Abstract
Purpose
The aim of our study was to assess whether multiparametric magnetic resonance imaging (MP-MRI) of the prostate with three parameters (PS3: T2-weighted, DWI, and DCE) benefits from an additional fourth parameter (PS4: including 1H-MRSI) in the detection and grading of prostate cancer (PCa) at 3 T.
Methods
MP-MRI was performed in 64 patients (mean 66.7 years, mean PSA 13 ng/ml). Reference standard was obtained by histopathology. Two readers independently evaluated the images. A summation score of each individual parameter for three parameters (PS3) and for four parameters (PS4) was calculated.
Results
In 52 (81.3 %) of 64 patients, histopathology confirmed a PCa. The diagnostic performance for PCa detection of PS4 (O1: 91.7 %, O2: 91.3 %) equaled that of PS3 (O1: 92.8 %, O2: 92.2 %, P > 0.05). Prediction of high-grade PCa by PS4 (O1: 75.1 %, O2: 74.7 %) was as good as with PS3 (O1: 75.1 %, O2: 72.8 %, P > 0.05). Kappa agreement between the two readers was substantial (0.734 PS4) to moderate (0.558 PS3).
Conclusions
MP-MRI with four parameters including 1H-MRSI does not increase the detection and grading of prostate cancer at 3 T compared to MP-MRI with three parameters. A sum score accurately detects PCa at 3 T without an endorectal coil and shows potential for the prediction of tumor grade.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Multiparametric magnetic resonance imaging (MP-MRI) of the prostate that includes T2-weighted (T2), diffusion-weighted imaging (DWI), dynamic contrast-enhanced MRI (DCE), and 3D proton spectroscopic imaging (1H-MRSI) has been established and has been shown to be of high diagnostic accuracy for the detection and for local staging of prostate cancer (PCa) [1].
In this setting, 1H-MRSI is considered an additional optional technique. Compared to the other three MRI parameters, 1H-MRSI is relatively time-consuming, often leading to motion artifacts and deteriorated data [2, 3]. Despite initial encouraging results, the additional value of 1H-MRSI has not been fully established [4]. Thus, several international societies strongly support continued research on the value of 1H-MRSI in MP-MRI of the prostate [5].
The aim of our study was to assess whether MP-MRI of the prostate with three parameters including DCE, DWI, and T2-weighted benefits from an additional fourth parameter (1H-MRSI) in the detection and grading of prostate cancer at 3 T.
Materials and methods
Patients
From May 2012 to June 2013, 64 consecutive patients without contraindications to MRI of the prostate were eligible for this prospective institutional review board-approved, single-center study. Written, informed consent was obtained from all patients.
Inclusion criteria were as follows: elevated prostate-specific antigen (PSA) levels >4.0 ng/ml; suspect findings at digital rectal examination; or TRUS and histopathological evaluation of the suspect finding. In case of negative biopsy, a 24-month follow-up was ensured.
Patients with a history of prostate therapy (e.g., brachytherapy) or therapy to other organs in the vicinity of the prostate, or hormonal therapy, were excluded.
MR imaging
In case of prior biopsy, MR imaging was always performed after at least 4–6 weeks to avoid artifacts caused by post-interventional hemorrhage [2]. All examinations were performed using the vendor-supplied combined spine array and body array receive-only coils on a 3-T MRI system (Tim Trio, Siemens Healthcare, Erlangen, Germany). No endorectal coil was used for the examinations. After emptying the bladder, the patients were positioned in the feet-first supine position. An antiperistaltic agent, 10 mg of hyoscine butylbromide (Buscopan®, Boehringer Ingelheim, GmbH, Germany), was injected i.m. To improve the image quality and avoid air bubbles, the rectum was filled with ultrasound gel.
The MP-MRI protocol included the following sequences:
Anatomical T2-weighted turbo spin echo in all three planes (TR/TE/TI 4000/101/230 ms; field of view (FOV) 200 mm; 20 slices at 3.0. mm; matrix 320; flip angle 150°; TA ≤ 4:10 per plane) GRAPPA factor 2.
Diffusion-weighted, single-shot, echo-planar imaging with inversion recovery fat suppression (DWI, TR/TE 3300/60 ms; spectrally adiabatic inversion recovery (SPAIR) fat suppression; FOV 260 mm; 20 slices at 3.6 mm; matrix 160; eight averages; b values of 0, 100, 400, and 800 s/mm2; TA 4:34 min) GRAPPA factor 2.
Three-dimensional proton magnetic resonance spectroscopic imaging (1H-MRSI, TR = 750 ms; TE = 145 ms; six averages; spectral fat and water suppression each with a bandwidth of 1.55 ppm; delta positions of water and fat set to 0 and −3.4 ppm, respectively). The effective spatial resolution was 7 by 7 by 7 mm, and TA 8:14 after manual first- and second-order shimming (1–3 min), resulting in typical full width at half maximum (FWHM) of the water peak of ≤25 Hz.
Dynamic contrast-enhanced (DCE) MRI was acquired using a view-sharing, three-dimensional, T1-weighted gradient echo sequence (TWIST), first acquiring a high-resolution k-space mask followed by repetitive subsampled central k-space acquisitions in order to achieve high spatial and temporal resolution (TR/TE 3.85/1.42; flip angle 12°; GRAPPA factor 2; 70 repetitions; TWIST k-space subsampling with central region A 30 % and sampling density 25 %, resulting in a temporal resolution of 4.22 s; FOV 260 mm; matrix 160). Before dynamic scanning, T1-mapping sequences using the variable flip-angle method were applied. (TR/TE 3.85/1.42; flip angle 2.5,10, 20°; matrix 256; FOV 260 mm; slice thickness 3.6 mm; four averages).
Gadoterate meglumine (Gd-DOTA, Dotarem®, Guerbet, France) was injected after three baseline scans intravenously as a bolus (0.2 ml/kg body weight) using a power injector at a flow rate of 4 ml/s, followed by a flush of 20 ml of saline solution.
The complete MP-MRI protocol magnet time per patient was approximately 40 min.
Image interpretation
Two radiologists (observer 1 = O1; observer 2 = O2) (K.P.−O1, P.B.−O2 with 12 and 7 years of MRI experience, respectively) independently assessed the images on a dedicated workstation (Syngo, Siemens Healthcare, Erlangen, Germany). Spectra were assessed on the same workstation by a dedicated spectroscopy toolbox with presets for baseline correction and spectra fitting. Artifacts were excluded by visual inspection of the acquired spectra. At the time of image interpretation, the radiologists were aware of only the PSA level and were blinded to clinical stage and histopathology.
Diagnostic criteria in MP-MRI
To describe the localization of the lesion, a 27-region scheme was used [2]. Subsequently, all lesions were grouped as peripheral zone or transition zone lesions. Each lesion suspected to be cancer was graded according to the PI-RADS classification system v1 for T2, DWI, DCE, and 1H-MRSI [2] (Fig. 1). For 1H-MRSI, the different Choline + Creatine/Citrate ratios in the peripheral and transition zones were considered. Here, a score from 1 (clinically significant cancer highly unlikely) to 5 (clinically significant cancer highly probable) was given for each single parameter.
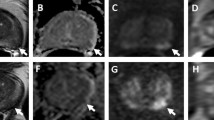
MP-MRI performed in a 71-year-old patient. Prostate-specific antigen (PSA) is 6.4 ng/ml at the time of imaging. a Axial 3-mm thick T2-weighted image of the middle third of the prostate. The observers identified a suspicious hypointense area in the right peripheral zone (arrows) (PI-RADS 5). b, c On DCE-MRI, a type 3 enhancement curve is appreciated inside the focally enhancing lesion (PI-RADS 5). d, e On the ADC map, the lesion presents as a focal area with low signal intensity with corresponding high signal intensity on b800 s/mm2 images (PI-RADS 5). f 1H-MRSI shows an elevated Choline/Citrate ratio in the suspicious region (PI-RADS 5). MP-MRI sum scoring according to PI-RADS with four parameters (PS4 19) and with three parameters (PS3 15) is rated true positive, indicating that the fourth parameter did not add any information. Histopathological workup revealed a high-grade PCa Gleason score 7 (4 + 3)
In addition, a simple sum of the individual MP-MRI parameters was calculated for each index lesion [6–13]. The index lesion was defined as the most suspect intraprostatic lesion with the highest sum score. The sum score was calculated for three-parameter MP-MRI (T2, DWI, and DCE; denoted as PS3), ranging between 3 and 15, and for four-parameter MP-MRI with optional 1H-MRSI (denoted as PS4), ranging between 4 and 20 (Fig. 2). We chose this approach, as the latest PI-RADS version (v2) does not include 1H-MRSI in its specific reporting recommendations. On the other hand, the used sum score has shown to be valid, robust, and reliable approach by several researchers [8–13].
MP-MRI performed in a 64-year-old patient. Prostate-specific antigen (PSA) is 5.8 ng/ml at the time of imaging. a Axial 3-mm thick T2-weighted image of the middle third of the prostate. The observers described an area with rather homogeneously low signal intensity and a well-defined margin in the transition zone (PI-RADS 2). b, c On DCE-MRI, a type 3 enhancement curve is appreciated inside the focally enhancing lesion (PI-RADS 5). d, e On the ADC map, the lesion presents as a focal area with low signal intensity with corresponding high signal intensity on b800 s/mm2 images (PI-RADS 4). f 1H-MRSI shows a Citrate peak exceeding the Choline peak two times (PI-RADS 1). MP-MRI sum score with four parameters (PS4 = 12) is rated false negative. On the other hand, MP-MRI with three parameters (PS3 11) is rated true positive. Histopathological workup revealed a PCa Gleason score 6 (3 + 3)
Standard of reference
Histopathology was defined as the standard of reference. Histopathology specimens were obtained by either transrectal ultrasound or MR-guided biopsy [14]. If radical prostatectomy was performed (13/64 patients), whole-mount step section was used as the reference standard. If the histopathology specimens were obtained by systematic TRUS, the findings from the biopsied sectors were correlated with MP-MRI in order to identify concordant suspicious lesions. This verification was performed after MP-MRI reading. Only lobes with positive biopsy and MP-MRI findings in the same sector were considered true positive (TP).
All specimens obtained from biopsy and the whole-mount step section were reviewed by an in-house pathologist (MS with 20 years of experience in uropathology) using the International Society of Urological Pathology-modified Gleason score classification [15].
Statistical analysis
Statistical analysis was performed using SPSS 22.0 (IBM Corp, NY, USA) and R 3.1.1 (R Foundation for Statistical Computing, Vienna, Austria). All calculations were performed on a per patient basis, requiring the reader to correctly localize the lesion (Obuchowski level 2 analysis [16]). Receiver operating characteristics (ROC) analysis was performed to determine the overall diagnostic accuracy of the sum scores as defined above (PS3, PS4). Sensitivity and specificity and their 95 % confidence intervals (CI) were calculated at appropriate cutoff levels. Inter-rater variability for ordinal parameters was assessed by quadratic κ coefficients. Spearman correlation analysis was performed to explore the association of the sum scores (PS3, PS4) with histopathological Gleason scores. A P value ≤0.05 was considered a significant result.
Results
Patients and lesions
The median age of our patient cohort was 65.4 years (ranging from 44 to 81 years), with a mean PSA level of 7.4 ng/ml (ranging from 1.0 to 36.9 ng/ml).
In 52 of 64 patients, a PCa index lesion was confirmed by histopathology. Twenty-nine (55.8 %) index lesions were classified as a high-grade PCa (primary Gleason pattern ≥4). Low-grade PCa was found in 23 (44.2 %) index lesions. Of all cancers, 44/52 (84 %) were located in the peripheral zone (PZ) and 8/52 (16 %) in the transition zone (TZ).
In 12 index lesions with a benign histopathology result, the most common referral diagnosis was prostatitis, in five (41.7 %) patients. Seven (58.3 %) index lesions were classified as benign and not otherwise specified.
Diagnostic performance for PCa detection
The area under the ROC curve (AUC) for MP-MRI with four parameters (PS4) equaled that of MP-MRI with three parameters (PS3: P = 0.928, O1; P = 0.922, O2) (Table 1). The results were similar for both observers (P = 0.937 PS4; P = 0.898 PS3) (Table 1).
The inter-reader agreement was substantial for PS4 (0.783) and for PS3 (0.638). Variations in sensitivity and specificity values at comparable PI-RADS sum score cutoff values are provided in Table 2.
Prediction of PCa Gleason score by MP-MRI
MP-MRI with three parameters and MP-MRI with four parameters were positively correlated with a PCa lesion’s Gleason score (Fig. 3). Considering the AUC for prediction of a PCa lesion’s Gleason score, MP-MRI with four parameters, the diagnostic performance of both observers was equal to that of MP-MRI with three parameters (P = 1, O1; P = 0.844, O2) (Table 1). The results were similar for both observers (P = 0.966 PS4; P = 0.812 PS3) (Table 1).
Discussion
Our results show that MP-MRI with four parameters that include 1H-MRSI does not increase the detection and grading of prostate cancer at 3 T compared to MP-MRI with three parameters. Applying a sum score can accurately detect PCa at 3 T without an endorectal coil and shows potential for prediction of tumor grade. Our results imply that omitting 1H-MRSI at 3 T without using an endorectal coil can considerably shorten prostate MRI examinations.
1H-MRSI is seen as an optional imaging parameter, but its value as a fourth parameter has not been fully established [2]. Thus, several international societies support continued research on the value of 1H-MRSI in MP-MRI of the prostate [5].
Röthke et al. examined 64 consecutive patients at 3T without an endorectal coil in a consensus reading approach. Using three parameters, specificity was 95.5 % and sensitivity was 71.4 %. When 1H-MRSI was included, a similar specificity and sensitivity of 94.7 and 75.8 %, respectively, were reported. A second group using four-parameter MP-MRI of the prostate did not calculate a sum score, but described a limited clinical utility for 1H-MRSI. Using three parameters, investigators have reported a sensitivity of 80–95 %, a specificity of 62–92 %, and an area under the ROC curve of 69–97 % [9, 10, 12, 17–19]. These findings are in good agreement with our results, showing similar sensitivity and specificity. When 1H-MRSI was added, no improvement in PCa detection and grading was achieved in our study.
In addition to the diagnostic accuracy for the detection of PCa, there is growing interest in image-guided risk stratification of PCa lesions by predicting the Gleason score. Prior publications demonstrated that MP-MRI of the prostate has the potential to differentiate between low- and high-grade PCa [20]. Again, adding 1H-MRSI did not improve the diagnostic performance of MP-MRI for tumor grading. We demonstrated a positive correlation between PCa Gleason scores and MP-MRI with three parameters and between MP-MRI with four parameters; the results did not differ. A higher PSsum score showed the potential to predict high-grade PCa. In the clinical setting, this seems to provide useful information for targeting the most significant lesion in the prostate, to overcome the problem of overdiagnosis of clinically insignificant disease [21, 22].
With regard to the detection and grading of prostate cancer, our findings are important, as data acquisition and interpretation of 1H-MRSI are time-consuming and thus detrimental, both in terms of examination costs and reading time [2, 3]. 1H-MRSI requires careful positioning of the examination voxel, saturation regions, and shimming. The acquisition time is long, making this sequence more vulnerable to patient motion. Spectra analysis is more time-consuming than standard-imaging sequences, as they require dedicated software and co-registration with anatomical images. Thus, our results are important, as they imply that omitting 1H-MRSI at 3 T without using an endorectal coil can considerably shorten prostate MRI examinations. Consequently, prior research supports our own finding: 1H-MRSI does not add incremental diagnostic information in 3-T MP-MRI of the prostate without the use of an endorectal coil.
There is an ongoing debate about using an endorectal coil in the clinical setting for the local detection of PCa on 1.5-T and on 3-T devices. At 1.5 T, an endorectal coil improves the 1H-MRSI qualities, leading to a higher sensitivity and specificity [23]. However, at 3 T, guidelines do not recommend an endorectal coil for cancer detection and grading protocols [2]. This can be explained by the fact that, for the three other parameters, sufficient diagnostic image quality and high sensitivity and specificity are achieved with MP-MRI. It has been suggested that the signal-to-noise ratio of peripheral gland 1H-MRSI may be up to tenfold higher using an endorectal coil compared to a surface body coil [4, 24].
In addition, the use of an endorectal coil requires additional time, incurs additional costs, can cause discomfort and minor complications, and is contraindicated in several circumstances [25]. We demonstrated, in the current study, that MP-MRI is feasible with a simple body coil at 3 T. In addition to patient comfort and equipment costs, this setting is clearly preferable in patients who choose radiation therapy as an alternative treatment option to radical prostatectomy [26, 27].
The PI-RADS scoring system has been established as a feasible method for the clinical routine and allows standardized reporting [8]. Such a system is intended to improve the inter-reader reproducibility [2]. The reproducibility of the sum score in our study, as measured by the kappa agreement, was substantial for PS3 and for PS4. One group reported the inter-reader agreement for each single parameter from moderate to substantial for T2, DWI, and DCE (0.55, 0.64, and 0.65, respectively) [18]. Another group published an inter-reader agreement for a sum score from 0.378 to 0.441, which included three readers with different levels of experience and the images were acquired on both 1.5- and 3.0-T devices [28]. These results are in good concordance with Rosenkrantz et al., who reported a moderate kappa agreement (0.41–0.49) for three readers on a 3.0-T device, without the use of an endorectal coil [8]. Consequently, our results are in accord with the literature, and underline a clinically reasonable level of reproducibility in reading MP-MRI of the prostate using a sum score.
The use of a sum score in the assessment of the diagnostic accuracy and grading of prostate cancer can be seen as a potential limitation. However, even the recent PI-RADS version (v2) does not provide any recommendation on how to integrate 1H-MRSI into the clinical evaluation process [5]. On the other hand, the sum score has been shown to be robust valid and reliable used by several researchers [9–11, 13]. The reference standard was not the same in all patients, as consecutive patients who fulfilled predefined inclusion and exclusion criteria were recruited from clinical practice. All lesions were histopathologically proven. Considering one image-related reference standard only (e.g., MR-guided biopsy) would bias the patient group toward lesions suspicious on MRI, possibly resulting in a lower specificity. However, considering whole-mounted specimens only would raise reader awareness for the presence of cancer, possibly resulting in a higher sensitivity [29]. Consequently, our results reflect what is to be expected in a real-life clinical setting. In addition, our patient-based Obuchowski level 2 analysis implies a possible bias toward an overestimated sensitivity when there are several lesions per patient. Further, specificity can be overestimated under certain conditions. Again, our approach reflects the clinical cancer detection scenario and thus the diagnostic performance in the clinical scenario.
Conclusions
In conclusion MP-MRI with four parameters that include 1H-MRSI does not increase the detection and grading of prostate cancer at 3 T compared to MP-MRI with three parameters. MP-MRI with three parameters can accurately detect PCa at 3 T without an endorectal coil, and shows potential for the prediction of tumor grade.
References
Hoeks CM, Barentsz JO, Hambrock T, Yakar D, Somford DM, Heijmink SW, Scheenen TW, Vos PC, Huisman H, van Oort IM, Witjes JA, Heerschap A, Futterer JJ (2011) Prostate cancer: multiparametric MR imaging for detection, localization, and staging. Radiology 261(1):46–66. doi:10.1148/radiol.11091822
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, Rouviere O, Logager V, Futterer JJ (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757. doi:10.1007/s00330-011-2377-y
Weinreb JC, Blume JD, Coakley FV, Wheeler TM, Cormack JB, Sotto CK, Cho H, Kawashima A, Tempany-Afdhal CM, Macura KJ, Rosen M, Gerst SR, Kurhanewicz J (2009) Prostate cancer: sextant localization at MR imaging and MR spectroscopic imaging before prostatectomy–results of ACRIN prospective multi-institutional clinicopathologic study. Radiology 251(1):122–133. doi:10.1148/radiol.2511080409
Scheenen TW, Heijmink SW, Roell SA, Hulsbergen-Van de Kaa CA, Knipscheer BC, Witjes JA, Barentsz JO, Heerschap A (2007) Three-dimensional proton MR spectroscopy of human prostate at 3 T without endorectal coil: feasibility. Radiology 245(2):507–516. doi:10.1148/radiol.2451061444
American College of Radiology. MR prostate imaging reporting and data system version 2.0. (2015). http://www.acr.org/Quality-Safety/Resources/PIRADS/
Schimmoller L, Quentin M, Arsov C, Hiester A, Kropil P, Rabenalt R, Albers P, Antoch G, Blondin D (2014) Predictive power of the ESUR scoring system for prostate cancer diagnosis verified with targeted MR-guided in-bore biopsy. Eur J Radiol 83(12):2103–2108. doi:10.1016/j.ejrad.2014.08.006
Rothke M, Blondin D, Schlemmer HP, Franiel T (2013) PI-RADS classification: structured reporting for MRI of the prostate. Rofo 185(3):253–261. doi:10.1055/s-0032-1330270
Rosenkrantz AB, Kim S, Lim RP, Hindman N, Deng FM, Babb JS, Taneja SS (2013) Prostate cancer localization using multiparametric MR imaging: comparison of prostate imaging reporting and data system (PI-RADS) and likert scales. Radiology 269(2):482–492. doi:10.1148/radiol.13122233
Roethke MC, Kuru TH, Schultze S, Tichy D, Kopp-Schneider A, Fenchel M, Schlemmer HP, Hadaschik BA (2014) Evaluation of the ESUR PI-RADS scoring system for multiparametric MRI of the prostate with targeted MR/TRUS fusion-guided biopsy at 3.0 Tesla. Eur Radiol 24(2):344–352. doi:10.1007/s00330-013-3017-5
Junker D, Quentin M, Nagele U, Edlinger M, Richenberg J, Schaefer G, Ladurner M, Jaschke W, Horninger W, Aigner F (2014) Evaluation of the PI-RADS scoring system for mpMRI of the prostate: a whole-mount step-section analysis. World J Urol. doi:10.1007/s00345-014-1370-x
Kayat Bittencourt L, Litjens G, Hulsbergen-van de Kaa CA, Turkbey B, Gasparetto EL, Barentsz JO (2015) Prostate cancer: the European society of urogenital radiology prostate imaging reporting and data system criteria for predicting extraprostatic extension by using 3-T multiparametric MR imaging. Radiology:141412. doi:10.1148/radiol.15141412
Schimmoller L, Quentin M, Arsov C, Hiester A, Buchbender C, Rabenalt R, Albers P, Antoch G, Blondin D (2014) MR-sequences for prostate cancer diagnostics: validation based on the PI-RADS scoring system and targeted MR-guided in-bore biopsy. Eur Radiol 24(10):2582–2589. doi:10.1007/s00330-014-3276-9
Schimmoller L, Quentin M, Arsov C, Hiester A, Kropil P, Rabenalt R, Albers P, Antoch G, Blondin D (2014) Predictive power of the ESUR scoring system for prostate cancer diagnosis verified with targeted MR-guided in-bore biopsy. Eur J Radiol. doi:10.1016/j.ejrad.2014.08.006
Polanec SH, Helbich TH, Margreiter M, Klingler HC, Kubin K, Susani M, Pinker-Domenig K, Brader P (2014) Magnetic resonance imaging-guided prostate biopsy: institutional analysis and systematic review. Rofo 186(5):501–507. doi:10.1055/s-0033-1355546
Epstein JI, Allsbrook WC, Amin MB, Egevad LL (2005) The 2005 international society of urological pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma. Am J Surg Pathol 29(9):1228–1242
Obuchowski NA, Meziane M, Dachman AH, Lieber ML, Mazzone PJ (2010) What’s the control in studies measuring the effect of computer-aided detection (CAD) on observer performance? Acad Radiol 17(6):761–767. doi:10.1016/j.acra.2010.01.018
Junker D, Schafer G, Edlinger M, Kremser C, Bektic J, Horninger W, Jaschke W, Aigner F (2013) Evaluation of the PI-RADS scoring system for classifying mpMRI findings in men with suspicion of prostate cancer. BioMed Res Int 2013:252939. doi:10.1155/2013/252939
Schimmoller L, Quentin M, Arsov C, Lanzman RS, Hiester A, Rabenalt R, Antoch G, Albers P, Blondin D (2013) Inter-reader agreement of the ESUR score for prostate MRI using in-bore MRI-guided biopsies as the reference standard. Eur Radiol 23(11):3185–3190. doi:10.1007/s00330-013-2922-y
Reisaeter LA, Futterer JJ, Halvorsen OJ, Nygard Y, Biermann M, Andersen E, Gravdal K, Haukaas S, Monssen JA, Huisman HJ, Akslen LA, Beisland C, Rorvik J (2014) 1.5-T multiparametric MRI using PI-RADS: a region by region analysis to localize the index-tumor of prostate cancer in patients undergoing prostatectomy. Acta Radiol. doi:10.1177/0284185114531754
Lawrentschuk N, Fleshner N (2009) The role of magnetic resonance imaging in targeting prostate cancer in patients with previous negative biopsies and elevated prostate-specific antigen levels. BJU Int 103(6):730–733. doi:10.1111/j.1464-410X.2008.08205.x
Lavery HJ, Droller MJ (2012) Do Gleason patterns 3 and 4 prostate cancer represent separate disease states? J Urol 188(5):1667–1675. doi:10.1016/j.juro.2012.07.055
Beauval JB, Ploussard G, Soulie M, Pfister C, Van Agt S, Vincendeau S, Larue S, Rigaud J, Gaschignard N, Roupret M, Drouin S, Peyromaure M, Long JA, Iborra F, Vallancien G, Rozet F, Salomon L, Members of Committee of Cancerology of the French Association of U (2012) Pathologic findings in radical prostatectomy specimens from patients eligible for active surveillance with highly selective criteria: a multicenter study. Urology 80(3):656–660. doi:10.1016/j.urology.2012.04.051
Futterer JJ, Engelbrecht MR, Jager GJ, Hartman RP, King BF, Hulsbergen-Van de Kaa CA, Witjes JA, Barentsz JO (2007) Prostate cancer: comparison of local staging accuracy of pelvic phased-array coil alone versus integrated endorectal-pelvic phased-array coils. Local staging accuracy of prostate cancer using endorectal coil MR imaging. Eur Radiol 17(4):1055–1065. doi:10.1007/s00330-006-0418-8
Futterer JJ, Scheenen TW, Huisman HJ, Klomp DW, van Dorsten FA, Hulsbergen-van de Kaa CA, Witjes JA, Heerschap A, Barentsz JO (2004) Initial experience of 3 Tesla endorectal coil magnetic resonance imaging and 1H-spectroscopic imaging of the prostate. Invest Radiol 39(11):671–680
Lee SH, Park KK, Choi KH, Lim BJ, Kim JH, Lee SW, Chung BH (2010) Is endorectal coil necessary for the staging of clinically localized prostate cancer? Comparison of non-endorectal versus endorectal MR imaging. World J Urol 28(6):667–672. doi:10.1007/s00345-010-0579-6
Lagendijk JJ, Raaymakers BW, Van den Berg CA, Moerland MA, Philippens ME, van Vulpen M (2014) MR guidance in radiotherapy. Phys Med Biol 59(21):R349–R369. doi:10.1088/0031-9155/59/21/R349
Bloch BN, Lenkinski RE, Helbich TH, Ngo L, Oismueller R, Jaromi S, Kubin K, Hawliczek R, Kaplan ID, Rofsky NM (2007) Prostate postbrachytherapy seed distribution: comparison of high-resolution, contrast-enhanced, T1- and T2-weighted endorectal magnetic resonance imaging versus computed tomography: initial experience. Int J Radiat Oncol Biol Phys 69(1):70–78. doi:10.1016/j.ijrobp.2007.02.039
Vache T, Bratan F, Mege-Lechevallier F, Roche S, Rabilloud M, Rouviere O (2014) Characterization of prostate lesions as benign or malignant at multiparametric MR imaging: comparison of three scoring systems in patients treated with radical prostatectomy. Radiology 272(2):446–455. doi:10.1148/radiol.14131584
Whiting P, Rutjes AW, Reitsma JB, Glas AS, Bossuyt PM, Kleijnen J (2004) Sources of variation and bias in studies of diagnostic accuracy: a systematic review. Ann Intern Med 140(3):189–202
Authors’ contribution
Stephan Polanec MD involved in project development and data collection/managing and wrote manuscript. Katja Pinker-Domenig MD involved in project development and data analysis and wrote manuscript. Peter Brader MD involved in data analysis and edited the manuscript. Dietmar Georg wrote and edited the manuscript. Shahrokh Shariat MD involved in project development, and wrote and edited the manuscript. Claudio Spick MD involved in data collection, and wrote and edited the manuscript. Martin Susani MD involved in data collection and data analysis. Thomas H. Helbich MD involved in project development and data managing, and wrote and edited the manuscript. Pascal A. Baltzer MD involved in project development and data analysis, and wrote and edited the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Polanec, S.H., Pinker-Domenig, K., Brader, P. et al. Multiparametric MRI of the prostate at 3 T: limited value of 3D 1H-MR spectroscopy as a fourth parameter. World J Urol 34, 649–656 (2016). https://doi.org/10.1007/s00345-015-1670-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-015-1670-9