Abstract
Objectives
The relative efficacy and safety of tofacitinib and baricitinib were assessed in patients with rheumatoid arthritis (RA) with an inadequate response to disease-modifying anti-rheumatic drugs (DMARDs) or biologics.
Methods
We performed a Bayesian network meta-analysis to combine direct and indirect evidence from randomized controlled trials (RCTs) to examine the efficacy and safety of tofacitinib and baricitinib in combination with DMARDs in RA patients with an inadequate DMARD or biologic response.
Results
Twelve RCTs including 5883 patients met the inclusion criteria. There were 15 pairwise comparisons including 10 direct comparisons of 6 interventions. Tofacitinib 10 mg + methotrexate (MTX) and baricitinib 4 mg + MTX were among the most effective treatments for active RA with an inadequate DMARD or biologic response, followed by baricitinib 2 mg + MTX, tofacitinib 5 mg + MTX, and adalimumab + MTX. The ranking probability based on the surface under the cumulative ranking curve (SUCRA) indicated that tofacitinib 10 mg + MTX had the highest probability of being the best treatment to achieve the ACR20 response rate (SUCRA = 0.865), followed by baricitinib 4 mg + MTX (SUCRA = 0.774), baricitinib 2 mg + MTX (SUCRA = 0.552), tofacitinib 5 mg + MTX (SUCRA = 0.512), adalimumab + MTX (SUCRA = 0.297), and placebo + MTX (SUCRA <0.001). No significant differences were observed in the incidence of serious adverse events after treatment with tofacitinib + MTX, baricitinib + MTX, adalimumab + MTX, or placebo + MTX.
Conclusions
In RA patients with an inadequate response to DMARDs or biologics, tofacitinib 10 mg + MTX and baricitinib 4 mg + MTX were the most efficacious interventions and were not associated with a significant risk of serious adverse events.
Zusammenfassung
Ziel
Bei Patienten mit rheumatoider Arthritis (RA) und unzureichendem Ansprechen auf krankheitsmodifizierende Medikamente („disease-modifying anti-rheumatic drugs“, DMARD) oder Biologika wurden die relative Wirksamkeit und Sicherheit von Tofacitinib und Baricitinib ermittelt.
Methoden
Die Autoren führten eine Bayes-Netz-Metaanalyse zur Kombination direkter und indirekter Evidenz aus randomisierten kontrollierten Studien („randomized controlled trials“, RCT) durch, die der Untersuchung der Wirksamkeit und Sicherheit von Tofacitinib und Baricitinib zusätzlich zu DMARD bei RA-Patienten mit unzureichendem Ansprechen auf DMARD oder Biologika diente.
Ergebnisse
Die Einschlusskriterien erfüllten 12 RCT mit 5883 Patienten. Es wurden 15 paarweise erfolgende Vergleiche einschließlich 10 direkter Vergleiche von 6 Interventionen durchgeführt. Tofacitinib 10 mg + Methotrexat (MTX) und Baricitinib 4 mg + MTX gehörten zu den wirksamsten Therapien bei aktiver RA mit unzureichendem Ansprechen auf DMARD oder Biologika, nächstwirksam waren Baricitinib 2 mg + MTX, Tofacitinib 5 mg + MTX und Adalimumab + MTX. Die auf dem SUCRA-Wert („surface under the cumulative ranking curve“) basierende Rangfolgewahrscheinlichkeit ergab für Tofacitinib 10 mg + MTX die größte Wahrscheinlichkeit, die beste Behandlung zur Erzielung einer Ansprechrate mit 20%iger Linderung der Symptome (ACR20) zu sein (SUCRA = 0,865); es folgten Baricitinib 4 mg + MTX (SUCRA = 0,774), Baricitinib 2 mg + MTX (SUCRA = 0,552), Tofacitinib 5 mg + MTX (SUCRA = 0,512), Adalimumab + MTX (SUCRA = 0,297) und Placebo + MTX (SUCRA <0,001). Bei der Inzidenz schwerer unerwünschter Ereignisse nach Behandlung mit Tofacitinib + MTX, Baricitinib + MTX, Adalimumab + MTX oder Placebo + MTX wurden keine signifikanten Unterschiede festgestellt.
Schlussfolgerung
Bei RA-Patienten mit unzureichendem Ansprechen auf DMARD oder Biologika stellten Tofacitinib 10 mg + MTX und Baricitinib 4 mg + MTX die wirksamsten Interventionen dar, sie waren dabei nicht mit einem signifikanten Risiko für schwere unerwünschte Ereignisse verbunden.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disease characterized by chronic synovial joint inflammation, which leads to disability and a decreased quality of life [38]. The intracellular pathways, which include the Janus kinases (JAKs—JAK1, JAK2, JAK3, and tyrosine kinase 2 [Tyk2]), are critical to immune cell activation, proinflammatory cytokine production, and cytokine signaling [14]. Proinflammatory cytokines—including the interleukins 2, 6, 12, 15, and 23, interferons, and granulocyte-macrophage colony-stimulating factor (GM-CSF), which act through the JAK family—are implicated in RA [36]. Small-molecule JAK inhibitors are being clinically developed for the treatment of RA [34].
Tofacitinib (CP-690,550) is an orally administered JAK inhibitor [6]. It selectively inhibits JAK-1, JAK-2, and JAK-3, with functional cellular specificity for JAK-1 and JAK-3 over JAK-2 [7, 30]. Tofacitinib effectively modulates adaptive and innate immunity [30]. Baricitinib is a potent, selective JAK1 and JAK2 inhibitor [37]. Baricitinib shows similar inhibitory activity against both JAK1 and JAK2, but decreased activity against JAK3 and tyrosine kinase 2 [15].
Several clinical trials have attempted to evaluate the efficacy and safety of tofacitinib and baricitinib in patients with active RA with an incomplete response to methotrexate (MTX) or biologics [3, 9, 13, 16, 20, 21, 23, 39,40,41, 44, 45]. All of these drugs have shown considerable efficacy in placebo-controlled trials, but the relative efficacy and safety of tofacitinib and baricitinib remain unclear due to a lack of head-to-head comparisons. In the absence of head-to-head trials of relevant comparators, it is necessary to combine evidence from randomized controlled trials (RCTs) of different treatments to derive an estimate of the effect of one treatment versus another. A network meta-analysis can assess the comparative efficacy of multiple interventions by combining evidence across a network of RCTs, even in the absence of head-to-head comparisons [4, 5], in contrast to a traditional meta-analysis [26, 28]. The present study aimed to use a network meta-analysis to investigate the relative efficacy and safety of tofacitinib and baricitinib in patients with active RA and an inadequate response to disease-modifying anti-rheumatic drugs (DMARDs) or biologics.
Materials and methods
Identification of eligible studies and data extraction
We conducted an exhaustive search for studies that examined the efficacy and safety of tofacitinib and baricitinib in patients with active RA who showed an inadequate response to DMARDs including MTX or biologics. A literature search was performed using MEDLINE, EMBASE, the Cochrane Controlled Trials Register, and the American College of Rheumatology (ACR) and European League against Rheumatism (EULAR) conference proceedings to identify available articles (up to April 2018). The following key words and subject terms were used in the search: “tofacitinib”, “baricitinib”, and “rheumatoid arthritis”. All references in the studies were reviewed to identify additional works not included in the electronic databases. RCTs were included if they met the following criteria: (1) the study compared tofacitinib or baricitinib with DMARDs including MTX to placebo + DMARDs including MTX for the treatment of active RA that responded inadequately to DMARDs or biologics; (2) the study provided endpoints for the clinical efficacy and safety of tofacitinib or baricitinib at 3 or 6 months; and (3) the study included patients diagnosed with RA based on the ACR criteria for RA [18] or the 2010 ACR/EULAR classification criteria [1]. The exclusion criteria were as follows: (1) the study included duplicate data; and (2) the study did not contain adequate data for inclusion. The efficacy outcome was the number of patients who fulfilled the ACR 20% improvement criteria (achieved an ACR20 response), and the safety outcome was the number of patients who experienced serious adverse events (SAEs). The following information was extracted from each study: first author, year of publication, country in which the study was conducted, dosages of tofacitinib and baricitinib, follow-up time when outcomes were evaluated, and efficacy and safety outcomes. Data were extracted from original studies by 2 independent reviewers. Any discrepancy between the reviewers was resolved by consensus or a third reviewer. We quantified the methodological quality of studies using Jadad scores [19] ranging from 0 to 5. Quality was classified as high (a score of 3–5) or low (a score of 0–2). We conducted this network meta-analysis in accordance with the guidelines provided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [31].
Evaluation of statistical associations for network meta-analysis
In RCTs that compared multiple doses of tofacitinib and baricitinib in different arms, the results from the different arms were analyzed simultaneously. The efficacy and safety of tofacitinib and baricitinib in different arms were ordered according to the probability of being ranked as the best performing regimen. We performed a Bayesian random-effects network meta-analysis using NetMetaXL [2] and WinBUGS statistical analysis program version 1.4.3 (MRC Biostatistics Unit, Institute of Public Health, Cambridge, UK). The Bayesian approach offers greater flexibility in the use of more complex models and different outcome types, thereby enabling the simultaneous comparison of all treatment options. We chose a random-effects model for the network meta-analysis, as it incorporates between-study variations and utilizes a conservative method. The random network model was selected prior to the statistical analysis. We used the Markov chain Monte Carlo method to obtain pooled effect sizes [4]. All chains were run with 10,000 burn-in iterations followed by 10,000 monitoring iterations. Information on relative effects was converted to a probability that a treatment is the best, second best, etc., or to the ranking of each treatment, called the surface under the cumulative ranking curve (SUCRA; [35]), which is expressed as a percentage. The SUCRA value is 1 when a treatment is certain to be the best and 0 when a treatment is certain to be the worst. SUCRA values enable the overall ranking of treatments for a particular outcome. SUCRA simplifies the information on the effect of each treatment into a single number, thereby facilitating decision-making. The league table arranges the presentation of summary estimates by ranking the treatments in the order of the most pronounced impact on the outcome under consideration, based on the SUCRA value [35]. We reported the pairwise odds ratio (OR) and 95% credible interval (CrI; or Bayesian confidence interval) and adjusted for multiple-arm trials. Pooled results were considered statistically significant if the 95% CrI did not contain the value 1.
Inconsistency assessment
Inconsistency refers to the extent of disagreement between direct and indirect evidence [8], and assessments of inconsistency are important when conducting a network meta-analysis because an inconsistency plot yields information that can help identify the loops in which the inconsistency is present [17]. We plotted the posterior mean deviance of the individual data points in the inconsistency model against the posterior mean deviance in the consistency model to assess the network inconsistency between the direct and indirect estimates in each loop [43]. A sensitivity test was performed by comparing the random and fixed effects models.
Results
Studies included in the meta-analysis
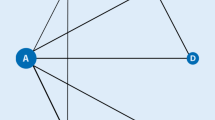
A total of 588 studies were identified through electronic or manual searches, and 17 studies were selected for a full-text review based on the title and abstract details. However, 5 of the 17 studies were excluded due to inclusion of DMARD-naïve RA patients [12, 25], monotherapy [10, 11], or a short-term follow-up period [22]. Thus, 12 RCTs that included 5883 patients (2964 events for efficacy and 206 events for safety) met the inclusion criteria [3, 9, 13, 16, 20, 21, 23, 39,40,41, 44, 45]. All of the RCTs provided data related to both efficacy and safety, except for 2 that showed only efficacy data for the tofacitinib and baricitinib groups [16, 23]. The evidence network diagram shows data pertaining to the number of studies that compared different treatments and the number of patients included in each treatment (Fig. 1, Table 1 and 2). While the recommended dosage of tofacitinib is 5 mg twice daily [42], some patients may benefit from an increased dose of 10 mg twice daily. Thus, we chose the dosages of 5 and 10 mg of tofacitinib twice daily. For phase II and III RCTs, the recommended dosage of baricitinib is 2 mg or 4 mg once daily. Therefore, we chose the dosages of 2 and 4 mg of baricitinib once daily. There were 15 pairwise comparisons including 10 direct comparisons and 6 interventions, including tofacitinib 5 mg + MTX, tofacitinib 10 mg + MTX, baricitinib 2 mg + MTX, baricitinib 4 mg + MTX, adalimumab 40 mg once every 2 weeks + MTX, and placebo + MTX for the network meta-analysis. The Jadad scores of the studies ranged from 3–5, indicating high study quality overall (Table 1 and 2). Relevant features of the studies included in the meta-analysis are provided in Table 1 and 2.
Evidence network diagram of network meta-analysis comparisons. The width of each edge is proportional to the number of randomized controlled trials comparing each pair of treatments, and the size of each treatment node is proportional to the number of randomized participants (sample size). A: Placebo + MTX, B: Tofacitinib 5 mg + MTX, C: Tofacitinib 10 mg + MTX, D: Adalimumab + MTX, E: Baricitinib 2 mg + MTX, F Baricitinib 4 mg + MTX
Network meta-analysis of the efficacy of tofacitinib and baricitinib in RCTs
Tofacitinib 10 mg + MTX is listed in the top left of the diagonal of the league table (OR, 3.58; 95% CrI, 2.69–5.12), because it was associated with the most favorable SUCRA for the ACR20 response rate, whereas placebo + MTX is listed in the bottom right of the diagonal of the league table because it was associated with the least favorable results (Fig. 2). All of the interventions achieved a significant ACR20 response compared with placebo + MTX (Fig. 2). A trend of greater efficacy with tofacitinib 10 mg + MTX, baricitinib 4 mg + MTX, baricitinib 2 mg + MTX, and tofacitinib 5 mg + MTX than with adalimumab + MTX was noted (Figs. 2 and 3). The ranking probability based on SUCRA indicated that tofacitinib 10 mg + MTX had the highest probability of being the best treatment in terms of the ACR20 response rate (SUCRA = 0.865), followed by baricitinib 4 mg + MTX (SUCRA = 0.774), baricitinib 2 mg + MTX (SUCRA = 0.552), tofacitinib 5 mg + MTX (SUCRA = 0.512), adalimumab + MTX (SUCRA = 0.297), and placebo + MTX (SUCRA <0.001; Table 3).
League tables showing the results of the network meta-analysis comparing the effects of all drugs including odds ratios and 95% credible intervals. a Efficacy: Odds ratio >1 indicates that the top-left treatment is better. b Tolerability: Odds ratio <1 indicates that the top-left treatment is better
Network meta-analysis of the safety of tofacitinib and baricitinib in RCTs
The number of SAEs in the adalimumab + MTX, baricitinib 2 mg + MTX, placebo + MTX, and baricitinib 4 mg + MTX groups tended to be lower than that in the tofacitinib 10 mg + MTX and tofacitinib 5 mg + MTX groups (Figs. 2 and 3). However, the number of SAEs did not differ significantly between the tofacitinib and baricitinib groups (Figs. 2 and 3). Ranking probability based on SUCRA values indicated that adalimumab + MTX, baricitinib 2 mg + MTX, and placebo + MTX had a higher probability of being the safest treatment (SUCRA = 0.877, 0.782, 0.475, respectively), followed by baricitinib 4 mg + MTX (SUCRA = 0.474), tofacitinib 10 mg + MTX (SUCRA = 0.208), and tofacitinib 5 mg + MTX (SUCRA = 0.184; Table 3).
Inconsistency and sensitivity analysis
The contributions to the deviance were likely to be similar and close to 1 for both models. Inconsistency plots assessing network inconsistencies between direct and indirect estimates showed a low possibility of inconsistencies that might significantly affect the results of the network meta-analysis (Fig. 4). In addition, the results of the random- and fixed-effects models yielded the same interpretation, indicating that the results of this network meta-analysis were robust (Fig. 3).
Discussion
Drugs that inhibit pathways may directly block cytokine signaling or indirectly modulate T‑cell functions through the suppression of the CD80/86 expression in the dendritic cells [24, 32]. Recent studies on the treatment of RA have focused on the small molecules that can inhibit intracellular kinases (such as those from the JAK family; [34]). Treatment trends have evolved to include the increasing use of new small molecules to target the JAK pathways.
Since patients with RA may receive tofacitinib or baricitinib when they are either refractory or intolerant to MTX or biologics or when they are contraindicated, it is important to determine the optimal treatment options for these patients. In addition to efficacy, the safety of tofacitinib and baricitinib is an important factor in selecting the therapeutic approach in patients with RA. We conducted a network meta-analysis to compare the efficacy and safety of tofacitinib and baricitinib in patients with active RA who show an inadequate response to DMARDs including MTX or biologics. With regard to efficacy, our network meta-analysis suggested that tofacitinib 10 mg + MTX and baricitinib 4 mg + MTX were the most effective treatments for active RA that responded inadequately to DMARDs or biologics, followed by baricitinib 2 mg + MTX, tofacitinib 5 mg + MTX, and adalimumab + MTX. Tofacitinib + MTX and baricitinib + MTX were more likely to be the best for achieving an ACR20 response, as compared to adalimumab + MTX. Although the reason for this finding was not identified, it was suggested to be differences in efficacies between JAK inhibitors and adalimumab. In terms of safety based on the number of SAEs, tofacitinib 10 mg + MTX and tofacitinib 5 mg + MTX were associated with more SAEs and had a lower probability of being optimal in terms of SAEs than adalimumab + MTX, baricitinib 2 mg + MTX, placebo + MTX, and baricitinib 4 mg + MTX. However, no significant difference was observed in the number of SAEs among the 6 interventions, suggesting comparable safety among the different tofacitinib and baricitinib regimens and the placebo.
The results of this network meta-analysis, which combined evidence from both direct and indirect comparisons for evaluation of the relative efficacy and safety of baricitinib, were in accordance with those of previous meta-analyses of direct comparisons showing that treatment with tofacitinib and baricitinib led to a statistically significant improvement according to the response criteria (ACR20) compared to placebo and that there were no statistically significant differences between tofacitinib and placebo in terms of SAEs [27, 29]. However, our network meta-analysis differs from previous meta-analyses in that we were able to generate a rank order for the relative efficacy and safety of tofacitinib and baricitinib in patients with active RA.
However, our results should be interpreted with caution, as this study had some limitations. First, the follow-up time points were limited to 3 or 6 months. Therefore, the follow-up duration was too short to evaluate the long-term effects, and longer comparative studies are needed. Second, the design and patient characteristics of the included trials were heterogeneous; therefore, there is a risk that the differences across the studies affected the results of the analysis. Third, this study did not comprehensively address the efficacy and safety outcomes of tofacitinib and baricitinib in RA patients. It focused solely on the effectiveness based on the number of patients that achieved an ACR20 response and on the safety according to the number of SAEs, without an assessment of various other outcomes [33].
In conclusion, we conducted a Bayesian network meta-analysis involving 12 RCTs and found that tofacitinib 10 mg + MTX and baricitinib 4 mg + MTX were the most efficacious interventions for RA patients with an inadequate response to DMARD or biologics therapy and that neither was associated with a significant risk of SAEs. Long-term studies are warranted to determine the relative efficacy and safety of tofacitinib and baricitinib in a large number of patients with active RA that is inadequately responsive to MTX or biologics.
References
Aletaha D, Landewe R, Karonitsch T et al (2008) Reporting disease activity in clinical trials of patients with rheumatoid arthritis: EULAR/ACR collaborative recommendations. Arthritis Care Res 59(10):1371–1377
Brown S, Hutton B, Clifford T et al (2014) A Microsoft-Excel-based tool for running and critically appraising network meta-analyses—an overview and application of NetMetaXL. Syst Rev 3(1):110
Burmester GR, Benda B, Gruben D et al (2013) Tofacitinib for rheumatoid arthritis—Authors’ reply. Lancet 381(9880):1812–1813
Caldwell DM, Ades A, Higgins J (2005) Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ 331(7521):897
Catalá-López F, Tobías A, Cameron C et al (2014) Network meta-analysis for comparing treatment effects of multiple interventions: an introduction. Rheumatol Int 34(11):1489–1496
Changelian PS, Flanagan ME, Ball DJ et al (2003) Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302(5646):875–878
Chrencik JE, Patny A, Leung IK et al (2010) Structural and thermodynamic characterization of the TYK2 and JAK3 kinase domains in complex with CP-690550 and CMP-6. J Mol Biol 400(3):413–433
Dias S, Welton NJ, Sutton AJ et al (2013) Evidence synthesis for decision making 4 inconsistency in networks of evidence based on randomized controlled trials. Med Decis Making 33(5):641–656
Dougados M, van der Heijde D, Chen Y‑C et al (2015) LB0001 Baricitinib, an oral Janus Kinase (JAK) 1/JAK2 inhibitor, in patients with active Rheumatoid Arthritis (RA) and an inadequate response to CDMARD therapy: results of the phase 3 RA-build study. Ann Rheum Dis 74(Suppl 2):79–79
Fleischmann R, Cutolo M, Genovese MC et al (2012) Phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) or adalimumab monotherapy versus placebo in patients with active rheumatoid arthritis with an inadequate response to disease-modifying antirheumatic drugs. Arthritis Rheum 64(3):617–629
Fleischmann R, Kremer J, Cush J et al (2012) Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med 367(6):495–507
Fleischmann R, Takeuchi T, Schlichting DE et al (2015) Baricitinib, methotrexate, or baricitinib plus methotrexate in patients with early rheumatoid arthritis who had received limited or no treatment with disease-modifying anti-rheumatic drugs (DMARDs): phase 3 trial results. In: Arthritis & Rheumatology. Wiley-Blackwell, Hoboken
Genovese MC, Kremer J, Zamani O et al (2016) Baricitinib in patients with refractory rheumatoid arthritis. N Engl J Med 374(13):1243–1252
Ghoreschi K, Laurence A, O’Shea JJ (2009) Janus kinases in immune cell signaling. Immunol Rev 228(1):273–287
Ghoreschi K, Laurence A, O’Shea JJ (2009) Janus kinases in immune cell signaling. Immunol Rev 228(1):273–287
Greenwald M, Fidelus-Gort R, Levy R et al (2010) A randomized dose-ranging, placebo-controlled study of INCB028050, a selective JAK1 and JAK2 inhibitor in subjects with active rheumatoid arthritis. Arthritis Rheum 62(Suppl 10):2172
Higgins J, Jackson D, Barrett J et al (2012) Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Res Synth Methods 3(2):98–110
Hochberg MC, Chang RW, Dwosh I et al (1992) The American College of Rheumatology 1991 revised criteria for the classification of global functional status in rheumatoid arthritis. Arthritis Rheum 35(5):498–502
Jadad AR, Moore RA, Carroll D et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17(1):1–12
Keystone EC, Taylor PC, Drescher E et al (2015) Safety and efficacy of baricitinib at 24 weeks in patients with rheumatoid arthritis who have had an inadequate response to methotrexate. Ann Rheum Dis 74(2):333–340
Kremer J, Li ZG, Hall S et al (2013) Tofacitinib in combination with nonbiologic disease-modifying antirheumatic drugs in patients with active rheumatoid arthritis: a randomized trial. Ann Intern Med 159(4):253–261
Kremer JM, Bloom BJ, Breedveld FC et al (2009) The safety and efficacy of a JAK inhibitor in patients with active rheumatoid arthritis: results of a double-blind, placebo-controlled phase IIa trial of three dosage levels of CP-690,550 versus placebo. Arthritis Rheum 60(7):1895–1905
Kremer JM, Cohen S, Wilkinson BE et al (2012) A phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) versus placebo in combination with background methotrexate in patients with active rheumatoid arthritis and an inadequate response to methotrexate alone. Arthritis Rheum 64(4):970–981
Kubo S, Yamaoka K, Kondo M et al (2013) The JAK inhibitor, tofacitinib, reduces the T cell stimulatory capacity of human monocyte-derived dendritic cells. Ann Rheum Dis 73:2192–2198
Lee EB, Fleischmann R, Hall S et al (2014) Tofacitinib versus methotrexate in rheumatoid arthritis. N Engl J Med 370(25):2377–2386
Lee Y‑H, Bae S‑C, Song G‑G (2012) Omega-3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: a meta-analysis. Arch Med Res 43(5):356–362
Lee Y, Bae S‑C (2018) Comparative efficacy and safety of baricitinib 2 mg and 4 mg in patients with active rheumatoid arthritis. Z Rheumatol 77(4):335–342
Lee Y, Bae S, Song G (2013) Hepatitis B virus (HBV) reactivation in rheumatic patients with hepatitis core antigen (HBV occult carriers) undergoing anti-tumor necrosis factor therapy
Lee YH, Bae S‑C, Song GG (2015) Comparative efficacy and safety of tofacitinib, with or without methotrexate, in patients with active rheumatoid arthritis: a Bayesian network meta-analysis of randomized controlled trials. Rheumatol Int 35(12):1965–1974
Meyer DM, Jesson MI, Li X et al (2010) Anti-inflammatory activity and neutrophil reductions mediated by the JAK1/JAK3 inhibitor, CP-690,550, in rat adjuvant-induced arthritis. J Inflamm (Lond) 7:41
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269
Norman P (2014) Selective JAK inhibitors in development for rheumatoid arthritis. Expert Opin Investig Drugs 23(8):1067–1077
Park Y‑W, Kim K‑J, Yang H‑I et al (2017) Comparing effectiveness rituximab (Mabthera®) to other second-line biologics for rheumatoid arthritis treatment in patients refractory to or intolerant of first-line anti-tumor necrosis factor agent: an observational study. J Rheum Dis 24(4):227–235
Roskoski R Jr. (2016) Janus kinase (JAK) inhibitors in the treatment of inflammatory and neoplastic diseases. Pharmacol Res 111:784–803
Salanti G, Ades A, Ioannidis JP (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64(2):163–171
Schindler C, Levy DE, Decker T (2007) JAK-STAT signaling: from interferons to cytokines. J Biol Chem 282(28):20059–20063
Shi JG, Chen X, Lee F et al (2014) The pharmacokinetics, pharmacodynamics, and safety of baricitinib, an oral JAK 1/2 inhibitor, in healthy volunteers. J Clin Pharmacol 54(12):1354–1361
Smolen JS, Aletaha D, McInnes IB (2016) Rheumatoid arthritis. Lancet 388(10055):2023–2038. https://doi.org/10.1016/S0140-6736(16)30173-8
Tanaka Y, Emoto K, Cai Z et al (2016) Efficacy and safety of baricitinib in Japanese patients with active rheumatoid arthritis receiving background methotrexate therapy: a 12-week, double-blind, randomized placebo-controlled study. J Rheumatol 43(3):504–511
Tanaka Y, Suzuki M, Nakamura H et al (2011) Phase II study of tofacitinib (CP-690,550) combined with methotrexate in patients with rheumatoid arthritis and an inadequate response to methotrexate. Arthritis Care Res 63(8):1150–1158
Taylor PC, Keystone EC, Van Der Heijde D et al (2015) Baricitinib versus placebo or adalimumab in patients with active rheumatoid arthritis (RA) and an inadequate response to background methotrexate therapy: results of a phase 3 study. In: Arthritis & rheuamtology. Wiley-Blackwell, Hoboken
Traynor K (2012) FDA approves tofacitinib for rheumatoid arthritis. Am J Health Syst Pharm 69(24):2120
Valkenhoef G, Lu G, Brock B et al (2012) Automating network meta-analysis. Res Synth Methods 3(4):285–299
van der Heijde D, Tanaka Y, Fleischmann R et al (2013) Tofacitinib (CP-690,550) in patients with rheumatoid arthritis receiving methotrexate: twelve-month data from a twenty-four-month phase III randomized radiographic study. Arthritis Rheum 65(3):559–570
van Vollenhoven RF, Fleischmann R, Cohen S et al (2012) Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N Engl J Med 367(6):508–519
Acknowledgements
This study was supported in part by a grant of the Korea Healthcare technology R&D Project, Ministry for Health and Welfare, Republic of Korea (HI15C2958).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.-C. Bae and Y. H. Lee have no financial or non-financial conflict of interest to declare.
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Redaktion
U. Müller-Ladner, Bad Nauheim
U. Lange, Bad Nauheim
Rights and permissions
About this article
Cite this article
Bae, SC., Lee, Y.H. Comparison of the efficacy and safety of tofacitinib and baricitinib in patients with active rheumatoid arthritis: a Bayesian network meta-analysis of randomized controlled trials. Z Rheumatol 78, 559–567 (2019). https://doi.org/10.1007/s00393-018-0531-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-018-0531-5