Abstract
Purpose
The treatment of patients with multisuture craniosynostosis is complex and patient-dependent. Cranial distraction osteogenesis is a relatively new procedure for treatment of these patients, with its use increasing in many centers. With this increased use comes an expanding range of indications. Surgical management of multisuture craniosynostosis in therapeutically immunosuppressed patients following a solid organ transplant presents unique challenges. We describe our experience with posterior cranial vault distraction in two patients with multisuture craniosynostosis that had previously undergone organ transplantation.
Methods
Two solid-organ transplant recipient patients with multisuture craniosynostosis were identified. A detailed examination of their medical/transplant history and perioperative details were recorded.
Results
The first patient was a 3-year-old girl who received a kidney transplantation in infancy and subsequently presented with a symptomatic Chiari malformation and papilledema. Imaging revealed pansynostosis. She underwent posterior cranial vault distraction extending into a Chiari decompression. Her postoperative course was complicated by distractor site infection at the beginning of consolidation, necessitating early removal of distractors. The second patient was a 2-year-old boy who received a heart transplantation at the age of 3 months and subsequently presented with head shape concerns. Imaging revealed bicoronal and sagittal craniosynostosis. He underwent a posterior cranial vault distraction without complication. Following removal of the distractors, he developed an infection at one of the distractor sites with associated fever and leukocytosis, necessitating washout and drain placement. Both patients achieved successful cranial vault expansion with distraction osteogenesis and at a 2-year follow-up do not have evidence of elevated intracranial pressure.
Conclusions
Immunosuppressive therapy has the potential to inhibit wound healing and place patients at risk for wound infection. Although we have demonstrated successful cranial vault expansion with distraction in two immunosuppressed children, extra care must be taken with these patients when placing semi-buried hardware. Specifically, prompt identification and proactive management of potential infectious complications is critical to applying this technique safely in these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Craniosynostosis is a condition characterized by premature fusion of the cranial sutures with negative sequelae on the health of those affected, including cranial dysmorphology, strabismus, facial asymmetry, and the risk of elevated intracranial pressure, vision changes, headaches, and developmental delay [1, 2]. These effects are correlated with localized and/or global cranial growth restrictions. Patients with multisuture craniosynostosis are at higher risk of developing elevated intracranial pressure than their single suture counterparts [1]. Posterior cranial vault distraction osteogenesis achieves greater intracranial volume expansion compared to fronto-orbital advancement [3]. Our institution performs posterior cranial vault distraction to treat patients with multisuture craniosynostosis in order to effect maximal cranial vault expansion, and thus treat existing or decrease the risk of elevated intracranial pressure development in these patients.
The applications of posterior cranial vault distraction to different populations with multisuture craniosynostosis are expanding [4, 5]. However, there is no information in the literature regarding the management of solid-organ transplant recipients who are subsequently diagnosed with multisuture craniosynostosis.
The immunosuppressive nature of transplant medications raises concern for both impaired healing and development of infection. Thus, we must take special care when performing surgery on solid organ transplant patients who are on chronic immunosuppressive therapy. The safety of elective surgery among solid organ transplant patients is mixed. While one study indicates that kidney transplant recipients experience a greater proportion of complications when undergoing elective surgery [6], another concludes that performing elective esthetic surgery in solid organ transplant recipients can be safely performed with close coordination with the patient’s medical team [7]. Given the paucity of information regarding this unique patient population, we seek to share our institutional experience. We herein describe our experience with two solid organ transplant recipients on therapeutic immunosuppressive medications who underwent cranial vault expansion surgery via distraction osteogenesis to treat multisuture craniosynostosis.
Patient 1
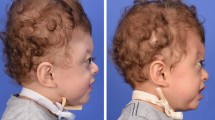
The first patient was a 3-year-old girl who presented to ophthalmology due to a six-month history of worsening headaches. She demonstrated a relatively normocephalic head shape with sloping of the forehead posteriorly (Figs. 1 and 2). On fundoscopic examination, she was found to have papilledema and was sent to the emergency department for imaging. Magnetic resonance imaging demonstrated a Chiari I malformation and skull computed tomography revealed pansynostosis (Fig. 3). The patient had undergone a renal transplantation at age two for anoxic renal injury sustained at birth. The patient’s immunosuppressive regimen consisted of mycophenolate, tacrolimus, and prednisone. The multidisciplinary recommendation from pediatric nephrology, neurosurgery, and craniofacial surgery was to proceed with posterior cranial vault distraction to relieve the elevated intracranial pressure and to maximize intracranial expansion. It was felt that a fronto-orbital advancement would not provide as much intracranial space as a posterior cranial vault distraction. Preoperatively, the patient was started on a standard regimen of Vitamin A to mitigate the wound healing effects of her prednisone regimen per the nephrology team [8]. A posterior cranial vault distraction was performed as previously described in the literature [9], except with the inferoposterior aspect of the osteotomy extending into an occipital bone Chiari decompression. Distraction was begun on postoperative day 4, and the patient was discharged on postoperative day 6. Distraction proceeded uneventfully at 1 mm/day until a distance of 34 mm was reached (Fig. 4). One week into the consolidation phase, the patient was seen in the clinic with concern for purulent drainage from the right distractor site. She was admitted for intravenous antibiotics and the distractors were removed the next day with placement of spanning plates to maintain the achieved cranial expansion. Intraoperative cultures grew Staphylococcus aureus and Enterobacter cloacae. She was discharged on culture-directed oral antibiotics per the infectious disease team. She presented 3 weeks after discharge from this hospitalization with erythema and a collection around the right side of the closed incision. She was again taken to the operating room for washout and removal of spanning plates. Cultures at this time were positive for Staphylococcus aureus and Candida parapsilosis. The patient was discharged several days later with appropriate antimicrobial coverage. She is currently doing well 2 years postoperatively with no return of papilledema and a relatively normocephalic head shape (Figs. 5 and 6).
Patient 2
Patient 2 was a 3-year-old boy who was referred to plastic surgery for evaluation of brachycephaly. Computer tomography revealed bicoronal and sagittal craniosynostosis. The patient had undergone an orthotopic heart transplant at four months of age for hypoplastic right ventricle and pulmonary atresia with intact ventricular septum. Prior to his transplant, he had undergone placement of Blalock-Taussig shunt, patent ductus arteriosus ligation, and atrial septectomy. The patient’s post-transplant immunosuppressive pharmacotherapy included mycophenolate and tacrolimus. A comprehensive microarray revealed no genetic abnormalities. The multidisciplinary recommendation from pediatric cardiology, pediatric cardiothoracic surgery, neurosurgery, and craniofacial surgery was to proceed with posterior cranial vault distraction to maximize intracranial expansion given the increased risk of developing intracranial hypertension with multisutural craniosynostosis. He underwent successful posterior craniotomy and cranial distractor placement. Distraction was begun on postoperative day 4, and the patient was discharged on postoperative day 6. Distraction proceeded uneventfully at 1 mm/day until a distance of 28 mm was reached. The distractors were removed after a 2-month consolidation period. Several days following the distractor removal a fluid collection was noted around one of the distractor sites with developing erythema. The patient was taken back to the operating room for formal washout of the surgical sites, and placement of subcutaneous drains. Intraoperative cultures grew Serratia marcescens. After recovering, the patient was discharged on appropriate antimicrobial coverage and healed without incident. He is currently doing well two years postoperatively with no return of papilledema and a relatively normocephalic head shape.
Discussion
We have presented two pediatric patients with multisuture craniosynostosis diagnosed after receiving a solid organ transplantation. We treated these patients within a 1-month time frame. Although their close presentation may have been a coincidence, it was an unusual occurrence in our experience and warrants dedicated description and discussion.
The incidence of craniosynostosis specifically among pediatric solid organ transplantation recipients has not been investigated or reported in the literature. We would presume that the rate of single-suture craniosynostosis among solid organ transplant patients would reflect those of the general population. Although multisuture craniosynostosis is known to occur with particular syndromes (e.g., Crouzon, Apert, Pfeiffer, Muenke, Sathre-Chotzen), neither of our patients had these diagnoses. Genetic testing revealed no defined abnormality.
There are a few surgical approaches to expanding the cranial vault among patients with multisuture craniosynostosis. Historically, patients with multisuture craniosynostosis associated with a syndrome were treated with cranial vault expansion in the form of a bifrontal orbital advancement [10,11,12]. However, cephalometric and cranial volumetric analyses demonstrate a greater volume of expansion with posterior vault remodeling and distraction as compared to anterior vault remodeling [3, 13,14,15,16,17,18,19,20,21]. The technique of vault distraction maximizes intracranial expansion through the gradual stretch of soft tissue, while harnessing the body’s ability to make bone through the process of distraction osteogenesis. This approach effectively treats existing and prevents the development of elevated intracranial pressure. In addition, this technique provides a stable cranial expansion that does not tend to relapse over time and minimizes the need for a repeat cranial vault expansion in the future. For these reasons, we treat patients with multisuture craniosynostosis at our institution with posterior vault distraction osteogenesis.
The application of posterior vault distraction in an immunocompromised patient presents a particular challenge in that the use of semiburied distractors results in prolonged communication between the environment and the surgical field. This results in an increased risk of surgical site infection in an already vulnerable patient. In both cases presented in this report, extensive multidisciplinary discussion was held between the plastic surgeon, neurosurgeon, the transplant team, and the family. Together, we prioritized maximal cranial expansion by proceeding with cranial vault distraction osteogenesis, while accepting the increased risk of infectious complications.
Successful cranial vault expansion with placement of semiburied distractors was successfully completed in both patients. Not only was the desired amount of expansion achieved, but both patients currently do not have evidence of elevated pressure at a 2-year follow-up. In addition, they have acceptable cranial morphologies. Despite these successes, both patients suffered infectious complications at some point during their course. The first patient had a unilateral infection while the distractors were still in place, and again 3 weeks after the distractors were removed. The second patient only had a unilateral infection after the distractors were removed. Both patients were on prolonged intravenous and subsequently oral antibiotics to fully treat the infections once the hardware was removed. At the authors’ institution, it is very uncommon to experience an infectious complication when performing posterior cranial vault distraction. Erythema and irritation around the distractor entry point of the scalp is common and frequently resolves with expectant management. The reported rate of distractor site infection in the literature is just under 7% [22]. Although many localized distractor site infections can be treated safely with oral antibiotics and local wound care among non-immunosuppressed patients, infections in transplant patients while on immunosuppressive therapy are unlikely amenable to this approach. Although both patients in this report underwent an unplanned surgical operation to treat localized infections at the distractor sites, the authors strongly feel that prompt diagnosis and surgical/medical intervention was the key to their successful treatment. Despite these infectious complications, both patients healed and maintained their solid organ transplant function throughout their course and did not require a cessation of immunosuppressive medications.
Minimizing the possibility of infection in immunosuppressed patients undergoing cranial distraction osteogenesis is a challenge. Reducing the immunosuppressive therapy, changing it, or holding it would potentially jeopardize the viability of the solid organ. In these two patients, this was not an option. Other attempts were made to minimize the risk of surgical site infection: First, Vitamin A was administered to mitigate the negative wound healing effects from corticosteroids in the first patient. Second, these patients were followed much closer throughout their postoperative course due to the team’s concern for infectious complications. Another possible prevention strategy would be to administer a longer prophylactic antibiotic course while the distractors are in place. This strategy has the clear disadvantage of encouraging multidrug-resistant organism growth, and the possibility of medication-related side effects. Finally, the most promising technologic advancement, would be the development and application of a completely buried cranial distractor in lieu of the semi-buried distractor. The availability of a completely buried distractor would allow the posterior vault distraction technique to be utilized, while minimizing the infectious risk associated with a semiburied distractor, particularly in immunosuppressed patients.
Given the experience with these patients, the following recommendations are put forth for managing cranial distraction in immunocompromised patients: (1) prophylactic perioperative administration of 1st-generation cephalosporin (or similar antibiotic if allergy) with continuation for 72 h postoperatively (institution protocol). (2) Placement of subgaleal drain. (3) Head wrap removal on postoperative day 2 with initiation of twice daily cleansing of distractor exit points with half strength hydrogen peroxide followed by application of bacitracin and removal of drain when daily output is less than 30 mL. (4) Weekly follow-up with patients until removal of distractors. (5) Removal of activation arms at the conclusion of activation phase to decrease the external hardware burden. (6) Removal of distractors after a 2-month consolidation period with liberal use of drains at the distractor sites to discourage fluid collection.
Conclusion
Cranial vault expansion with distraction osteogenesis can be effectively applied in immunosuppressed pediatric patients who have undergone a solid organ transplant. However, both patients in our series suffered infectious complications at the surgical site requiring surgical intervention and prolonged antibiotic course. Steps taken to minimize the risk of infectious complications, as well as the development of buried cranial distractors, will allow for optimized application of cranial vault distraction in the immunosuppressed pediatric population.
Data availability
Not applicable
Code availability
Not applicable
References
Renier D, Sainte-Rose C, Marchac D, Hirsch JF (1982) Intracranial pressure in craniostenosis. J Neurosurg 57:370–377. https://doi.org/10.3171/jns.1982.57.3.0370
Kapp-Simon KA, Speltz ML, Cunningham ML, Patel PK, Tomita T (2007) Neurodevelopment of children with single suture craniosynostosis: a review. Childs Nerv Syst 23:269–281
Derderian CA, Wink JD, McGrath JL et al (2015) Volumetric changes in cranial vault expansion: comparison of fronto-orbital advancement and posterior cranial vault distraction osteogenesis. Plast Reconstr Surg 135:1665–1672. https://doi.org/10.1097/PRS.0000000000001294
Lin LO, Zhang RS, Hoppe IC, Paliga JT, Swanson JW, Bartlett SP, Taylor JA (2019) Onset and resolution of Chiari malformations and hydrocephalus in syndromic craniosynostosis following posterior vault distraction. Plast Reconstr Surg 144:932–940. https://doi.org/10.1097/PRS.0000000000006041
Li J, Gerety PA, Xu W, Bartlett SP, Taylor JA (2016) A perioperative risk comparison of posterior vault distraction osteogenesis in an older pediatric population. J Craniofac Surg 27:1165–1169. https://doi.org/10.1097/SCS.0000000000002795
Palamuthusingam D, Kunarajah K, Pascoe EM, Johnson DW, Hawley CM, Fahim M (2020) Postoperative outcomes of kidney transplant recipients undergoing non-transplant-related elective surgery: a systematic review and meta-analysis. BMC Nephrol 21:21. https://doi.org/10.1186/s12882-020-01978-4
Vandegrift MT, Nahai F (2016) Is aesthetic surgery safe in the solid organ transplant patient? An international survey and review. Aesthet Surg J 36
Polcz ME, Barbul A (2019) The role of vitamin A in wound healing. Nutr Clin Pract 34:695–700
White N, Evans M, Dover S et al (2009) Posterior calvarial vault expansion using distraction osteogenesis. Childs Nerv Syst 25:231–236. https://doi.org/10.1007/s00381-008-0758-6
Taylor JA, Bartlett SP (2017) What’s new in syndromic craniosynostosis surgery? Plast Reconstr Surg 140:82e–93e. https://doi.org/10.1097/PRS.0000000000003524
Zhang RS, Wes AM, Naran S, Hoppe IC, Sun J, Mazzaferro D, Bartlett SP, Taylor JA (2018) Posterior vault distraction osteogenesis in nonsyndromic patients: an evaluation of indications and safety. J Craniofac Surg 29:566–571. https://doi.org/10.1097/SCS.0000000000004230
Derderian CA, Bartlett SP (2012) Open cranial vault remodeling. J Craniofac Surg 23:229–234. https://doi.org/10.1097/scs.0b013e318241b93a
Nowinski D, di Rocco F, Renier D, SainteRose C, Leikola J, Arnaud E (2012) Posterior cranial vault expansion in the treatment of craniosynostosis. Comparison of current techniques. Child’s Nervous System 28:1537–1544. https://doi.org/10.1007/s00381-012-1809-6
Choi M, Flores RL, Havlik RJ (2012) Volumetric analysis of anterior versus posterior cranial vault expansion in patients with syndromic craniosynostosis. J Craniofac Surg 23:455–458
Goldstein JA, Paliga JT, Wink JD, Low DW, Bartlett SP, Taylor JA (2013) A craniometric analysis of posterior cranial vault distraction osteogenesis. Plast Reconstr Surg 131:1367–1375. https://doi.org/10.1097/PRS.0b013e31828bd541
Serlo WS, Ylikontiola LP, Lähdesluoma N, Lappalainen OP, Korpi J, Verkasalo J, Sàndor GKB (2011) Posterior cranial vault distraction osteogenesis in craniosynostosis: estimated increases in intracranial volume. Childs Nerv Syst 27:27–633. https://doi.org/10.1007/s00381-010-1353-1
ter Maaten NS, Mazzaferro DM, Wes AM, Naran S, Bartlett SP, Taylor JA (2018) Craniometric Analysis of frontal cranial morphology following posterior vault distraction. J Craniofac Surg 29:1169–1173. https://doi.org/10.1097/SCS.0000000000004473
Mazzaferro DM, ter Maaten NS, Wes AM, Naran S, Bartlett SP, Taylor JA (2019) A craniometric analysis of the posterior cranial base after posterior vault distraction. J Craniofac Surg 30:1692–1695. https://doi.org/10.1097/SCS.0000000000005496
Shimizu A, Komuro Y, Shimoji K, Miyajima M, Arai H (2016) Quantitative analysis of change in intracranial volume after posterior cranial vault distraction. J Craniofac Surg 27:1135–1138. https://doi.org/10.1097/SCS.0000000000002739
Salokorpi N, Vuollo V, Sinikumpu JJ, Satanin L, Nestal Zibo H, Ylikontiola LP, Pirttiniemi P, Sándor GK, Serlo W (2017) Increases in cranial volume with posterior cranial vault distraction in 31 consecutive cases. Neurosurgery 81:803–811. https://doi.org/10.1093/neuros/nyx125
Bauder AR, Wink JD, Swanson JW, Derderian C, Bartlett SP, Taylor JA (2015) An analysis of posterior vault distraction and its effects on the posterior fossa and cranial base. Plast Reconstr Surg 136:52–53. https://doi.org/10.1097/01.prs.0000472342.36900.c7
Greives MR, Ware BW, Tian AG, Taylor JA, Pollack IF, Losee JE (2016) Complications in posterior cranial vault distraction. Ann Plast Surg 76:211–215. https://doi.org/10.1097/SAP.0000000000000518
Author information
Authors and Affiliations
Contributions
All authors contributed to the design, data collection, and manuscript preparation.
Corresponding author
Ethics declarations
Ethics approval
Deemed “exempt” from the Institutional Review Board of the University of Mississippi Medical Center.
Consent to participate
Not applicable; the study was retrospective.
Consent for publication
All patients provided written consent for use of pictures in publications.
Conflict of interest
No authors report any conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sullivan, J.S., Snider, A.E., Farrington, J. et al. Posterior cranial vault distraction osteogenesis in the immunocompromised patient. Childs Nerv Syst 37, 2313–2318 (2021). https://doi.org/10.1007/s00381-021-05202-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-021-05202-y