Abstract
Arachnoid cysts are usually asymptomatic, benign lesions commonly occurring in the middle cranial fossa. However, the cysts may rupture in rare cases causing intracystic or subdural hemorrhages with significant mass effect. We report two cases of middle cranial fossa arachnoid cyst with subdural hemorrhage with very different clinical course. The first case presented with significant mass effect with cerebral herniation and had significant neurological morbidity post-surgery. The second case had minimal symptoms and was managed conservatively with offer of elective surgery. The report underscores the importance of prompt diagnosis and appropriate surgical intervention in managing arachnoid cysts with hemorrhage, highlighting the potential for diverse clinical presentations and outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Arachnoid cysts are benign, cystic, intracranial space-occupying lesions (SOL) that occur in children and young adults [1]. They are usually congenital in origin, however, some cases of acquired arachnoid cysts have been reported in literature [2]. Arachnoid cysts most commonly occur in the middle cranial fossa and have a male preponderance [1]. They are usually asymptomatic but sometimes can cause headaches and bony remodeling. Rarely, the arachnoid cysts can rupture and present with intracystic or subdural hemorrhage (SDH). Features of raised intracranial pressure may be present in these cases and warrant urgent surgery. Herein, we present two cases of subdural hemorrhage in middle cranial fossa arachnoid cyst in children with very different clinical courses.
Case 1
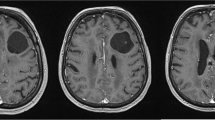
A 12-year-old boy was brought to the emergency department of our hospital with a history of headache for 2 days associated with multiple episodes of vomiting. He had history of generalized tonic–clonic seizure about 6 h before presentation. He did not have any history of trauma, or fever. On examination, his GCS was E1V2M3, right-sided blown pupil. He was not moving his right-sided limbs on stimulation. Non-contrast computed tomography (NCCT) scan done at presentation showed the presence of a subdural hemorrhage on the right side with an SOL containing haemorrhagic fluid in the right middle cranial fossa region with thinning of the overlying temporal bone (Fig. 1A). A hypodense area in the right posterior cerebral artery (PCA) region was present. A diagnosis of a ruptured arachnoid cyst (Galassi type II) with subdural hemorrhage and uncal herniation causing PCA infarct was made.
A Case 1: NCCT showing subacute subdural hemorrhage with hemorrhage within the cyst and significant midline shift. B Case 1: postoperative NCCT showing the decompressive craniectomy status with relief of mass effect. C Case 1: diffusion weighted scan showing infarcts in the right PCA territory, splenium of corpus callosum, and posterior limb of right internal capsule. D Case 2: NCCT showing a subacute subdural hemorrhage in right frontal region. E Case 2: NCCT showing the right middle cranial fossa arachnoid cyst. F Case 2: T2 weighted MRI showing the right middle cranial fossa arachnoid cyst with a vessel coursing along it
He was taken up for urgent right-sided decompressive craniectomy. The SDH was evacuated and the cyst fenestrated. The decision of decompressive craniectomy was taken considering that the patient had already herniated with a developing PCA infarct. Post-operatively, the patient had improvement in his GCS to E4VtM6, however, he required a tracheostomy and had right-sided hemiparesis. Post-operative magnetic resonance imaging (MRI) showed the presence of an infarct in the right PCA territory, splenium of the corpus callosum, posterior limb of the right internal capsule and left midbrain (Fig. 1B, 1C). The rehabilitation was continued and he was discharged to home care.
Case 2
A 14-year-old boy presented to the emergency department with a history of fall while playing basketball 1 day back. Following the fall, he was disoriented for about an hour. He did not have any history of loss of consciousness, convulsion, or limb weakness. On examination, his GCS was E4V5M6, pupils were bilaterally equal and reacting to light. He did not have any focal neurodeficit. NCCT and MRI of the head showed the presence of a subacute right subdural hemorrhage along with a right middle cranial fossa arachnoid cyst (Galassi type I) (Fig. 1D–F). There was no evidence of hemorrhage within the cyst. The patient was managed conservatively in the present admission with later elective surgery offered.
Discussion
Arachnoid cysts are intra-arachnoidal fluid-filled malformations. Their prevalence within the general population is about 1.2% with a male preponderance [3]. Prevalence in children is higher than the general population at about 2.6% with similar male preponderance as in the general population [4]. The cause of formation of arachnoid cysts is not known clearly. Congenital splitting of the arachnoid membrane during the fetal period is considered as the likely causative factor. In the particular case of middle cranial fossa arachnoid cysts, the likely mechanism is the failure of fusion of the embryonic frontal and temporal meninges resulting in duplication of arachnoid within the Sylvian fissure [5]. Arachnoid cysts usually expand in size by secretion of fluid secondary to an osmotic gradient or due to the presence of a ball valve mechanism that allows the ingress of fluid but prevents egress [6]. Middle cranial fossa is noted as the common location of intracranial arachnoid cysts followed by the posterior fossa [5, 6]. Galassi et al. has classified middle cranial fossa arachnoid cysts into three types [7]. Type I cysts are small spindle-shaped cysts, type II cysts are moderate-sized cysts, and type III cysts are large cysts filling the whole of the middle cranial fossa.
Most arachnoid cysts are asymptomatic, about 6.8% of arachnoid cysts are symptomatic in the pediatric population [5]. Symptomatic arachnoid cysts most commonly present with macrocephaly, headache, hydrocephalus, and increased intracranial pressure. Rarely, arachnoid cysts can present with intracystic hemorrhage or subdural hemorrhage [6]. Hemorrhage most commonly occurs in middle cranial fossa arachnoid cysts, particularly of the type II variety. The mechanism of hemorrhage in these cysts is not clear. Risk factors identified are head trauma and the large size of the cysts [6]. Two putative mechanisms have been proposed, firstly, unsupported bridging veins on the wall of the arachnoid cyst are prone to rupture causing hemorrhage. Secondly, the arachnoid cyst is ruptured causing a subdural effusion in which subsequent head trauma causes a subdural hemorrhage. In the literature review by Liu et al., the hemorrhage was most commonly subacute or chronic [8]. The presentation was headache and vomiting suggestive of raised intracranial pressure. Cases presenting with seizures have also been reported in the literature. Cress et al. have reported that arachnoid cysts with a diameter of 5 cm or more have a 16.5 times more likelihood of rupture or hemorrhage [9].
The diagnosis is confirmed with the help of a CT scan or MRI [6]. Depending on the duration of presentation, the hemorrhage may be hyperdense, hypodense, or isodense. In delayed presentation, the isodense or hypodense subdural hemorrhage may make it difficult to identify the arachnoid cyst, and an MRI may be helpful. Ochi et al. have suggested that thinning of the overlying temporal bone is diagnostic of middle cranial fossa arachnoid cysts in such cases [10].
Surgery is the treatment of choice in arachnoid cysts with hemorrhage. The three main surgical options for the management of arachnoid cysts are microsurgical fenestration, open fenestration, and cystoperitoneal shunting. Cystoperitoneal shunting can cause overdrainage, shunt dependency and the hemorrhagic component is likely to cause shunt blockage. Cyst fenestration, either microsurgically or endoscopically is considered to be the initial surgical treatment of choice [11]. In a meta-analysis conducted by Chen et al., the rate of symptom improvement was comparable between patients undergoing microsurgical fenestration and those undergoing endoscopic fenestration [12]. However, the rate of reduction in cyst size was slightly less in the endoscopic fenestration group compared to microscopic group but the difference was not statistically significant. Microscopic fenestration on the other hand, had a much more incidence of short-term post-surgical complications in the form of subdural fluid collections. Some authors have proposed that burr hole drainage of the SDH is adequate as it relieves the symptoms of raised ICP. However, only burr hole drainage is associated with a re-surgery in a significant number of patients [8]. Liu et al. suggest craniotomy and evacuation of subdural hemorrhage, excision of the cyst and establishing communication between cyst and CSF cisterns prevent rebleed and relieve mass effect [8]. In the first case described, we performed a decompressive craniectomy with microscopic cyst fenestration considering the emergent presentation of the patient with signs of herniation and possibility of PCA infarct secondary to herniation.
Conclusion
Subdural hemorrhage in an arachnoid cyst is a rare occurrence but it can cause significant morbidity and neurological impairment if the disease process is left untreated. Prompt recognition and treatment should be undertaken to achieve an optimum outcome.
References
Albuquerque FC, Giannotta SL (1997) Arachnoid cyst rupture producing subdural hygroma and intracranial hypertension. Neurosurgery 41(4):951–956
Guzel A, Tatli M, Kilincer C, Yilmaz F (2007) Posttraumatic intraventricular arachnoid cyst accompanied by pseudo meningoencephalocele in a child. J Clin Neurosci 14(12):1210–1213
Weber F (2018) The prevalence of intracranial arachnoid cysts. InArachnoid Cysts (pp. 95–100). Academic Press
Al-Holou WN, Yew AY, Boomsaad ZE, Garton HJ, Muraszko KM, Maher CO (2010) Prevalence and natural history of arachnoid cysts in children. J Neurosurg Pediatr 5(6):578–585
Wester K (1999) Peculiarities of intracranial arachnoid cysts: location, sidedness, and sex distribution in 126 consecutive patients. Neurosurgery 45(4):775
Jafrani R, Raskin JS, Kaufman A, Lam S (2019) Intracranial arachnoid cysts: pediatric neurosurgery update. Surg Neurol Int 10
Galassi E, Tognetti F, Gaist G, Fagioli L, Frank F, Frank G (1982) CT scan and metrizamide CT cisternography in arachnoid cysts of the middle cranial fossa: classification and pathophysiological aspects. Surg Neurol 17(5):363–369
Liu Z, Xu P, Li Q, Liu H, Chen N, Xu J (2014) Arachnoid cysts with subdural hematoma or intracystic hemorrhage in children. Pediatr Emerg Care 30(5):345–351
Cress M, Kestle JR, Holubkov R, Riva-Cambrin J (2013) Risk factors for pediatric arachnoid cyst rupture/hemorrhage: a case-control study. Neurosurgery 72(5):716–722
Ochi M, Morikawa M, Ogino A, Nagaoki K, Hayashi K (1996) Supratentorial arachnoid cyst and associated subdural hematoma: neuroradiologic studies. Eur Radiol 6:640–644
Choi JW, Lee JY, Phi JH, Kim SK, Wang KC (2015) Stricter indications are recommended for fenestration surgery in intracranial arachnoid cysts of children. Childs Nerv Syst 31:77–86
Chen Y, Fang HJ, Li ZF, Yu SY, Li CZ, Wu ZB, Zhang YZ (2016) Treatment of middle cranial fossa arachnoid cysts: a systematic review and meta-analysis. World Neurosurg 1(92):480–490
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Dr. Debajyoti Datta and Dr. Debajyoti Pathak. The first draft of the manuscript was written by Dr. Debajyoti Datta and all authors commented on the previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent to participate
Written informed consent was obtained from the parents for publication.
Conflict of interest
The authors did not receive support from any organization for the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Datta, D., Pathak, D., Ghosh, P. et al. Subdural hemorrhage in middle cranial fossa arachnoid cysts: a report of two cases at two ends of the spectrum. Childs Nerv Syst 40, 263–266 (2024). https://doi.org/10.1007/s00381-023-06107-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-023-06107-8