Abstract
Purpose
Transanal minimally invasive surgery (TAMIS) is gaining worldwide popularity as an alternative for the transanal endoscopic microsurgery (TEMS) method for the local excision of rectal polyps and selected neoplasms. Data on patient reported outcomes regarding short-term follow-up are scarce; data on functional outcomes for long-term follow-up is non-existent.
Methods
We used the fecal incontinence severity index (FISI) to prospectively assess the fecal continence on the intermediate-term follow-up after TAMIS. The primary outcome measure is postoperative fecal continence. Secondary outcome measures are as follows: perioperative and intermediate-term morbidity.
Results
Forty-two patients (m = 21:f = 21), median age 68.5 (range 34–94) years, were included in the analysis. In four patients (9.5%), postoperative complications occurred. The median follow-up was 36 months (range 24–48). Preoperative mean FISI score was 8.3 points. One year after TAMIS, mean FISI score was 5.4 points (p = 0.501). After 3 years of follow-up, mean FISI score was 10.1 points (p = 0.01). Fecal continence improved in 11 patients (26%). Continence decreased in 20 patients (47.6%) (mean FISI score 15.2 points, [range 3–31]).
Conclusions
This study found that the incidence of impaired fecal continence after TAMIS is substantial; however, the clinical significance of this deterioration seems minor. The present data is helpful in acquiring informed consent and emphasizes the need of proper patient information.
Functional results seem to be comparable to results after TEMS. Furthermore, we confirmed TAMIS is safe and associated with low morbidity.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
The rectum is a challenging area for endoluminal surgery, partly due to limited accessibility. Rectal polyps may be excised by a variety of transanal techniques: the submucosal resection described by Parks and Stuart [1] in 1973 or the parachute technique popularized by Francillon and co-workers in 1974 [2]. These local procedures can however be technically challenging; access is limited and problems are encountered regarding bleeding from unreachable sites in the rectum. Transanal endoscopic microsurgery was first described by Buess et al. in 1983 and is currently the preferred method for local excision of large polyps end selected rectal cancers because of its safety, high incidence of complete resections, and ability to treat polyps throughout the entire rectum as was elegantly shown by the group of de Graaf [3] [4, 5]. This technique facilitates full-thickness local excision of large polyps and selected rectal cancers. However, the specialized instruments for transanal endoscopic microsurgery (TEMS) are expensive and the technique is known to have a long learning curve [6, 7]. The functional results after TEMS are fairly well investigated, yielding acceptable results [8,9,10,11,12]. Transanal minimally invasive surgery (TAMIS) is a relatively new technique that is gaining worldwide popularity among colorectal surgeons, utilizing a disposable flexible port in combination with conventional laparoscopic instruments for the local excision of rectal masses [13, 14]. Although not supported by scientific evidence, it is thought that the technique of TAMIS is more easy to learn [15]. Moreover, no specialized instruments are needed, making this technique more readily available and less expensive. To keep expenses low, some authors even reported the successful use of a “glove” port [16, 17]. While no sphincter lesions have been reported following TAMIS in literature [13, 18], to date, the literature on the short-term functional outcome after TAMIS is limited. Recently, data on the short-time fecal continence after TAMIS were published by our group as well as by the group of Verseveld et al. [19, 20]. We concluded that short-term functional results of transanal minimally invasive surgery were excellent and comparable to functional results using the dedicated transanal endoscopic microsurgery equipment. To the best of our knowledge, no data on fecal continence after longer periods of follow-up exist; we therefore aim to assess the fecal continence of TAMIS after an intermediate term of follow-up using the fecal incontinence severity index (FISI) in the present study [21]. Based on the conclusion of our earlier study, we hypothesize that no deterioration of fecal continence will occur over a longer period of follow-up after TAMIS.
Patients and methods
Consecutive patients who were eligible for local transanal surgery were included in our study population. Patients were eligible for the present study if they had sessile rectal adenomas with any degree of dysplasia, low risk cT1-carcinomas, rectal lesions that were deemed not suitable for endoscopic polypectomy, or more invasive tumors for patients who were unsuitable for rectal resection. Patients with an ostomy and patients in whom TAMIS was converted to laparoscopy were excluded. Patients whose postoperative histology reports showed a carcinoma greater than T1 for which they underwent additional surgery were also excluded from the present analysis.
Preoperative assessment included a digital rectal examination, a flexible colonoscopy, a tumor biopsy, and a rigid rectoscopy. The latter was performed to determine the height and the location of the lesion as well as to assess whether transanal resection was feasible. Preoperatively, endoanal sonography was performed to exclude any deep invasion (>T1) in all patients. In case of signs suggesting an invasive carcinoma or when a lesion larger than 30 mm was found on either endoanal sonography or during rectoscopy, patients were staged with pelvic magnetic resonance imaging (MRI). Follow-up for all patients consisted of a 3-monthly physical examination and a flexible sigmoidoscopy, supplemented by polyp screening as described by national guidelines. In the case of T1 invasive carcinomas, the follow-up also included an annual pelvic MRI.
Patient characteristics were entered into a prospective database for quality control purposes. A written informed consent was obtained from all individual participants included in the study. The study was approved by the Institutional Ethics Committee.
The present report was prepared in concordance with STROBE guidelines (http://www.equator-network.org/reporting-guidelines/strobe/).
Surgical procedure
TAMIS was performed or supervised by one certified colorectal surgeon (D.Z.), trained in transanal endoscopic microsurgery as well as laparoscopic surgery. Methods were described in detail in our earlier report [19].
Data collection
The following data was prospectively collected: patient demographics, details on preoperative diagnostic examinations, surgical details, histopathological results, postoperative complications (defined as postoperative hemorrhage requiring readmission, re-intervention, or transfusion, abscesses and/or fistulae, urinary tract infections, mortality), hospital stay, readmissions, pre- and postoperative FISI scores, and follow-up data (local recurrences and long-term morbidity). The Clavien-Dindo classification for surgical complications was used. For the purpose of this study, intermediate-term follow-up was defined as a minimum length of 2 years of follow-up.
The first FISI detailed questionnaire was completed by the patient together with the treating physician during the preoperative consultation. The FISI included four types of leakage: gas, mucus, liquid, and solid stool. Incidence was defined as one to three times per month, once per week, twice per week, once per day, and twice per day [21]. The FISI scores ranged from 0 (total continence) to 61 (complete incontinence on a daily basis). Postoperatively, fecal continence was assessed at every outpatient consultation and entered the database. Patients who were discharged from surgical follow-up were asked to complete a paper questionnaire, which was send to their home address. The preoperative FISI scores were compared to the postoperative scores. In this study, abnormal continence was defined as any FISI score higher than zero.
Statistical analysis
Our primary endpoint was the possible change in fecal continence at the intermediate-term follow-up after TAMIS. Secondary endpoints were to assess perioperative and intermediate-term morbidity. All data were collected on a Microsoft Excel spreadsheet (Microsoft Corporation, Redmond, Washington, USA).
Descriptive statistics were used in describing the study population (demographic and clinical characteristics). Statistical significance was defined as p < 0.05. The changes in FISI score were evaluated by using the non-parametric Wilcoxon signed-rank test. To determine which factors were associated with an improvement or a decline in the pre- and postoperative FISI scores, a multivariate linear regression analysis was performed. The statistical analysis was performed using SPSS 17.0 (SPSS, Inc., Chicago, IL, USA) for Windows.
Results
Between October 2011 and March 2014, 49 patients underwent a TAMIS procedure for neoplasms or benign polyps throughout the rectum. Of these, five patients were excluded from the study (Fig. 1). Of the remaining 44 patients, 42 (m = 21 (50%):f = 21 (50%)) completed all the FISI follow-up moments (i.e., response rate 95%) and were included in the present analysis (Fig. 1). Patient characteristics are depicted in Table 1. The median age was 68.5 years [range 34–94]. The median distance of the lesions from the dentate line was 7.5 cm [range 0–19].
The median operation duration was 63 min [range 17–211]. The rectal defect was closed in 34 cases (80.9%). Closure was deemed unnecessary due to distal localization of the defect in the remaining eight cases.
The median follow-up was 36 months [range 24-48 months]. During this period, one patient (2.3%) had recurrent local disease, as she developed an in situ carcinoma 9 months after a microscopic complete (R0) excision with TAMIS for a T1 carcinoma. A successful re-excision was performed with TAMIS and the patient remained recurrence free 47 months later.
Histopathology
A median surface area of 19.2 cm2 [range 6–56] was resected. The histopathological results of the resected specimens showed adenoma in 26 cases (61.9%), in situ carcinoma in five patients (11.9%), and invasive carcinoma in 11 patients (26.2%). One of the invasive carcinomas was staged as a T2 rectal carcinoma necessitating additional surgery; however, this patient declined any additional surgical or adjuvant chemo-radiation. The remaining invasive carcinomas that were found were staged as T1. None of the patients were treated with neoadjuvant therapy. There were no fragmented excisions. Four specimens (9.5%) showed microscopically positive margins; however, all showed adenomas in the resection margin. All patients received a close follow-up attention with frequent sigmoidoscopies and biopsies; no additional surgery was performed.
Postoperative course
Patients were discharged after a median hospital stay of 1 day [range 1–24]. Postoperative complications occurred in four patients (9.5%). Three patients were readmitted for postoperative hemorrhage and were successfully treated with conservative measures; no reoperations or blood transfusions were necessary (Clavien-Dindo grade I). One patient developed a pelvic abscess, subsequent sepsis, and a perianal fistula. This patient received antibiotics and the abscess was drained surgically and the fistula healed spontaneously. The patient was discharged after a total hospital stay of 24 days (Clavien-Dindo grade III).
Fecal continence
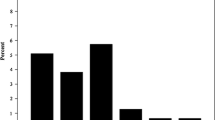
The observed differences in the mean FISI scores during short-term follow-up were 8.3 points pre-TAMIS [range 0–35] vs 5.4 points 1 year post-TAMIS [range 0–20]; this difference was not statistically different (p = 0.501). After intermediate-term follow-up, a decrease in FISI score was observed from 5.4 points [range 0–20] at 1 year post-TAMIS to 10.1 points [range 0–31] 3 years after TAMIS. This difference was statistically significant (p = 0.01). These results are summarized in Fig. 2. Of the 27 patients (m:f = 13:14) with normal continence pre-TAMIS (FISI = 0), 17 patients (63.0%) suffered deterioration of their fecal continence over a period of 3 years (mean FISI score 14.2). Of these patients, 11 patients suffered only minor incontinence (incontinence to flatus or soiling). However, six patients encountered more severe incontinence to liquid stool. Fortunately, none of these patients suffered incontinence for solids, and in the patients encountering incontinence to liquids, actual loss was very infrequent (once monthly or less frequent). A total of 14 patients reached a normal FISI score postoperatively. The results are summarized in Fig. 3a.
Overall pre- and postoperative FISI scores. Values are mean (± SEM). Higher FISI scores indicate a worse anorectal functioning. FISI scores range from 0 (total continence) to 61 (complete incontinence to a solid stool on a daily basis). TAMIS transanal minimally invasive surgery, FISI fecal incontinence severity index
a. Functional outcomes 3 years after transanal minimally invasive surgery (TAMIS). b. Detailed overview, depicting number of patients (in parentheses) with impaired and normal continence pre-TAMIS (left side) and post-TAMIS (right side). Change in degree of continence after TAMIS is shown in the arrows for each individual patient. Incontinence for flatus, liquids, or soiling was encountered once monthly or less frequent. No incontinence for solids was reported
Before the operation, 15 patients (m:f = 9:6) (35.7%) had an abnormal continence (any FISI score higher than 0) (mean FISI score 21.4, range 4–40). After 3 years of follow-up, a total of 28 patients (66.6%) had impaired continence (mean FISI score 15.2, range 3–31). Of these 15 patients, 8 patients complained of minor incontinence before the operation, whereas 7 patients complained of more severe incontinence. After the operation, four patients regained normal function. Four patients did not encounter a significant improvement in their continence status. Two patients encountered a further decrease of their continence. These results are summarized in Fig. 3b.
Of the total amount of patients with fecal incontinence after TAMIS, no significant age difference could be observed when compared to those patients with an improved or an unchanged FISI score (64.8 vs 63.5 years; p = 0.607). There was no significant difference in the tumor distance from the anal verge (5.5 vs 6.1 cm; p = 0.567). No difference in tumor size could be observed between the two groups (21.1 vs 28.2 cm2; p = 0.206). A univariate linear regression analysis resulted in no significant association of independent variables with either an improvement or a decline of pre- and postoperative FISI scores at the medium-term follow-up. A multivariate regression analysis was performed to predict fecal incontinence from gender, age, tumor size, and distance from the anal verge. These variables did not predict the FISI score: F (4, 35) = 95.7, p = 0.740, R 2 = 0.053. All four variables did not add statistically significant to the prediction.
Discussion
In the current study, we analyzed the influence of TAMIS on fecal continence in 42 patients undergoing TAMIS. While there are two previous studies that reported on this subject [19, 20], our study is the first study focusing on the impact of TAMIS on fecal continence after a longer follow-up period.
In a previous study conducted by our group, continence improved in 88% of the patients, 11 months after TAMIS. These findings were confirmed by Verseveld; in their study, the FISI scores improved in 79% of the patients, 6 months after TAMIS. Both studies reported a deterioration of the anorectal function in 5 and 21% of the patients, respectively. No significant predictors of this deteriorated anorectal function were determined. It was hypothesized that this difference in observed anorectal function could be associated with the relatively short period of follow-up. Contrary to the hypothesis of the present study, we found that statistically significantly more patients had some degree of fecal incontinence 3 years after TAMIS when compared to the total number of patients with impaired continence 1 year after TAMIS.
Since the introduction of TEMS [22], local excision gained a wide acceptance while regarded being safe, effective, and associated with less postoperative morbidity in terms of defecation disorders and anterior resection syndromes when compared to anterior resection with total mesorectal excisions [10, 12, 23, 24]. In 2009, transanal minimally invasive surgery (TAMIS) was developed [13]. Several research groups have already proven that TAMIS is safe and feasible, leading to a rapid worldwide adoption by colorectal surgeons.
During the implementation of TAMIS in our hospital, different transanal platforms were used. In the search for an optimal system, different systems were tried, that had a ring of comparable rigidity incorporated. The ease of use and the quality of pneumorectum using all system was good.
Since TAMIS is considered to be the successor, or a suitable alternative to TEMS, a comparison of functional results between these modalities is relevant. Outcome data in this area is limited; most of the data is relatively short term (6–12 months) [10, 23, 25]. Therefore, reliable conclusions cannot be drawn from these direct comparisons. However, results on follow-up data after TEMS, reported by several other research groups, seem to be comparable with those reported after TAMIS [26]. Studies that conclude this are summarized in Table 2.
A relatively new technique that is gaining more acceptance in the treatment of early rectal neoplasms, particularly in Asian countries where this technique was born, is the ESD (endoscopic submucosal dissection). On theoretical grounds, it may be beneficial to use this novel technique in the future for patients suspected of benign disease with low index of suspicion and who abandoned the TAMIS technique. However, this technique proves difficult to master with a long and laborious learning curve. In European countries, there is currently limited experience and comparative data is lacking making this technique no suitable alternative for TAMIS yet [27].
It has been postulated that anorectal continence is maintained by the combination of multiple physiological mechanisms and is therefore seldom disrupted by one single factor. The internal anal sphincter is responsible for most of the anal resting pressure and chiefly responsible for the fecal continence. This function is supported by the anal mucosal folds and the hemorrhoidal cushions [28, 29]. Damage to the internal sphincter may therefore lead to significant fecal incontinence. The external anal sphincter reinforces the aforementioned structures during voluntary squeeze [30, 31]. Damage or weakening of the external sphincter can cause urge-related incontinence or fecal incontinence for liquid stool. Damage to the hemorrhoidal plexus can resolve in a poor anal “seal” and thus incontinence for flatus and liquids [31].
The logical primary factor developing fecal incontinence after TAMIS must therefore be the surgically induced trauma. Upon speculating on the factors influencing the anorectal function in the intermediate- or long-term follow-up after TAMIS, one of the post-surgery hypotheses states that some part of the fecal incontinence is related to a disturbed rectoanal inhibitory reflex, leading to disturbed rectoanal coordination. Authors describing this hypothesis believe the main risk factor for developing this disturbed sphincter reflex is the extent of the resection (> 50% of the rectal wall circumference). Furthermore, some authors believe the depth (full thickness) of the resection in combination with the location of the tumor (internal anal sphincter defects) influences rectal wall compliance and maximal tolerable rectal volume [9]. These results however are based upon low number of patients after TEMS, and short-term follow-up therefore solid conclusions cannot be drawn. However, causality between the extent and the depth of the resection and the compliance of the rectal wall sounds plausible. Furthermore, it is likely that stretch to one or both sphincters can have a detrimental effect as well. A similar mechanism has been described in hemorrhoidal and fistula surgery [32, 33]. Based on this theory, it could be advocated that TAMIS may have a less detrimental influence on fecal continence, since a flexible port is used in lieu of a rigid operating rectoscope as in TEMS. Unfortunately, the present data do not support this postulate.
If these hypotheses are true and there is a relation to permanent damage of the anorectal apparatus, fecal incontinence should be more likely to occur in tumors located close to the anal verge and should be less severe for tumors resected higher in the anal canal. Unfortunately, the present study could not demonstrate any association between location of the tumor and FISI scores. Moreover, it is difficult to explain why patients with decreased continence before TAMIS would encounter an improvement of their continence after local excision. Even though no clinical evidence is available, we postulate that the presence of a large polyp or tumor may induce a continuous anal sphincter inhibitory reflex that may result in a decreased continence preoperative. This would explain why continence can be improved after local excision.
Some authors have previously stated that transanal operations lasting over 2 h account for a significant worsening of occurrence of fecal incontinence [8]. In the present series, only three procedures lasted over 2 h. Two of these patients had normal preoperative FISI scores; only one patient experienced a worsening after 3 years of follow-up. One of these patients had impaired fecal incontinence pre-TAMIS. Despite an operation time of 139 min, the FISI score was normalized after the TAMIS treatment.
There is an ongoing debate on the possibility of an age-related anal sphincter pressure change [34, 35]. Some studies report a decrease of 30–40% in both man and woman over 70 years of age, when compared to patients younger than 30 years of age. Other studies that examined anal pressures have reported only insignificant decrease with age [36]. In the present study, however, analyses for age differences between continent and incontinent patients showed no statistical significant difference.
Moreover, some studies show thickening of the internal anal sphincter due to fibrosis during aging [37, 38]. Fibrotic muscle fibers generate less resting pressure leading to a lower volume and pressure threshold for the desire to defecate. Maybe the development of scar and fibrotic tissues after TAMIS plays a role in the development of anorectal dysfunction on the medium-term follow-up. This may be the reason why studies focusing on the short-term follow-up after TAMIS find less patients with impaired incontinence when compared to the present study.
Interestingly, the present study does not show a statistically significant difference in the mean FISI score in the total study population, 3 years following TAMIS. However, the total number of patients with an impaired anorectal function did increase from 15 patients (35.7%) pre-TAMIS to 28 patients (66.6%), 3 years after TAMIS. At first glance, the lack of change in overall FISI scores can be deceiving. This can be explained by the larger part of patients in whom continence improved. In the questionnaires, only minor incontinence was noted by the study subjects, even though we believe the incidence of impaired continence cannot be ignored. Since this impairment is not nearly as severe as the impairment reported after low anterior resection [39] [40] and reported functional results after TAMIS are superior to the functional results reported after total mesorectal excision [41, 42], we believe these current study results are sufficient reason for TAMIS to be the procedure of choice for patients with rectal adenomas and selected carcinomas.
A shortcoming of the present study is the lack of quality-of-life measurements, in addition to assessment of anorectal functioning. The addition of quality-of-life measurements would possibly depict patient perspectives in a better way and may attribute to clinical significance. The authors are well aware of significant impact of fecal incontinence on quality of life and admit to this limitation of the present study. However, the FISI questionnaire used in our study is an easy-to-use validated instrument, using patient input to weigh type and severity of fecal incontinence. Furthermore, it has been shown to have an excellent correlation with quality-of-life measures (lifestyle, restriction, depression, and embarrassment) [43]. In past years, this questionnaire has proven to be a highly sensitive measuring tool that enables assessment of functional recovery (or deterioration) over time [43]. Investigations concerning the effect of TAMIS on quality of life after TAMIS are currently being performed by our group.
In our opinion, even when considering the limitations of our study and the ongoing ambiguity on the cause of fecal incontinence, it is clear that the incidence of persistent minor impairment of continence should not be overlooked. Furthermore, there does not seem to be a difference between the results reported after TAMIS or those reported after TEMS. Patients should be informed that a risk of permanent (minor) anorectal function impairment exists. In this regard, we believe that future studies on TAMIS should focus more on the functional outcomes and less on the safety and feasibility of this technique.
Conclusion
This study found that the risk of developing minor functional impairment is considerable. No less than two thirds of all patients with preoperative normal continence will encounter impairment of their fecal continence. Even though these disturbances can be considered minor, especially when compared to far more invasive procedures such as low anterior resection, they deserve to be discussed in detail. However, functional results after TAMIS seem to be comparable to the results after TEMS in the intermediate-term follow-up. Furthermore, the present study confirms that transanal minimally invasive surgery for rectal adenomas and early rectal cancer is safe and is associated with a low morbidity in the intermediate term. In our opinion, future studies following TAMIS should focus on the functional outcome and the quality of life in the long term and not so much on the feasibility and the safety issues.
References
Parks AG, Stuart AE (1973) The management of villous tumours of the large bowel. Br J Surg 60(9):688–695
Francillon J, Moulay A, Vignal J, Tissot E (1974) Excision of carcinomas of the rectal ampulla via the inferior route. The parachute technic. Nouv Press Med 3(21):1365–1366
Buess G, Hutterer F, Theiss J, Bobel M, Isselhard W, Pichlmaier H (1984) A system for a transanal endoscopic rectum operation. Der Chirurg; Zeitschrift fur alle Gebiete der operativen Medizen 55(10):677–680
Moore JS, Cataldo PA, Osler T, Hyman NH (2008) Transanal endoscopic microsurgery is more effective than traditional transanal excision for resection of rectal masses. Dis Colon Rectum 51(7):1026–1030; discussion 1030-1021. https://doi.org/10.1007/s10350-008-9337-x
de Graaf EJ, Burger JW, van Ijsseldijk AL, Tetteroo GW, Dawson I, Hop WC (2011) Transanal endoscopic microsurgery is superior to transanal excision of rectal adenomas. Color Dis : Off J Assoc Coloproctology G B Irel 13(7):762–767. https://doi.org/10.1111/j.1463-1318.2010.02269.x
Barendse RM, Dijkgraaf MG, Rolf UR, Bijnen AB, Consten EC, Hoff C, Dekker E, Fockens P, Bemelman WA, de Graaf EJ (2013) Colorectal surgeons’ learning curve of transanal endoscopic microsurgery. Surg Endosc 27(10):3591–3602. https://doi.org/10.1007/s00464-013-2931-6
Maslekar S, Pillinger SH, Sharma A, Taylor A, Monson JR (2007) Cost analysis of transanal endoscopic microsurgery for rectal tumours. Color Dis 9(3):229–234. https://doi.org/10.1111/j.1463-1318.2006.01132.x
Dafnis G, Pahlman L, Raab Y, Gustafsson UM, Graf W (2004) Transanal endoscopic microsurgery: clinical and functional results. Color Dis : Off J Assoc Coloproctology G B Irel 6(5):336–342. https://doi.org/10.1111/j.1463-1318.2004.00629.x
Herman RM, Richter P, Walega P, Popiela T (2001) Anorectal sphincter function and rectal barostat study in patients following transanal endoscopic microsurgery. Int J Color Dis 16(6):370–376
Doornebosch PG, Gosselink MP, Neijenhuis PA, Schouten WR, Tollenaar RA, de Graaf EJ (2008) Impact of transanal endoscopic microsurgery on functional outcome and quality of life. Int J Color Dis 23(7):709–713. https://doi.org/10.1007/s00384-008-0442-z
Planting A, Phang PT, Raval MJ, Brown CJ (2013) Transanal endoscopic microsurgery: impact on fecal incontinence and quality of life. Can J Surg 56(4):243–248. https://doi.org/10.1503/cjs.028411
Allaix ME, Rebecchi F, Giaccone C, Mistrangelo M, Morino M (2011) Long-term functional results and quality of life after transanal endoscopic microsurgery. Br J Surg 98(11):1635–1643. https://doi.org/10.1002/bjs.7584
Albert MR, Atallah SB, deBeche-Adams TC, Izfar S, Larach SW (2013) Transanal minimally invasive surgery (TAMIS) for local excision of benign neoplasms and early-stage rectal cancer: efficacy and outcomes in the first 50 patients. Dis Colon Rectum 56(3):301–307. https://doi.org/10.1097/DCR.0b013e31827ca313
Atallah S, Albert M, Larach S (2010) Transanal minimally invasive surgery: a giant leap forward. Surg Endosc 24(9):2200–2205. https://doi.org/10.1007/s00464-010-0927-z
Barendse RM, Doornebosch PG, Bemelman WA, Fockens P, Dekker E, de Graaf EJ (2012) Transanal employment of single access ports is feasible for rectal surgery. Ann Surg 256(6):1030–1033. https://doi.org/10.1097/SLA.0b013e3182523b31
Alessandro C, Daniela M, Michele M, Andrea T, Gianmarco G, Massimo S, Orazio Z, Fabio G, Giuseppe T (2012) Glove port technique for transanal endoscopic microsurgery. Int J Surg Oncol 2012:383025. https://doi.org/10.1155/2012/383025
Hompes R, Ris F, Cunningham C, Mortensen NJ, Cahill RA (2012) Transanal glove port is a safe and cost-effective alternative for transanal endoscopic microsurgery. Br J Surg 99(10):1429–1435. https://doi.org/10.1002/bjs.8865
Lee TG, Lee SJ (2013) Transanal single-port microsurgery for rectal tumors: minimal invasive surgery under spinal anesthesia. Surg Endosc. https://doi.org/10.1007/s00464-013-3184-0
Schiphorst AH, Langenhoff BS, Maring J, Pronk A, Zimmerman DD (2014) Transanal minimally invasive surgery: initial experience and short-term functional results. Dis Colon Rectum 57(8):927–932. https://doi.org/10.1097/DCR.0000000000000170
Verseveld M, de Graaf EJ, Verhoef C, van Meerten E, Punt CJ, de Hingh IH, Nagtegaal ID, Nuyttens JJ, Marijnen CA, de Wilt JH, Group CS (2015) Chemoradiation therapy for rectal cancer in the distal rectum followed by organ-sparing transanal endoscopic microsurgery (CARTS study). Br J Surg 102(7):853–860. https://doi.org/10.1002/bjs.9809
Rockwood TH, Church JM, Fleshman JW, Kane RL, Mavrantonis C, Thorson AG, Wexner SD, Bliss D, Lowry AC (1999) Patient and surgeon ranking of the severity of symptoms associated with fecal incontinence: the fecal incontinence severity index. Dis Colon Rectum 42(12):1525–1532
Buess G, Kipfmuller K, Hack D, Grussner R, Heintz A, Junginger T (1988) Technique of transanal endoscopic microsurgery. Surg Endosc 2(2):71–75
Kennedy ML, Lubowski DZ, King DW (2002) Transanal endoscopic microsurgery excision: is anorectal function compromised? Dis Colon Rectum 45(5):601–604
Barendse RM, Oors JM, de Graaf EJ, Bemelman WA, Fockens P, Dekker E, Smout AJ (2013) The effect of endoscopic mucosal resection and transanal endoscopic microsurgery on anorectal function. Color Dis : Off J Assoc Coloproctology G B Irel 15(9):e534–e541. https://doi.org/10.1111/codi.12311
de Graaf EJ, Doornebosch PG, Tetteroo GW, Geldof H, Hop WC (2009) Transanal endoscopic microsurgery is feasible for adenomas throughout the entire rectum: a prospective study. Dis Colon Rectum 52(6):1107–1113. https://doi.org/10.1007/DCR.0b013e3181a0d06d
Restivo A, Zorcolo L, D'Alia G, Cocco F, Cossu A, Scintu F, Casula G (2016) Risk of complications and long-term functional alterations after local excision of rectal tumors with transanal endoscopic microsurgery (TEM). Int J Color Dis 31(2):257–266. https://doi.org/10.1007/s00384-015-2371-y
Marin-Gabriel JC, Fernandez-Esparrach G, Diaz-Tasende J, Herreros de Tejada A (2016) Colorectal endoscopic submucosal dissection from a Western perspective: today’s promises and future challenges. World J Gastrointest Endosc 8(2):40–55. https://doi.org/10.4253/wjge.v8.i2.40
Frenckner B, Euler CV (1975) Influence of pudendal block on the function of the anal sphincters. Gut 16(6):482–489
Rao SS (2004) Pathophysiology of adult fecal incontinence. Gastroenterology 126(1 Suppl 1):S14–S22
Gibbons CP, Bannister JJ, Trowbridge EA, Read NW (1986) An analysis of anal sphincter pressure and anal compliance in normal subjects. Int J Color Dis 1(4):231–237
Gibbons CP, Trowbridge EA, Bannister JJ, Read NW (1986) Role of anal cushions in maintaining continence. Lancet 1(8486):886–888
van Tets WF, Kuijpers JH, Tran K, Mollen R, van Goor H (1997) Influence of Parks’ anal retractor on anal sphincter pressures. Dis Colon Rectum 40(9):1042–1045
Zimmerman DD, Gosselink MP, Hop WC, Darby M, Briel JW, Schouten WR (2003) Impact of two different types of anal retractor on fecal continence after fistula repair: a prospective, randomized, clinical trial. Dis Colon Rectum 46(12):1674–1679. https://doi.org/10.1097/01.DCR.0000098911.72691.27
Gundling F, Seidl H, Scalercio N, Schmidt T, Schepp W, Pehl C (2010) Influence of gender and age on anorectal function: normal values from anorectal manometry in a large caucasian population. Digestion 81(4):207–213. https://doi.org/10.1159/000258662
Loening-Baucke V, Anuras S (1985) Effects of age and sex on anorectal manometry. Am J Gastroenterol 80(1):50–53
Rao SS, Hatfield R, Soffer E, Rao S, Beaty J, Conklin JL (1999) Manometric tests of anorectal function in healthy adults. Am J Gastroenterol 94(3):773–783. https://doi.org/10.1111/j.1572-0241.1999.00950.x
Huebner M, Margulies RU, Fenner DE, Ashton-Miller JA, Bitar KN, DeLancey JO (2007) Age effects on internal anal sphincter thickness and diameter in nulliparous females. Dis Colon Rectum 50(9):1405–1411. https://doi.org/10.1007/s10350-006-0877-7
Lewicky-Gaupp C, Hamilton Q, Ashton-Miller J, Huebner M, DeLancey JO, Fenner DE (2009) Anal sphincter structure and function relationships in aging and fecal incontinence. Am J Obstet Gynecol 200(5):559):e551–e555. https://doi.org/10.1016/j.ajog.2008.11.009
Ziv Y, Zbar A, Bar-Shavit Y, Igov I (2013) Low anterior resection syndrome (LARS): cause and effect and reconstructive considerations. Tech Coloproctol 17(2):151–162. https://doi.org/10.1007/s10151-012-0909-3
Emmertsen KJ, Laurberg S, Rectal Cancer Function Study G (2013) Impact of bowel dysfunction on quality of life after sphincter-preserving resection for rectal cancer. Br J Surg 100(10):1377–1387. https://doi.org/10.1002/bjs.9223
Doornebosch PG, Tollenaar RA, Gosselink MP, Stassen LP, Dijkhuis CM, Schouten WR, van de Velde CJ, de Graaf EJ (2007) Quality of life after transanal endoscopic microsurgery and total mesorectal excision in early rectal cancer. Color Dis 9(6):553–558. https://doi.org/10.1111/j.1463-1318.2006.01186.x
De Graaf EJ, Doornebosch PG, Tollenaar RA, Meershoek-Klein Kranenbarg E, de Boer AC, Bekkering FC, van de Velde CJ (2009) Transanal endoscopic microsurgery versus total mesorectal excision of T1 rectal adenocarcinomas with curative intention. Eur J Surg Oncol 35(12):1280–1285. https://doi.org/10.1016/j.ejso.2009.05.001
Cavanaugh M, Hyman N, Osler T (2002) Fecal incontinence severity index after fistulotomy: a predictor of quality of life. Dis Colon Rectum 45(3):349–353
Acknowledgements
The authors would like to thank M. De Jong MD, PhD for her comments that improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
A written informed consent was obtained from all individual participants included in the study. The study was approved by the Institutional Ethics Committee.
The present report was prepared in concordance with STROBE guidelines (http://www.equator-network.org/reporting-guidelines/strobe/).
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Clermonts, S.H.E.M., van Loon, Y.T., Schiphorst, A.H.W. et al. Transanal minimally invasive surgery for rectal polyps and selected malignant tumors: caution concerning intermediate-term functional results. Int J Colorectal Dis 32, 1677–1685 (2017). https://doi.org/10.1007/s00384-017-2893-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-017-2893-6