Abstract
Spinal teratomas are infrequent lesions in the pediatric population. These lesions can be extradural, intradural or intramedullary. We present a case of an 8-month-old boy that was assessed for underdevelopment of motor milestones. The neurologic examination revealed hyporeflexia, decreased sensation and flaccid paraplegia. MRI of the spine revealed two simultaneous and independent lesions in the extradural and intradural compartment. A laminectomy was performed for the T4-T7 vertebrae with total resection of both lesions. The histopathological analysis confirmed both lesions to be mature cystic teratomas. At the 1-year follow-up, the patient remained with no recovery of neurological function. A debate takes place regarding the etiology of formation of these lesions in the spine. The simultaneous presentation of two independent lesions in this patient could contribute to define the flawed migration of germ cells theory as the etiology for formation of teratomatous lesions in the spine.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Teratomas are lesions comprising tissues arising from all three germ cell layers: endoderm, mesoderm, and ectoderm, scarcely seen in the pediatric central nervous system (CNS) accounting for only 0.1% of tumors [1]. Spinal teratomas are rarely seen in children with only a few cases reported (Table 1). They can be located in the intradural, extradural or intramedullary regions with neurological symptoms arising from compression and displacement of nervous tissue [1, 2]. Whereas the morphological and histological composition of the lesions is very variable, most teratomas contain fatty tissue making it an imaging hallmark for its diagnosis and positioning magnetic resonance imaging (MRI) as the preferred tool for the evaluation of its features [3, 4]. The emergence of technology, surgical techniques and the growing experience of surgeons makes complete resection as the main goal for most spinal tumors and teratomas are no exception to this [5]. Although an established mechanism for the formation of spinal teratomas has not been elucidated, some theories are more prone to match its clinical and anatomical characteristics. The purpose of this paper is to present the case of an 8-month-old boy with two teratomas in the thoracic spine and to the best of our knowledge, this is the first report of two independent simultaneous extra and intradural teratomas in a pediatric patient. With this article, we aim to contribute to the available cases in the literature and seek to collaborate in the consolidation of its formation.
Case presentation
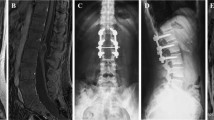
An 8-month-old male with a previous history of three episodes of urinary tract infections was admitted for in-patient care. During hospital stay, the patient was noted with underdevelopment of motor milestones, as he dragged his legs while crawling and was unable to stand. The rest of developmental milestones were appropriate according to age. The patient was referred to our department for further evaluation after hospital discharge. The neurologic examination revealed hyporeflexia, decreased sensation and flaccid paraplegia besides urinary symptoms. Magnetic resonance imaging (MRI) showed two medullary lesions in the thoracic region from T5–T8 with heterogeneous enhancement after administration of intravenous (IV) gadolinium (Fig. 1) and were described as two independent lesions. The first mass was described as an extradural fusiform lesion with fatty tissue components (Fig. 1). The intradural lesion appeared as an intradural extramedullary lobulated neoplasm with spinal cord, foraminal and dentate ligament compression. The lesion showed hyperintense regions in T1 sequence and heterogeneous characterization in T2 with no suppression in fat saturation imaging (Fig. 1).
Preoperative (A) T1-weighted and (B) T2- weighted images of the thoracic spine with evidence of the extradural (yellow arrow) and subdural (red arrow) lesions. Intraoperative ultrasonography depicting (C) extradural lesion in axial plane and (D) intradural lesion in sagittal plane. (E) Axial and (F) Sagittal planes with evidence of GTR of both lesions through intraoperative ultrasonography. (G) T1 and (H) T2-weighted postoperative images showing GTR of both lesions
A laminectomy was performed for the T4–T7 vertebrae revealing an extradural lesion composed of sebaceous material, hair and dental organs extending from T4 to T7 enclosed in a fibrous capsule (Fig. 2). Subsequent durotomy exposed a round sebaceous lesion of 10 mm diameter with compression of the spinal cord (Fig. 3).
Gross total resection (GTR) was achieved for both lesions with the aid of intraoperative ultrasonography (Fig. 1). Histopathological analysis of the tissue revealed both lesions to be comprised of mature elements from the three germ cell layers. None of the lesions showed features of malignancy. Final diagnosis was established as mature teratoma for both the extradural and intradural tumors. During hospital stay, the patient remained with no changes and was discharged 4 days later without complications. In the 1-year-follow-up, he remained with no motor nor sensitive function in the lower extremities; however, spasticity and hyperreflexia were noted. Recovery of neurological function in the lower extremities is yet to be determined in a longer-term follow-up.
Discussion
Spinal tumors account for 5.1% of all brain and other CNS neoplasms in the 0–19 years age group [5, 6]. Teratomas are rare neoplasms in the pediatric population and can arise in any location near the midline; still, patients in infancy or early childhood most often present these lesions in the sacrococcygeal region and only a few reported in the thoracic spine [7, 8]. Computed tomography (CT) can identify calcifications, fat and soft tissue but MRI remains as the gold standard imaging tool allowing better assessment of their morphology [9]. Teratomas need to go under histological differentiation to be classified in mature or immature [4], with the great majority of lesions exhibiting benign characteristics with 1 to 2% of these tumors undergoing malignant transformation [10]. The age of presentation is diverse and an early onset of the symptoms may be due to a congenital origin which would strictly imply its presence since gestation; however, its definition remains arbitrary and its use has been variable, utilizing it for lesions between 28 days to 12 months after birth [11].
We performed an analysis with available cases in the literature comparing age, location, degree of invasion, clinical manifestation, course of treatment and prognosis (Table 1).
Only one article was excluded due to incomplete clinical information. Children with thoracic teratomas presented with motor or sensitive neurological deficits that can improve completely or partially after surgical resection [9, 12,13,14,15,16,17,18]. Motor symptoms in infants were found to manifest as underdevelopment of motor milestones. In contrast, older children referred pain, numbness, and gait disturbances. Only one case was reported to present as recurrent aseptic meningitis due to rupture of the lesion [19]. Different variables influence the prognosis and presentation in each patient, such as age at onset of symptoms, size of the lesion, location and extra-spinal invasion of the lesion and concomitant malformations which may be directly related to spinal teratomas, but independent causality cannot be excluded (Table 1) [15, 16, 18, 20,21,22,23,24,25]. To the best of our knowledge, their relationship with teratomas is yet undetermined and we consider that further investigations with an accurate statistical methodology are still impeded due to the low number of cases reported to achieve statistical power and great heterogeneity between these. As in adults, functional prognosis in children is relatively good following surgery regardless of intramedullary, extradural or intradural location [20]. Preoperative assessment of invasion to nearby structures and degree of compression to neurovascular structures with MRI could help predict motor and sensitive outcomes in patients. Surgery is still the mainstay treatment for teratomas as histological typification is necessary. GTR is encouraged to seek remission and clinical recovery. Remaining lesions have a risk of relapse going up to 18% [22] and chemotherapy or radiotherapy should be reserved for tumors exhibiting immature or malignant components [3].
The etiology of spinal teratomas is still under discussion by different authors and no established cause is available. According to the notion of the misplaced germ cell theory, the improper location of multipotent germ cells during the neural tube migration may prompt the development of spinal teratoma; however, a second hypothesis establishes a dysembryonic causality for its origin [12, 23, 25]. Midline position, occasional intramedullary location and determination of the sex chromatin of these lesions make the misplaced germ cell theory the most likely etiology but certain limitations, such as the number of cases available for genomic studies, make it extremely difficult to determine [21, 24]. Our case is the first report of simultaneous teratomatous lesions in the spine of a pediatric patient in both the intradural and extradural compartment. This presentation makes us consider flawed migration of germ cells more likely to give origin to this lesion due to the presence of both lesions at the same level of the spine with structural independency between them giving an anatomical explanation rather than pure chance of pluripotential cell rests at the same level of the spine as a dysembryonic origin would require [25]. Dysembryonic theory is more supported by the presence of dysraphism and lesions in proximity with structures originating from the caudal cell mass [12]. Upper lesions without dysraphisms, as in this case, do not match with the associated characteristics and are better supported by flawed migration of germ cells.
We acknowledge that the discussion of a single case with these characteristics does not confirm nor reject any theory for its etiology, but we consider that it can open new lines of discussion for its resolution. Limitations to this study include the interpretation of possible outcomes with a limited pool of patients as well as the lack of a statistical analysis. Further reports in the field may contribute to a more accurate evaluation of the data and the elaboration of more robust reviews. Multicentric collaborations of possible nonreported cases could help to pool a greater number of patients and analyze the characteristics of these lesions to explore the relationship between spinal teratomas and the cell migration theory, or in the other case, explore alternative mechanisms.
Conclusion
Spinal teratomas are rare neoplasms in children with high heterogeneity in its location and extension. The lack of a proper statistical analysis precludes the objective analysis of outcomes and prognosis among patients. Surgical treatment should be performed as it can provide improvement and allow histological analysis for its typification. This is the first reported case of an extradural and intradural teratoma in a pediatric patient. The simultaneous presentation of two lesions in the extradural and intradural compartment with no relationship or connection between them can help to elucidate and further analyze the theory of its etiology.
Data Availability
Data is provided within the manuscript or supplementary information files
References
Işık N, Balak N, Silav G, Elmaci I (2008) Pediatric intramedullary teratomas. Neuropediatrics 39:196–199. https://doi.org/10.1055/s-0028-1112116
Frühwald MC, Rutkowski S (2011) Tumors of the central nervous system in children and adolescents. Dtsch Ärztebl Int. https://doi.org/10.3238/arztebl.2011.0390
Goyal N, Kakkar A, Singh PK et al (2013) Intracranial teratomas in children: a clinicopathological study. Childs Nerv Syst 29:2035–2042. https://doi.org/10.1007/s00381-013-2091-y
Peterson CM, Buckley C, Holley S, Menias CO (2012) Teratomas: a multimodality review. Curr Probl Diagn Radiol 41:210–219. https://doi.org/10.1067/j.cpradiol.2012.02.001
Shimony N, Fehnel K, Abbott IR, Jallo GI (2023) The evolution of spinal cord surgery: history, people, instruments, and results. Childs Nerv Syst 39:2687–2700. https://doi.org/10.1007/s00381-023-06128-3
Ostrom QT, Patil N, Cioffi G et al (2020) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013–2017. Neuro-Oncol 22:iv1–iv96. https://doi.org/10.1093/neuonc/noaa200
Barksdale EM, Obokhare I (2009) Teratomas in infants and children. Curr Opin Pediatr 21:344–349. https://doi.org/10.1097/MOP.0b013e32832b41ee
Azizkhan RG, Caty MG (1996) Teratomas in childhood: Curr Opin Pediatr 8:287–292. https://doi.org/10.1097/00008480-199606000-00018
Basmaci M, Hasturk AE, Pak I (2011) Cystic mature teratoma of the thoracic region in a child: an unusual case. J Neurosci Rural Pract 02:186–189. https://doi.org/10.4103/0976-3147.83591
Guo J-X, Zhao J-G, Bao Y-N (2022) Adult sacrococcygeal teratoma: a review Medicine (Baltimore) 101:e32410. https://doi.org/10.1097/MD.0000000000032410
Hwang SW, Su JM, Jea A (2012) Diagnosis and management of brain and spinal cord tumors in the neonate. Semin Fetal Neonatal Med 17:202–206. https://doi.org/10.1016/j.siny.2012.03.001
Park S, Kim K, Wang K-C et al (2010) Spinal epidural teratoma: review of spinal teratoma with consideration on the pathogenesis: case report. Neurosurgery 67:E1818–E1825. https://doi.org/10.1227/NEU.0b013e3181f846ca
Acar O, Güney Ö, Eser O (2003) Epidural thoracal teratoma in a 7-month-old girl. Childs Nerv Syst 19:126–128. https://doi.org/10.1007/s00381-002-0681-1
Choi S-J, Choi H-J, Hong J-T et al (2004) Intraspinal extradural teratoma mimicking neural sheath tumor in infant. Childs Nerv Syst 20:123–126. https://doi.org/10.1007/s00381-003-0821-2
Kaneko M, Ohkawa H, Iwakawa M, Ikebukuro K (1999) Extensive epidural teratoma in early infancy treated by multi-stage surgery. Pediatr Surg Int 15:280–283. https://doi.org/10.1007/s003830050578
Murovic JA, De Armond S, Nagashima T et al (1986) Cell kinetics analysis in a case of teratoma of the thoracic spine. J Neurosurg 65:331–334. https://doi.org/10.3171/jns.1986.65.3.0331
Quon JL, Grant RA, Huttner AJ, Duncan CC (2014) Thoracic epidural teratoma: case report and review of the literature. Clin Med Insights Pathol 7:CPath.S14723. https://doi.org/10.4137/CPath.S14723
Deckey DG, Fernandez A, Lara NJ et al (2021) Pediatric extramedullary epidural spinal teratomas: a case report and review of the literature. Case Rep Orthop 2021:1–8. https://doi.org/10.1155/2021/6702972
Mpayo LL, Liu X-H, Xu M et al (2014) Extramedullary spinal teratoma presenting with recurrent aseptic meningitis. Pediatr Neurol 50:655–657. https://doi.org/10.1016/j.pediatrneurol.2014.02.005
Schmidt RF, Casey JP, Gandhe AR et al (2017) Teratoma of the spinal cord in an adult: report of a rare case and review of the literature. J Clin Neurosci 36:59–63. https://doi.org/10.1016/j.jocn.2016.10.022
Oosterhuis JW, Stoop H, Honecker F, Looijenga LHJ (2007) Why human extragonadal germ cell tumours occur in the midline of the body: old concepts, new perspectives. Int J Androl 30:256–264. https://doi.org/10.1111/j.1365-2605.2007.00793.x
Göbel U, Calaminus G, Engert J et al (1998) Teratomas in infancy and childhood. Med Pediatr Oncol 31:8–15. https://doi.org/10.1002/(SICI)1096-911X(199807)31:1%3c8::AID-MPO2%3e3.0.CO;2-H
Rewcastle NB (1964) Teratomatous cysts of the spinal canal: with “sex chromatin” studies. Arch Neurol 11:91. https://doi.org/10.1001/archneur.1964.00460190095007
al-Sarraj, Parmar, Dean, et al (1998) Clinicopathological study of seven cases of spinal cord teratoma: a possible germ cell origin. Histopathology 32:51–56. https://doi.org/10.1046/j.1365-2559.1998.00324.x
Koen JL, McLendon RE, George TM (1998) Intradural spinal teratoma: evidence for a dysembryogenic origin: report of four cases. J Neurosurg 89:844–851. https://doi.org/10.3171/jns.1998.89.5.0844
Kahn M, Iacob S, Bach SE et al (2022) A rare neonatal intramedullary immature teratoma undiagnosed in utero: a case report. Pediatr Neurosurg 57:50–55. https://doi.org/10.1159/000519768
Ishiguro S, Nagashima T, Yamamoto K et al (2011) An infantile large and extensive intramedullary mature spinal teratoma -case report-: —case report—. Neurol Med Chir (Tokyo) 51:393–396. https://doi.org/10.2176/nmc.51.393
Yoshioka F, Shimokawa S, Masuoka J et al (2018) Extensive spinal epidural immature teratoma in an infant: case report. J Neurosurg Pediatr 22:411–415. https://doi.org/10.3171/2018.4.PEDS17676
Shimauchi M, Yamakawa Y, Maruoka N, Hamada K (1988) Intramedullary teratoma of the thoracic spinal cord associated with anomalies of the vertebrae and ribs: —case report—. Neurol Med Chir (Tokyo) 28:1005–1009. https://doi.org/10.2176/nmc.28.1005
Senayli A, Deniz FE, Sezer T et al (2004) A case of bladder exstrophy and thoracal intramedullary teratoma. J Pediatr Surg 39:e9–e11. https://doi.org/10.1016/j.jpedsurg.2004.05.036
Gressot LV, Mohila CA, Jea A et al (2014) Cervicothoracic nonterminal myelocystocele with mature teratoma: case report. J Neurosurg Pediatr 13:204–208. https://doi.org/10.3171/2013.12.PEDS13408
Rainey SC, Kandikattu BS (2018) Progressive feeding intolerance secondary to a congenital spinal teratoma in a four-week-old female. Cureus. https://doi.org/10.7759/cureus.3281
Sancak R, Dagdemir A, Tasdemir HA et al (2001) A thoracal spinal cord teratoma associated with Taussig-Bing anomaly in a newborn. Teratology 63:77–78. https://doi.org/10.1002/1096-9926(200102)63:2%3c77::AID-TERA1012%3e3.0.CO;2-C
Funding
No funding was obtained for this study.
Author information
Authors and Affiliations
Contributions
Omar R. Ortega-Ruiz wrote the original draft as well as edition of the manuscript. Performed the research and analysis of the data. Carlos D. Acevedo-Castillo: writing of the original draft. Luis Alejandro Pérez-Ruano edited and reviewed the manuscript and the preparation of the figures shown in the manuscript. Enrique Caro-Osorio edited and reviewed the manuscript and should be recognized for the administration of the project.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial nor any non-financial competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ortega-Ruiz, O.R., Acevedo-Castillo, C.D., Pérez‑Ruano, L.A. et al. Simultaneous spine extradural and intradural teratomas in a pediatric patient: A rare presentation with insights in the flawed migration of germ cells theory. Childs Nerv Syst 40, 1011–1017 (2024). https://doi.org/10.1007/s00381-024-06317-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-024-06317-8