Abstract
Purpose
To evaluate on a lesion-by-lesion basis Narrow-Band Imaging flexible videoscopy (NBI-FV) in the detection of cancer compared to White-Light Imaging flexible videoscopy (WLI-FV).
Methods
WLI-FV and NBI-FV were sequentially performed in patients scheduled for TURBT for primary bladder cancer. Suspicious findings were individually harvested and characterized under WLI-FV (suspicious/non-suspicious) and NBI-FV (5-point Likert scale) and pathology. The primary objective was to determine if NBI-FV informed at least 20% more cancer lesions than WLI-FV (Relative true-positive rate > 1.19). A minimum of 120 specimens was to be analyzed to reach 90% power.
Results
Of 147 specimens taken in 68 patients, 101 were found suspicious under WLI-FV and 64 (64/101, 63.4%) confirmed as cancer. Of the 46 lesions undetected by WLI-VF, 16 were found positive for cancer (16/46, 34.8%). For NBI-FV, a significant increase in positive samples was observed with increments in Likert scale (p < 0.0002). Relative true-positive rate was 1.22 (95% CI 1.12–1.39)—NBI-FV detected 22% more cancer lesions compared to WLI-FV. Relative false-positive rate was 1.35 (95% CI 1.19–1.59).
Conclusion
Researching alterations in mucosa and microvasculature by narrow-band imaging flexible videoscopy augmented by 22% the detection of cancer foci and contributed to the objective of complete resection of all visible lesions. Conversely, it entailed a 35% increase in false-positive results compared to white-light imaging, although the structured analysis of narrow-band imaging findings might be used to grade suspicion according to the Likert scale and balance the risk of a false-positive result to the benefit of demonstrating cancer.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Most cases of bladder cancer do not exhibit detrusor invasion at first diagnosis befitting the definition of non-muscle-invasive bladder cancer (NMIBC) for which optimal transurethral resection of the bladder tumor (TURBT) is crucial to risk stratification and treatment [1, 2]. However, the results of TURBT are highly heterogeneous [3] with suggestion that early recurrence might relate to imperfect visualization and incomplete excision of the tumors [4].
Since Folkman, the ability to elicit angiogenesis from the host microvasculature is considered one hallmark of cancer [5]. Narrow-Band Imaging (NBI) was developed to take advantage of this using for illumination 415 and 540 nm wavelengths that are selectively absorbed by hemoglobin and facilitate the visualization of the sub-mucosa microvasculature [6]. In addition, while the normal bladder mucosa is thin and transparent under White-Light Imaging (WLI), the increased scattering of the light by accumulated nuclei and organelles may reveal areas of increased thickness or disorganization of the urothelium. This phenomenon is digitally reinforced and shifted to the red in NBI that thereby also provides insights into the structure of the mucosa. Improved detection with NBI was consistently observed across all fields of endoscopy, notably for bladder tumors [7, 8].
However, the thorough analysis of the entire bladder is necessary before engaging resection with the 12° or 30° lenses that are needed to visualize the resection loop but are restrained in field and depth of view and in access to the entirety of the bladder lining [9, 10]. The objective of this study was to evaluate on a lesion-by-lesion basis the contribution of NBI obtained by flexible videoscopy (NBI-FV) to the detection of cancer in patients due for primary TURBT, compared to WLI flexible videoscopy (WLI-FV).
Patients and methods
Patients and study design (Fig. 1a)
Patients scheduled for TURBT were enrolled (6/2013-9/2014) in a multicentre study after informed consent. All investigators had over 10 years of experience in NMIBC detection and treatment. Following the National Institute of Health [11], this study that was designed to validate the diagnostic performances of a CE-marked diagnostic instrument did not qualify as a clinical trial and was therefore not registered. The Institutional Review Board of each institution approved the study.
a Flow chart of the prospective matched pair assessment of the relative true-positive and relative true-negative rates of cancer detection by narrow-band imaging, compared to white-light imaging. b Cancer distribution of white-light imaging and narrow-band imaging findings in 147 lesions detected during flexible videoscopy (LS Likert 5-score scale)
Patients were sequentially imaged in WLI and NBI with the Olympus CYF-VH flexible HD video scope and high-definition TV monitor (Olympus Europa SE & Co, Hamburg). The same investigator performed the two procedures to control inter-operator variability and to follow the real-life nature of the study. Suspicious findings were positioned on bladder diagrams. Under WLI-FV, the lesions were considered as either suspicious when they were found by the investigator to warrant biopsy or resection, or non-suspicious. On the contrary, for NBI-FV the suspicion was scored on the Likert 5-score scale (LS) from not (score 1), to highly suspicious (score 5). Representative pictures organized along the alterations observed in the structures of the mucosa and microvasculature are presented in Fig. 2a. All suspicious findings were individually resected and processed for pathological examination by the Institution pathologist, blinded to the techniques used for detection. Additional resections or biopsies could be taken at the discretion of the investigators. As opposed to the only previous report on flexible cystoscopy in the context of primary NMIBC [12], no biopsies were taken in areas found unsuspicious on both WLI and NBI-FV.
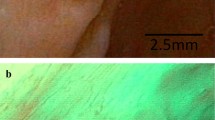
A Representative cases of NBI-FV findings organized along increasing abnormalities in the urothelium structure and microvasculature and graded according to Likert 5-score scale. LS1-not suspicious: Well-organized microvasculature organized along a geometric pattern underneath the transparent (non-visible) mucosal layer. LS2-mildly suspicious: Isolated alterations of microvasculature or epithelium. (a) Reddish area that demonstrates mucosal thickening above a well-organized, non-suspicious microvasculature. (b) Dense microvasculature but no tortuosity. LS3-moderately suspicious: (a) Raised urothelium that obscures the vision of the capillaries. (b) Reddish patch of thick mucosa with dense microvasculature. (c) Dense and tortuous microvasculature but no definite abnormalities of the mucosa. LS4-suspicious: (a) Micro-papillary epithelium with dense microvasculature. (b) Large veins and thick mucosa in the vicinity of a papillary tumour. LS5-highly suspicious: Exophytic lesion above a dense and tortuous capillary network. B Distribution of positive samples in relation to Likert 5-score scale
2002 AJCC-TNM staging system and 1973 WHO manual on histological typing of urinary bladder tumors were used for reporting stage and grade [13].
Statistical methodology
As highlighted by the photodynamic diagnosis literature [14], WLI considered as the diagnostic standard is far from optimal. It is, therefore, not possible to ascertain the presence or absence of cancer on the basis of WLI endoscopy alone whose unbiased estimates of the diagnostic characteristics remain elusive.
Such limitations are classical when individuals negative for a diagnostic test are not verified. However, when comparing two systems of diagnosis, unbiased estimates of the relative true-positive and relative true-negative rates of one system in relation to another can be obtained [15]. Therefore, contrary to previous reports cancer detection under NBI-FV was not assessed in terms of sensitivity or specificity but of relative True-Positive Rate (rTPR), with relative True-Negative Rate (rTNR) as the second endpoint [15]. 95% confidence intervals (95%CI) of rTPR and rTNR were computed according to the modified Wald method [16]. Detection figures according to NBI-FV Likert scores were compared using the Kruskall–Wallis test.
Sample size was obtained from the confidence interval theory [17, 18]. Detection rate with WLI-FV was approximated to 80% and it was hypothesized that to have clinical significance, NBI-FV would have to detect at least 20% more cases than WLI-FV (rTPR-NBI/WLI > 1.19). With a unilateral type I error rate of 2.5%, 120 lesions were required to obtain a power of 90%. Considering that most cancers are multifocal, no more than 80 patients were necessary to ascertain whether NBI-FV improved detection relative to WLI-FV.
Results
147 specimens were obtained in 68 patients [Mean and (95% CI); age: 70.8 years (63.4–74.2), % of male patients: 80.9% (69.9–88.5), current smokers: 51.5% (39.8–62.9), Fig. 1b]. Eighty lesions were confirmed positive at pathology (80/147; 55.8%, Table 1). In eleven patients, NBI-FV detected fourteen cancer lesions that had been overlooked by WLI-VF with upgrading or upstaging in six patients (Fig. 1b; Table 1). Of the seven additional lesions found suspicious in the trigon and bladder neck areas, two were positive for cancer. Flexible videoscopy findings in relation to pathology are summarized in Table 2. A significant increase (p < 0.0002) in the proportion of samples confirmed positive for cancer was observed with increments in the Likert scale (Fig. 2a).
The relative true-positive rate was 1.22 (95% CI 1.12–1.39) that is that NBI-FV detected 22% more cancer lesions compared to WLI-FV. The relative false-positive rate was 1.35 (95% CI 1.19–1.59), which indicated a 35% increase in false-negative specimens with Narrow-Band Imaging, compared to White-Light Imaging (Table 2). Positive predictive values for WLI-FV and NBI-FV were 63.4% (95% CI 53.6–72.1) and 59.1% (95% CI 50.6–67.1), respectively. Negative predictive values were 65.2% (95% CI 50.7–77.4) and 86.7% (95% CI 60.9–97.5).
WLI-FV sensitivity and specificity were 80.0% (95% CI 69.6–88.1%) and 44.8% (95% CI 32.6–57.4%), respectively, while the corresponding NBI-FV values were 97.5% (95% CI 91.3–99.7%) and 19.4% (95% CI 10.8–30.9%) demonstrating higher sensitivity but lower specificity for NBI-FV (both p < 0.001).
Discussion
In the best-case scenario where the limitations of rigid cystoscopy were overcome by the wide field and multiple angles of view allowed by flexible endoscopy and the optimized vision provided by chip-on-tip technology and high-definition monitor, white-light imaging failed to detect at least one cancer lesion out of five (16/80, 20.0%). Alternately, NBI-FV detected 22% more cancer lesions compared to WLI-FV, meeting the primary objective of the study (relative true-positive rate: 1.22). It also corrected the misallocation of grade or stage in a minority of patients (6/68, 8.8%).
This observation was in keeping with a recent meta-analysis, where compared to WLI, NBI was shown to carry additional detection rates of 9.9% and 18.6% in per-patient and per-lesion analysis, respectively [19]. In view of such limitations, the recent NICE guidelines concluded that at the time of TURBT, WLI should not be used as single modality but complemented by NBI, photodynamic diagnosis or a urinary biomarker test [2]. Here, we extended to the operating theater the clinical relevance of NBI flexible videoscopy already reported in the fields of primary diagnosis [7] and follow-up [20].
However, NBI-FV specificity (19.4%) was degraded compared to the already imperfect specificity of WLI-FV (44.8%). By design, the specimens were taken only when any one of the two imaging modalities was found suspicious. The number of negative findings was, therefore, not inflated with negative protocol biopsies and the specificity figures, that is the proportion of specimens found negative for cancer in non-suspicious areas, were lower than those reported in series that integrated random biopsies of non-suspicious mucosa [12, 21]. We believe that the present figures offered a better reflection of the clinical real life where inflammation and metaplastic conditions are not uncommon and random biopsies hardly recommended. In spite of that, NBI-FV entailed a 35% increase in false-negative specimens, which in the specific context of patients already due for TURBT would constitute an acceptable trade-off for optimized surgery.
NBI may highlight only two of the six distinct micro-environmental barriers that are overcome by premalignant lesions to establish cancer, that is overcoming contact inhibition and the resulting increase in thickness and angiogenesis to control acidosis [22]. Although investigators were not given recommendations to characterize their increments in suspicion, representative cases of non-papillary lesions drawn from the intraoperative records collected at the time of surgery were retrospectively organized along increasing degrees of abnormalities in the urothelium and microvasculature (Fig. 2a). By allowing the structured analysis of NBI findings, this instrument would constitute a valuable tool in the translation of NBI into the clinical routine of TURBT. Indeed, one major limitation in endoscopy comes from the fact that while the detailed analysis of the lesions may translate into a large spectrum of suspicions, the ultimate decision of the surgeon remains dichotomous to resect or leave untouched any given area of interest. While this decision is quite straightforward for typical cases of large exophytic tumors, it may be difficult in small or non-papillary abnormalities. We observed that the Likert score of NBI findings was significantly correlated to the probability of cancer in the biopsy or resection specimens (Fig. 2b) and could be used to balance the benefit of demonstrating cancer to the risk of false-positive result.
The demonstration of relevant clinical outcomes in the photodynamic diagnosis [14] or narrow-band imaging [19, 23] literatures required prolonged and costly trials. We offer that the present methodology may be better suited to the constraints of modern endoscopy where ameliorations in scopes, light sources, software and monitors drive a rapid evolution in manufacturer’s catalogs. While conformity with safety standards is certified for all new products (CE marking in Europe, FDA approval in USA), clinical relevance is more often inferred than proven. The present evaluation addressed detection, a crucial prerequisite in all urology guidelines [2, 4], taking advantage of the methodology of relative true-positive and negative rates that was designed to address the uncertain nature of a negative diagnostic test when it is not verified [15]. Moreover, this design did not constitute a clinical trial according to the most recent National Institute of Health definition [11], a key advantage to streamline clinical evaluation and accelerate the introduction of modern endoscopy technology into the clinics.
Although efforts were paid to control imprecision in design and analysis, some limitations must be acknowledged.
First, to follow the clinical routine where it is considered as an add-on to WLI, the same operator conducted NBI-FV after WLI-FV, thereby taking advantage of WLI-FV to inform on NBI-FV detection of cancer lesions. To address this bias of sequence, Shen randomly allocated the rank order of imaging between two separate surgeons. He still observed a significant 15% increase in sensitivity for NBI-FV, compared to WLI-FV (92.9% vs. 77.7%). Another recent multicentre study using the same design confirmed similar improvement [24].
Second, the complex biology of NMIBC where cancer-primed cell may show little morphological changes [22, 25] supports a continuum of endoscopic presentations from a normal looking mucosa to minute alterations in thickness and microvasculature to self-evident exophytic tumors. Conversely, pathologists must dichotomize their understanding of the specimens as either cancer or non-cancer, a demanding decision as shown by the large inter-observer variability coefficients reported in most studies [26]. This is most exacting for flat urothelial lesions that are being dismembered into a wide array of entities, some with clear premalignant potential [27]. In keeping with the real-life nature of the study, neither central review nor recommendations as to the use of immunochemistry for select cases were set, which may have influenced in either way the correct allocation of lesions of minute pathological alterations and impacted sensitivity and sensitivity performances of both modalities.
Last, the benefit on recurrence and progression of improved detection may be debated. This was not addressed here, although two meta-analyses confirmed NBI as a valuable adjunct to WLI to control recurrence rates at 3 and 12 months after TURBT [19, 23] while reduction in recurrence rates was observed in the subset of low-risk patients in the sole randomized controlled trial comparing NBI to WLI [28].
Conclusion
Researching alterations in mucosa and microvasculature by narrow-band imaging flexible videoscopy augmented by 22% the detection of cancer foci and contributed to the objective of complete resection of all visible lesions. Conversely, it entailed a 35% increase in false-positive results compared to white-light imaging, although the structured analysis of narrow-band imaging findings might be used to grade suspicion according to the Likert scale and balance the risk of a false-positive result to the benefit of demonstrating cancer.
References
Sylvester RJ, van der Meijden AP, Oosterlinck W, Witjes JA, Bouffioux C, Denis L, Newling DW, Kurth K (2006) Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol 49(3):466–477 (discussion 75-7)
Power NE, Izawa J (2016) Comparison of guidelines on non-muscle invasive bladder cancer (EAU, CUA, AUA, NCCN, NICE). Bladder Cancer 2(1):27–36
Brausi M, Collette L, Kurth K, van der Meijden AP, Oosterlinck W, Witjes JA, Newling D, Bouffioux C, Sylvester RJ, Group EG-UTCC (2002) Variability in the recurrence rate at first follow-up cystoscopy after TUR in stage Ta T1 transitional cell carcinoma of the bladder: a combined analysis of seven EORTC studies. Eur Urol 41(5):523–531
Babjuk M, Bohle A, Burger M, Capoun O, Cohen D, Comperat EM, Hernandez V, Kaasinen E, Palou J, Roupret M, van Rhijn BW, Shariat SF, Soukup V, Sylvester RJ, Zigeuner R (2017) EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2016. Eur Urol 71(3):447–461
Folkman J, Merler E, Abernathy C, Williams G (1971) Isolation of a tumor factor responsible for angiogenesis. J Exp Med 133(2):275–288
Gono K, Obi T, Yamaguchi M, Ohyama N, Machida H, Sano Y, Yoshida S, Hamamoto Y, Endo T (2004) Appearance of enhanced tissue features in narrow-band endoscopic imaging. J Biomed Opt 9(3):568–577
Bryan RTBL, Wallace DM (2008) Narrow-band imaging flexible cystoscopy in the detection of recurrent urothelial cancer of the bladder. BJU Int 101:702–705 (discussion 5–6)
Cauberg EC, Kloen S, Visser M, de la Rosette JJ, Babjuk M, Soukup V, Pesl M, Duskova J, de Reijke TM (2010) Narrow band imaging cystoscopy improves the detection of non-muscle-invasive bladder cancer. Urology 76(3):658–663
Jones S, Larchian W (2012) Non-muscle-invasive bladder cancer (Ta, T1, and CIS). In: Kavoussi LR, Novick AC, Partin AW, Peters CA (eds) Campbell-walsh urology tenth edition IV. Elsevier Saunders, Philadelphia, pp 2335–2354
Ragsdale C, Hreha H, Hardesty JS, Yune JJ, Chan PJ, Siddighi S (2017) What is the best lens angle for rigid diagnostic cystoscopy? A comparison of 30-degree and 70-degree lenses. Female Pelvic Med Reconstr Surg 24(5):371–374
NIH’s definition of a clinical trial (2018)https://grants.nih.gov/policy/clinical-trials/definition.htm Bethesda, Maryland 20892 (USA) [cited 26 Mar 2018]
Shen YJ, Zhu YP, Ye DW, Yao XD, Zhang SL, Dai B, Zhang HL, Zhu Y (2012) Narrow-band imaging flexible cystoscopy in the detection of primary non-muscle invasive bladder cancer: a “second look” matters? Int Urol Nephrol 44(2):451–457
Mostofi FK, Sobin LH, Torloni H (1973) Histological typing of urinary bladder tumours. World Health Organization, Geneva, p 1973
Kausch I, Sommerauer M, Montorsi F, Stenzl A, Jacqmin D, Jichlinski P, Jocham D, Ziegler A, Vonthein R (2010) Photodynamic diagnosis in non-muscle-invasive bladder cancer: a systematic review and cumulative analysis of prospective studies. Eur Urol 57(4):595–606
Chock CIL, Berry G, Glasziou P (2010) Comparing dichotomous screening tests when individuals negative on both tests are not verified. J Clin Epidemiol 50(11):1211–1217
Agresti A, Coull B (1998) Approximate is better than “exact” for interval estimation of binomial proportions. Am Stat 52(2):119–126
Alonzo TA, Pepe MS, Moskowitz CS (2002) Sample size calculations for comparative studies of medical tests for detecting presence of disease. Stat Med 21(6):835–852
Alonzo TA, Kittelson JM (2006) A novel design for estimating relative accuracy of screening tests when complete disease verification is not feasible. Biometrics 62(2):605–612
Xiong Y, Li J, Ma S, Ge J, Zhou L, Li D, Chen Q (2017) A meta-analysis of narrow band imaging for the diagnosis and therapeutic outcome of non-muscle invasive bladder cancer. PLoS One 12(2):e0170819
Herr HW, Donat SM (2008) A comparison of white-light cystoscopy and narrow-band imaging cystoscopy to detect bladder tumour recurrences. BJU Int 102(9):1111–1114
Tatsugami K, Kuroiwa K, Kamoto T, Nishiyama H, Watanabe J, Ishikawa S, Shinohara N, Sazawa A, Fukushima S, Naito S (2010) Evaluation of narrow-band imaging as a complementary method for the detection of bladder cancer. J Endourol 24(11):1807–1811
Gatenby RA, Gillies RJ (2008) A microenvironmental model of carcinogenesis. Nat Rev Cancer 8(1):56–61
Lee JY, Cho KS, Kang DH, Jung HD, Kwon JK, Oh CK, Ham WS, Choi YD (2015) A network meta-analysis of therapeutic outcomes after new image technology-assisted trans-urethral resection for non-muscle invasive bladder cancer: 5-aminolaevulinic acid fluorescence vs hexylaminolevulinate fluorescence vs narrow band imaging. BMC Cancer 15:566
Ye Z, Hu J, Song X, Li F, Zhao X, Chen S, Wang X, He D, Fan J, Ye D, Xing J, Pan T, Wang D (2015) A comparison of NBI and WLI cystoscopy in detecting non-muscle-invasive bladder cancer: a prospective, randomized and multi-center study. Sci Rep 5:10905
Curtius K, Wright NA, Graham TA (2018) An evolutionary perspective on field cancerization. Nat Rev Cancer 18(1):19–32
Soukup V, Capoun O, Cohen D, Hernandez V, Babjuk M, Burger M, Comperat E, Gontero P, Lam T, MacLennan S, Mostafid AH, Palou J, van Rhijn BWG, Roupret M, Shariat SF, Sylvester R, Yuan Y, Zigeuner R (2017) Prognostic performance and reproducibility of the 1973 and 2004/2016 World Health Organization grading classification systems in non-muscle-invasive bladder cancer: a European association of urology non-muscle invasive bladder cancer guidelines panel systematic review. Eur Urol 72(5):801–813
Lopez-Beltran A, Marques RC, Montironi R, Reymundo C, Fonseca J, Cheng L (2015) Dysplasia and carcinoma in situ of the urinary bladder. Anal Quant Cytopathol Histpathol 37(1):29–38
Naito S, Algaba F, Babjuk M, Bryan RT, Sun YH, Valiquette L, de la Rosette J (2016) The clinical research office of the endourological society (CROES) Multicentre randomised trial of narrow band imaging-assisted transurethral resection of bladder tumour (TURBT) versus conventional white light imaging-assisted TURBT in primary non-muscle-invasive bladder cancer patients: trial protocol and 1-year results. Eur Urol 70(3):506–515 (Group CNBIGS)
Acknowledgements
Dr Hannah Levy for editorial assistance, Mr Gamot, urology nurse, for his input in the collection of representative NBI and WLI-FV cases.
Author information
Authors and Affiliations
Contributions
LPD: Protocol development, data collection, RZ: Protocol development, data collection, JMG: Data collection, JPR: Protocol development, data collection, MR: Data collection, TF: Protocol development, data analysis, BM: Protocol development, data collection, data analysis, manuscript writing.
Corresponding author
Ethics declarations
Conflict of interest
Dalgaard: none, Zare: none, Gaya: none, Joan Palou Redorta: Participation to Olympus Europe advisory board, Mathieu Roumiguié: none, Thomas Filleron: none, Bernard Malavaud: Participation to Olympus Europe advisory board, Honoraria from Olympus Europe for speaking at Symposia.
Research involving human participants
The study was approved by institutional ethics committee and performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Dalgaard, L.P., Zare, R., Gaya, J.M. et al. Prospective evaluation of the performances of narrow-band imaging flexible videoscopy relative to white-light imaging flexible videoscopy, in patients scheduled for transurethral resection of a primary NMIBC. World J Urol 37, 1615–1621 (2019). https://doi.org/10.1007/s00345-018-2537-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2537-7