Abstract
Objective
Osteoprotegerin (OPG) is considered an important biomarker in cardiovascular (CV) disease. CV disease is the most common cause of mortality in patients with rheumatoid arthritis (RA), a consequence of accelerated atherosclerosis. The present study aimed to evaluate the relationship of serum OPG levels to arterial stiffness, carotid intima-media thickness (CIMT), and clinical and laboratory indices in RA patients.
Patients and methods
Included in the study were 68 RA patients with no history or signs of CV disease and 48 healthy subjects Disease activity was assessed by the 28-joint disease activity score (DAS28) in RA patients. Serum OPG level was measured using enzyme-linked immunosorbent assay (ELISA). Carotid femoral pulse wave velocity (PWV) was measured as an index of arterial stiffness and CIMT was evaluated by carotid ultrasonography.
Results
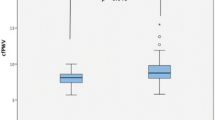
The mean serum OPG level was significantly higher in RA patients than controls (p < 0.001). Mean PWV and CIMT were also significantly increased in RA patients compared to controls (both p < 0.001). In RA patients, serum OPG level was significantly correlated with PWV and CIMT, as well as rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP) antibody; but not with DAS28, high-sensitivity C-reactive protein (hsCRP), or erythrocyte sedimentation rate.
Conclusion
Serum OPG levels were increased and correlated with CIMT and PWV in RA patients. In addition to PWV and CIMT, OPG may be a useful biomarker for CV risk management in RA patients.
Zusammenfassung
Ziel
Osteoprotegerin (OPG) gilt als relevanter Biomarker für kardiovaskuläre Erkrankungen. Als Konsequenz der bei rheumatoider Arthritis (RA) beschleunigt verlaufenden Atherosklerose ist eine kardiovaskuläre Erkrankung die häufigste Ursache für Mortalität bei RA-Patienten. Evaluiert wurden mögliche Beziehungen zwischen OPG-Konzentrationen, Gefäßsteifigkeit, Wanddicke der A. carotis (CIMT) sowie klinischen und laborchemischen Indizes bei RA.
Patienten und Methoden
Eingeschlossen in die Studie wurden 68 RA-Patienten ohne Anamnese bzw. Symptome einer kardiovaskulären Erkrankung und 48 gesunde Individuen. Bei den RA-Patienten wurde die Erkrankungsaktivität mit dem DAS28 („28-joint disease activity score2) errechnet. Die Serum-OPG-Konzentrationen wurden mit einem ELISA („enzyme-linked immunosorbent assay“) gemessen. Die Carotis-Femoralis-Pulswellengeschwindigkeit (cfPWV) wurde als Index für die Gefäßsteifigkeit bestimmt, und die CIMT wurde sonographisch ermittelt.
Ergebnisse
Die durchschnittlichen Serum-OPG-Konzentrationen waren bei den RA-Patienten signifikant höher als im Kontrollkollektiv (p < 0,001), ebenso die durchschnittlichen Werte für PWV und CIMT (jeweils p < 0,001). Bei den RA-Patienten bestanden zwischen Serum-OPG-Konzentrationen, PWV, CIMT sowie RF(Rheumafaktor)- und Anti-CCP(Antikörper gegen zyklisch citrulliniertes Peptid)-Konzentrationen signifikante Korrelationen, nicht dagegen mit DAS28, hsCRP (hoch sensitives C-reaktives Protein) und Erythrozytensedimentierungsgeschwindigkeit.
Schlussfolgerung
Bei den RA-Patienten waren die Serum-OPG-Konzentrationen erhöht und korrelierten mit CIMT und PWV. Neben PWV und CIMT kann auch OPG als Biomarker für das kardiale Risikomanagement bei RA hilfreich sein.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Rheumatoid arthritis (RA) is one of the most prevalent inflammatory diseases in the general population. While the most important pathogenic lesion is inflammatory synovitis, extra-articular features are equally important, considering the fact that inflammation has a systemic magnitude [1]. Mortality is higher among RA patients compared to the general population and the majority of premature deaths are attributable to cardiovascular (CV) disease [2]. CV morbidity is also enhanced, with an increased prevalence of all stages of atherogenesis—from endothelial dysfunction to increased thickness and plaque in carotid arteries, and ultimately to fatal and nonfatal myocardial infarction and stroke [3, 4, 5, 6, 7, 8]. Classic risk factors for CV diseases in the general population—such as hypertension, dyslipidemia, diabetes, and smoking—also appear to be important in RA, but only account in part for the excess CV risk [9]. Chronic inflammation plays an important role in the pathogenesis of both atherosclerosis and RA and accentuates established CV risk factors [2].
Carotid intima-media thickness (CIMT) of the common carotid artery is a useful noninvasive anatomical structural measure of subclinical CV disease and the most popular technique used to study early structural changes in the arterial wall [10]. Increased CIMT has been reported in RA patients compared to controls [11, 12]. Large artery stiffness is known to increase in patients with atherosclerosis and is both a surrogate marker and an independent risk factor for atherosclerosis [13]. In chronic inflammatory diseases, elevated proinflammatory cytokines may contribute to the increased risk for CV disease by promoting vascular changes and increased arterial stiffness. Arterial stiffness can be assessed noninvasively by measurement of pulse wave velocity (PWV), a simple and reproducible method [14].
Osteoprotegerin (OPG) is a member of the tumor necrosis factor (TNF) family and is known to inhibit osteoclastogenesis [15]. Clinical studies have shown an association between OPG and vascular atherosclerotic diseases. OPG was also independently associated with coronary artery calcification in RA patients with long-standing disease [16]. To our knowledge, there is no available study reporting on the relationship between OPG and PWV in RA patients without CV disease or risk factors. Therefore, the aim of this study was to evaluate OPG levels in RA patients compared to healthy controls and to assess their correlation with arterial stiffness measured by carotid femoral PWV, CIMT, and other laboratory and disease activity indices.
Materials and methods
Patients
For this cross-sectional study, 68 consecutive patients (age 48.4 ± 8.1 years; 56 women and 12 men) who fulfilled the 2010 American College of Rheumatology (ACR)/European League against Rheumatism (EULAR) classification criteria [17] for RA were recruited. All patients were taking conventional synthetic disease-modifying antirheumatic drugs (DMARD) including methotrexate, sulfasalazine, leflunomide, and hydroxychloroquine either as monotherapy or in combination, and 11 patients also were treated with biologics. Patients with known prior CV disease (coronary angina, myocardial infarction, cerebral ischemic stroke, or peripheral vascular disease), hypertension, diabetes mellitus, or hyperlipidemia were excluded from the study. As normal controls, 48 age- and gender-matched healthy volunteers (age 46.6 ± 6.2 years; 37 women and 11 men), who had no rheumatic or CV disease, were included. Written informed consent was obtained from all participants prior to the study. The study protocol was approved by the local Ethics Committee of our institution and performed in accordance with the principles stated in the Declaration of Helsinki.
Clinical assessment
Demographics of all participants were recorded. Disease activity was assessed by the 28-joint disease activity score (DAS28) in RA patients [18]. Pain intensity was evaluated using a 100-mm horizontal visual analog scale (VAS). Erythrocyte sedimentation rate (ESR) was recorded. Based on DAS28, the patients were subdivided into three subgroups: mild (DAS28 ≤ 3.2), moderate (3.2 < DAS28 ≤ 5.1), and severe (DAS28 > 5.1).
A single experienced cardiologist, who was blind to patient clinical data, performed vascular assessments for both the patients and controls. Reported blood pressures were all obtained in the morning after an overnight fast and at the same visit as patients’ vascular assessment. Resting blood pressures were measured in the seated position using an automated sphygmomanometer.
Laboratory analysis
Overnight fasting blood samples were obtained from all subjects for determination of glucose, total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and triglyceride (TG) levels. Glucose and lipid profile were determined by standard methods.
Anti-cyclic citrullinated peptide (anti-CCP) antibody was measured by enzyme-linked immunosorbent assay (ELISA), using a commercial kit according to the manufacturer’s instructions. A result was considered positive for anti-CCP antibodies if the titer was above 15 IU/ml. Serum levels of rheumatoid factor (RF)-IgM were measured by the nephelometric method, and a result was considered positive for RF when its concentration was above 20 IU/ml. Serum high-sensitivity C-reactive protein (hsCRP) level was measured with an immunoturbidimetric method using an Abbott auto-analyzer (Architect C1600; Abbott, USA). The normal hsCRP range was defined as ≤ 0.5 mg/dl.
Determination of serum osteoprotegerin
Blood samples from patients and controls were collected after an overnight fast. Serum was obtained after centrifugation and stored at − 70 ℃ until analysis. The concentration of OPG was measured using a commercially available human OPG ELISA kit (eBioscience, Vienna, Austria) according to the instructions provided by the manufacturer. Absorbance was measured at a wavelength of 450 nm using an ELISA reader. The levels of OPG were recorded in pg/ml. The sensitivity of the OPG assay was 2.5 pg/ml.
Assessment of carotid intima-media thickness
Ultrasound imaging examination of the carotid arteries was performed using a high-resolution ultrasonography scanner (VingMed Vivid 3; GE Medical Systems, Horten, Norway) with a 7-MHz linear array transducer. Measurements were performed on the right and left carotid arteries with the subject in the supine position. The region 1 cm proximal to the carotid bifurcation was identified, and the CIMT of the far wall was evaluated as the distance between the lumen–intima interface and the media–adventitia interface. The CIMT measurement was obtained from four contiguous sites at 1-mm intervals on each carotid artery, and the mean value of all eight measurements (in millimeters) was calculated and used for analysis.
Assessment of pulse wave velocity
Carotid femoral PWV was measured as an index of arterial stiffness. Assessments were performed after the subjects had rested for 15 min in the supine position in a quiet room. The carotid femoral PWV was calculated from the measurements of pulse transmission time and the distance between the two recording sites by a validated noninvasive device (SphygmoCor, AtCor Medical, Sydney, Australia). The distances from the sternal notch to the right carotid artery and from the sternal notch to the right femoral artery were measured. The distance traveled by the pulse wave over the surface of the body, as determined with a tape measure, was divided by the transit time; the carotid femoral PWV was then calculated automatically in meters/second (m/s).
Statistical analysis
All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS), version 13.0, for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables are presented as the mean ± standard deviation or median (95 % confidence interval, CI). The normality of the distribution for all variables was assessed by the Kolmogorov–Smirnov test. Intergroup comparisons were made using Student’s t-test for normally distributed variables and the Mann–Whitney U test for nonparametric variables. Analyses of evaluated parameters in subgroups with RA were performed using a one-way analysis of variance and the Kruskal–Wallis test. To assess the correlations between variables, Spearman’s rank or Pearson’s correlation analysis were used according to data distribution. Values of p < 0.05 were considered statistically significant.
Results
The demographic and clinical characteristics of patients and controls are shown in Tab. 1. Patients and controls did not significantly differ in terms of age or gender (48.4 ± 8.1 vs. 46.6 ± 6.2 years, p = 0.183; female/male 56/12 vs. 37/11, p = 0.642, respectively), but the body mass index (BMI) was significantly higher in RA patients than controls (p = 0.01). The mean disease duration in RA patients was 5.6 ± 3.9 years.
The comparison of laboratory parameters, PWV, and CIMT between patients and controls is shown in Tab. 2. TG and LDL-C levels did not differ between groups (p > 0.05), whereas TC, HDL-C, blood glucose, and systolic and diastolic blood pressures were significantly higher in RA patients compared to controls (all p < 0.05). Patients with RA demonstrated significantly higher levels of hsCRP and ESR compared to controls (1.2 ± 1.4 vs. 0.2 ± 0.1 mg/dl, p < 0.001; 28.9 ± 17.9 vs. 8.7 ± 4.6 mm/h, p < 0.001, respectively). The mean serum OPG level was significantly higher in RA patients than in controls (116.9 ± 92.7 vs. 57.1 ± 12.2 pg/ml, p < 0.001). The mean PWV and CIMT values were also significantly increased in RA patients compared to controls (8.2 ± 1.9 vs. 6.2 ± 1.2 m/s, p < 0.001; 0.80 ± 0.12 vs. 0.57 ± 0.07 mm, p < 0.001, respectively).
Although the patients with severe disease activity had the highest level of OPG, there was no statistically significant difference in mean serum OPG levels among subgroups with low, mild, and severe disease activity (98.9 ± 81.1, 118.6 ± 85.7, and 148.2 ± 126.6 pg/ml, respectively; p = 0.296); nor did the mean CIMT and PWV values demonstrate significant differences among RA subgroups (CIMT: 0.80 ± 0.14, 0.81 ± 0.11, and 0.78 ± 0.13 mm; p = 0.814; PWV: 8.6 ± 2.3, 7.9 ± 1.5, and 7.9 ± 2 m/s; p = 0.390, respectively).
In RA patients, serum OPG levels were significantly correlated with PWV and CIMT (r = 0.273, p = 0.02; r = 0.335, p = 0.005, respectively; Fig. 1 a, b). Serum OPG levels also demonstrated significant correlations with RF and anti-CCP antibody, but not with DAS28, hsCRP, or ESR (p = 0.007, p = 0.007, p = 0.202, p = 0.179, p = 0.237, respectively; Tab. 3). In addition, CIMT was significantly correlated with hsCRP, PWV, and patient age, but not with ESR or DAS28 (r = 0.263, p = 0.031; r = 0.460, p < 0.001; r = 0.405, p = 0.001; r = 0.192, p = 0.118; r = 0.007, p = 0.954, respectively). While PWV showed no significant correlation with hsCRP, ESR, DAS28, or disease duration (all p > 0.05), it was significantly correlated with patient age (r = 0.336, p = 0.005). Serum lipid profiles were not correlated with any other studied parameters (all p > 0.05; data not shown).
Discussion
There is increasing evidence from controlled clinical trials that patients with RA present more extensive atherosclerosis and coronary calcification than those without [19]. A prospective Dutch study has also demonstrated that the magnitude of CV risk in RA may equal the CV risk in patients with type 2 diabetes [20]. In this study, the CV risk in RA patients with no traditional risk factors was investigated with regard to the association of serum osteoprotegerin level with PWV and CIMT.
CIMT is an important marker for early, preclinical atherosclerosis and a predictor of future CV events [21]. RA is considered an independent risk factor for intima-media thickening of the common carotid and femoral arteries [19, 22]. Additionally, in previously reported studies in RA patients, CIMT demonstrated significant correlations with traditional CV risk factors [23, 24], disease severity [11], and CRP [25] and ESR values [26]. CIMT ≥ 0.60 mm is a common marker of atherosclerosis and the presence of carotid plaques is the best indication of advanced atherosclerosis [10]. In accordance with previous studies, the mean CIMT in this study was higher in RA patients compared to controls (0.80 vs. 0.57 mm). In addition, CIMT demonstrated statistically significant correlations with hsCRP and age in patients with RA, but no correlation with ESR, DAS28, or lipid profile.
Arterial stiffness is an important arterial phenotype and an independent predictor of CV disease [27]. In this study, arterial stiffness was evaluated by carotid femoral PWV, the established gold-standard measure of aortic stiffness, and a significantly greater PWV was observed in patients with RA as compared to controls. Similar results also have been reported in other studies [28, 29, 30]. In contrast, a study by Fan et al. [13] found no significant difference in terms of brachial ankle PWV among groups including patients with RA, inflammatory bowel disease, and healthy subjects. Increased arterial stiffness has also been linked with disease duration [31, 32], disease activity scores [33], patient age, and CRP values [30] in RA patients. However, the latter studies included patients with traditional CV risk factors. In the present study, PWV was only significantly correlated with patient age, and showed no significant correlation with hsCRP, ESR, or DAS28. Similar to our results, PWV was not significantly correlated with disease duration, biochemical parameters of inflammation (ESR, CRP), or DAS28 in a study by Pieringer et al. [29]. As in our study design, RA patients with traditional CV risk factors were excluded from the Pieringer study to eliminate possible confounders. We additionally found significant correlations between PWV and CIMT in patients with RA.
Osteoprotegerin is currently defined as an important biomarker in CV disease. By acting as a soluble decoy receptor competing for receptor activator of nuclear factor κB ligand (RANKL), OPG prevents receptor activator of nuclear factor κB (RANK)–RANKL interactions, and thus osteoclast differentiation and bone resorption. Mechanisms supporting a pro-atherosclerotic role for OPG include its in vitro ability to enhance expression of endothelial cell adhesion molecules and subsequent infiltration of leukocytes and monocytic cells. Furthermore, OPG might contribute to endothelial dysfunction by blocking RANKL signaling, which is able to activate protective intracellular endothelial pathways such as the nitric oxide synthase pathway. OPG also influences the production of other agents important in plaque stability, such as matrix metalloproteinases [34]. A recent systematic update including 14 studies with clearly defined cohorts qualified for review concluded that OPG concentration is associated with the presence and severity of stable coronary artery disease, acute coronary syndrome, and cerebrovascular disease [35].
RA patients with no CV risk factors had higher OPG levels and increased CIMT and PWV values
In the present study, serum OPG concentration was evaluated along with CIMT and carotid femoral PWV in RA patients. Consistent with previous studies, serum OPG levels were significantly higher in RA patients compared to controls. Furthermore, statistically significant, albeit not strong, correlations of serum OPG with PWV, CIMT, and age were also demonstrated. In fact, RA patients with CV disease or risk factors were excluded from this study. In addition, our patients had short disease duration (mean 5.6 years), moderate disease activity (3.73), and relatively low CRP values (1.2 mg/dl). It is known that preclinical atherosclerosis is associated with CV risk factors, disease duration, disease severity, and level of CRP [1]. These factors may explain the weak correlation between serum OPG levels and cardiac parameters. Nevertheless, to our knowledge, this study is the first to report the correlation of OPG with carotid femoral PWV in RA patients. We also observed significant correlations of OPG with RF and anti-CCP antibodies. However, no correlations were identified between serum OPG and disease activity indices such as ESR, hsCRP, and DAS28. All of the patients were on drug treatment, including conventional synthetic or biologic DMARDs. These treatment regimens could have influenced the association of OPG levels with inflammatory markers. A small number of previous studies have evaluated OPG as an indicator of CV disease in RA patients. In a study by Asanuma et al. [16], similar to our results, higher OPG concentrations were observed in patients with early and long-standing RA as compared to control subjects. This study also detected significant correlations of OPG with ESR, hsCRP, DAS28, and disease duration. However, contrary to our study design, the Asanuma study included patients and controls with CV risk factors such as hypertension, smoking, diabetes, and family history of coronary artery disease. Furthermore, the patients had older age, longer disease duration, and higher CRP values than those in our study. A later study also found that serum OPG level was significantly higher in RA patients with carotid plaques than in those without plaques, and independently associated with carotid plaques [36]. In a recent uncontrolled study by Dessein et al. [37] that included 30 patients with severe RA, OPG concentrations were significantly associated with both endothelial activation and carotid atherosclerosis, and this finding was consistent before and after infliximab infusion. This study also included patients with traditional CV risk factors. In addition, similar to our results, there were no associations of OPG with ESR, DAS28, or CRP in their study.
Limitations
This study has some limitations. First, the study design was cross-sectional, and a longitudinal study would have been more conclusive. Second, the number of patients with low and severe disease activity was relatively small, and the mean disease duration was short. Therefore, the results may not be generalizable to all RA patients. Third, it was not possible to evaluate patients before and after treatment with DMARDs and biologics, which might have reflected the significance of effective anti-inflammatory therapy in risk management strategies for CV disease in patients with RA.
Conclusion
This study demonstrated that RA patients with no CV risk factors had higher levels of OPG and increased CIMT and PWV values compared to controls, and detected significant correlations of OPG with CIMT and PWV in this population. However, further follow-up studies involving large samples are required to clarify the diagnostic, predictive, and prognostic value of OPG for the risk management of CV disease in RA patients.
References
Tanasescu C, Jurcut C, Jurcut R et al (2009) Vascular disease in rheumatoid arthritis: from subclinical lesions to cardiovascular risk. Eur J Intern Med 20:348–354
Peters MJ, Symmons DP, McCarey D et al (2010) EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann Rheum Dis 69(2):325–331
Han C, Robinson DWJ, Hackett MV et al (2006) Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J Rheumatol 33:2167–2172
Roman MJ, Devereux RB, Schwartz JE et al (2005) Arterial stiffness in chronic inflammatory diseases. Hypertension 46:194–199
Roman MJ, Moeller E, Davis A, et al (2006) Preclinical carotid atherosclerosis in patients with rheumatoid arthritis. Ann Intern Med 144:249–256
Solomon DH, Karlson EW, Rimm EB et al (2003) Cardiovascular morbidity and mortality in women diagnosed with rheumatoid arthritis. Circulation 107:1303–1307
Turesson C, Jarenros A, Jacobsson L (2004) Increased incidence of cardiovascular disease in patients with rheumatoid arthritis: results from a community based study. Ann Rheum Dis 63:952–955
Vaudo G, Marchesi S, Gerli R et al (2004) Endothelial dysfunction in young patients with rheumatoid arthritis and low disease activity. Ann Rheum Dis 6:31–35
Dessein PH, Norton GR, Woodiwiss AJ et al (2007) Influence of nonclassical cardiovascular risk factors on the accuracy of predicting subclinical atherosclerosis in rheumatoid arthritis. J Rheumatol 34:943–951
Gonzalez-Gay MA, Gonzalez-Juanatey C, Vazquez-Rodriguez TR et al (2008) Endothelial dysfunction, carotid intima-media thickness, and accelerated atherosclerosis in rheumatoid arthritis. Semin Arthritis Rheum 38(2):67–70
Kumeda Y, Inaba M, Goto H et al (2002) Increased thickness of the arterial intima-media detected by ultrasonography in patients with rheumatoid arthritis. Arthritis Rheum 46:1489–1497
Park YB, Ahn CW, Choi HK et al (2002) Atherosclerosis in rheumatoid arthritis: morphologic evidence obtained by carotid ultrasound. Arthritis Rheum 46:1714–1719
Fan F, Galvin A, Fang L et al (2014) Comparison of inflammation, arterial stiffness and traditional cardiovascular risk factors between rheumatoid arthritis and inflammatory bowel disease. J Inflamm 11(1):29
Boutouyrie P, Tropeano AI, Asmar R et al (2002) Aortic stiffness is an independent predictor of primary coronary events in hypertensive patients: a longitudinal study. Hypertension 39(1):10–15
Venuraju SM, Yerramasu A, Corder R et al (2010) Osteoprotegerin as a predictor of coronary artery disease and cardiovascular mortality and morbidity. J Am Coll Cardiol 55(19):2049–2061
Asanuma Y, Chung CP, Oeser A et al (2007) Serum osteoprotegerin is increased and independently associated with coronary-artery atherosclerosis in patients with rheumatoid arthritis. Atherosclerosis 195(2):135–141
Aletaha D, Neogi T, Silman AJ et al (2010) Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581
Prevoo ML, Hof MA van’t, Kuper HH et al (1995) Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 38:44–48
Soeiro Ade M, Haddad M, Almeida MC de et al (2012) Rheumatoid arthritis and cardiovascular disease: what is known about this relationship and what can currently be done for affected patients? Rev Port Cardiol 31(3):225–232
Halm VP van, Peters MJ, Voskuyl AE et al (2009) Rheumatoid arthritis versus type 2 diabetes as a risk factor for cardiovascular disease: a cross-sectional study. Ann Rheum Dis 68:1395–1400
Nurmohamed MT (2009) Cardiovascular risk in rheumatoid arthritis. Autoimmun Rev 8(8):663–667
Stamatelopoulos KS, Kitas GD, Papamichael CM et al (2010) Subclinical peripheral arterial disease in rheumatoid arthritis. Atherosclerosis 212(1):305–309
Carotti M, Salaffi F, Mangiacotti M et al (2007) Atherosclerosis in rheumatoid arthritis: the role of high-resolution B mode ultrasound in the measurement of the arterial intima-media thickness. Reumatismo 59:38–49
Rincón I del, Freeman GL, Haas RW et al (2005) Relative contribution of cardiovascular risk factors and rheumatoid arthritis clinical manifestations to atherosclerosis. Arthritis Rheum 52:3413–3423
Gonzalez-Gay MA, Gonzalez-Juanatey C, Piñeiro A et al (2005) High-grade C-reactive protein elevation correlates with accelerated atherogenesis in patients with rheumatoid arthritis. J Rheumatol 32:1219–1223
Rincón I del, Williams K, Stern MP et al (2003) Association between carotid atherosclerosis and markers of inflammation in rheumatoid arthritis patients and healthy subjects. Arthritis Rheum 48:1833–1840
Jain S, Khera R, Corrales-Medina VF, Townsend RR et al (2014) Inflammation and arterial stiffness in humans. Atherosclerosis 237(2):381–390
Roman MJ, Devereux RB, Schwartz JE et al (2005) Arterial stiffness in chronic inflammatory diseases. Hypertension 46:194–199
Pieringer H, Stuby U, Pohanka E et al (2010) Arterial stifness in a muscular artery in women with longstanding rheumatoid arthritis compared with healthy controls and patients with traditional cardiovascular risk factors. Rheumatol Int 30:1335–1339
Mäki-Petäjä KM, Hall FC, Booth AD et al (2006) Rheumatoid arthritis is associated with increased aortic pulse-wave velocity, which is reduced by anti-tumor necrosis factor-α therapy. Circulation 114:1185–1192
Klocke R, Cockcroft JR, Taylor GJ et al (2003) Arterial stiffness and central blood pressure, as determined by pulse wave analysis, in rheumatoid arthritis. Ann Rheum Dis 62:414–418
Arosio E, De Marchi S, Rigoni A et al (2007) Forearm haemodynamics, arterial stiffness and microcirculatory reactivity in rheumatoid arthritis. J Hypertens 25:1273–1278
Turesson C, Jacobsson L, Rydén Ahlgren A et al (2005) Increased stiffness of the abdominal aorta in women with rheumatoid arthritis. Rheumatology 44:896–901
Van Campenhout A, Golledge J (2009) Osteoprotegerin, vascular calcification and atherosclerosis. Atherosclerosis 204(2):321–329
Hosbond SE, Poulsen TS, Diederichsen AC et al (2012) Osteoprotegerin as a marker of atherosclerosis: a systematic update. Scand Cardiovasc J 46(4):203–211
Asanuma YF, Shimada Y, Kouzu N et al (2013) Serum osteoprotegerin concentration is associated with carotid atherosclerotic plaque in patients with rheumatoid arthritis. Mod Rheumatol 23(2):269–275
Dessein PH, López-Mejias R, González-Juanatey C et al (2014) Independent relationship of osteoprotegerin concentrations with endothelial activation and carotid atherosclerosis in patients with severe rheumatoid arthritis. J Rheumatol 41(3):429–436
Compliance with ethical guidelines
Conflict of interest. M.S. Beyazal, T. Erdoğan, G. Devrimsel, A.K. Türkyılmaz, M.C. Cüre, M. Beyazal, and I. Sahin state that there are no conflicts of interest.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beyazal, M., Erdoğan, T., Devrimsel, G. et al. Relationship of osteoprotegerin to pulse wave velocity and carotid intima-media thickness in rheumatoid arthritis patients. Z Rheumatol 75, 723–728 (2016). https://doi.org/10.1007/s00393-015-1675-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-015-1675-1