Abstract
Purpose
Transrectal ultrasound (US) imaging is paramount to the successful completion of prostate biopsies. Certain US features have been associated with prostate cancer (PCa), but their utility remains controversial. We explored the role of multiparametric US (mpUS) in the detection of clinically significant PCa.
Methods
We performed a retrospective cohort study to contrast the findings of prostate MRI and mpUS. Patients who underwent MRI, US and biopsy between 2015 and 2021 were included. Biopsies were performed using a systematic approach (12 cores), as well as with MRI (4 cores/lesion) and US (1 core/lesion) targeting. The US features analyzed consisted of: calcifications, hypoechoic lesions and power or color Doppler positivity. Gleason 3 + 4 or higher was used as to define true positives. Measures of diagnostic accuracy were calculated for the different imaging modalities.
Results
The final cohort included 74 patients, of which 24 (32.4%) had clinically significant PCa. The concordance between MRI and US was 63.5%. Seven individuals with discordant results had clinically significant PCa. MRI alone was more sensitive (87.5% vs 75%) but less specific (28% vs 32%) than US alone. An all-inclusive approach considering any suspicious US or MRI finding had a sensitivity of 95.8%. A more restrictive approach, targeting lesions noted in both US and MRI, yielded the highest specificity (50.0%) and accuracy (55.4%).
Conclusion
Biopsy targeting based on US findings can provide additional diagnostic information that may increase sensitivity or specificity. Additional research into this topic could open the door to a more personalized approach to prostate biopsy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
For several decades, prostate cancer (PCa) diagnosis was largely dependent on a non-targeted 12-core biopsy technique. The introduction of multiparametric magnetic resonance imaging (mpMRI) and mpMRI fusion biopsy have changed our ability to detect more clinically significant neoplasms with a targeted approach [1]. mpMRI has not only demonstrated superior sensitivity and accuracy in the detection of clinically significant PCa compared to a non-targeted approach, but its use does not preclude the combination of this technique with a systematic 12-core sampling method. The inclusion of both sampling modalities during prostate biopsy has been used frequently to achieve improved detection and has now been widely adopted in clinical practice [2].
Although the added value of mpMRI fusion biopsy has been clearly established, real-time ultrasound (US) guidance remains paramount to the successful conduction of both systematic and fusion biopsies. Transrectal US performed at the time of prostate biopsy provides real-time anatomical guidance as well as potentially useful diagnostic information. For example, it has been previously shown that hypoechoic lesions identified on US can represent prostatic neoplasms [3], leading some authors to propose the inclusion of a targeted approach based on US findings in addition to the standard 12-core sampling method [4]. Other US features have been shown to be associated with the presence of PCa; for example, color Doppler positivity has been associated with PCa positivity as well as higher Gleason grade [5]. Similarly, power Doppler positivity has been reported to be a positive predictor of disease [6]. However, it remains controversial whether imaging findings on transrectal ultrasound provide diagnostic information that results in improved PCa detection or diagnostic accuracy when combined with mpMRI fusion biopsy, and previous studies have mostly focused on individual US features [7].
Given the paucity of data regarding this question, we performed a retrospective cohort study with a pragmatic design to determine the utility of a US-targeted prostate biopsy approach. Several US-based parameters and features were considered suspicious for PCa and used to target prostate biopsies. We contrast the findings of this multiparametric US-based targeting approach with mpMRI findings and assess the utility of both imaging modalities in detecting clinically significant disease.
Methods
Data collection and patient selection
Data were extracted from our prospectively maintained institutional PCa database, and included patients who underwent ultrasound-guided prostate biopsy at Virginia Mason Medical Center from January 2015–September 2021. Data were cross-referenced with the prospectively collected MRI database from the same institution. Individuals 18 years or older who had a mpMRI of the prostate followed by transrectal US-guided biopsy within 6 months were included in the analysis. All biopsies were performed by the same experienced urologic oncologist (C.P.) who was not blinded to the mpMRI results. A single US operator was utilized with the goal of minimizing inter-operator variability. After initial patient selection, manual review of the medical records was performed for accuracy. Demographic, clinicopathologic and imaging data were collected and managed using REDCap tools [8, 9]. All the individuals included provided written informed consent and all research-related activities were pre-approved by our independent institutional ethics review board.
Multiparametric magnetic resonance imaging
Prior to biopsy, all patients had PSA testing, digital rectal exams, and a mpMRI using an endorectal coil, with a 3 T MRI scanner (GE Siemens Symphony with TIMS). This was subsequently read by a radiologist specializing in prostate MRI interpretation. MRI positivity was defined as PI-RADS 3 or greater according to PI-RADS v2 [10]. MRI fusion was performed on the UroNav System (v2.2.1807.3d6a4180) by the same urologist performing the biopsy. All recorded lesions were analyzed separately and results later combined at the patient level for the primary analysis.
Multiparametric ultrasound (mpUS)
Multiparametric ultrasound (mpUS) of the prostate was performed using the Philips HD11XE model with a C95ec rectal probe at the time of transrectal US. mpUS lesion positivity was defined as the presence of any of the following features: (1) significant hypoechoic areas on b-mode ultrasound with a level of suspicion greater than 2 on a 1–5 scale, (2) increased flow on power or color Doppler when compared to the majority of the surrounding parenchyma [11, 12] or (3) calcified lesions. The mpUS was performed and interpreted by the same urologist performing the biopsy (C.P). After formal MRI/US image fusion, suspicious lesions were identified and evaluated with different US modalities prior to targeting (see above). No formal image fusion was performed between the different mpUS maps; for lesions not identified on MRI, cognitive fusion of the different US maps was performed by the urologist operating the US.
Biopsy sampling
Systematic 12 core biopsies were obtained in all individuals without a prior recent biopsy (i.e., last biopsy < 1 year ago). Samples were systematically obtained from the right mid-gland, right mid-lateral gland, right apex of the gland, right lateral apex of the gland, right base of the gland, right base of the lateral portion of the gland and then mirrored on the other side of the prostate. A total of four cores were obtained from each lesion identified on mpMRI, while a single core was obtained from lesions identified only on mpUS. If a particular sextant was found to have a lesion on ultrasound, the lesion was targeted and the sextant was not rebiopsied during systematic sampling.
Pathologic review
Pathologic review was performed by an experienced genitourinary pathologist. For each individual, the most aggressive histologic finding reported on the pathology report was considered for analysis. The pathologic results considered for analysis included high-grade prostatic intraepithelial neoplasia (hgPIN), atypical small acinar proliferation (ASAP), and PCa with its corresponding Gleason grade. Biopsies showing any of these findings were considered ‘non-negative’. Biopsies showing prostate cancer Gleason grade 3 + 3 or higher were considered ‘positive’, while those showing Gleason 3 + 4 or higher were considered as harboring ‘clinically significant’ disease. For the primary analysis, each individual was considered separately and the presence of clinically significant disease was noted regardless of the number of lesions or positive biopsy cores.
Statistical analysis
Individuals were grouped into four categories based on mpMRI and mpUS positivity. Correlation between the imaging findings and pathology results was then determined at the patient level. Basic frequencies and summary measures were calculated and compared between the four imaging groups. Numerical results were summarized using medians and interquartile ranges (IQR). Nonparametric statistical testing was conducted to compare the four diagnostic subgroups with regard to clinically relevant covariates. Krukal–Wallis tests were used to compare differences in the distribution of numerical variables, while Fisher’s exact tests were used for categorical variables. The utility of MRI, US and their combination with regard to detection of clinically significant PCa was explored by constructing 2 × 2 confusion matrices. Positive results noted on pathologic review (i.e., Gleason score > = 3 + 4) were considered true positives (TP). Measures of sensitivity, specificity and accuracy were then calculated for each imaging modality and different combinations. Data formatting was conducted using Microsoft Excel for Mac v.16.53 and all statistical analysis was done using the R platform v. 4.0.0 (R Core Team, 2019).
Results
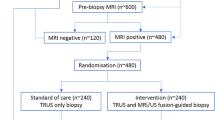
The final cohort consisted of 74 male patients who underwent both MRI and US-guided biopsy within 6 months. Participants were categorized into four groups for analysis based on their MRI and US findings (Fig. 1) and their baseline characteristics compared (Table 1). The median age of the sample was 66.5 years (IQR 62.4 to 70.9) and the median PSA level was 6.44 (IQR 4.46, 8.76). The median time from PSA testing to MRI was 2.0 months (IQR 1.1, 3.4) and the median time from MRI to biopsy was 1.5 months (IQR 0.7, 2.1). The timeline of the different diagnostic tests performed in each patient are shown individually (Fig. 2).
Timeline of diagnostic studies performed. Bar plot depicting the studies assessed for each participant and the time when these were performed. Results are displayed in months with regard to the date of MRI used in the study. Both the time from PSA to MRI (crosses) and time from MRI to biopsy (bars) are shown for each participant. The colors of the bars represent the maximum PIRADS score noted on MRI, with darker colors indicating higher scores. PIRADS prostate imaging-reporting and data system, MRI magnetic resonance imaging
No differences were observed between the four diagnostic groups in the time from PSA testing to MRI (Kruskal–Wallis, p = 0.6) nor in the time from MRI to biopsy (Kruskal–Wallis, p = 0.5). Of the 74 individuals included, 35 (47.3%) had prior biopsies and 18 (24.3%) had a prior positive biopsy (i.e., showing PCa Gleason 3 + 3 or higher). A total of 24 individuals (32.4%) were found to have clinically significant PCa, while 58 (78.4%) had a non-negative biopsy. Baseline characteristics were compared between the four diagnostic groups used for analysis. Individuals in the MRI( +)/US( +) group were found to have a significantly lower rate of prior biopsy compared to the other groups (34.1% vs 50.0, 90.9 and 50.0% for MRI + /US-, MRI-/US + and MRI-/US-, respectively, Fisher’s exact, p = 0.007). No significant differences in the other baseline characteristics studied were observed between the four diagnostic groups (Table 1). Notably, the distribution of PSA values was found to be comparable between the four diagnostic groups (Kruskal–Wallis, p = 0.4) (Fig. 3a). Similarly, no differences were observed between the four diagnostic groups in the proportion of non-negative biopsies (Fisher’s exact, p = 0.1), nor in the proportion of individuals with clinically significant PCa (Fisher’s exact, p = 0.5) (Fig. 3b).
PSA and biopsy results by MRI and US imaging findings. A Comparison of the distribution of PSA-values among participants with different imaging findings. B Breakdown of pathologic review results among participants with different imaging findings. Each bar represents the entirety of individuals in a given group, the colored bars represent the proportion of individuals with a given histologic result, all results are normalized to 100% (i.e., 1.00 proportion). ASAP atypical small acinar proliferation, MRI magnetic-resonance imaging, PCa prostate cancer, hg.PIN high-grade prostate intra-epithelial neoplasia, PIRADS prostate imaging-reporting and data system, PSA prostate-specific antigen, US ultrasound
MRI and ultrasound findings were compared at the patient-level with regard to the rate of detection of clinically significant PCa. The concordance between MRI, US and biopsy findings was evaluated (Fig. 4). Most individuals (n = 47, 63.5%) had concordant MRI and US results (i.e., both positive or both negative). Of the 41 individuals in which both MRI and US were positive, 16 (39.0%) were found to have clinically significant PCa. Of the 6 individuals with negative results in both imaging studies, only one was found to have clinically significant disease (16.7%). Of the 27 individuals with discordant results, 7 had clinically significant prostate cancer (25.9%). Five of these were only identified with MRI and two only with US.
Concordance between MRI, ultrasound, and pathology. Euler plot showing the concordance between MRI, US, and biopsy results in the study cohort. The areas of the ellipses represent the number of individuals in each group, with overlapping regions representing positive results in two or more tests. Results are also shown numerically (i.e., n individuals). The red circle corresponds to individuals who had negative results in all three tests. MRI magnetic resonance imaging, US ultrasound
Finally, we assessed the diagnostic accuracy of the different imaging modalities and their combination in detecting Gleason 3 + 4 or greater disease (Fig. 5). MRI alone was found to have a sensitivity of 87.5% but a specificity of only 28%. US alone was found to be less sensitive (75%) and slightly more specific (32%) than MRI alone. Overall, accuracy was lower for US alone compared to MRI alone (47.3 vs 45.9%). Next, we evaluated an approach in which positive results by either MRI or US were considered. As expected, this resulted in increased sensitivity (95.8%) but decreased specificity (10.0%). Notably, this approach also resulted in lower overall accuracy (37.8%). On the other hand, consideration of only positive results in both imaging modalities resulted in greater specificity (50.0%) and greater accuracy (55.4%) than each imaging modality alone, at the expense of lower sensitivity (66.7%).
Comparison of different imaging approaches in the detection of clinically significant prostate cancer. Bar plot showing the sensitivity, specificity and accuracy estimates when comparing the different imaging modalities in the detection of prostate cancer Gleason > = 3 + 4 by biopsy. Positive results in both MRI and US seemed to increase biopsy accuracy and specificity. Targeting of any suspicious lesion noted on imaging resulted in increased sensitivity while negatively impacting specificity. MRI magnetic resonance imaging, US ultrasound
Discussion
PCa diagnosis has progressed in the past decade to focus on the detection of clinically meaningful disease, and mpMRI has been established as an important pre-biopsy tool to improve detection [13]. However, ultrasound guidance remains the operational tool of choice at the time of performing a prostate biopsy and its diagnostic utility remains controversial. We used a pragmatic retrospective study design to assess the impact of detailed multiparametric ultrasonographic evaluation in the detection of Gleason grade ≥ 7 disease during mpMRI fusion biopsy of the prostate.
Recent studies have demonstrated that systematic biopsies are an integral part of the diagnostic algorithm. Patients with a low (< 15%) predicted chance of malignancy outside of the index lesion still showed a 7% rate of missed clinically significant prostate cancer when a systematic transrectal prostate biopsy was omitted. When omitted, only 16% of their cohort was spared a systematic (non-targeted) transrectal US-guided biopsy [14]. A similar result was identified in a trial published by Van der Leest et al. in 2019, where patients underwent a combined approach that included both non-targeted and MRI-guided biopsies. Clinically significant cancers were detected 30% of the time with both modalities, as opposed to 23% with systematic biopsy alone and 25% with MRI alone [15]. This was further confirmed in a study from 2020 suggesting that, combined systematic biopsy and MRI fusion guidance reduced both over and under diagnosis of PCa [16]. Our results are consistent with prior literature in that certain lesions are not readily visible with either US nor MRI. One individual with PCa Gleason > = 3 + 4 and 5 with less aggressive biopsy findings did not show any positive findings in either MRI or US. Notably, three of these individuals underwent transperineal saturation biopsies (i.e., 24 cores obtained systematically) which might have a better yield than a standard 12-core approach.
The operational need for US guidance during prostate biopsy (with or without MRI-based targeting) and the benefits that systematic non-targeted biopsies provide in PCa detection, make transrectal ultrasound fundamental for the successful completion of a prostate biopsy regardless of MRI targeting. Thus, it is not surprising that investigators have attempted to obtain additional value from the US images routinely obtained during prostate biopsy. In previous work, it was noted that the addition of ultrasound targeted lesions during MRI fusion biopsy resulted in a slight increase in finding clinically significant PCa [17]. Other studies have looked at the added value of targeting lesions seen on ultrasound at the time of MRI fusion biopsy. The results showed that there is added benefit to performing additional biopsies of hypoechoic areas seen on US. The area under the curve (AUC) of the receiver-operating characteristic (ROC) curve in this study was 0.85 for lesions targeted by both MRI and ultrasound vs 0.80 for US alone and 0.83 for MRI alone [18].
Our study demonstrates that mpUS assessment during mpMRI fusion biopsy of the prostate provides tangible benefits in the detection of clinically significant disease. In terms of overall detection, mpUS aided in detecting additional patients with clinically significant disease than mpMRI fusion biopsy alone. Although only 2 additional patients with Gleason > = 7 disease were detected by considering the mpUS results, this represents a roughly 10% increase in detection. These results are not clinically insignificant, particularly when noting the minimal costs and effort associated with the inclusion of mpUS results during standard transrectal US-guided biopsy. Other studies have also reported potential benefits in using US results in conjunction with mpMRI fusion biopsy. In the MRI-FIRST study, 21% of patients had a normal MRI [7]. These patients were subsequently biopsied, and 5 patients of the 53 who had “normal” MRIs had clinically significant PCa. Furthermore, 5.2% of the cohort would have been missed if systematic biopsy was skipped. This study defined systematic biopsy as including up to 2 hypoechoic lesions identified on transrectal US [7]. Overall, the combination of the two biopsy techniques yielded better results than one or the other alone.
The results from this study are consistent with previous literature in that a combined approach using both mpUS and mpMRI can improve PCa detection as well as diagnostic accuracy of clinically significant disease. The potential implementation of an approach that considers only lesions that are positive by both US and MRI yielded greater specificity and accuracy in the detection of clinically significant disease. While the use of a less restrictive ‘include-all’ approach, where any lesion identified in either US or MRI was considered positive, resulted in greatly improved sensitivity which approached nearly 100% for the detection of clinically significant disease (only 1 patient missed by both imaging modalities). Multiparametric transrectal US offered the opportunity to perform a multi-feature evaluation of the prostate looking for suspicious findings at the same time that it allowed guidance of non-targeted biopsies and MRI fusion targeting, rendering it a viable alternative that might increase the diagnostic utility of prostate needle biopsy in general and potentially allow for personalized decision-making at the time of targeting suspicious prostatic lesions. It is important to note that a pragmatic approach was chosen for the design of this study, focusing on standard US modalities (i.e., b-mode, color Doppler, power Doppler). However, these have been reported not to be the most sensitive for PCa detection. In fact, extensive research in recent years has shown that other modalities such as elastography or contrast-enhanced US can serve as powerful tools for the detection of PCa [19, 20], and their inclusion in predictive models has been shown to boost classification performance [21,22,23]. Although not included in this study, future research efforts exploring the utility of mpUS could benefit from the inclusion of these additional image modalities.
Limitations of the study include its retrospective nature as well as relatively small sample size which render it susceptible to selection bias. However, sample size is comparable to prior studies in this area [7, 15]. Additionally, while the urologist performing the ultrasound has significant experience in the evaluation of prostatic lesions on ultrasound, this may not be generalizable to other urologists who might require additional training in the interpretation of mpUS findings. We opted for an approach that would minimize inter-operator variability given that transrectal US of the prostate is an operator-dependent test and the interpretation of its findings requires considerable expertise. Although the ultrasound features included might differ in their value to detect clinically significant PCa, we were unable to assess them independently due to lack of statistical power. It is possible that the results observed are due to overrepresentation of specific US features, in which case a multiparametric US assessment would not be required. However, determination of the clinical value of every single ultrasonographic feature was outside the scope of our study and future research endeavors with larger sample sizes should investigate this issue further. Furthermore, although the lesions identified on MRI and US were recorded separately in each sextant, results were then aggregated for analysis. Therefore, the concordance rates between the two imaging modalities must be interpreted with caution as these could be overestimated when considering the whole gland instead of each lesion separately. Finally, it must also be considered that results could be influenced by sampling bias due to the fact that more cores were obtained when targeting MRI-positive lesions compared to US-positive lesions.
Conclusion
Multiparametric US of the prostate seems to provide tangible benefits in terms of improved detection and diagnostic accuracy for clinically significant PCa. The results from this pragmatic study suggest that mpUS can be used as an adjunct to MRI-targeted and systematic non-targeted biopsies to improve either sensitivity or specificity depending on the approach used. Implementation of this technology during the conduction of prostate needle biopsy appears to be feasible and cost-effective, however, further studies are needed to optimize its use and determine the diagnostic value of the different US features evaluated.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Carroll PR, Parsons JK, Andriole G, Bahnson RR, Barocas DA, Castle EP et al (2015) NCCN clinical practice guidelines prostate cancer early detection, version 2.2015. J Natl Compr Canc Netw 3(12):1534–1561
Verma S, Rosenkrantz AB, Choyke P, Eberhardt SC, Eggener SE, Gaitonde K et al (2017) Commentary regarding a recent collaborative consensus statement addressing prostate MRI and MRI-targeted biopsy in patients with a prior negative prostate biopsy. Abdom Radiol (NY) 42(2):346–349
Dähnert WF, Hamper UM, Eggleston JC, Walsh PC, Sanders RC (1986) Prostatic evaluation by transrectal sonography with histopathologic correlation: the echopenic appearance of early carcinoma. Radiology 158(1):97–102
Gosselaar C, Roobol MJ, Roemeling S, Wolters T, van Leenders GJLH, Schröder FH (2008) The value of an additional hypoechoic lesion-directed biopsy core for detecting prostate cancer. BJU Int 101(6):685–690
Nelson ED, Slotoroff CB, Gomella LG, Halpern EJ (2007) Targeted biopsy of the prostate: the impact of color Doppler imaging and elastography on prostate cancer detection and Gleason score. Urology 70(6):1136–1140
Ezquer A, Ortega Hrescak MC, Sanagua C, Roggia-Rebullida P, López R, Cenice F et al (2015) Transrectal Doppler ultrasound during prostate biopsy: clinical utility and limitations. Actas Urol Esp 39(1):13–19
Rouvière O, Puech P, Renard-Penna R, Claudon M, Roy C, Mège-Lechevallier F et al (2019) Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol 20(1):100–109
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42(2):377–381
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L et al (2019) The REDCap consortium: building an international community of software platform partners. J Biomed Inform 95:103208. https://doi.org/10.1016/j.jbi.2019.103208
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ et al (2016) PI-RADS prostate imaging - reporting and data system: 2015, version 2. Eur Urol 69(1):16–40
Feleppa EJ, Mamou J, Porter CR, Machi J (2011) Quantitative ultrasound in cancer imaging. Semin Oncol 38(1):136–150
Lee HY, Lee HJ, Byun S-S, Lee SE, Hong SK, Kim SH (2009) Classification of focal prostatic lesions on transrectal ultrasound (TRUS) and the accuracy of TRUS to diagnose prostate cancer. Korean J Radiol 10(3):244–251
Bjurlin MA, Carroll PR, Eggener S, Fulgham PF, Margolis DJ, Pinto PA et al (2020) Update of the standard operating procedure on the use of multiparametric magnetic resonance imaging for the diagnosis, staging and management of prostate cancer. J Urol 203(4):706–712
Dell’Oglio P, Stabile A, Soligo M, Brembilla G, Esposito A, Gandaglia G et al (2020) There is no way to avoid systematic prostate biopsies in addition to multiparametric magnetic resonance imaging targeted biopsies. Eur Urol Oncol 3:112–118. https://doi.org/10.1016/j.euo.2019.03.002
van der Leest M, Cornel E, Israël B, Hendriks R, Padhani AR, Hoogenboom M et al (2019) Head-to-head comparison of transrectal ultrasound-guided prostate biopsy versus multiparametric prostate resonance imaging with subsequent magnetic resonance-guided biopsy in biopsy-naïve men with elevated prostate-specific antigen: a large prospective multicenter clinical study. Eur Urol 75(4):570–578
Ahdoot M, Wilbur AR, Reese SE, Lebastchi AH, Mehralivand S, Gomella PT et al (2020) MRI-targeted, systematic, and combined biopsy for prostate cancer diagnosis. N Engl J Med 382(10):917–928
Shakir NA, Siddiqui MM, George AK, Kongnyuy M, Ho R, Fascelli M et al (2017) Should hypoechoic lesions on transrectal ultrasound be sampled during magnetic resonance imaging-targeted prostate biopsy? Urology 105:113–117
Garcia-Reyes K, Nguyen HG, Zagoria RJ, Shinohara K, Carroll PR, Behr SC et al (2018) Impact of lesion visibility on transrectal ultrasound on the prediction of clinically significant prostate cancer (Gleason score 3 + 4 or greater) with transrectal ultrasound-magnetic resonance imaging fusion biopsy. J Urol 199(3):699–705
Rouvière O, Melodelima C, Hoang Dinh A, Bratan F, Pagnoux G, Sanzalone T et al (2017) Stiffness of benign and malignant prostate tissue measured by shear-wave elastography: a preliminary study. Eur Radiol 27(5):1858–1866
Mannaerts CK, Engelbrecht MRW, Postema AW, van Kollenburg RAA, Hoeks CMA, Savci-Heijink CD et al (2020) Detection of clinically significant prostate cancer in biopsy-naïve men: direct comparison of systematic biopsy, multiparametric MRI- and contrast-ultrasound-dispersion imaging-targeted biopsy. BJU Int 126(4):481–493
Loch T, Leuschner I, Genberg C, Weichert-Jacobsen K, Küppers F, Yfantis E et al (1999) Artificial neural network analysis (ANNA) of prostatic transrectal ultrasound. Prostate 39(3):198–204
Tokas T, Grabski B, Paul U, Bäurle L, Loch T (2018) A 12-year follow-up of ANNA/C-TRUS image-targeted biopsies in patients suspicious for prostate cancer. World J Urol 36(5):699–704
Wildeboer RR, Mannaerts CK, van Sloun RJG, Budäus L, Tilki D, Wijkstra H et al (2020) Automated multiparametric localization of prostate cancer based on B-mode, shear-wave elastography, and contrast-enhanced ultrasound radiomics. Eur Radiol 30(2):806–815
Acknowledgements
Acknowledgments of people, grants, funds, etc. or “Not applicable”.
Author information
Authors and Affiliations
Contributions
NJ: Manuscript writing, manuscript editing, data collection, data management. JF: Manuscript editing. HK: Data analysis, manuscript editing. RGDN: data analysis, manuscript writing, manuscript editing. OH: Manuscript editing, supervision. JPF: Manuscript editing. CP: Manuscript editing, project conception, supervision, project management, data collection.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jung, N., DiNatale, R.G., Frankel, J. et al. The role of multiparametric ultrasound in the detection of clinically significant prostate cancer. World J Urol 41, 663–671 (2023). https://doi.org/10.1007/s00345-022-04122-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-022-04122-z