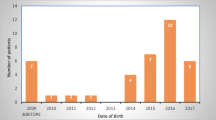
Abstract
Purpose
Recurrent severe hypoglycemic attacks often persist even after performing pancreatectomy for medically unresponsive congenital hyperinsulinism (CHI). In this study, we present our experience with redo pancreatectomy for CHI.
Methods
We reviewed all children who underwent pancreatectomy for CHI between January 2005 and April 2021 in our center. A comparison was made between patients whose hypoglycemia was controlled after primary pancreatectomy and patients who required reoperation.
Results
A total of 58 patients underwent pancreatectomy for CHI. Refractory hypoglycemia after pancreatectomy occurred in 10 patients (17%), who subsequently underwent redo pancreatectomy. All patients who required redo pancreatectomy had positive family history of CHI (p = 0.0031). Median extent of initial pancreatectomy was lesser in the redo group with borderline level of statistical significance (95% vs. 98%, p = 0.0561). Aggressive pancreatectomy at the initial surgery significantly (p = 0.0279) decreased the risk for the need to redo pancreatectomy; OR 0.793 (95% CI 0.645–0.975). Incidence of diabetes was significantly higher in the redo group (40% vs. 9%, p = 0.033).
Conclusion
Pancreatectomy with 98% extent of resection for diffuse CHI, especially with positive family history of CHI, is warranted to decrease the chance of reoperation for persistent severe hypoglycemia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Congenital hyperinsulinism (CHI) is known to be the most common cause of persistent hypoglycemia in infants [1]. The disease is characterized by inappropriate secretion of insulin, resulting in severe hypoglycemic attacks. Consequently, any delay in diagnosis and management puts the patient under high risk of morbidity and mortality [2, 3]. Complete resolution of the hypoglycemia might be achieved by supportive therapy and/or medications alone [4]. However, surgical intervention is often required in refractory cases, poor compliance to medical therapy, or in case a discrete pancreatic lesion was detected and deemed resectable [5]. The optimal extent of pancreatic resection that would control the hypoglycemia and not compromise the endocrine and exocrine function has not been agreed upon [5,6,7]. Due to high rates of refractory persistent disease after partial pancreatectomy for the diffuse form of CHI, and the need to redo the procedure, surgeons became more aggressive in the extent of initial pancreatectomy [8]. However, even after subtotal or near-total pancreatectomy, recurrent severe hypoglycemic attacks occurred in between 11 and 50% of the cases, which also required further resection [7,8,9,10,11,12]. The purpose of this study is to review our experience in pancreatectomy for CHI in the last 16 years to investigate the incidence of redo pancreatectomy, identify the potential factors that may predict the need for reoperation, and determine the long-term incidence of diabetes and pancreatic exocrine insufficiency (PEI).
Methods
A retrospective chart review was conducted at our tertiary center, King Faisal Specialist Hospital and Research Center, Riyadh, Saudi Arabia. The population of interest for this study were all pediatric patients aged 0–14 years who underwent pancreatectomy to manage CHI between January 2005 and April 2021. We excluded all patients who were operated outside our center. Also, all patients with histopathology results not consistent with CHI were excluded. This study was approved by the Research Ethics Committee at our institution.
Data included in the analysis were: gestational age, gender, family history of CHI, parental consanguinity, genetic analysis, age and weight at time of pancreatectomy, surgical approach and extent of resection, placement of gastrostomy tube, histopathology results, blood sugar control, need of pancreatic enzyme replacement therapy, need of redo pancreatectomy and time elapsed between the initial and redo surgery.
The diagnosis of CHI was established when a patient had a non ketotic hypoglycemia (blood sugar level less than 36 mg/dL), hyperinsulinemia (plasma insulin level more than 3 mU/L) and there was a need for high rate glucose infusion (> 10 mg/kg/min) to maintain blood sugar level above 54 mg/dL [5]. After confirming the diagnosis, a combination of frequent enteral feedings, diazoxide, glucagon and/ or octreotide was used for management. Those who required intravenous glucose infusion to maintain adequate blood sugar level or developed symptoms of hypoglycemia despite maximum medical therapy were candidates for surgical intervention in the form of near-total or subtotal pancreatectomy depending on the surgeon`s preference and experience [11]. After reviewing the work by Reyes and colleagues in 1992 and our operative reports, we defined the extent of our surgical resection as the following. Subtotal pancreatectomy (85% of the pancreas) involves the resection of pancreatic tissue distal to the right of the superior mesenteric vessels (SMV). Near-total pancreatectomy (95% of the pancreas) constitutes removal of the tail, body, uncinated process, and part of the pancreatic head. Additionally, when the head of pancreas is shaved off the duodenal sweep and bile duct leaving only a rim of pancreatic tissue, it is considered as 98% pancreatectomy [13]. Surgical intervention was considered successful if the hypoglycemia subsided or came under control with medications. For the patients who showed signs of recurrence after pancreatectomy (Refractory hypoglycemia unresponsive to maximum medical therapy) redo pancreatectomy was indicated. This consisted of shaving or cauterizing the remnant or hypertrophied pancreatic tissue. Likewise, it was considered successful if the hypoglycemia subsided or came under control with medications. The long-term outcomes that we looked for were blood sugar control status and pancreatic exocrine function. PEI was considered positive if the patient had evidence of oily stool and was started on pancreatic enzyme replacement therapy by the endocrinology team.
Statistical analysis was performed using Statistical Analysis System (SAS) statistical software package version 9.4 (SAS Institute Inc., Cary, NC, USA). Continuous variables are reported as mean ± standard deviation (SD) for normally distributed data or median and interquartile range (IQR) for non-parametric variables whereas categorical variables are reported as frequencies (n) and percentages. Comparison between patients who were successfully managed with primary pancreatectomy and those who needed redo pancreatectomy was performed using Student’s t test/ANOVA or non-parametric (Mann–Whitney U/Kruskal–Wallis) test as appropriate for continuous variables, while categorical variables were compared by Chi-square test or Fisher’s exact probability test when deemed necessary. Logistic regression was used to test predictors of the need to redo pancreatectomy. The level of significance was set at a p value < 0.05.
Results
During the 16-year study period, between January 2005 and April 2021, a total of 58 patients underwent subtotal or near-total pancreatectomy for the diagnosis of CHI. There were 28 males (48%) and 30 females (52%). Most of the patients were full term with median gestational age of 37 weeks. Parental consanguinity reported in 82% of the cases while positive family history of CHI was found in 58%. Genetic testing was done for 43 patients. ABCC8 gene was the most common mutated gene and found in 39 patients (91%). Median (IQR) age at presentation was 1 (1–2) day with mostly nonspecific symptoms like lethargy, apnea and seizures. All patients after confirming the diagnosis of CHI were given the maximum chance for their hypoglycemia to be controlled with frequent enteral feeds and medications before proceeding for pancreatectomy. Median (IQR) age and weight at time of pancreatectomy were 8 (6–24) weeks and 5.5 (4.6–8.2) kilograms, respectively. Surgical approach was laparoscopic in 55 patients (98%), one (2%) was converted from laparoscopic to open due to technical difficulties. The median (IQR) extent of surgical resection was 98% (95%-98%) of the pancreas. In addition to the pancreatectomy procedure, gastrostomy tube was performed in 10 patients (10%). There were no iatrogenic injuries to the common bile duct or duodenum. Median (IQR) post-operative hospital stay was 9 (6.6–11) days. At day of discharge, euglycemia was achieved with or without medications in 49 patients (94%), while 3 patients (6%) were diabetic and started on insulin. Histopathological examination revealed diffuse form of CHI in all examined specimens 58 (100%). Patient characteristics and comparison are listed in Table 1.
Recurrence of symptomatic refractory hypoglycemia after pancreatectomy occurred in 10 patients (17% of the total series), for which, redo pancreatectomy was indicated and performed. Median (IQR) time elapsed between the first and redo pancreatectomy was 26 (12–45) weeks at median (IQR) age of 48 (28–72) weeks. The initial extent of pancreatectomy in the first procedure were 85%, 95% and 98% of the pancreas in 3, 4 and 3 patients, respectively. The redo surgery was done laparoscopically in 8 patients, open in 1 patient and laparoscopic converted to open in 1 patient. Surgery performed was completion to 98% pancreatectomy for those who underwent initially less than that, and removal of hypertrophied pancreatic tissue found for those had initial 98% pancreatectomy. Afterward, one patient underwent third exploration and redo pancreatectomy for persistence refractory hypoglycemia. Patient characteristics at time of redo are presented in Table 2.
Univariant analysis of patient characteristics before the recurrence of refractory CHI, revealed that positive family history of CHI was significantly higher in the redo group (p = 0.0031). It is also noticed that those who required redo pancreatectomy underwent initial lesser extent of surgical resection in the first procedure compared to those who were successfully managed with primary pancreatectomy with borderline level of statistical significance (p = 0.0561). There were no statistically significant differences for the remaining variables (Table 1).
Logistic regression model showed that higher extent of surgical resection at the initial surgery significantly decreases the risk for the need to redo pancreatectomy (OR 0.793, 95% CI 0.645–0.975, p = 0.0279). Other variables were not statistically significant to predict the need to redo pancreatectomy (Table 3).
Blood sugar control and pancreatic exocrine status upon follow-up are summarized in Table 4. Median (IQR) follow-up period was 70 (33–110) months. Control of the hypoglycemia was achieved with or without medications in 45 patients (85%), whereas 8 patients (15%) became insulin dependent diabetic. PEI was evident in 6 (12%) patients. Incidence of diabetes was significantly higher in the redo group (p = 0.033). However, there was no statistically significant difference in regards to PEI between the two groups.
Discussion
Infantile hypoglycemia has a wide variety of differential diagnoses. CHI is one of the most important differentials in case of a persistently low blood sugar level [1]. The frequency of the disease is reported as one in 50,000 live births, but in highly consanguineous communities, it can reach as high as one in 2500 live births [11, 14]. The hallmark of CHI is inappropriate over secretion of insulin that leads to severe hypoglycemia. Since hypoglycemia in children, and especially neonates, can result in severe irreversible neurological damage and possible death, the condition should be detected and managed without any delay [2, 3]. Medical management consists of frequent enteral feedings, continuous intravenous infusion of glucose and administration of diazoxide, glucagon, and octreotide as appropriate. These modes of medical management can be used as single agents or in combination as needed. Many patients gradually improve, and their need for medications to maintain appropriate blood sugar level decrease as they grow up. Complete resolution of the condition occurs in some cases [4, 11]. The most common indication to proceed for surgical intervention is failure of maximum medical management, which was the case in all of our patients. Other indications include, poor compliance with medical therapy or the presence of a resectable focal pancreatic lesion. The latter is usually identified by dedicated imaging modalities such as 18F-DOPA-PET/ CT [15].
When it comes to molecular genetics and inheritance, CHI is caused by a defect in key genes that regulate insulin secretion form β-cells of the pancreas. So far, 12 different genes, namely; ABCC8, KCNJ11, GLUD1, GCK, HADH, SLC16A1, HNF4A, HNF1A, HK1, PGM1 and PMM2 have been identified to play a role in regulating insulin secretion [16]. ABCC8 gene mutation is known to be the most commonly mutated gene in cases of CHI and was present in 91% of our genetically tested patients. A close percentage, around 81%, was reported in a previous Saudi study by Al-Agha [17]. In addition to that, family history of CHI was positive in all of our patients who required redo surgery with a statically significant result (p = 0.0031). Thus, genetic evaluation and further understanding of the pattern of inheritance are important as they may give a clue to direct the management of CHI and predict the response of medical or surgical interventions [14, 17].
Looking into histopathology, there are two main subtypes of CHI in the literature; diffuse and focal. The diffuse form is slightly more prevalent (60–70% of cases) [16, 18]. The clinical implication of differentiating the two forms either preoperatively or intraoperatively is to direct the type and extent of surgical intervention. The focal form can be potentially cured by localized and limited resection of the lesion, while the diffuse form requires more aggressive resection up to near-total pancreatectomy [1, 18, 19]. In our series, all of the resected specimens revealed the diffuse form; we never encountered the focal form of CHI. This might be explained by the genetic background in our community and the high rate of consanguineous marriage [17]. Therefore, it is our routine practice, with coordination with our colleagues from the endocrinology team, to proceed immediately to laparoscopic subtotal or near-total pancreatectomy when surgical intervention is indicated without investigating for the focal form. The only exception is when a discrete mass was identified intraoperatively before dissection.
The optimal amount of pancreatic tissue resection to control the hypoglycemia in the diffuse form of CHI that does not negatively impact the pancreatic endocrine and exocrine functions is difficult to appreciate [5,6,7]. In addition, accurate objective measures that define the volume of the pancreas to guide the extent of resection are lacking. Thus, cases of over or under resection are common. These factors explain the variation of outcomes reported in the literature after pancreatectomy for CHI [1]. Relying on the anatomical relationship of the pancreas to the SMV as described by Reyes and colleagues is the most commonly used method by surgeons to guide and define the extent of resection. Nonetheless, the surgeon should consider that the precise total volume and dimensions of the pancreas are not always in a fixed anatomical relationship to the SMV. Visualization of the whole pancreas is crucial to have a more accurate estimation about the extent of the resection [13].
Dealing with diazoxide unresponsive hypoglycemia after subtotal or near-total pancreatectomy for diffuse CHI is challenging and often requires redo pancreatectomy [20]. It is reported between 25 and 50% of the cases in the literature [7, 9,10,11,12]. Such high rate of recurrence could be explained by the over estimation of how much pancreatic tissue was resected initially, or due to intentional over preservation of pancreatic tissue when the surgeon worries about injuring adjacent vital structures at the expense of performing sufficient resection [1, 13]. In our review, we had a recurrence rate of 17%. We believe that our tendency to perform aggressive resections, since we always encounter diffuse CHI, has led to lower recurrence rate compared to what is reported. The association between the extent of resection and need to reoperate is well demonstrated in our data. The more aggressive pancreatic resection performed for diffuse CHI, the less likelihood the patient will comeback with medically unresponsive hypoglycemia necessitating him to undergo a redo pancreatectomy.
Gastrostomy tube insertion is often performed in patients with CHI to aid in hypoglycemia control by continuous feeding regimen or dextrose administration [11]. It is postulated in the literature that gastric procedures including gastrostomy tube insertions and Nissen fundoplication may predispose patients to post prandial hypoglycemia due to dumping syndrome effect or increased level of incretin hormones, particularly glucagon-like peptide-1 (GLP-1). It is thought that these factors may play a role in the pathophysiology of hyperinsulinemic hypoglycemia and possibly pancreatic hyperplasia [21,22,23,24]. We thought of this physiologic reaction as a possible contributing factor in increasing the chance of recurrence after pancreatectomy. However, in our comparison, there was no difference between the two groups, whether they had gastrostomy tube or not. More investigations and higher number of patients may reveal further information.
Long-term outcomes, namely, pancreatic endocrine and exocrine functions may change over time, thus, the reported long-term results vary between the studies depending on the follow-up period [9]. While diabetes developed in a significantly higher percentage in the redo group (p = 0.033), there was no statistically significant difference in regard to PEI. It is expected that both diabetes and PEI incidence would increase with longer follow-up period. Unfortunately, eliminating these complications seem to be inevitable when aggressive pancreatectomy is performed. Indeed, priority should be given to the hypoglycemia control at any expense since neurodevelopmental outcomes are the greatest concerns [12].
Limitations of our study were mainly attributed to the retrospective study design and include that some of the retrieved data were missed. Additionally, the relatively short follow-up period does not reflect the actual ultimate outcomes of our management.
Conclusion
CHI is a heterogeneous disease in regards to genetics, histopathology, response to medical and surgical intervention, and the long-term outcomes. It is common to perform redo pancreatectomy for diffuse CHI to control refractory hypoglycemia at the expense of risking sufficient pancreatic functions. Our data suggest that when surgical intervention is needed for diffuse CHI, surgeons should consider the maximum possible extent (up to 98%) of pancreatectomy, especially for those with family history of CHI since they are at a high risk of having recurrent refractory disease. Both the initial and redo pancreatectomy are feasible to be performed laparoscopically, and iatrogenic injuries can be avoided with meticulous steps and experience. Developing new medical therapies in the future to control diffuse CHI is essential to limit the extent of pancreatectomy or even to completely avoid it.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Al-Shanafey S, Habib Z, AlNassar S (2009) Laparoscopic pancreatectomy for persistent hyperinsulinemic hypoglycemia of infancy. J Pediatr Surg 44:134–138. https://doi.org/10.1016/j.jpedsurg.2008.10.120. (Discussion 138)
Menni F, de Lonlay P, Sevin C et al (2001) Neurologic outcomes of 90 neonates and infants with persistent hyperinsulinemic hypoglycemia. Pediatrics 107:476–479. https://doi.org/10.1542/peds.107.3.476
Carcassonne M, DeLarue A, LeTourneau JN (1983) Surgical treatment of organic pancreatic hypoglycemia in the pediatric age. J Pediatr Surg 18:75–79. https://doi.org/10.1016/s0022-3468(83)80277-2
Worth C, Yau D, Salomon Estebanez M et al (2020) Complexities in the medical management of hypoglycaemia due to congenital hyperinsulinism. Clin Endocrinol 92:387–395. https://doi.org/10.1111/cen.14152
Goel P, Choudhury SR (2012) Persistent hyperinsulinemic hypoglycemia of infancy: an overview of current concepts. J Indian Assoc Pediatr Surg 17:99–103. https://doi.org/10.4103/0971-9261.98119
Gross RE (1953) The surgery of infancy and childhood. WB Saunders, Philadelphia, p 574
Redkar R, Karkera PJ, Krishnan J et al (2015) Subtotal pancreatectomy for congenital hyperinsulinism: our experience and review of literature. Indian J Surg 77:778–782. https://doi.org/10.1007/s12262-013-0999-9
Schönau E, Deeg KH, Huemmer HP et al (1991) Pancreatic growth and function following surgical treatment of nesidioblastosis in infancy. Eur J Pediatr 150:550–553. https://doi.org/10.1007/BF02072204
Meissner T, Wendel U, Burgard P et al (2003) Long-term follow-up of 114 patients with congenital hyperinsulinism. Eur J Endocrinol 149:43–51. https://doi.org/10.1530/eje.0.1490043
Schiller M, Krausz M, Meyer S et al (1980) Neonatal hyperinsulinism—surgical and pathologic considerations. J Pediatr Surg 15:16–20. https://doi.org/10.1016/S0022-3468(80)80395-2
Ackermann AM, Palladino AA (2015) Managing congenital hyperinsulinism: improving outcomes with a multidisciplinary approach. RRED 5:103–117. https://doi.org/10.2147/RRED.S56608
Lord K, Radcliffe J, Gallagher PR et al (2015) High risk of diabetes and neurobehavioral deficits in individuals with surgically treated hyperinsulinism. J Clin Endocrinol Metab 100:4133–4139. https://doi.org/10.1210/jc.2015-2539
Reyes GA, Fowler CL, Pokorny WJ (1993) Pancreatic anatomy in children: emphasis on its importance to pancreatectomy. J Pediatr Surg 28:712–715. https://doi.org/10.1016/0022-3468(93)90039-N
Stanley CA (2016) Perspective on the genetics and diagnosis of congenital hyperinsulinism disorders. J Clin Endocrinol Metab 101:815–826. https://doi.org/10.1210/jc.2015-3651
Mohamed Z, Arya VB, Hussain K (2012) Hyperinsulinaemic hypoglycaemia:genetic mechanisms, diagnosis and management. J Clin Res Pediatr Endocrinol 4:169–181. https://doi.org/10.4274/jcrpe.821
Demirbilek H, Hussain K (2017) Congenital hyperinsulinism: diagnosis and treatment update. J Clin Res Pediatr Endocrinol 9:69–87. https://doi.org/10.4274/jcrpe.2017.S007
Al-Agha AE, Ahmad IA (2013) Characterization of the ABCC8 gene mutation and phenotype in patients with congenital hyperinsulinism in western Saudi Arabia. Saudi Med J 34:1002–1006
Pierro A, Nah SA (2011) Surgical management of congenital hyperinsulinism of infancy. Semin Pediatr Surg 20:50–53. https://doi.org/10.1053/j.sempedsurg.2010.10.009
Hussain K (2008) Diagnosis and management of hyperinsulinaemic hypoglycaemia of infancy. Horm Res 69:2–13. https://doi.org/10.1159/000111789
Adzick NS, De Leon DD, States LJ et al (2019) Surgical treatment of congenital hyperinsulinism: Results from 500 pancreatectomies in neonates and children. J Pediatr Surg 54:27–32. https://doi.org/10.1016/j.jpedsurg.2018.10.030
Danowitz M, De Leon DD (2022) The role of GLP-1 signaling in hypoglycemia due to hyperinsulinism. Front Endocrinol 13:863184. https://doi.org/10.3389/fendo.2022.863184
Palladino AA, Sayed S, Levitt Katz LE et al (2009) Increased glucagon-like peptide-1 secretion and postprandial hypoglycemia in children after Nissen fundoplication. J Clin Endocrinol Metab 94:39–44. https://doi.org/10.1210/jc.2008-1263
Patti ME, McMahon G, Mun EC et al (2005) Severe hypoglycaemia post-gastric bypass requiring partial pancreatectomy: evidence for inappropriate insulin secretion and pancreatic islet hyperplasia. Diabetologia 48:2236–2240. https://doi.org/10.1007/s00125-005-1933-x
Bizzarri C, Cervoni M, Crea F et al (2011) Dumping syndrome: an unusual cause of severe hyperinsulinemic hypoglycemia in neurologically impaired children with gastrostomy. Minerva Pediatr 63:67–71
Acknowledgements
We would like to thank Qutaiba N. M. Shah Mardan, MBBS, MRCS(Eng) for proof reading and language assistance.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by [Ali Al-Ameer] and [Afrah Alsomali]. The first draft of the manuscript was written by [Ali Al-Ameer]. The final draft was reviewed and adjusted by [Zakaria Habib]. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Ethical approval was waived by the local Research Ethics Committee of King Faisal Specialist Hospital and Research Center in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
Informed consent
A waiver of informed consent was granted by the institutional review board for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Al-Ameer, A., Alsomali, A. & Habib, Z. Incidence, predictors and outcomes of redo pancreatectomy in infants with congenital hyperinsulinism: a 16-year tertiary center experience. Pediatr Surg Int 39, 183 (2023). https://doi.org/10.1007/s00383-023-05470-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s00383-023-05470-6