6 responses were significantly higher in the JAK inhibitor group than in the placebo group. Other efficacy outcomes, such as ASAS partial remission, Bath Ankylosing Spondylitis Disease Activity Index (BASDAI50), Ankylosing Spondylitis Disease Activity Score (ASDAS), Spondyloarthritis Research Consortium of Canada (SPARCC) Magnetic Resonance Imaging (MRI) scores, and Bath Ankylosing Spondylitis Functional Index (BASFI) were also significantly higher in the JAK inhibitor group compared to the placebo group. The JAK inhibitors significantly improved disease activity (ASAS partial remission, BASDAI50, ASDAS), function (BASFI), and MRI outcomes (SPARCC MRI spine). However, the incidence of adverse events (AEs) and serious adverse events (SAEs), and the rate of withdrawal attributed to AEs did not differ between the JAK inhibitor and placebo groups.
Conclusion
JAK inhibitors were effective in active AS patients exhibiting an inadequate response or intolerance to two or more NSAIDs, without the risk of SAEs; this suggests that based on our data, studies are warranted to further investigate the use of JAK inhibitors for treating AS.
Zusammenfassung
Ziel
In der vorliegenden Studie war es das Ziel, die Sicherheit und Wirksamkeit von Januskinase(JAK)-Inhibitoren bei Patienten mit ankylosierender Spondylitis (AS) zu untersuchen.
Methoden
Dazu führten die Autoren eine Bayes-Netzwerk-Metaanalyse durch, für die direkte und indirekte Daten aus randomisierten kontrollierten Studien (RCT) verwendet wurden, und untersuchten die Sicherheit und Wirksamkeit von JAK-Inhibitoren bei Patienten mit aktiver AS, die ein unzureichendes Ansprechen oder eine Intoleranz auf 2 oder mehr nichtsteroidale Antiphlogistika (NSAID) zeigten.
Ergebnisse
Die RCT umfassten 406 Patienten (203 Versuchsteilnehmer und 203 Kontrollen) aus 3 Studien zu Upadacitinib, Filgotinib und Tofacitinib. Das Ansprechen gemäß Assessment of SpondyloArthritis International Society mit 20 % Verbesserung (ASAS20), ebenso ASAS40 und ASAS5/6, war in der JAK-Inhibitor-Gruppe signifikant höher als in der Placebogruppe. Auch andere Ergebnisse in Bezug auf die Wirksamkeit, wie eine ASAS-Teilremission, Bath Ankylosing Spondylitis Disease Activity Index (BASDAI50), Ankylosing Spondylitis Disease Activity Score (ASDAS), Scores in der Magnetresonanztomographie (MRT) gemäß Spondyloarthritis Research Consortium of Canada (SPARCC) und Bath Ankylosing Spondylitis Functional Index (BASFI), waren in der JAK-Inhibitor-Gruppe signifikant höher als in der Placebogruppe. Die JAK-Inhibitoren führten zu einer signifikant verbesserten Krankheitsaktivität (ASAS-Teilremission, BASDAI50, ASDAS), Funktion (BASFI) und MRT-Ergebnissen (SPARCC-Scores für MRT der Wirbelsäule). Jedoch unterschieden sich die Inzidenz von Nebenwirkungen (AE) und schweren AE (SAE) sowie die Rate derer, die aufgrund von AE aus der Studie ausschieden, nicht zwischen der JAK-Inhibitor- und der Placebogruppe.
Schlussfolgerung
JAK-Inhibitoren waren – ohne das Risiko von SAE – bei Patienten mit aktiver AS wirksam, die ein unzureichendes Ansprechen auf 2 oder mehr NSAID oder eine entsprechende Intoleranz aufwiesen; demzufolge sind auf Basis der vorgestellten Daten Studien gerechtfertigt, in denen die Anwendung von JAK-Inhibitoren zur Therapie der AS weiter untersucht wird.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory disease, which is accompanied by enthesis and inflammation of the spinal and sacroiliac joints, and eventually induces bone and joint erosion. Additionally, it progressively contributes to new bone growth, syndesmophytes, and ankylosis, thereby resulting in increased structural damage, fatigue, and decreased quality of life (QOL) [1,2,3]. Non-steroidal anti-inflammatory drugs (NSAIDs) are prescribed as first-line pharmacological treatment for AS. Biological disease-modifying antirheumatic drugs (DMARDs), i.e., anti-tumor necrosis factor (TNF) therapies and secukinumab, an interleukin-17A (IL-17A) inhibitor, are prescribed for treating AS patients who do not respond well to NSAIDs [4]. However, the absence or failure of response to current treatments remains a major concern in some patients. In about 40% of patients with AS, anti-TNF treatment either fails to achieve sufficient disease management or induces undesirable side effects [5]. There is also an unmet requirement for elucidating alternate mechanisms of action to effectively manage AS treatment.
Several cytokines, including those involved in the IL-23/IL-17 axis, signal through the Janus kinase (JAK) family of tyrosine kinases. The JAK pathway is thus a potential therapeutic target in AS. Tofacitinib, an oral inhibitor of JAK [6], selectively inhibits JAK1, JAK2, and JAK3, and exhibits a specificity for JAK1 and JAK3 over JAK2. Upadacitinib was developed to confer higher selectivity for JAK1 than for JAK2, JAK3, and Tyk2 [7]. Likewise, filgotinib, an inhibitor of JAK1, was developed to confer higher selectivity for JAK1 than for others.
Several clinical studies have been conducted to determine the effectiveness and safety of JAK inhibitors in active AS patients exhibiting insufficient response or intolerance toward two or more NSAIDs [8,9,10]. In this study, we aimed to improve the precision and accuracy of the effectiveness and safety estimates of JAK inhibitors in active AS patients exhibiting an inadequate response or intolerance to two or more NSAIDs, through meta-analysis [11,12,13] of the results of randomized clinical trials (RCTs). The findings of such a strategy would encourage the regular evaluation of the publicly available data.
Materials and methods
Identification of eligible studies and data extraction
We conducted an extensive search for RCTs investigating the use of JAK inhibitors for the treatment of AS. We initially performed a literature search using MEDLINE, EMBASE, and the Cochrane Controlled Trials Registry to identify eligible publications (until October 2020) to be included in this study. To perform the searches, the following search terms were used: “ankylosing spondylitis” and “JAK inhibitor.” All references in the selected research articles were further verified to find relevant studies that were not included in the online repositories. The following criteria were used for selecting the RCTs: (1) comparison of JAK inhibitors with placebo for the treatment of active AS patients exhibiting inadequate response or intolerance to two or more NSAIDs, and (2) reporting of the clinical effectiveness and safety endpoints of JAK inhibitors with placebo at 12–14 weeks. The following criteria were used for exclusion: (1) duplicate data and (2) lack of data needed for inclusion. The efficacy endpoints included the following: Assessment of SpondyloArthritis International Society 20% improvement (ASAS20) response rate; ASAS40 response rate; ASAS5/6; ASAS partial remission; Ankylosing Spondylitis Disease Activity Score (ASDAS) with C‑reactive protein (CRP) major response (improvement ≥2.0) and clinical improvement (≥1.1); proportion of patients with clinically relevant improvement (decrease of ASDAS from baseline ≥1 · 1), major improvement (decrease of ASDAS from baseline ≥2 · 0), or inactive disease (ASDAS <1 · 3), ASDAS low disease activity (defined as less than 2 · 1); BASDAI50; Bath AS Functional Index (BASFI); change from baseline in Spondyloarthritis Research Consortium of Canada (SPARCC) score of SI joints and spine (six most severely affected discovertebral units). The safety outcomes were analyzed based on the following parameters: the number of patients experiencing adverse events (AEs), the number of patients experiencing serious adverse events (SAEs), and the number of withdrawn patients owing to adverse events. The following details were collected from each report: first author, year of publication, dosage of JAK inhibitor, number of patients treated with JAK inhibitor with placebo, and safety and efficacy results at 12–14 weeks post JAK inhibitor administration. We assessed the methodological quality of the selected studies using the Jadad score [14]. The Jadad score measures random assignment, blinding, and patient withdrawal and dropout rates, and it varies from zero to five. Quality was categorized as high (a score of 3–5) or low (a score of 0–2). We performed a meta-analysis in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [15].
Evaluation of statistical associations
The effect size of the study outcomes was represented as odds ratio (OR) for dichotomous data or standardized mean difference (SMD) for continuous data and the corresponding 95% confidence intervals (95% CIs). We tested the differences and heterogeneities within and between the samples using Cochran’s Q‑statistics [16]. The heterogeneity method was applied to measure the null hypothesis, which stated that both experiments measured the same effect. If relevant Q‑statistics (p < 0.10) indicated variance among the analyses, the random impact model was used to perform the meta-analysis; otherwise, the fixed impact model was used. The fixed impact model suggests that all experiments estimate the same underlying effect and recognize only differences within the sample. We quantified the effect of heterogeneity using the following equation: I2 = 100% × (Q − df) /Q [17], where I2 assesses the level of inconsistency between the studies and determines whether the percentage of the total variation across the studies is due to the heterogeneity rather than by chance. I2 varies from 1% to 100%; however, I2 values of 25%, 50%, and 75% are referred to as low, moderate, and high values, respectively. Statistical analyses were performed using the Applied Meta-Analysis Software System (Biosta, Englewood, NJ, USA).
Evaluation of publication bias
Funnel plots are usually generated to detect bias in publications. However, as funnel plots require a large number of studies with varying sizes and individual decisions, we assessed publication bias using Egger’s linear regression test [18], which tests funnel plot asymmetry using a normal logarithm OR scale.
Results
Studies selected to perform the meta-analysis
Initially, 370 articles were identified using online and manual searches, of which 10 were chosen for full-text review based on their title and abstract. However, 7 of the 10 articles were excluded as they contained redundant data, non-RCT data, or no outcome data. Therefore, only three RCTs met the inclusion criteria for performing the meta-analysis [8,9,10]. These three selected studies included a total of 406 patients (203 experimental subjects and 203 controls), and included the investigation of upadacitinib, filgotinib, and tofacitinib. The JAK inhibitors were used at the following dosages: upadacitinib 15 mg once daily, filgotinib 200 mg once daily, and tofacitinib 5 mg twice daily. The Jadad score across all the studies ranged from 3 to 4, thereby indicating high quality. All patients underwent standard therapy, and the related aspects of the trials used in the meta-analysis are listed in Table 1.
Meta-analysis to assess the efficacy of JAK inhibitor for treating AS
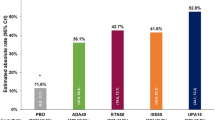
The ASAS20 response was significantly higher in the JAK inhibitor group than in the placebo group (OR = 3.762, 95% CI 2.474–5.721, p < 0.001; Table 2; Fig. 1). ASAS40 and ASAS5/6 responses were also substantially higher in the JAK inhibitor group than in the placebo group (Table 2; Fig. 1). Other efficacy outcomes in the JAK inhibitor group, such as ASAS partial remission, BASDAI50, ASDAS, SPARCC MRI scores, and BASFI, were also significantly higher in the placebo group (Table 2; Fig. 1).
Meta-analysis of the a ASAS20, b ASAS40, and c ASDAS clinically important improvement response rates for JAK inhibitors versus placebo treatments in patients with AS. ASAS20 Assessment of SpondyloArthritis International Society 20% improvement, ASAS40 Assessment of SpondyloArthritis International Society 40% improvement, ASDAS Ankylosing Spondylitis Disease Activity Score, JAK Janus kinase
Meta-analysis to assess the safety of JAK inhibitors for treating AS
The incidence of AEs did not differ between the JAK inhibitor and placebo groups (Table 2; Fig. 2). Additionally, no differences were identified in SAEs and withdrawal owing to the difference in AEs between the JAK inhibitor and placebo groups (Table 2; Fig. 2). There was a non-serious deep vein thrombosis in the calf of a man aged 53 years who had a heterozygous factor V Leiden mutation, diagnosed 3 days after the patient’s last dose of filgotinib. However, there was no further thromboembolic event in upadacitinib and tofacitinib RCTs. Herpes zoster infection was not reported in any of the three RCTs.
Heterogeneity and publication bias
Heterogeneity between the studies was not found in most of the meta-analyses assessing the safety and efficacy of JAK inhibitors, excluding the ASDAS major response, SPARCC spine score, SPARCC SI joint score, and BASFI. The cause of heterogeneity was identified to be the difference in the magnitude of the effect size, and not its direction. It was difficult to compare the funnel map, which is typically used to identify the reporting bias, as the number of studies included was very limited. However, no evidence of publication bias was detected (Egger’s regression test p-values >0.1).
Discussion
In this meta-analysis, we extracted data from three RCTs that compared JAK inhibitors to placebo in AS. All efficacy results, including ASAS20, ASAS40, and ASAS5/6 responses, were substantially higher in the JAK inhibitor group than in the placebo group. In comparison, there was little disparity between the frequency of AEs and SAEs and the AE withdrawal rate between the JAK inhibitor and the placebo groups. JAK inhibitors reached the endpoints demonstrating substantial improvements in disease activity (ASAS partial remission, BASDAI50, ASDAS), work (BASFI), and MRI (SPARCC MRI spine) outcomes. JAK inhibitors decreased disease activity and symptoms and signs more significantly than placebo in patients with active AS who did not respond to NSAIDs, and were well tolerated. Two studies are phase II studies compared to one phase II/III study. Tofacitinib and filgotinib were evaluated in phase II studies, while upadacitinib was assessed in a phase II/III study, which is ongoing to evaluate long-term safety and efficacy of upadacitinib treatment in AS. These data with JAK inhibitors in AS are very promising and indicate that JAK inhibitors could be used as a potential therapeutic alternative for AS.
Currently, the therapeutic regimes for patients with AS who do not respond well to NSAIDs are restricted to TNF inhibitors and secukinumab [4]. AS patients with allergic responses to IL-17A and/or TNF inhibitor cannot undergo these treatments [19] and the use of IL-17 antagonists is not recommended in patients with active inflammatory bowel disease [20]. Considering these unmet needs, our data suggest that JAK inhibitors can serve as an effective and safe treatment regimen for patients with active AS who do not respond well to NSAIDs; these findings also encourage further study on the use of JAK inhibitors for managing AS.
The JAK-STAT cascade regulates the proliferation and cytokine network associated with different T‑cell subpopulations, such as Th17 cells and the IL-23/IL-17 cytokine axis [21]. As the IL-23/IL-17 cytokine axis plays a vital role in the pathogenesis of AS, JAK inhibitors are predicted to have therapeutic potential for AS. Inhibition of TNF‑α has been shown to be an efficient strategy in the management of AS. However, the IL-23/IL-17 cytokine is not specifically inhibited by JAK inhibitors, but it has been shown that the selective inhibition of JAK can result in secondary inhibition of additional pathways that do not rely on JAK1 signaling [22].
There are certain limitations to be considered in the present meta-analysis. First, the number of experiments involved was limited and the possibilities for sampling mistakes and publishing prejudices cannot be excluded. While we did not find publication bias, it should be noted that publishing bias is difficult to eliminate with full confidence, particularly when the number of studies involved is limited, and only three RCTs were included in our analysis. Second, long-term findings have not been included in this meta-analysis. The follow-up time of the included trials was 14 weeks, and therefore, follow-up studies conducted for a longer duration are expected to be included in the future analyses. Third, variation in clinical characteristics, such as ethnicity, sex, age, and AS severity, complicates the results of meta-analysis and could have influenced our analysis.
Conversely, this meta-analysis study also has several strengths, as it is up to date and included all the existing information. The number of AS patients in each sample varied from 103 to 187, but the combined analysis comprised 406 patients. Compared to the individual research papers, we provided more reliable evidence by increasing the statistical power and resolution through combining the findings of independent analyzes [23]. This is—to the best of our knowledge—the first meta-analysis assessing the efficacy and safety of JAK inhibitors in treating patients with active AS despite failure of NSAID treatment. Our meta-analysis has provided detailed reliable evidence across all accessible RCTs regarding the use of JAK inhibitors in AS with statistical significance and addresses the variability of the outcomes of independent analyzes across current literature [24,25,26]. Therefore, these might be the best available data in this field [24].
Conclusively, upon performing this meta-analysis, we found that treatment with JAK inhibitor was successful in patients with active AS who demonstrated an insufficient response or intolerance toward two or more NSAIDs and JAK inhibitor therapy. While the long-term efficacy and safety of JAK inhibitors needs to be assessed, they were found to exhibit potential therapeutic effects against AS. Further long-term research is required in this field to better assess the safety and efficacy of JAK inhibitors for treating AS.
References
Sieper J, Rudwaleit M, Khan MA, Braun J (2006) Concepts and epidemiology of spondyloarthritis. Best Pract Res Clin Rheumatol 20:401–417
Lee YH, Song GG (2018) Overall and sex-specific mortality in psoriatic arthritis and ankylosing spondylitis: a meta-analysis. J Rheum Dis 25(3):197–202
Koo BS, Lim JW, Shin JH, Kim T‑H (2018) Characteristics of uveitis in patients with ankylosing spondylitis in Korea: a single-center survey. J Rheum Dis 25:28–33
van der Heijde D, Ramiro S, Landewé R, Baraliakos X, Van den Bosch F, Sepriano A, Regel A, Ciurea A, Dagfinrud H, Dougados M (2017) 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann Rheum Dis 76:978–991
Baeten D, Baraliakos X, Braun J et al (2013) Anti-interleukin-17A monoclonal antibody secukinumab in treatment of ankylosing spondylitis: a randomised, double-blind, placebo-controlled trial. Lancet 382:1705–1713
Changelian PS, Flanagan ME, Ball DJ et al (2003) Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302:875–878
Nakase T, Wada H, Minamikawa K et al (1994) Increased activated protein C‑protein C inhibitor complex level in patients positive for lupus anticoagulant. Blood Coagul Fibrinolysis 5:173–177
van der Heijde D, Song I‑H, Pangan AL, Deodhar A, Van den Bosch F, Maksymowych WP, Kim T‑H, Kishimoto M, Everding A, Sui Y (2019) Efficacy and safety of upadacitinib in patients with active ankylosing spondylitis (SELECT-AXIS 1): a multicentre, randomised, double-blind, placebo-controlled, phase 2/3 trial. Lancet 394:2108–2117
van der Heijde D, Baraliakos X, Gensler LS, Maksymowych WP, Tseluyko V, Nadashkevich O, Abi-Saab W, Tasset C, Meuleners L, Besuyen R (2018) Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active ankylosing spondylitis (TORTUGA): results from a randomised, placebo-controlled, phase 2 trial. Lancet 392:2378–2387
van der Heijde D, Deodhar A, Wei JC, Drescher E, Fleishaker D, Hendrikx T, Li D, Menon S, Kanik KS (2017) Tofacitinib in patients with ankylosing spondylitis: a phase II, 16-week, randomised, placebo-controlled, dose-ranging study. Ann Rheum Dis 76:1340–1347
Lee YH (2015) Meta-analysis of genetic association studies. Ann Lab Med 35:283–287
Lee YH, Song GG (2019) YKL-40 levels in rheumatoid arthritis and their correlation with disease activity: a meta-analysis. J Rheum Dis 26:257–263
Lee YH, Song GG (2020) Associations between circulating interleukin-17 levels and systemic lupus erythematosus and between Interleukin-17 gene polymorphisms and disease susceptibility: a meta-analysis. J Rheum Dis 27:37–44
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ (1996) Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control Clin Trials 17:1–12
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151:264–269
Davey Smith G, Egger M (1997) Meta-analyses of randomised controlled trials. Lancet 350:1182
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Burmester GR, Kremer JM, Van den Bosch F, Kivitz A, Bessette L, Li Y, Zhou Y, Othman AA, Pangan AL, Camp HS (2018) Safety and efficacy of upadacitinib in patients with rheumatoid arthritis and inadequate response to conventional synthetic disease-modifying anti-rheumatic drugs (SELECT-NEXT): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 391:2503–2512
Fleischmann R, Pangan AL, Song IH, Mysler E, Bessette L, Peterfy C, Durez P, Ostor AJ, Li Y, Zhou Y (2019) Upadacitinib versus placebo or adalimumab in patients with rheumatoid arthritis and an inadequate response to methotrexate: results of a phase III, double-blind, randomized controlled trial. Arthritis Rheumatol 71:1788–1800
Seif F, Khoshmirsafa M, Aazami H, Mohsenzadegan M, Sedighi G, Bahar M (2017) The role of JAK-STAT signaling pathway and its regulators in the fate of T helper cells. Cell Commun Signal 15:23
Sornasse T, Sokolove J, McInnes I (2019) THU0181 treatment with upadacitinib results in the normalization of key pathobiologic pathways in patients with rheumatoid arthritis: biomarker results from the phase 3 select-next and select-beyond studies. Ann Rheum Dis 78:365–366
Lee YH (2018) An overview of meta-analysis for clinicians. Korean J Intern Med 33:277
Lee Y‑H, Bae S‑C, Song G‑G (2012) Omega‑3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: a meta-analysis. Arch Med Res 43:356–362
Lee YH, Bae S‑C, Choi SJ, Ji JD, Song GG (2012) Associations between TNFAIP3 gene polymorphisms and rheumatoid arthritis: a meta-analysis. Inflamm Res 61:635–641
Song GG, Lee YH (2019) Causal association between bone mineral density and osteoarthritis: a Mendelian randomization study. J Rheum Dis 26:104–110
Anderson JJ, Baron G, van der Heijde D, Felson DT, Dougados M (2001) Ankylosing spondylitis assessment group preliminary definition of short-term improvement in ankylosing spondylitis. Arthritis Rheum 44:1876–1886
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Y. H. Lee and G. G. Song declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Additional information
Redaktion
U. Müller-Ladner, Bad Nauheim
U. Lange, Bad Nauheim
Rights and permissions
About this article
Cite this article
Lee, Y.H., Song, G.G. Janus kinase inhibitors for treating active ankylosing spondylitis: a meta-analysis of randomized controlled trials. Z Rheumatol 81, 71–76 (2022). https://doi.org/10.1007/s00393-020-00948-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-020-00948-3