Abstract
Objectives
Paediatric cerebellar pilocytic astrocytomas (PA) (WHO grade 1) are amongst the most common of childhood brain tumours and are generally amenable to resection, with surgery alone being curative in the majority of cases. There is, however, a lack of consensus regarding the frequency and duration of post-treatment MRI surveillance for these tumours. This is important, as follow-up imaging is a significant use of resources and often associated with patient and family anxiety. We have assessed the utility of MRI surveillance in the detection of cerebellar PA recurrence at our regional paediatric neurosurgical centre.
Materials and methods
The tumour register at Alder Hey Children’s Hospital was searched to identify all patients diagnosed between 2007 and 2017, with a confirmed histopathological diagnosis of cerebellar PA. Patient demographics, surgical outcome, number of MRI scans and length of follow-up were recorded for each patient.
Results
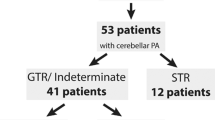
Forty patients met the inclusion criteria. The mean age at diagnosis was 7.8 years (range 2 to 17 years). Complete surgical resection (CR), confirmed by post-operative MRI, was achieved in 36 of the 40 patients, including all 31 cases from 2009 and later for which intraoperative MRI (iMRI) was utilised. There was one case of recurrence after CR (at 2.2 years) out of the 36 cases, whereas all 4 patients with initial partial resections had progressive growth of their tumours and required second surgical interventions.
Conclusion
This series confirms the very low likelihood of recurrence for completely resected cerebellar PAs and suggests that in such cases the duration and frequency of surveillance imaging could be limited to a maximum of 2.5 to 3 years of follow-up imaging. This report also indicates improved complete resection rates over time, probably associated with technical advances including the routine in-house use of iMRI in 2009.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
During childhood, low-grade gliomas (LGG) are the most common tumours of the central nervous system (CNS). Such tumours occur throughout the CNS, with those of the cerebellar hemispheres and optic pathways being the most common. A number of histological sub-types exist, of which pilocytic astrocytomas (PAs) (WHO grade I) are the majority, with peak incidence occurring between the ages of 5 and 9 years [1]. Tumorigenesis of this group of cancers is commonly characterised by activated BRAF protein kinase mutations and the hallmark duplication of the 7q34 locus, which typifies this low-grade disease [2]. Although complete surgical resection of the cerebellar examples without chemotherapy or radiotherapy remains the standard treatment, further development in the understanding of the underlying disease biology may soon herald the development and introduction of more targeted oncogenetic therapies.
Surgery is the mainstay of treatment for cerebellar LGG. Complete surgical resection (CR), confirmed in post-operative magnetic resonance images (MRI), is achievable in the majority of cases, following which the likelihood of disease reoccurrence is extremely low. This finding is consistently reported from both single institution [3,4,5] and group-wide [6, 7] reports, with figures suggestive that over 90% of patients remain disease free 10 years from diagnosis following complete tumour excision. With regard to surveillance imaging to detect relapse, the timely recognition of subclinical progression is said to help ensure safer further reoperation and ultimately result in improved neurosurgical outcomes and possible survival than if treatment was commenced once signs and symptoms suggestive of relapse were present [8]. In some cases, for example where there is local invasion of adjacent vital structures such as the brain stem, surgery may be challenging and partial resection to prevent significant morbidity is appropriate [9].
A period of post-operative MRI surveillance imaging is thus considered a standard of care; however, the optimal frequency and length of follow-up remains a salient and somewhat controversial topic [10]. There are very few studies that have addressed this issue, as reflected in a recent systematic review undertaken in the UK by the CRUK Clinical Trials unit [11]. A pilot study at our tertiary neuro-oncology centre, performed between 1988 and 2007 (unpublished data), found no evidence of relapse in 37 patients with cerebellar LGG who underwent complete surgical resection. As a result of these findings, duration of follow-up was reduced from 5 to 2.5 years within our department. The aim of this study was therefore to replicate this evaluation of the utility of MRI surveillance imaging in the detection of paediatric LGG recurrence, as validation of this decision and to assess if any additional change should be made to the current protocol.
Methods
To identify suitable patients, the cancer register at Alder Hey Children’s NHS Foundation Trust was searched. The register documents all patients with a tumour diagnosis that have received care within the centre. Cases were selected from 2007 onwards to prevent overlap with the previous pilot study referred to above and to assess how the change in the cerebellar low-grade glioma tumour surveillance protocol has affected current practice.
Patients with a histopathologically confirmed diagnosis of cerebellar PA were selected from the database. Histological diagnoses such as oligodendroglioma, pilomyxoid astrocytoma and mixed glial–neuronal tumours were excluded from this series. Patients were included in the analysis if they had undergone surgical resection for treatment of their disease at our centre.
For each patient, the dates of any related surgical excision, intraoperative or post-operative MRI scans were recorded. The length of follow-up between the date last seen by the neuro-oncology team and the date of the first surgery could then be calculated. Progression or disease relapse was defined radiologically, and medical notes were consulted to give context to the findings. In patients with a partially resected tumour, if there was progression of disease, time to progression was recorded. In the one patient with evidence of relapse following CR, transition had been made to an adult service, and this centre was contacted to assess if the patient was stable or if there was evidence of active disease on neuroimaging.
Results
Patient demographics and tumour characteristics
Forty patients fulfilled the inclusion criteria of this study. Of these 40 cases, 23 (57.5%) were male and 17 (42.5%) were female. The mean age at diagnosis was 7.8 years (range 2 to 17 years). The broad anatomical location of the lesions within the posterior fossa was recorded for each case. The majority of tumours were recorded as arising laterally within the cerebellar hemispheres (n = 25) with a further 15 cases developing in the midline.
Initial surgery and follow-up
The majority (36) of patients underwent complete surgical excision at the time of the first surgery, a finding that was confirmed radiologically with MRI during the intraoperative or immediate post-operative period. The remaining 4 (10%) cases, however, were deemed to have evidence of residual disease in scans and were therefore classified as partial resections.
In the 36 patients with a surgical CR, 29 patients have had MRI surveillance for at least 2.5 years and 7, all more recent cases, less than this period. Overall, during the surveillance period following surgery, patients underwent an average of 6.5 MRI scans. The mean length of follow-up from the date of the first surgery until the date last seen in clinic was 4.0 years (range 0.8 to 10.5 years) and the mean duration of surveillance imaging was 3.05 years (range 0.8 to 5.6 years).
Intraoperative MRI (iMRI) was introduced in December 2009. For this series, 31 of 40 patients underwent iMRI and in all cases complete resection was obtained (Fig. 1 ). The 4 cases with partial resection occurred in the group of 9 patients who did not undergo iMRI.
Pre-operative T1-weighted post-contrast image in the sagittal plane (a) shows a complex solid/multicystic posterior fossa pilocytic astrocytoma in a 10-year-old boy with evidence of heterogenous enhancement. T1-weighted post-contrast image on intraoperative MRI scan (b) demonstrates complete resection of the tumour. The open occipital craniotomy site is noted posteriorly (white arrow)
All 40 patients included in this study were alive at the time of data collection.
Completely resected tumours
Of the 36 cases in which total excision of tumour was achieved, to date, only one patient developed recurrent tumour. In this particular case, relapse was manifest as a 2-mm nodule occurring 2.2 years following surgery. Due to the size of the lesion and lack of any symptomatology, no further re-resection has been undertaken or adjuvant therapy initiated at the time of this report. The patient is currently under surveillance by an adult service.
Partially resected tumours
In the 4 patients with partially resected tumours, all required further surgery for progression of residual disease (Fig. 2 ). In all 4 cases, disease progression occurred within the first 12 months following initial surgery, the mean time to progression being 5.4 months. In those that required a second surgery, only one of these patients had an element of residual disease following re-resection. The most recent imaging suggests disease is stable in this patient at present. All incidences of progressive disease were diagnosed from surveillance imaging.
Pre-operative T1-weighted post-contrast image in the sagittal plane (a) shows a cystic posterior fossa pilocytic astrocytoma in a 3-year-old boy with evidence of a thick rim of enhancement anteriorly (white arrows). T1 post-contrast image on the 48-h post-operative scan (b) demonstrates irregular enhancement (arrowheads) anterior to the surgical cavity (*). This was suspicious of residual tumour. An early 3-month follow-up scan was performed and the T1 post-contrast image (c) confirms the presence of residual tumour which increased in size demonstrating a rim enhancing cystic lesion (open arrows)
Discussion
It is clear from the above data that complete surgical resection of cerebellar pilocytic astrocytoma is curative in the vast majority of paediatric patients, a finding consistent with previous reports from single institutions [3,4,5]. Shaw and Wishoff summarised the results from five clinical trials (adults and children) for low-grade glioma and showed a greater than 95% progression-free survival (PFS) in the 338 patients having a complete resection out of a total of 518 cases but with significantly lesser PFS in those with residual disease [6]. Similarly, Wishoff et al. presented the results of a large observational study of paediatric low-grade glioma and reported a 95% 8-year PFS for completely resected cortical pilocytic astrocytomas compared with 58% for less than completely resected tumours [7]. The incidence of relapse within the present series is consistent with such data, with one confirmed case out of 36 CR patients. This data also matches those from other studies examining the utility of post-operative surveillance imaging [8, 12]. Vassilyadi et al. also suggest that the low likelihood of reoccurrence so often seen may render intensive surveillance imaging obsolete in this sub-set of patients, as does the study by Alford et al. [10, 13]. The current protocol at our centre for CR patients suggests scans are performed at 6, 18 and 30 months. The fact that many centre surveillance scans are performed more frequently and for a longer time after surgery may reflect an opinion that the incidence of relapse after CR is higher than described in the literature and that more aggressive surveillance is prudent [12, 14,15,16].
It should be noted that all of the surgeries that resulted in partial tumour resection occurred prior to 2010. It may be that the likelihood of complete excision is now increased possibly due to the advent of intraoperative MRI, which commenced at our centre in October 2009. All surgeries that occurred from this point onwards have resulted in total tumour excision. As well as the introduction of this service, refinement of surgical techniques and the level of experience gained by the involved surgeons over time may also be a contributing factor to attainment of complete resection.
In the patients in whom progression of residual disease was identified, time to progression was < 12 months following initial surgery in all cases. This is also seen in a number of reports that highlight the importance of the first year of follow-up in subtotal resections [8, 10, 13]. Where there is a lesser need for intensive surveillance in completely resected tumours, there is certainly an argument that can be made for a greater degree of vigilance in partially resected lesions. The frequency and length of such a surveillance regime requires further study but at present should perhaps be treated on a case-by-case basis.
The aim of this report was to describe one institution’s experience of the utility of surveillance imaging following a change in our local protocol. As such, the sample size of patients is relatively small as only the cases from this centre have been included. In addition, the patient follow-up is relatively short. Collaboration with other groups may lead to more robust data in the future.
Overall, it is evident the changes made to surveillance imaging of cerebellar LGG at this centre in 2007 were appropriate. Thus, based on our findings, this study endorses a limited surveillance imaging protocol for completely resected cerebellar LGG, with post-op scans at 6, 18 and 30 months and then stopping.
Reducing the number of MRIs performed has benefits for both the patient and family alike by preventing unnecessary stress and anaesthesia (if required) as well as having financial implications that would allow for reallocation of resources.
References
Sievert AJ, Fisher MJ (2009) Pediatric low-grade gliomas. J Child Neurol 24:1397–1408
Sievert AJ, Lang S-S, Boucher KL, Madsen PJ, Slaunwhite E, Choudhari N, Kellet M, Storm PB, Resnick AC (2013) Paradoxical activation and RAF inhibitor resistance of BRAF protein kinase fusions characterizing pediatric astrocytomas. Proc Natl Acad Sci 110:5957–5962
Gajjar A, Sanford RA, Heideman R, Jenkins JJ, Walter A, Li Y, Langston JW, Muhlbauer M, Boyett JM, Kun LE (1997) Low-grade astrocytoma: a decade of experience at St. Jude Children’s Research Hospital. J Clin Oncol 15:2792–2799
Fisher BJ, Leighton CC, Vujovic O, Macdonald DR, Stitt L (2001) Results of a policy of surveillance alone after surgical management of pediatric low grade gliomas. Int J Radiat Oncol Biol Phys 51:704–710
Fisher PG, Tihan T, Goldthwaite PT, Wharam MD, Carson BS, Weingart JD, Repka MX, Cohen KJ, Burger PC (2008) Outcome analysis of childhood low-grade astrocytomas. Pediatr Blood Cancer 51:245–250
Shaw EG, Wishoff JH (2003) Prospective clinical trials of intracranial low-grade glioma in adults and children. Neuro-Oncology 5(3):153–160
Wisoff JH, Sanford RA, Heier LA, Sposto R, Burger PC, Yates AJ, Holmes EJ, Kun LE (2011) Primary neurosurgery for pediatric low-grade gliomas: a prospective multi-institutional study from the Children’s Oncology Group. Neurosurgery 68(6):1548–1554
Sutton LN, Cnaan A, Klatt L, Zhao H, Zimmerman R, Needle M, Molloy P, Phillips P (1996) Postoperative surveillance imaging in children with cerebellar astrocytomas. J Neurosurg 84:721–725
Steinbok P, Hentschel S, Cochrane DD, Kestle JRW (1996) Value of postoperative surveillance imaging in the management of children with some common brain tumors. J Neurosurg 84:726–732
Alford R, Gargan L, Bowers DC, Klesse LJ, Weprin B, Koral K (2016) Postoperative surveillance of pediatric cerebellar pilocytic astrocytoma. J Neuro-Oncol 130:149–154
Stevens SP, Main C, Bailey S, Pizer B, English M, Phillips R, Peet A, Avula S, Wilne S, Wheatley K, Kearns PR, Wilson JS (2018) The utility of routine surveillance screening with magnetic resonance imaging (MRI) to detect tumour recurrence/progression in children with low grade central nervous system (CNS) tumours: a systematic review. J Neuro-Oncol 139:507–522
Dorward IG, Luo J, Perry A, Gutmann DH, Mansur DB, Rubin JB, Leonard JR (2010) Postoperative imaging surveillance in pediatric pilocytic astrocytomas. J Neurosurg Pediatr 6:346–352
Vassilyadi M, Shamji MF, Tataryn Z, Keene D, Ventureyra E (2009) Postoperative surveillance magnetic resonance imaging for cerebellar astrocytoma. Can J Neurol Sci 36:707–712
Udaka YT, Yeh-Nayre LA, Amene CS, VandenBerg S, Levy ML, Crawford JR (2013) Recurrent pediatric central nervous system low-grade gliomas: the role of surveillance neuroimaging in asymptomatic children. J Neurosurg Pediatr 11:119–126
Ogiwara H, Bowman RM, Tomita T (2012) Long-term follow-up of pediatric benign cerebellar astrocytomas. Neurosurgery 70:40–47
Saunders DDE, Phipps KPK, Wade AM, Hayward RD (2005) Surveillance imaging strategies following surgery and/or radiotherapy for childhood cerebellar low-grade astrocytoma. J Neurosurg 102:172–178
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McAuley, E., Brophy, H., Hayden, J. et al. The benefit of surveillance imaging for paediatric cerebellar pilocytic astrocytoma. Childs Nerv Syst 35, 801–805 (2019). https://doi.org/10.1007/s00381-019-04078-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-019-04078-3