Abstract
Purpose
Hepatocellular carcinoma (HCC), the second most common pediatric malignant liver tumor after hepatoblastoma, represents 1% of all pediatric tumors.
Methods
A retrospective study was conducted on children with HCC treated at our center from March 2002 to October 2022, excluding those with inadequate follow-up or records. Demographic data, initial complaints, alpha-fetoprotein (AFP) values, underlying disease, size and histopathological features of the masses, chemotherapy, and long-term outcomes were analyzed.
Results
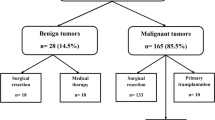
Fifteen patients (8 boys, 7 girls) with a mean age of 11.4 ± 4.1 years (0.8–16.4 years) were analyzed. The majority presented with abdominal pain, with a median AFP of 3.9 ng/mL. Hepatitis B cirrhosis in one patient (6.6%) and metabolic disease (tyrosinemia type 1) in two patients (13.3%) were the underlying diseases. Histopathological diagnoses were fibrolamellar HCC (n:8; 53.3%), HCC (n:6; 40%). Four of the 15 patients underwent liver transplantation, and 9 underwent surgical resection. Due to late diagnosis, two patients were considered inoperable (13.3%). The survival rate for the four patients who underwent liver transplantation was found to be 75%.
Conclusion
Surgical treatment of various variants of HCC can be safely performed in experienced centers with a multidisciplinary approach, and outcomes are better than in adults.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary tumors of the liver are rarely seen in the pediatric age group and constitute about 1% of all pediatric malignancies [1, 2]. Hepatoblastoma is the most common malignant liver tumor (48%) in children, followed by hepatocellular carcinoma [1, 3, 4]. HCC accounts for 27% of all liver tumors and 4% of all pediatric liver transplantations [5]. HCC is generally associated with poor survival and is mainly seen in adults, more often in males, whereas in the pediatric age group, it is usually seen in older children/adolescents [2, 6].
Surgical resection is the mainstay treatment for HCC. For children diagnosed with non-metastatic HCC, numerous pediatric studies emphasize the significance of complete tumor excision, ensuring clear surgical margins during liver resection, or opting for liver transplantation as the most definitive treatments [7,8,9,10]. In this study, we aimed to evaluate the findings obtained through a detailed evaluation of the demographics, tumor characteristics, and surgical treatment options to provide insights about the outcomes of pediatric HCC patients.
Methods
Study design
Following the approval of the institutional review board, a retrospective analysis was conducted on 15 pediatric patients diagnosed with hepatocellular carcinoma treated at our institution from March 2002 to October 2022. We conducted an analysis of demographic data, lesion size, underlying disease, radiological features, bridging therapy, and the postoperative outcomes.
A total of 15 patients (8 boys and 7 girls) were included in the study. All patients had a histopathologically confirmed diagnosis, either through a tru-cut or a surgical biopsy. Preoperative AFP values, abdominal ultrasonography (USG), computerized tomography (CT), and/or magnetic resonance imaging (MRI) were evaluated for all patients. The PRE-Treatment Extent of Tumor (PRETEXT) [11] system designed by International Society of Paediatric Oncology (SIOPEL), was use for tumor staging. Analysis of radiologic images (CT scans and MRI) obtained at the time of diagnosis was performed to determine both the size and number of liver lesions, and to assign an appropriate PRETEXT score following the adaptations by the Children’s Oncology Group (COG) to the original 2005 PRETEXT guidelines. The patient cohort (n = 15) was then assigned into groups based on their treatment pathways: “Mass/Liver Resection Group” (n = 9), “Liver Transplantation Group” (n = 4), and “Inoperable Group” (n = 2). Their respective PRETEXT stages were recorded with a breakdown as follows: in the mass/liver resection group, two patients were PRETEXT 1, six patients PRETEXT 2, and one patient PRETEXT 3. Conversely, all patients in the transplant group were PRETEXT 3 and inoperable group was PRETEXT 4, respectively.
Milan Criteria [10] was considered when deciding for liver transplantation. For patients with a single hepatocellular carcinoma to be eligible for liver transplantation, the tumor should not exceed 5 cm in diameter. In patients with multiple tumors, there could be no more than three tumors, none exceeding 3 cm in diameter. Patients with evident or suspected tumor invasion of blood vessels or lymph nodes were excluded from the study.
Underlying etiological factors were considered for liver transplantation. Two patients were identified as having metabolic liver diseases as the underlying cause for liver transplantation, while one patient was diagnosed with Hepatitis B (HBV) cirrhosis. In these three patients, HCC was not a preoperative diagnosis but discovered at liver explant pathology.
Statistical analysis
The statistical analysis was performed using SPSS for Mac (IBM SPSS Statistics 26) and Excel 2023 (Microsoft Excel for Mac).
All statistical evaluations were carried out on a cohort of 15 pediatric patients diagnosed with HCC. Demographic and clinical variables were analyzed using descriptive statistics. Continuous data, such as age and alpha-fetoprotein (AFP) levels, were presented as means and ranges, while categorical data, such as gender and tumor type, were summarized as frequencies and percentages.
Kaplan–Meier method was used to calculate 1-, 3-, and 5-year overall survival (OS) and disease-free survival (DFS) rates. The survival analyses were further stratified by surgical approach (mass/liver resection vs. liver transplantation), as well as by preoperative and postoperative use of chemotherapy. The log-rank test was applied to identify any significant differences in survival outcomes across these subgroups. The effect of various prognostic factors, including histopathological subtypes, neoadjuvant and adjuvant chemotherapy, and surgical margin status, on 5-year OS was evaluated using the log-rank test. Additionally, we used the log-rank test to assess the impact of metastatic spread, specifically to the lung and bone, on patient survival.
Statistical significance was set at a p-value of less than 0.05. All analyses were conducted using R version 3.6.3, and p-values were two-sided.
Results
Patient characteristics
There were eight boys (53.3%) and seven girls and (46.6%) in the series. Mean age was 11.4 ± 4.1 years with a range of 0.8–16.4 years. Demographic variables are summarized in Table 1, and patient details are given in Table 2. Histopathological evaluations of the tumor characteristics revealed that three patients (20%) had well-differentiated HCC (WD-HCC), two patients (13.3%) had conventional HCC (C-HCC), eight patients (53.3%) had fibrolamellar HCC (FL-HCC), and one patient (6.6%) had undifferentiated HCC (U-HCC). One patient was identified with HCC based on HBV cirrhosis.
Clinical presentations
Nine (60%) of the patients underwent mass/liver resection, and four patients (26.6%) underwent liver transplantation. Two patients (13.3%) were not suitable for surgical treatment due to advanced stage-widespread disease. Neo-adjuvant chemotherapy was given to 40% of the patients. Clean tumor-free surgical margin was achieved in all patients who underwent surgical resection. All patients who had total tumor excision with primary liver resection were observed to have low AFP values at the time of admission (n = 9, median AFP: 2.78 ng/ml, range: 1.2–21).
Ten patients received postoperative adjuvant chemotherapy, with a 5-year survival rate of 70%. For those patients who received liver transplantation; a 5-year survival rate of 75% was achieved. In inoperable patients, all showed metastatic disease and received neoadjuvant chemotherapy. Chemotherapy treatment was planned individually, with sorafenib treatment prominent in patients with local negative surgical margins, and chemotherapy protocols containing cisplatin and doxorubicin applied in advanced stage patients.
Nasocerebral mucormycosis developed in one of the patients who underwent liver resection resulting in the patient’s demise. The 5-year survival rate among inoperable patients was found to be 0%. For one patient, who was PRETEXT stage 3, mass resection was initially performed to gain time due to the unavailability of a donor. However, during this waiting period, recurrence developed, and the patient later underwent liver transplantation. The same patient also underwent transarterial radioembolization (TARE) until liver transplantation for bridging therapy.
The most common metastases were seen in the lung and bone, occurring at a rate of 26.6%. Overall, the 5-year survival rate of pediatric HCC patients was determined to be 66.6%. Results of Kaplan–Meier analysis revealed that PRETEXT stage and chemotherapy type were observed to be independent risk factors for 5-year survival (Figs. 1, 2). According to the ROC analysis, it was observed that the average age and AFP values of the patients were independent risk factors for 5-year survival (Fig. 3).
We have 2 cases that have recurrence of the disease (Patient 10, 14), one of them was unable to undergo liver transplantation because lack of donor. He was subsequently put on a bridge treatment while waiting for a donor. However, during this time, a recurrence was observed (Patient 10). The other patient did undergo liver transplantation, but in the second-year post-operation, metastases were detected in the bone and transplanted liver, which were considered as a recurrence (Patient 14).
In our research, one patient (Patient 1) was found to have lung metastasis in the 5th year after liver transplantation, and another patient (Patient 9) was found to have lymph node metastasis in the 2nd year after liver resection. Both patients underwent metastasectomy. While two of our patients were not eligible for surgery due to distant organ metastases (Patients 12 and 15), the other patient was found to have distant organ metastases in the 2nd year after liver resection (Patient 13).
In univariate Cox regression analysis, age, neoadjuvant chemotherapy AFP, and PRETEXT staging systems were observed to be associated with 5-year survival. According to the results of multivariate Cox regression analysis, neoadjuvant chemotherapy [HR = 2.840, 95% confidence interval (CI): 0.915–4.671, P < 0.001], AFP value [HR = 1.719 95% confidence interval (CI): 1.427–1.915, P < 0.001], and PRETEXT staging systems were observed to be independent positive factors for 5-year survival (Table 3).
Discussion
Hepatocellular carcinoma is a very rare malignancy in the pediatric age group. In adults, HCC predominantly develops on a cirrhotic liver background [5]. However, in the pediatric age group, it can occur on two different bases: the more commonly encountered form (70%) is sporadic/de-novo HCC that develops in normal liver tissue. Histopathologically, sporadic HCC can be classified into “Conventional”, “Undifferentiated” (HCC-NOS- HCC with elements of hepatoblastoma), and “Fibrolamellar” HCC. The fibrolamellar variant of HCC occurs in 24% of cases and generally has a more favorable prognosis [12]. The remaining 30% of HCC mostly arises from chronic liver diseases, cirrhosis, and underlying metabolic, infectious, and vascular liver diseases (hemochromatosis, hereditary tyrosinemias, alpha-1 antitrypsin deficiency, glycogen storage diseases), chronic cholestasis (progressive familial intrahepatic cholestasis), and chronic viral hepatitis, as well as biliary atresia [13]. Tyrosinemia and perinatally acquired hepatitis B infection are major risk factors [4, 13, 14]. Pediatric HCC is more common in males than in females (0.45; 0.37/1 million). Of these, 12.9% are children under 5 years of age, and 34% are adolescents between the ages of 15 and 19 [15]. In our series, only 4 of our 15 patients (28.5%) had underlying liver disease, supporting the fact that most pediatric HCC patients are not related to hepatic cirrhosis but are ‘de-novo’ [16].
Clinical symptoms and signs generally include abdominal pain and a mass in the abdomen, hepatosplenomegaly, gastroesophageal reflux, jaundice, and signs of liver failure [13]. AFP was found to be elevated in over 90% of HCC patients in a relatively large recent series of 65 patients under 20 years of age [17]. AFP correlates with tumor size, thus representing an indirect marker for treatment response [18]. AFP is high in 55–65% of pediatric HCC patients and normal in 25% [19]. The tumor size at diagnosis in pediatric HCC is generally larger compared to adults and falls outside the classic criteria used for surgical resection in adults [20]. On the contrary, in our series, all patients who had total tumor excision with primary liver resection were observed to have normal to low AFP values at the time of admission (n = 9, AFP: 2.78 ng/ml, range: 1.2–21).
Macroscopic and microscopic features of pediatric and adult HCC are similar [16, 21, 22]. The two most common pathological subtypes are the classical trabecular variant and the fibrolamellar variant, but recently an intermediate form (indistinguishable between hepatoblastoma and hepatocellular carcinoma) has been also defined [23]. In our series, one patient was identified with this indistinguishable type (7.1%).
According to publications from adult series, adult variant of fibrolamellar HCC has a better prognosis than pediatric HCC [24, 25]. However, our experience has not confirmed this; in our study, the 5-year survival of fibrolamellar variant HCC patients was found to be higher than adult series with a rate of 87.5%. This discrepancy between adults and children may be attributed to the distinct biological behavior of the tumor in pediatric cases, which is generally less aggressive.
Surgical treatment, either through liver resection or transplantation, is essential for achieving relapse-free survival in hepatocellular carcinoma (HCC) patients [26]. Liver transplantation (LT) is particularly critical for those with chronic liver disease and HCC, offering a dual benefit of removing the tumor and addressing the underlying liver condition [27]. Despite its advantages, the use of LT in pediatric cases is not fully supported by adult-oriented staging systems like the Milan Criteria. Research indicates that LT could be more advantageous for pediatric patients not meeting these criteria, with survival rates ranging from 72 to 83% [28,29,30]. However, the scarcity of liver grafts and the high HCC recurrence rates limit LT’s broader application. Additionally, the long wait times exacerbate tumor progression, leading to high dropout rates from transplant lists. Locoregional therapies, such as radiofrequency ablation (RFA), transarterial radioembolization (TARE), and transarterial chemoembolization (TACE), are used to manage disease progression and bridge the gap to transplantation [31, 32]
One of the most important issues here is the recurrence rate after transplantation. This rate has been reported as 10–30% in studies [33]. Recurrence significantly reduces post-transplant survival, and treatment options are very limited in these patients [34]. The recurrence rate of our existing 4 transplantation patients is 25% and this has also been confirmed in our study. Overall survival is not encouraging in studies, but we found a 5-year survival of 66.6% in our series.
Specialized centers with expertise in hepatobiliary surgery, liver transplantation, and pediatric oncology are needed for pediatric HCC patients, who require both surgical and oncological aggressive treatment.
Limitations
Present study has several limitations. The relatively small sample size of 15 patients may confine the generalization of the findings. Additionally, the study’s retrospective design relied on existing medical records, which may introduce recall and selection bias.
The single-center nature of our study means that the results reflect the specific characteristics, the experience, and.
the treatment practices of our institution. It would be beneficial to conduct multicenter studies for a broader perspective. Moreover, the follow-up periods for some patients were limited, which might have prevented a comprehensive understanding of long-term survival and recurrence rates.
The cohort’s heterogeneity, with patients having varying stages of hepatocellular carcinoma (HCC), different underlying conditions, and diverse treatment pathways presents another challenge for this study.
This diversity mirrors real-world clinical scenarios but complicates direct comparisons and limits the potential for specific subgroup analyses.
Addressing these limitations in future research could provide more definitive insights into the efficacy of the surgical technique.
Conclusion
The effectiveness of surgical treatment in pediatric hepatocellular carcinoma patients and the favorable outcomes of liver resection and liver transplantation have been demonstrated in this study. The use of postoperative adjuvant chemotherapy and individualized chemotherapy protocols has increased survival rates. However, it should be emphasized that the treatment options for inoperable patients are limited and that more studies are needed.
Data availability
Data supporting the findings of this study are provided within the manuscript.
References
Angelico R, Grimaldi C, Saffioti MC, Castellano A, Spada M (2018) Hepatocellular carcinoma in children: hepatic resection and liver transplantation. Transl Gastroenterol Hepatol 3:59–59. https://doi.org/10.21037/tgh.2018.09.05
Ranganathan S, Lopez-Terrada D, Alaggio R (2020) Hepatoblastoma and pediatric hepatocellular carcinoma: an update. Pediatr Dev Pathol 23(2):79–95. https://doi.org/10.1177/1093526619875228
Schmid I, von Schweinitz D (2017) Pediatric hepatocellular carcinoma: challenges and solutions. JHC 4:15–21. https://doi.org/10.2147/JHC.S94008
Moore SW, Millar AJW, HadleyLarry GP et al (2004) Hepatocellular carcinoma and liver tumors in South African children: a case for increased prevalence. Cancer 101(3):642–649. https://doi.org/10.1002/cncr.20398
Khanna R, Verma SK (2018) Pediatric hepatocellular carcinoma. WJG 24(35):3980–3999. https://doi.org/10.3748/wjg.v24.i35.3980
Lau CSM, Mahendraraj K, Chamberlain RS (2015) Hepatocellular carcinoma in the pediatric population: a population based clinical outcomes study involving 257 patients from the surveillance, epidemiology, and end result (SEER) database (1973–2011). HPB Surg 2015:1–10. https://doi.org/10.1155/2015/670728
Murawski M, Weeda VB, Maibach R et al (2016) Hepatocellular carcinoma in children: does modified platinum- and doxorubicin-based chemotherapy increase tumor resectability and change outcome? lessons learned from the SIOPEL 2 and 3 studies. JCO 34(10):1050–1056. https://doi.org/10.1200/JCO.2014.60.2250
Nagtegaal ID, Odze RD, Klimstra D et al (2020) The 2019 WHO classification of tumours of the digestive system. Histopathology 76(2):182–188. https://doi.org/10.1111/his.13975
Austin MT, Leys CM, Feurer ID et al (2006) Liver transplantation for childhood hepatic malignancy: a review of the united network for organ sharing (UNOS) database. J Pediatr Surg 41(1):182–186. https://doi.org/10.1016/j.jpedsurg.2005.10.091
Mazzaferro V, Regalia E, Doci R et al (1996) Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 334(11):693–700. https://doi.org/10.1056/NEJM199603143341104
Roebuck DJ, Aronson D, Clapuyt P et al (2007) 2005 PRETEXT: a revised staging system for primary malignant liver tumours of childhood developed by the SIOPEL group. Pediatr Radiol 37(2):123–132. https://doi.org/10.1007/s00247-006-0361-5
Allan BJ, Wang B, Davis JS et al (2014) A review of 218 pediatric cases of hepatocellular carcinoma. J Pediatr Surg 49(1):166–171. https://doi.org/10.1016/j.jpedsurg.2013.09.050
Kelly D, Sharif K, Brown RM, Morland B (2015) Hepatocellular carcinoma in children. Clin Liver Dis 19(2):433–447. https://doi.org/10.1016/j.cld.2015.01.010
Meyers RL (2007) Tumors of the liver in children. Surg Oncol 16(3):195–203. https://doi.org/10.1016/j.suronc.2007.07.002
Darbari A (2003) Epidemiology of primary hepatic malignancies U.S. children. Hepatology 38(3):560–566. https://doi.org/10.1053/jhep.2003.50375
Moore SW, Hesseling PB, Wessels G, Schneider JW (1997) Hepatocellular carcinoma in children. Pediatric Surg Int 12:266–270
Wang J, Mao Y, Liu Y et al (2017) Hepatocellular carcinoma in children and adolescents: clinical characteristics and treatment. J Gastrointest Surg 21(7):1128–1135. https://doi.org/10.1007/s11605-017-3420-3
Liu C (2013) Value of α-fetoprotein in association with clinicopathological features of hepatocellular carcinoma. WJG 19(11):1811. https://doi.org/10.3748/wjg.v19.i11.1811
Katzenstein HM, Krailo MD, Malogolowkin MH et al (2002) Hepatocellular carcinoma in children and adolescents: results from the pediatric oncology group and the children’s cancer group intergroup study. JCO 20(12):2789–2797. https://doi.org/10.1200/JCO.2002.06.155
deVilledeGoyet J, Meyers RL, Tiao GM, Morland B (2017) Beyond the Milan criteria for liver transplantation in children with hepatic tumours. Lancet Gastroenterol Hepatol 2(6):456–462. https://doi.org/10.1016/S2468-1253(17)30084-5
Kew MC (1989) Hepatocellular carcinoma with and without cirrhosis. Gastroenterology 97(1):136–139
Friedman LS (1999) The risk of surgery in patients with liver disease. Hepatol 29(6):1617–23. https://doi.org/10.1002/hep.510290639
Prokurat A, Kluge P, Kościesza A, Perek D, Kappeler A, Zimmermann A (2002) Transitional liver cell tumors (TLCT) in older children and adolescents: a novel group of aggressive hepatic tumors expressing beta-catenin. Med Pediatr Oncol 39(5):510–518. https://doi.org/10.1002/mpo.10177
Ramai D, Ofosu A, Lai JK, Gao ZH, Adler DG (2021) Fibrolamellar hepatocellular carcinoma: a population-based observational study. Dig Dis Sci 66(1):308–314. https://doi.org/10.1007/s10620-020-06135-3
Czauderna P (2002) Adult type vs. childhood hepatocellular carcinoma are they the same or different lesions? Biology, natural history, prognosis, and treatment. Med Pediatr Oncol 39(5):519–523. https://doi.org/10.1002/mpo.10178
Ziogas IA, Benedetti DJ, Matsuoka LK et al (2021) Surgical management of pediatric hepatocellular carcinoma: an analysis of the national cancer database. J Pediatr Surg 56(4):772–777. https://doi.org/10.1016/j.jpedsurg.2020.06.013
Nagai H, Matsui T, Kanayama M et al (2011) Multimodal therapy for liver cirrhosis patients with advanced hepatocellular carcinoma. Cancer Chemother Pharmacol 68(1):139–145. https://doi.org/10.1007/s00280-010-1465-z
Romano F, Stroppa P, Bravi M et al (2011) Favorable outcome of primary liver transplantation in children with cirrhosis and hepatocellular carcinoma: liver transplantation in children with hepatocellular carcinoma. Pediatr Transplant. https://doi.org/10.1111/j.1399-3046.2011.01528.x
Ismail H, Broniszczak D, Kaliciński P et al (2009) Liver transplantation in children with hepatocellular carcinoma do Milan criteria apply to pediatric patients? Pediatr Transplant 13(6):682–692. https://doi.org/10.1111/j.1399-3046.2009.01062.x
Beaunoyer M, Vanatta JM, Ogihara M et al (2007) Outcomes of transplantation in children with primary hepatic malignancy. Pediatr Transplant 11(6):655–660. https://doi.org/10.1111/j.1399-3046.2007.00751.x
Sacco R, Conte C, Tumino E et al (2016) Transarterial radioembolization for hepatocellular carcinoma: a review. JHC 3:25–29. https://doi.org/10.2147/JHC.S50359
Lang H, Sotiropoulos GC, Dömland M et al (2005) Liver resection for hepatocellular carcinoma in non-cirrhotic liver without underlying viral hepatitis. Br J Surg 92(2):198–202. https://doi.org/10.1002/bjs.4763
Fernandez-Sevilla E, Allard MA, Selten J et al (2017) Recurrence of hepatocellular carcinoma after liver transplantation: is there a place for resection. Liver Transplant 23(4):440–447. https://doi.org/10.1002/lt.24742
Welker MW, Bechstein WO, Zeuzem S, Trojan J (2013) Recurrent hepatocellular carcinoma after liver transplantation-an emerging clinical challenge: Recurrent hepatocellular carcinoma after liver transplantation. Transpl Int 26(2):109–118. https://doi.org/10.1111/j.1432-2277.2012.01562.x
Acknowledgements
The authors would like to thank Prof Dr Funda Yılmaz from the Department of Pathology, Ege University School of Medicine for contributing to the case-by-case discussions and providing insights to scientific content of the manuscript.
Author information
Authors and Affiliations
Contributions
M.K. performed all the data collection, and was involved in the drafting and critical revision of the manuscript for important intellectual content. U.C. contributed to the study design, data analysis, interpretation of results. E.A. contributed to the data collection and was involved in drafting the manuscript. D.N. provided the histopathological data, interpreted the pathology findings. O.E. designed and conceptualized the study, supervised the project, contributed to the data interpretation, and was involved in drafting and critically revising the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karayazili, M., Celtik, U., Ataseven, E. et al. Evaluation of surgical strategies and long-term outcomes in pediatric hepatocellular carcinoma. Pediatr Surg Int 40, 144 (2024). https://doi.org/10.1007/s00383-024-05721-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s00383-024-05721-0