Abstract
Purpose
The best management of pediatric unruptured brain arteriovenous malformations (ubAVM) should be further elucidated.
Methods
Pediatric patients who presented to our facility with ubAVM and were treated nonconservatively were retrospectively analyzed regarding demographics, presenting symptoms, AVM characteristics, treatment modality, and outcome.
Results
Thirteen patients were included. There were 9 male and 4 female patients with an average age (± SD) of 7.8 ± 4.6 years. The most common presenting symptom was seizure. The mean Spetzler Martin (SM) grade was 3.2. Seven patients were treated by surgical excision, 8 by embolization, 6 by radiosurgery, and during the mean follow-up period of 8.9 years (2.6–14.4 years), there were no hemorrhage. Complete ubAVM obliteration was achieved in all 7 patients (100%) who underwent surgical excision. AVM was obliterated in 5 out of 9 patients whose SM grade was 3 or 4. Modified Rankin scale was good (mRS 0–2) in all 13 patients.
Conclusions
Since the lifetime hemorrhage rate of pediatric ubAVM is high, it may be better to proceed with nonconservative treatment, especially when surgical excision is amenable. Nonconservative treatment could be an appropriate treatment with acceptable neurological outcome even in those with moderate to high-grade AVMs in selected cases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A randomized trial of unruptured brain arteriovenous malformations (ARUBA) showed that standalone medical management is superior to interventional therapy. However, this trial excluded those less than 18 years old and the follow-up period was only 33 months [1]. Since there are few reports on long-term outcome of pediatric unruptured brain arteriovenous malformations (ubAVM), the best management has not been established yet. The purpose of this case series is to further elucidate the long-term outcome of nonconservative treatment in pediatric ubAVM.
Methods
We retrospectively analyzed those who were referred to our facility with unruptured brain arteriovenous malformation (ubAVM) between 2002 and 2013. Exclusion criteria were multiple AVMs and less than 1 year follow-up. For those who were followed up by other facility, we did the telephone interview to their parents. In our facility, we treat all isolated ubAVM either by open surgery, embolization, stereotactic radiosurgery, or combination of those. The primary treatment modality is an open surgery with or without preoperative embolization if functionally acceptable outcome is expected. The secondary modalities are embolization or stereotactic radiosurgery. Analysis of the medical charts included the initial presenting symptoms, SM grade, presence of aneurysm, postoperative modified Rankin scale (mRS), treatment morbidity or mortality, and obliteration status. AVM features of computed tomography (CT), magnetic resonance image (MRI), and digital subtraction angiogram (DSA) were analyzed by radiologists in our facility. We classified mRS 0–2 into good postoperative status. A Kaplan-Meier survival curve was drawn for the time to recurrence since radiologically confirmed obliteration. We used the chi-square test for categorical variables, and a p value of less than 0.05 was considered as statistically significant.
Results
Thirteen patients were identified, and all were treated nonconservatively. Two patients were excluded for multiple ubAVMs and an early lost to follow-up. All patients had not undergone prior treatment for AVM. The summary of the patients and patient characteristics were shown in Tables 1 and 2. There were 9 males and 4 females. The mean age at diagnosis was 7.8 years old (SD 4.6). The most common presenting symptom was seizure (46%), which was consistent with previous studies. Six patients presented with seizure, 3 with headache, 1 with tachypnea, 1 with progressive weakness, and AVM was found incidentally in 2 patients, where CT scan in one patient showed intracranial calcification and MRI in the other one showed flow void in T2 sequence. As for Spetzler Martin (SM) grade, 1 was grade 1, 2 were grade 2, 5 were grade 3, 4 were grade 4, and 1 was grade 5. The mean SM grade was 3.2. All AVMs were found in the supratentorial location. Seven patients were treated by surgical excision, 8 by embolization, and 6 by stereotactic radiation. Intraoperative angiogram was not used, and 6 out of all 7 cases where excision was done, postoperative DSA within 6 months, showed no residual signs of AVM (86%). In 1 case (case number 4), it showed some residual abnormal vessels near a deep eloquent area, which was decided to be treated by radiosurgery. The remaining abnormal vessels were confirmed obliterated by DSA done in 3 years. The outcome of AVMs where surgical excision was performed is summarized in Table 3. There were 6 cases, where AVMs were treated only by embolization or radiosurgery, and the total duration since the initial treatment was 54 patient-years (mean follow-up of 9.0 years) and 1 patient resulted in obliteration (Table 4). None was treated by embolization alone. The follow-up period from the initial presentation was 116 patient-years (mean follow-up of 8.9 years). Obliteration of AVM was confirmed in 8 cases (62%), and 5 had residual AVM. No patients ruptured during the follow-up period and all patients achieved good mRS of 0–2. In 2 cases, AVM recurred 4 and 5 years after the confirmation of obliteration on DSA, respectively (recurrence incidence of 0.05 person-year). Figure 1 shows the Kaplan-Meier curve describing the time to recurrence since angiographically confirmed obliteration after surgical excision. There was no statistical significance between embolization before surgical excision and recurrence (p value = 0.22; not shown), or between sex and recurrence (p value = 0.54; not shown).
Illustrative cases
Case 2
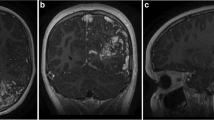
A 9-year-old boy presented with persistent headache. MRI showed a lesion in the left occipital lobe suspicious of AVM (Fig. 2a). DSA confirmed an AVM of about 3.0 cm × 1.5 cm, fed by a branch of posterior cerebral artery, and drained into the straight sinus (Fig. 2b). SM grade was 4. After the feeder embolization, AVM excision was performed. The obliteration was confirmed by DSA in 6 months (Fig. 2c). He was followed by an annual MRI. However, 4 years after the DSA, MRI showed evidence of recurrence. AVM recurrence was confirmed by DSA in the left occipital lobe (Fig. 2d). Again, surgical excision was performed. The obliteration was confirmed by DSA 6 months after the surgery. The latest MRI taken 2 years after the 2nd surgery showed no evidence of AVM recurrence (Fig. 2e).
Case 2. Axial T2-weighted MRI shows a lesion in the left occipital lobe (arrow) (a). Left internal carotid artery injection angiogram shows an AVM (b). The same angiogram as (b) done in 6 months from the excision shows complete obliteration of the previous AVM (c). Angiogram done in 4 years shows recurrence (arrow) (d). MRI taken 2 years after the 2nd excision showed no evidence of AVM recurrence (e)
Case 7
A 13-year-old boy with a past history of occasional headache, transient episodes of motor aphasia, and loss of consciousness in the past few years presented with altered mental status. MRI showed an AVM in the left frontal lobe (Fig. 3a). DSA confirmed an AVM of about 4.7 cm × 2.3 cm, fed by a branch of middle cerebral artery, and drained into the sphenoparietal sinus as well as superior sagittal sinus via cortical veins (Fig. 3b). SM grade was 4. Surgical excision of AVM was performed. Postoperative DSA in 6 months showed obliteration of AVM (Fig. 3c). An annual MRI has revealed no signs of recurrence for 6 years (Fig. 3d).
Case 7. Axial T2-weighted MRI shows a lesion in the left frontal lobe (a). Left internal carotid artery injection angiogram shows an AVM (b). The same angiogram as (b) done in 6 months from the excision shows complete obliteration of the previous AVM (c). MRI taken 6 years after surgery showed no signs of AVM recurrence (d)
Discussion
AVMs in pediatric patients have a high risk of rupture considering the cumulative hemorrhagic risk of their later life [2]. Although there are many studies regarding ubAVM comparing the outcomes between conservative and nonconservative management [3, 4], the patients’ age at diagnosis is over 15 years old. To the authors’ knowledge, few studies are published regarding the management outcome of younger patients. In this study, the mean age of patients was 7.8 years old all under the age of 15 years old. All underwent nonconservative treatment and the posttreatment neurological status was good (mRS of 0–2). AVM was obliterated in 8 out of 13 patients (62%) in total and all 4 patients (100%) after surgical excision whose SM grade was 3 or 4 (Table 3). In an ARUBA study, adults (18 years old or older) were randomized into nonconservative and conservative therapy groups. The annual risk of hemorrhage in those allocated into the interventional group was 10.7% compared with 3% of the medical group during the mean follow-up of 33 months, concluding it is better to treat ubAVM in a conservative manner [1]. However, our study suggests that it may be better to proceed with nonconservative treatment even in those with moderate to high-grade AVMs in selected cases and that the result of ARUBA may not simply apply to the management guide of pediatric ubAVMs. The risk of annual rupture of ubAVM is reported to be 0.6–4.3% [2, 5,6,7,8,9,10]. And the annual hemorrhagic risk of pediatric ubAVM is 0.6–4.0% [2, 6]. In our study, there was no rupture during the mean f/u of 9 years (2.6–14.4 years). The annual hemorrhage rate in our study was 0%. This may be due to the fact that the number of subjects was small and that the follow-up duration is not long enough. Another possibility is that radiosurgery is steadily making the AVMs unlikely to rupture on the way to the complete obliteration even if AVM still exists radiologically. In our study, all 6 patients treated by non-excisional therapy did not result in hemorrhage during the mean follow-up of 9.0 years despite the fact only 1 (17%) resulted in angiographical obliteration (Table 4). This result is consistent with a larger study done by Darsaut et al., where all 24 high-grade AVMs treated non-surgically did not result in obliteration. They also showed that there was no significant difference in the hemorrhagic risk between obliterated AVMs and non-obliterated ones [6]. So, our study suggests it is difficult to pursue radiological cure in high-grade ubAVM by radiosurgery with or without embolization.
After surgical excision of AVM, DSA performed within 6 months demonstrated obliteration in 6 out of 7. However, out of the 6 obliterated cases, recurrences were found in 2 cases during follow-up. The incidence of recurrent AVM in our series was 0.05 person-years, which was consistent with 0.08 from a larger retrospective study [11]. In their study, they showed that if DSA about 1 year after the resection was negative, no recurrence was found in DSA 5 years after the resection. In 2 recurrent cases in our study, the postoperative DSA was performed in less than 6 months after the excision. If DSA had been performed 1 year after the resection, recurrence might have been found earlier than MRA. The way of follow-up in pediatric patients with AVM is different from that of adults, since recurrence rarely occurs after the complete excision in adults [12]. In pediatric AVMs, periodical follow-up with imaging study is needed to prevent an AVM recurrence from presenting with hemorrhage. In the 2 recurrent cases, both patients were asymptomatic when the recurrence was found. We have to weigh the risk of complications associated with cerebral DSA against the risk of AVM recurrence. The complication rate associated with cerebral DSA was 0.4% (95% confidence interval, 0.012–2.29%) [13], which means that no complications would occur in 95.4–99.2% if DSA were done twice within a year (postoperative DSA immediately after the resection and in one year). On the other hand, the recurrence rate of AVM which obliteration was confirmed in a postoperative DSA is reported as 3.5–14.3% [11, 12, 14,15,16,17,18]. Considering these figures, the repeat DSAs, one immediately after the resection and the other one in a year, may reasonably be done. There are several case reports on AVM recurrence in pediatric population, but in most cases, second DSA in 1 year from the surgery had not been done [16, 19,20,21,22,23,24]. The exact pathogenesis for AVM recurrence has not been fully elucidated. There are several postulated mechanisms including de novo appearance and angiographically occult lesion with subsequent growth by hemodynamic stress, hidden compartments, or a “reserve” nidus that is visible after some hemodynamic change [12, 23, 25].
The limitations of our study are the small number of cohorts and not long-term follow-up. A larger cohort with longer follow-up needs to be done. And the possibility that those with negative findings on the 1 year postoperative DSA will have no recurrence should be also investigated in future studies.
Conclusions
In pediatric ubAVMs, the result of ARUBA may not simply apply to the management guide of pediatric ubAVMs.
References
Mohr JP, Parides MK, Stapf C, Moquete E, Moy CS, Overbey JR, al-Shahi Salman R, Vicaut E, Young WL, Houdart E, Cordonnier C, Stefani MA, Hartmann A, von Kummer R, Biondi A, Berkefeld J, Klijn CJ, Harkness K, Libman R, Barreau X, Moskowitz AJ, International ARUBA investigators (2014) Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicenter, non-blinded, randomized trial. Lancet 383(9917):614–621
Yang W, Anderson-Keightly H, Westbroek EM, Caplan JM, Rong X, Hung AL (2016) Long-term hemorrhagic risk in pediatric patients with arteriovenous malformations. J Neurosurg Pediatr 18(3):329–338
Al-Shahi Salman R, White PM, Counsell CE, du Plessis J, van Beijnum J, Josephson CB et al (2014) Outcome after conservative management or intervention for unruptured brain arteriovenous malformations. JAMA 311(16):1661–1669
Halim AX, Johnson SC, Singh V, McCulloch CE, Bennett JP, Achrol AS et al (2004) Longitudinal risk of intracranial hemorrhage in patients with arteriovenous malformation of the brain within a defined population. Stroke 35(7):1697–1702
Brown RD Jr, Wiebers DO, Forbes G, O’Fallon WM, Piepgras DG, Marsh WR et al (1988) The natural history of unruptured intracranial arteriovenous malformations. J Neurosurg 68(3):352–357
Darsaut TE, Guzman R, Marcellus ML, Edwards MS, Tian L, Do HM, Chang SD, Levy RP, Adler JR, Marks MP, Steinberg GK (2011) Management of pediatric intracranial arteriovenous malformations: experience with multimodality therapy. Neurosurgery 69(3):540–556
Graf CJ, Perret GE, Torner JC (1983) Bleeding from cerebral arteriovenous malformations as part of their natural history. J Neurosurg 58(3):331–337
Gross BA, Du R (2013) Natural history of cerebral arteriovenous malformations (2013) a meta-analysis. J Neurosurg 118(2):437–443
Kim H, Al-Shahi Salman R, McCulloch CE, Stapf C, Young WL, MARS Coinvestigators (2014) Untreated brain arteriovenous malformation: patient-level meta-analysis of hemorrhage predictors. Neurology 83(7):590–597
Ondra SL, Troupp H, George ED, Schwab K (1990) The natural history of symptomatic arteriovenous malformations of the brain: a 24-year follow-up assessment. J Neurosurg 73(3):387–391
Lang SS, Beslow LA, Bailey RL, Vossough A, Ekstrom J, Heuer GG, Storm PB (2012) Follow-up imaging to detect recurrence of surgically treated pediatric arteriovenous malformations. J Neurosurg Pediatr 9(5):497–504
Kader A, Goodrich JT, Sonstein WJ, Stein BM, Carmel PW, Michelsen WJ (1996) Recurrent cerebral arteriovenous malformations after negative postoperative angiograms. J Neurosurg 85(1):14–18
Burger IM, Murphy KJ, Jordan LC, Tamargo RJ, Gailloud P (2006) Safety of cerebral digital subtraction angiography in children: complication rate analysis in 241 consecutive diagnostic angiograms. Stroke 37(10):2535–2539
Andaluz N, Myseros JS, Sathi S, Crone KR, Tew JM Jr (2004) Recurrence of cerebral arteriovenous malformations in children: report of two cases and review of the literature. Surg Neurol 62(4):324–330
Klimo P Jr, Rao G, Brockmeyer D (2007) Pediatric arteriovenous malformations: a 15-year experience with an emphasis on residual and recurrent lesions. Childs Nerv Syst 23(1):31–37
Nerva JD, Kim LJ, Barber J, Rockhill JK, Hallam DK, Ghodke BV, Sekhar LN (2016) Outcomes of multimodality therapy in pediatric patients with ruptured and unruptured brain arteriovenous malformation. Neurosurgery 78(5):695–707
Rath EB, Felipe CA, Robert FS, Harold LR, Cameron GM, Joseph MZ (2006) Surgical management of arteriovenous malformations in children. J Neurosurg 105(2 Suppl):88–93
Rubin D, Santillan A, Greenfield JP, Souweidane M, Riina HA (2010) Surgical management of pediatric cerebral arteriovenous malformations. Childs Nerv Syst 26(10):1337–1344
Codd PJ, Mitha AP, Ogilvy CS (2008) A recurrent cerebral arteriovenous malformation in an adult. J Neurosurg 109(3):486–491
Freudenstein D, Duffner F, Ernemann U, Rachinger J, Grote E (2001) Recurrence of a cerebral arteriovenous malformation after surgical excision. Cerebrovasc Dis 11(1):59–64
Hino A, Fujimoto M, Iwamoto Y, Takahashi Y, Katsumori T (1999) An adult case of recurrent arteriovenous malformation after “complete” surgical excision: a case report. Surg Neurol 52(2):156–158
McCarthy C, Kaliaperumal C, O’Sullivan M (2012) Recurrence of a paediatric arteriovenous malformation 9 years postcomplete excision: case report and review of literature. BMJ Case Rep 25:2012
Musluman AM, Cavusoglu H, Yilmaz A, Aydin Y (2011) Recurrent cerebral arteriovenous malformation with a posterior inferior artery aneurysm. Turk Neurosurg 21(4):674–679
Weil AG, Li S, Zhao JZ (2011) Recurrence of a cerebral arteriovenous malformation following complete surgical resection: a case report and review of the literature. Surg Neurol Int 2:175
Akimoto H, Komatsu K, Kubota Y (2003) Symptomatic de novo arteriovenous malformation appearing 17 years after the resection of two other arteriovenous malformations in childhood: case report. Neurosurgery 52(1):228–231
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hong, S., Ogiwara, H. Long-term outcomes in pediatric unruptured brain arteriovenous malformation treated by nonconservative management: a single center analysis. Childs Nerv Syst 35, 1363–1369 (2019). https://doi.org/10.1007/s00381-019-04221-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-019-04221-0