Abstract
Background
A critical and unmet therapeutic need is the prompt and reliable exclusion of acute myocardial infarction (AMI), which would allow for prompt discharge from the emergency department. High-sensitivity troponin (HS-TnT) combined with copeptin has been proposed to expedite the diagnostic exclusion of AMI in addition to its predictive usefulness in the intermediate and long-term outcomes of AMI. The European Society of Cardiology recommends the Global Registry of Acute Coronary Events (GRACE) as a prognostic score to manage acute coronary syndrome (ACS) without ST segment elevation. The purpose of this study was to compare the diagnostic accuracy of HS-TnT combined with copeptin in ruling out AMI compared to HS-TnT alone. By combining a low GRACE score (108) with negative HS-TnT (14 ng/L) and copeptin (14 pmol/L), non-ST and ACS were reliably ruled out, including non-ST segment elevation MI and unstable angina.
Results
The present research included nine studies with a total of 13,232 participants. The negative predictive value (NPV) for copeptin and HS-TnT was found to be slightly higher in combination (62–99%) than for HS-TnT alone (60–99%). The sensitivity of copeptin ranged from 0% to 100%, with a pooled sensitivity of 0.79 (CI 95% 0.76, 0.82). The specificity of copeptin ranged from 13% to 100%, with a pooled specificity of 0.89 (CI 95% 0.85, 0.91), a pooled positive likelihood ratio (PLR) of 9.86 (CI 95% 4.42, 22.02), and a pooled negative likelihood ratio (NLR) of 0.08. (95% CI 0.01, 0.44).
Conclusions
The use of combined negative copeptin and highly sensitive troponin testing in low-to-intermediate risk patients with suspected acute coronary syndrome resulted in a quick discharge with a safe and rapid rule out of non-ST + ACS.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Acute myocardial infarction (AMI) is a difficult diagnosis to determine due to the narrow therapeutic window and need for immediate attention [1, 2]. Chest pain is common and most often caused by non-cardiac conditions. Atypical presentations of acute coronary syndrome (ACS), as well as the unreliability of the presence or absence of cardiovascular risk factors, make the diagnosis of ACS difficult in patients with chest pain syndrome, particularly in those without ST elevation on electrocardiograms.
Several cardiac biomarkers have been proposed for use in the early diagnosis of these patients in the emergency department. Despite that troponin assays are highly specific, their delayed rise (up to 6–12 h) limits their effectiveness as a diagnostic tool and reduces their overall negative predictive value (NPV) [1, 2]. Furthermore, most patients with chest pain require serial troponin measurements over several hours before A non-ST-elevation myocardial infarction (NSTEMI) is ruled out (Table 1).
Copeptin is a 39-amino acid glycopeptide derived from the C-terminus of the arginine vasopressin (AVP) hormone precursor (CT-proAVP) [3]. Copeptin levels peak early (0–1 h) after AMI and then return to baseline within 12–36 h in patients with early presentation of AMI. Thus, copeptin improves the early detection of non-ST segment elevation myocardial infarction (MI) and the prediction of infarct size, cardiac function alteration, and, most importantly, prognosis after AMI.
Objectives
To compare the diagnostic accuracy of copeptin plus troponin versus troponin alone in the prompt rule out of acute myocardial infarction and assess the prognostic value of the combined measurement on post-myocardial infarction outcomes.
Methods
The literature review was conducted from June 2021 to April 2022 using the databases PubMed Central, Embase, and Web of Science as well as the Cochrane Library. The keywords “copeptin AND troponin AND acute myocardial infarction” and their synonyms were used. The search was comprehensive, employing an umbrella method, and reference lists from the articles retrieved were cross-checked.
Study selection
The reference management software Endnote 7.4 was used for all identified studies. Two reviewers examined the title, abstract, and type for each identified article.
The included studies fulfilled the following criteria: full text articles, peer-reviewed articles, and any observational or interventional articles with no language restriction. The following articles were excluded: reviews, case reports, editorials, letters, comments, conference abstracts, systematic reviews and/or meta-analyses; animal studies; duplicate studies; irrelevant population; irrelevant index test; and irrelevant outcomes.
A third reviewer intervened in cases of disagreement between the two reviewers, and differences were discussed until a consensus was reached. The full texts of the chosen articles were acquired, which were then rescreened and evaluated more thoroughly for eligibility using the same exclusion criteria.
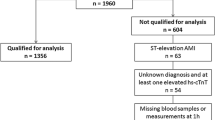
After exclusion, nine articles were selected for the final review. Two authors independently reviewed selected articles. All disagreements were resolved with a discussion between the two authors and mutually agreed upon by the authors. A third independent reviewer provided input as needed.
Study design
The study was conducted in accordance with the principles outlined by the Systematic Reviews of Diagnostic Test Accuracy as well as Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA).
Data extraction
The two reviewers obtained the characteristics and results of the selected studies. They contacted the authors of studies lacking data. Despite this outreach, studies with lacking data were excluded from the meta-analysis. Data regarding true-positive, false-positive, false-negative, and true-negative results for individual studies were obtained. The following variables were extracted from the studies: first author, year of publication, country, study population, inclusion period, and patients’ baseline characteristics. Furthermore, ethical approval was not required for this study as already available databases were used to obtain data and patients were not directly involved.
Assessment of methodological quality
The two reviewers assessed the methodological quality of the primary studies at the study level. They accessed patient selection, index test, reference standard, flow, and timing using a checklist adapted from the Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) tool. The potential for incorporation bias was the primary methodological issue in determining whether to include studies. Thus, Elevation of copeptin (cTn), which is a part of non-ST elevation myocardial infarction (NSTEMI), was included as one of the index tests in this study.
Statistical analysis
For each primary study, estimates of sensitivity and specificity points and corresponding 95% confidence intervals (CI) were calculated from extracted data for cTn alone and cTn with copeptin. We used SPSS Statistics to perform a bivariate.
The statistical significance for hypothesis testing was set at 0.05 for two-tailed heterogeneity testing and at 0.10 for two-tailed tests. Dichotomous variables were reported as proportions (%), whereas continuous variables are reported as the mean (standard deviation [SD]) or median (interquartile range [IQR]).
Summary diagnostic accuracy estimates
The summary estimates of sensitivity, specificity, positive likelihood ratios (PLRs), and negative likelihood ratios (NLRs) were derived from bivariate mixed-effect regression model parameter estimates.
Sensitivity analyses
A sensitivity analysis was performed for all studies A subgroup analysis was also performed for studies comparing levels of troponin alone versus troponin combined with copeptin only in MI patients.
Results
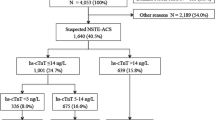
Our literature search yielded nine relevant articles [4,5,6,7,8,9,10,11,12] with studies from 2013 to 2020. A total of 13,232 patients were studied in these articles. Copeptin levels were measured in all of them, and specific characteristics were noted.
Copeptin levels were measured in all the studies within 12 h of the cardiovascular events, making comparisons between them easier. Three of the studies were even more specific, obtaining lab results within 4 h of the event’s occurrence.
Copeptin was the most studied of all available markers. The sensitivity ranged from 0 to 100%, with a pooled sensitivity of 0.79 (CI 95% 0.76, 0.82). The specificity ranged from 13 to 100%, with a pooled specificity of 0.89 (CI 95% 0.85, 0.91), a pooled PLR of 9.86 (CI 95% 4.42, 22.02), and a pooled NLR of 0.08 (CI 95% 0.01, 0.44) (Fig. 1, 2, 3, 4). Figure 5 depicts the summary receiver operating characteristic (ROC) plot as well as an overview of the studies, including their summary sensitivity and specificity with areas under the curve (AUCs) of 0.9816 and Q* = 0.9403.
In the pooled analysis of the studies, the combination of copeptin and troponin showed a slightly higher sensitivity (86–97%) compared troponin alone (70–97%). However, the specificity was slightly lower in the combination group (62–79%) than in the troponin group (80–95%). The combined measurement of copeptin and troponin levels had a slightly higher NPV (62–99%) than measurements of troponin levels alone (60–99%) (Fig. 6).
Discussion
Copeptin is a neuroendocrine peptide derived from the C-terminus of the pre-pro-hormone of arginine vasopressin (AVP). Copeptin is released with AVP) after hemodynamic or osmotic stimuli. It is released into the circulation following AMI, first with an immediate rise that is then followed by a rapid decline within 5 days.
According to studies involving a large proportion of ST elevation myocardial infarction (STEMI) patients, the combination of copeptin and troponin has a high NPV of AMI, allowing rapid rule out of AMI in the emergency department [14]. Copeptin increases in the first 6 h, unlike cardiac troponins, which typically increase at 12 h. As a result, the use of copeptin is inadequate in the first 1–3 h as a rule-in marker for AMI and in ruling out patients with acute chest pain and a history of coronary artery disease (CAD) [15].
According to Gu et al., cardiac troponin-T (cTnT) levels rise within 6–9 h of symptom onset, with a sensitivity of 39–43% in the first 3 h [16]. In the CHOPIN trial, the inclusion of copeptin allowed 58% of patients with non-diagnostic electrocardiogram (ECGs) to be ruled out for an AMI with a negative predictive value (NPV) of greater than 99% and a decrease in the time for AMI diagnosis from 3 to 1.8 h on average (43% reduction). The addition of another biomarker to cTnT, such as copeptin, may be more informative in detecting myocardial ischemia [17]. Furthermore, the additive effect accelerates clinical decision-making in ruling out non-AMI patients without the need for extended monitoring times extended monitoring times and serial blood sampling for repeated cTnT measurements [18, 19].
A metanalysis from Hyungoo et al. revealed a high sensitivity (100%) of copeptin with troponin measured in the first 6 h and a high NPV of copeptin with troponin-I measured in the first 6 h (100%) [20]. Furthermore, Bohyn et al. used copeptin, high-sensitivity troponin T, and GRACE scores to rule out AMI quickly [21]. Finally, in another study conducted by Ray et al., participants with a prior history of CAD and negative cTn on admission were evaluated, and the researchers found that copeptin was superior to conventional cTn in patients with acute chest pain and a prior history of CAD [22].
Impact of sex, age, and comorbidities on copeptin diagnostic accuracy
According to our findings, the male gender has a significant influence on the diagnostic accuracy of copeptin levels. The pooled analysis revealed that 8,269 of the 13,232 patients enrolled in our studies were males. This is consistent with a systematic review from Lipinski et al., which found that men made up 64% of the enrolled patients [13]. Our enrolled patients had an average age of 63.8 years. This is also consistent with Lipinski et al., in which the average age of the studied population was 64 years [13].
Of the patients included, 65.95% had hypertension as a comorbidity. Lipinski et al. also reported a weighted mean of 63.0%. Furthermore, 23.41% of the patient population in our study was diabetic. Smoking also seemed to have an impact on copeptin diagnostic accuracy, as 27.60% of the patient population were smokers. This is also consistent with Lipinski et al., where 28.6% were smokers. Our results from the lipid profile were also in line with Lipinski et al., with 42.06% of the patients having hyperlipidemia [13].
Most clinical studies used a copeptin cutoff of 14 pmol/l [23,24,25]. Keller et al. tested several copeptin cutoff values in a large reference population (n = 1386), revealing that the 99th percentile value was 18.9 pmol/l, the 97.5th percentile value was 13 pmol/l, and the 95th percentile value was 9.8 pmol/l. Thus, the lower the cutoff value, the higher the NPV for AMI diagnosis. However, a 95th percentile value (10 pmol/l) may be preferable to avoid false-negative results. [26, 27]. A plasma copeptin cutoff level of 21.1 pg/ml might be used for early rule-in of myocardial infarction, which can later be confirmed by troponin measurement.
Conclusions
Combining copeptin and troponin I improves diagnostic accuracy in the emergency department for chest pain that appears within 12 h of onset. Copeptin allows for the rapid exclusion of NSTEMI, adding value to standard cTn in patients with acute chest pain and a history of CAD. However, even when using a standard troponin assay, the sensitivity of this combination is insufficient to rule out NSTEMI safely.
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Morgenthaler NG, Struck J, Jochberger S, Dünser MW (2008) copeptin clinical use of a new biomarker. Trends Endocrinol Metab 19(2):43–49
Vargas KG, Kassem M, Mueller C, Wojta J, Huber K (2016) Copeptin for the early rule-out of non-ST-elevation myocardial infarction. Int J Cardiol 223:797–804
Mueller C, Giannitsis E, Möckel M, Huber K, Mair J, Plebani M, Thygesen K, Jaffe AS, Lindahl B (2017) Biomarker Study Group of the ESC Acute Cardiovascular Care Association. Rapid rule out of acute myocardial infarction: novel biomarker-based strategies. Eur Heart J Acute Cardiovasc Care 6(3):218–222
Llorens P, Sánchez M, Herrero P, Martín-Sánchez FJ, Piñera P, Miró O (2014) COPED study investigators The utility of copeptin in the emergency department for non-ST-elevation myocardial infarction rapid rule out: COPED-MIRRO study. Eur J Emerg Med 21(3):220–229
Charpentier S, Lepage B, Maupas-Schwalm F, Cinq-Frais C, Bichard-Bréaud M, Botella JM, Elbaz M, Lauque D (2013) Copeptin improves the diagnostic performance of sensitive troponin I-Ultra but cannot rapidly rule out non-ST-elevation myocardial infarction at presentation to an emergency department. Ann Emerg Med 61(5):549-558.e1
Wildi K, Zellweger C, Twerenbold R, Jaeger C, Reichlin T, Haaf P, Faoro J, Giménez MR, Fischer A, Nelles B, Druey S, Krivoshei L, Hillinger P, Puelacher C, Herrmann T, Campodarve I, Rentsch K, Steuer S, Osswald S, Mueller C (2015) Incremental value of copeptin to highly sensitive cardiac Troponin I for rapid rule-out of myocardial infarction. Int J Cardiol 190:170–176
Sebbane M, Lefebvre S, Kuster N, Jreige R, Jacques E, Badiou S, Dumont R, Cristol JP, Dupuy AM (2013) Early rule out of acute myocardial infarction in ED patients: value of combined high-sensitivity cardiac troponin T and ultrasensitive copeptin assays at admission. Am J Emerg Med 31(9):1302–1308.
O’Malley RG, Bonaca MP, Scirica BM, Murphy SA, Jarolim P, Sabatine MS, Braunwald E, Morrow DA (2014) Prognostic performance of multiple biomarkers in patients with non-ST-segment elevation acute coronary syndrome: analysis from the MERLIN-TIMI 36 trial (Metabolic Efficiency With Ranolazine for Less Ischemia in Non-ST-Elevation Acute Coronary Syndromes-Thrombolysis In Myocardial Infarction 36). J Am Coll Cardiol 63(16):1644–1653
Giannitsis E, Slagman A, Hamm CW, Gehrig S, Vollert JO, Huber K (2020) Copeptin combined with either non-high sensitivity or high sensitivity cardiac troponin for instant rule-out of suspected non-ST segment elevation myocardial infarction. Biomarkers 25(8):649–658
Giannitsis E, Clifford P, Slagman A, Ruedelstein R, Liebetrau C, Hamm C, Honnart D, Huber K, Vollert JO, Simonelli C, Schröder M, Wiemer JC, Mueller-Hennessen M, Schroer H, Kastner K, Möckel M (2019) Multicenter cross-sectional observational registry to monitor the safety of early discharge after rule-out of acute myocardial infarction by copeptin and troponin: the Pro-Core registry.BMJ Open 9(7):e028311.
Sukul D, Bonaca MP, Ruff CT, Kosowsky J, Conrad M, Murphy SA, Sabatine MS, Jarolim P, Morrow DA (2014) Diagnostic performance of copeptin in patients with acute nontraumatic chest pain: BWH-TIMI ED chest pain study. Clin Cardiol 37(4):227–232
El Shafey WEDH, Ahmedy IA (2016) Diagnostic values of Copeptin as a novel cardiac marker in relation to traditional markers in acute myocardial infarction.Clinical Trials and Regulatory Science in Cardiology19:13–19.
Giannitsis E, Kehayova T, Vafaie M, Katus HA (2011) Combined testing of high-sensitivity troponin T and copeptin on presentation at prespecified cutoffs improves rapid rule-out of non-ST-segment elevation myocardial infarction.Clin Chem 57(10):1452–1455.
Lipinski MJ, Escárcega RO, D’Ascenzo F, Magalhães MA, Baker NC, Torguson R, Chen F, Epstein SE, Miró O, Llorens P, Giannitsis E, Lotze U, Lefebvre S, Sebbane M, Cristol JP, Chenevier-Gobeaux C, Meune C, Eggers KM, Charpentier S, Twerenbold R, Mueller C, Biondi-Zoccai G, Waksman RA (2014) systematic review and collaborative meta-analysis to determine the incremental value of copeptin for rapid rule-out of acute myocardial infarction. Am J Cardiol 113(9):1581–1591
Chenevier-Gobeaux C, Sebbane M, Meune C, Lefebvre S, Dupuy AM, Lefèvre G, Peschanski N, Ray P (2019) Is high-sensitivity troponin, alone or in combination with copeptin, sensitive enough for ruling out NSTEMI in very early presenters at admission? A post hoc analysis performed in emergency departments. BMJ Open 9(6):e023994
Gu YL, Voors AA, Zijlstra F, Hillege HL, Struck J, Masson S, Vago T, Anker SD, van den Heuvel AF, van Veldhuisen DJ, de Smet BJ (2011) Comparison of the temporal release pattern of copeptin with conventional biomarkers in acute myocardial infarction. Clin Res Cardiol 100(12):1069–1076
Pedersen CK, Stengaard C, Søndergaard H, Dodt KK, Hjort J, Bøtker MT, Terkelsen CJ (2018) A multicentre, randomized, controlled open-label trial to compare an Accelerated Rule-Out protocol using combined prehospital copeptin and in-hospital high sensitive troponin with standard rule-out in patients suspected of acute Myocardial Infarction - the AROMI trial.Trials 19(1):683.
Kim KS, Suh GJ, Song SH, Jung YS, Kim T, Shin SM, Kang MW, Lee MS (2020) Copeptin with high-sensitivity troponin at presentation is not inferior to serial troponin measurements for ruling out acute myocardial infarction. Clin Exp Emerg Med 7(1):35–42
Reinhold T, Giannitsis E, Möckel M, Frankenstein L, Vafaie M, Vollert JO, Slagman, (2018) A Cost analysis of early discharge using combined copeptin/cardiac troponin testing versus serial cardiac troponin testing in patients with suspected acute coronary syndrome. PLoS ONE 13(8):e0202133
Shin H, Jang BH, Lim TH, Lee J, Kim W, Cho Y, Ahn C, Choi KS (2018) Diagnostic accuracy of adding copeptin to cardiac troponin for non-ST-elevation myocardial infarction: A systematic review and meta-analysis. Plos one13(7): e0200379.
Bohyn E, Dubie E, Lebrun C, Jund J, Beaune G, Lesage P, Belle L, Savary D (2014) Expeditious exclusion of acute coronary syndrome diagnosis by combined measurements of copeptin, high-sensitivity troponin, and GRACE score. Am J Emerg Med 32(4):293–296
Ray P, Charpentier S, Chenevier-Gobeaux C, Reichlin T, Twerenbold R, Claessens YE, Jourdain P, Riou B, Mueller C (2012) Combined copeptin and troponin to rule out myocardial infarction in patients with chest pain and a history of coronary artery disease. Am J Emerg Med 30(3):440–448
Reichlin T, Schindler C, Drexler B, Twerenbold R, Reiter M, Zellweger C, Moehring B, Ziller R, Hoeller R, Rubini Gimenez M, Haaf P, Potocki M, Wildi K, Balmelli C, Freese M, Stelzig C, Freidank H, Osswald S, Mueller C (2012) One-hour rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. Arch Intern Med 172(16):1211–1218
Ay MO, Erenler AK, Dogan T, Yetim M (2017) Diagnostic value of copeptin in acute myocardial infarction. Eur Rev Med Pharmacol Sci 21(7):1576–1582
Twerenbold R, Boeddinghaus J, Nestelberger T, Wildi K, Rubini Gimenez M, Badertscher P, Mueller C (2018) How to best use high-sensitivity cardiac troponin in patients with suspected myocardial infarction. Clin Biochem 53:143–155
Li F, Guo L, Hu Y, Li Z, Liu J, He J, Cui H (2020) Multiplexed chemiluminescence determination of three acute myocardial infarction biomarkers based on microfluidic paper-based immunodevice dual amplified by multifunctionalized gold nanoparticles. Talanta 207:120346
Thygesen K, Mair J, Katus H, Plebani M, Venge P, Collinson P, Lindahl B, Giannitsis E, Hasin Y, Galvani M, Tubaro M, Alpert JS, Biasucci LM, Koenig W, Mueller C, Huber K, Hamm C, Jaffe AS (2010) Study group on biomarkers in cardiology of the ESC working group on acute cardiac care recommendations for the use of cardiac troponin measurement in acute cardiac care. Eur Heart J 18:2197–21204
Acknowledgements
Daad Hassan SharafEldin, MD.
Funding
Not Applicable.
Author information
Authors and Affiliations
Contributions
All the authors have shared work in collecting, analyzing, and writing the research paper. SE and AA were major contributors in writing the manuscript, while DM, MV and AE were major contributors in collecting needed information from different centers. The manuscript was under the supervision of TM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no competing interests.
Ethics approval and consent to participate
Not Applicable.
Consent for publication
Not Applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elseidy, S.A., Awad, A.K., Mandal, D. et al. Copeptin plus troponin in the rapid rule out of acute myocardial infarction and prognostic value on post-myocardial infarction outcomes: a systematic review and diagnostic accuracy study. Heart Vessels 38, 1–7 (2023). https://doi.org/10.1007/s00380-022-02123-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-022-02123-x