Abstract
Salinization and sodication present significant threats to crop productivity in many parts of the world including Pakistan. Cultivating halophytes like quinoa presents a viable solution for the profitable use of salt-affected lands. This study specifically examines the performance and salt tolerance mechanism of four quinoa accessions under varying salinity and sodicity levels. In addition to a control group, different combinations of electrical conductivity and sodium adsorption ratio (ECe dS m−1 SAR levels) were established: 10:20, 10:40, 20:20, and 20:40, achieved by using a mixture of salts. Most of the quinoa cultivars exhibited robust growth, with the exception of GLN-22, which proved unable to withstand high levels of salinity and sodicity, resulting in a 78% reduction in yield. GLN-29, on the other hand, demonstrated superior performance across all levels of salinity and sodicity. UAF-Q7 excelled under conditions of high salinity and low sodicity compared to equivalent salinity levels but elevated sodicity. Meanwhile, GLN-33 exhibited enhanced growth under elevated sodicity levels but struggled in the face of high salinity stress. In terms of nutrient uptake, GLN-29 displayed a higher accumulation of Na+ (32%) in older leaves compared to younger ones, alongside elevated levels of antioxidant activity at all salinity and sodicity levels. Notably, GLN-29 exhibited excellent adaptation to both high salinity and sodicity levels, resulting in the highest grain yield (14.75 g/pot) and the salt tolerance mechanism was associated with highly efficient K+ retention and transport of Na+ to older leaves. This underscores the necessity for further comprehensive field studies to ascertain its suitability for the sustainable utilization of salt-affected soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The uncontrolled increase in the global population in recent decades has posed serious threat to sustainable agriculture and food security. Land resources are shrinking progressively due to land-degradation. Poor soil fertility, soil salinity, water logging, soil erosion, soil pollution, deficiency of essential minerals, and steady reduction of organic matter content are key features of soil degradation. Among these, soil salinity and sodicity is considered as the most critical issue of agriculture sector particularly in countries with arid to semi-arid climatic conditions (Akram et al. 2021). The salt-affected area of semi-arid and arid regions occupies about 1 billion hectares (Syed et al. 2021) and in spite all the efforts put forward by scinetific community, it is incerasing with variable rates across the world. The total area of 831 Mha in the world is salt-affected of which 397 Mha comprises saline soils and 434 Mha have sodic soils (Hasanuzzaman et al. 2014). Approximately 6.30 Mha area of Pakistan is salt-affected and out of which 1.89, 1.85, 1.02 and 0.028 Mha are saline, permeable saline-sodic, impermeable saline-sodic and sodic, respectively (Hussain et al. 2020).
Salt stress affects 20% of total farm land and 50% of irrigated regions around the world. Growth and productivity of crops are negatively affected on salt-affected soils. The crop growth and yield in salt-affected soils is mainly affected due to osmotic stress, ionic toxicity, nutrient deficiency and poor physical conditions of soils (Munns and Tester 2008; Abbas et al. 2015). Leaf gas exchange, chlorophyll and relative water contents are severely decreased due to high levels of salts in soil solution (Flowers and Colmer 2015; Abbas et al. 2017). Plants growing in salt-affected soils also suffer from oxidative stress due to the over production of several reactive oxygen species (ROS) (Abbas et al. 2017). Macromolecules such as carbohydrates, lipids, protein, and cell membranes are damaged due to cytotoxic nature of ROS. Due to lipid peroxidation, cell membranes are ruptured and results in plant death (Abbas et al. 2017). To overcome the ROS actions, antioxidant enzymes activities are increased such as superoxide dismutase, peroxidase, and catalase in plants (Parvez et al. 2020; Rehman et al. 2019). Under salinity stress, the accumulation of sugars and other compatible solutes (e.g., proline) allows plants to maintain their cellular turgor pressure necessary for cell expansion and growth under stress conditions; they also act as osmo-protectants. Proline is also considered the only osmolyte able to scavenge free radicals thereby ensuring membrane stabilization and preventing protein denaturation during severe osmotic stress (Szabados and Savouré 2010).
The productive utilization of degraded salt-affected soils is crucial to meet the needs of expanding population of the world. Depending upon type of salt-affected soils and availability of management package, salt-affected soils can be reclaimed using different physical, chemical, and biological methods. One option for counteracting the salt stress is the enhancement of salt tolerance potential of glycophytes through genetic engineering or plant breeding (Negrão et al. 2017). However, this option is time consuming and little success has been reported in developing salt tolerance and growing such corps at large areas. Another approach is to utilize such soils by cultivating halophytes (Panta et al. 2014). According to Adolf et al. (2013), the second option is the most promising approach. However, halophytes have great genetic variability regarding their salt tolerance potential (Ruiz et al. 2016). For examples, Dicotyledonous halophytes have shown maximum growth around 150 mM NaCl concentration, whereas monocotyledonous halophytes have maximum growth around 50 mM NaCl (Adolf et al. 2013).
The most promising example of dicotyledonous halophytes is quinoa (Chenopodium quinoa Willd. L.). It has the capacity to grow even at 400 mM salt concentration (Riaz et al. 2020) and can produce highly nutritious food grains (Afzal et al. 2023). Moreover, it can grow well in highly salt-affected soils with limited water supply and has potential as supplementer/alternative human food source with high nutritional value (Jacobsen 2017). In light of the current changing climate scenario and also due to extraordinary nutritional profile, and adaptability to adverse climate, quinoa has recently received a considerable attention of the progressive farmers and researchers from the arid and semi-arid regions to be used as an alternative food crop for ensuring the issue of food security and also to efficiently utilizing the saline-sodic soils (Adolf et al. 2013; Afzal et al. 2023).
Intensive research has been done on quinoa to evaluate its growth and yield potential on saline soils however to best of our knowledge, limited information is available regarding its cultivation on soils with varying levels of salinity and sodicity. Therefore, the current study was designed to address this research gap and to explore the comparative growth, yield, phenological responses, and potential salt tolerance mechanisms of different available quinoa genotypes under saline-sodic conditions. We hypothesize that genotypic variations may be a feasible approach to reduce the salinity and sodicity levels.
Materials and Methods
Experimental Design
Present study was conducted at the Wire House at Institute of Soil and Environmental Sciences, University of Agriculture Faisalabad (UAF), Pakistan during the months of November to May 2020–2021 to test the performance of three genotypes (GLN-22, GLN-29, GLN-33) and one variety UAF-Q7 of quinoa crop under saline-sodic conditions. The soil used in the study was collected from student experimental area of Institute of Soil and Environmental Sciences, UAF and characterized using methods described by U.S. Salinity Laboratory Staff (1954) and Bottomley et al. (2020) for various physico-chemical properties before start of the experiment (Table 1). The sieved soil (10 kg) was filled in each ceramic pot lined internally with polythene sheet before filling with soil and the hole at bottom was also plugged with cork to prevent leaching. Using quadratic equation, required salt concentrations were calculated to develop different salinity: sodicity (ECe:SAR) levels i.e., 10:20; 10:40; 20:20; and 20:40 (dS m−1:mmol L−1)1/2 along with control and each with three replications (Haider and Ghafoor 1992). The pots were saturated to soil saturation percentage using distilled water along with calculated amounts of salts and incubated for one month. After incubation, 10 seeds of quinoa in each pot were sown at the depth of 2.50 cm. Each pot was supplemented with P and K at 60 kg ha−1 as basal dose using diammonium phosphate (DAP) and sulphate of potash (SOP) fertilizers, while N dose was applied at 75 kg ha−1 using urea (half dose at sowing and half along with 2nd irrigation). Each pot was irrigated with canal water throughout the experiment. After seed emergence, only 3 plants in each pot were retained till maturity.
Phenological and Physiological Attributes
The phenological data (emergence of cotyledon and true leaves, visible floral bud, anthesis, end of flowering, physiological maturity) were recorded as and when needed. For recording of data, a specific phenological stage is considered completed when half of the plant population completed that stage (Stanschewski et al. 2021).The leaf area of the quinoa plant during the vegetative growth stage was measured using a LICOR LI-3000 leaf area meter (Hunt 1978). Physiological parameters like Transpiration and photosynthetic rates were recorded using an infrared gas analyzer (IRGA) (Analytical development company, Hoddeson, UK) in the morning during the vegetative growth stage. Chlorophyll content was assessed at the vegetative stage using a chlorophyll meter (Minolta SPAD-502 DL meter Japan).
Fully expanded top 2nd leaf of the plant was used to determine the relative water content (RWC) of the quinoa plant. 0.5 g of fresh leaf sample was immediately immersed in Petri plates filled with distilled water (DW) for 4 h. Subsequently, leaves were removed from the Petri plates, dried with tissue paper, and their turgid weights were recorded. The samples were then placed in an oven at 70 °C for 48 h until a constant weight was achieved. The dry weights were measured using weighing balance (OHAUS digital weighing balance PX233). The RWC was determined in leaf samples using the equation (Tahjib-Ul-Arif et al. 2018).
The membrane stability index (MSI) was determined using fully expanded younger leaves. The fresh weight of these leaves was measured after rinsing them with distilled water. In a test tube containing fresh leaf cuttings of equal size (0.2 g), 10 mL of distilled water was added. After 30 min, these test tubes were placed in a water bath at 40 °C. The first electrical conductivity (EC1) reading was taken after 30 min. EC2 was noted 15 min after placing the samples back in the water bath at a temperature of 100 °C, and the MSI was calculated according to the method outlined by Sairam et al. (2002).
Ionic Analysis
Each quinoa genotype’s fully expanded younger and older leaves as well as their respective roots samples were taken from each salt-treated and non-treated pots. Roots and shoots were ground separately. The ground plant samples (0.5 g) were acid digested with a diacid mixture (HNO3 and HClO4 in 2:1 ratio). The digestates were cooled, filtered, and diluted up to 50 mL using distilled water. The ionic concentrations (Na+ and K+) of leaf and root samples were measured using a flame photometer (Sherwood, Japan, Model 410) (Shavrukov et al. 2009).
Antioxidants Assay
Fully expanded younger leaves (60 days after emergence) were removed from each pot. Leaf samples were collected in the morning before 5:00 am, and enveloped in aluminum foil and stored in plastic zipper-bags, then placed in ice box and subsequently frozen till analysis. Within two days of leaves collection, antioxidants were determined. In a pre-chilled pestle and mortar, leaf samples (0.1 g) were grounded in 1 mL phosphate buffer (50 mM; pH 7.8). Grounded material was transferred into pre-chilled Eppendorf tubes, which were centrifuged (Clandon, T53, England) for 20 min at 15,000 rpm and the supernatant was collected to measure the activities of antioxidant enzymes, superoxide dismutase (SOD) (Giannopolitis and Ries 1977), peroxidase (POD) and catalase (CAT) (Chance and Maehly 1955) by taking absorbance at 560 and 240 nm, correspondingly. Proline contents in fresh leaves were also determined using the method described by Bates et al. (1973).
Yield and Yield-Related Attributes
Crop was harvested at maturity and plant tissue samples were collected for ionic composition and biochemical analyses. Filter paper was used to dry inflorescences and other parts of plants were dried out at 25–30 °C. The seeds were threshed manually after drying. The remaining plant parts were dried at 65 °C till constant weight was achieved and the dry matter was given. Plant height, main panicle length, width, no. of branches per plant, total dry biomass per plant and given yield/plant, total grain yield and thousand seeds weight was recorded. Panicle shape of quinoa plant was also recorded according to the protocol given by Bertero et al. (1996). Harvest index was also calculated after harvesting of crop by using standard procedures.
Statistical Analysis
Data obtained from the experiment were analyzed statistically using 2-way analysis of variance (ANOVA), while significance of treatments was compared at 5% probability level using least significant difference (LSD) test using XLSTAT v2019, USA (Steel 1997).
Results
Phenological Responses
The statistical analysis of growth stage data revealed significant effects of salinity and sodicity on quinoa growth. Genotypes GLN-29 and GLN-33 exhibited the shortest duration to reach each growth stage, closely followed by UAF-Q7. In contrast, GLN-22 took the longest time from cotyledon emergence to maturity due to the imposed salinity and sodicity stress (Table 2).
The data in Table 2 illustrates the influence of varying salinity and sodicity levels on the germination percentage (%) of quinoa genotypes in both the control (normal soil) and saline-sodic soils. As salinity and sodicity levels increased, germination of all tested genotypes significantly decreased (i.e., up to 43% in GLN-22 at ECO20:SAR40) compared to the control treatment. Notably, GLN-29 exhibited the highest germination percentage (56.6%).
Physiological Responses
The physiological parameters of quinoa plants exhibited adverse effects at different ECe:SAR levels compared to the control. Among these, the maximum decrease in relative water content (RWC) was observed in GLN-22 at EC:SAR 20:40 (78% reduction compared to the control), followed by decreases of 59, 36, and 10% at ECe:SAR levels 20:20, 10:40, and 10:20, respectively (Fig. 1b). Similarly, the maximum reduction in membrane stability index was recorded in genotype GLN-22 at ECe:SAR 20:20 (20% decrease), followed by decreases of 9, 7, and 1% at ECe:SAR levels 10:20, 10:40, and 20:40, respectively (Fig. 1c). In terms of photosynthetic rate, the most significant decrease was observed in GLN-22 at ECe:SAR 20:40 (83% reduction compared to the respective control), followed by decreases of 60, 39, and 15% at ECe:SAR levels 20:20, 10:40, and 10:20. Notably, GLN-29 experienced a 2% increase in photosynthetic rate at ECe:SAR 20:40 (Fig. 2a). The transpiration rate exhibited the maximum reduction in GLN-22 at ECe:SAR 20:40 (86% decrease), followed by decreases of 66, 63, and 53% at ECe:SAR levels 20:20, 10:40, and 10:20, respectively (Fig. 2b). Furthermore, the SPAD value saw the most significant reduction in UAF-Q7 (94%) at ECe:SAR 20:40, followed by decreases of 19, 17, and 4% at ECe:SAR levels 20:20, 10:40, and 10:20, respectively. Interestingly, no reduction in SPAD value was recorded in genotype GLN-33 at EC:SAR 10:20 (Fig. 2c).
Ionic Analysis
The concentration of Na+ in both older and younger leaves, as well as roots, of quinoa genotypes exhibited an increase, while the concentration of K+ showed a decrease with rising ECe:SAR levels. In older leaves, generally GLN-29 accumulate higher amount of Na+ in its older leaves at ECe:SAR(20:40) but if we talk about % age increase of Na+ accumulation with the increment in salinity and sodicity among all quinoa genotypes then, the most significant rise in Na+ concentration (49% relative to the control) was observed in GLN-33 at ECe:SAR (20:40), followed by increases of 19, 19, and 8% at ECe:SAR levels 20:20, 10:40, and 10:20, respectively (Fig. 3a). Conversely, the most substantial decrease in K+ concentration (49% compared to the control) was recorded in older leaves of UAF-Q7 at ECe:SAR (20:40) (Fig. 3b). For younger leaves, the same case was observed as older leaves i.e. GLN-22 accumulate highest quantity of Na+ in younger leaves but according to % age increase with the increasing salinity and sodicity levels, the highest increase in Na+ concentration was noted in UAF-Q7, showing a 56% rise compared to the respective control, at ECe:SAR (20:40). This was followed by increases of 28, 26, and 14% at ECe:SAR levels 20:20, 10:40, and 10:20, respectively (Fig. 4a). Meanwhile, the concentration of K+ in younger leaves of UAF-Q7 significantly decreased (− 63% compared to its control) (Fig. 4b). In terms of Na+/K+ ratio, the highest value was recorded in older leaves of GLN-29 at the highest ECe:SAR level. In the case of younger leaves, the highest Na+/K+ ratio was found in GLN-22 at ECe:SAR level of 20:40 (Figs. 3c, 4c). It's worth noting that the roots of all genotypes exhibited lower Na+ levels and higher K+ levels. According to % age increase in Na+ concentration with increasing salinity and sodicity (ECe:SAR), UAF-Q7 showed the highest Na+ levels, reaching 82% at ECe:SAR (20:40) compared to its control, followed by ECe:SAR (10:20, 10:40, and 20:20) with corresponding increases of 63, 35, and 1% (Fig. 5a). Overall GLN-22 showed maximum accumulation of Na+ at highest ECe:SAR level i.e., 20:40. The concentration of K+ was most significantly decreased in the roots of GLN-22, which were 59% lower than the respective control at ECe:SAR (20:40) (Fig. 5b). Na+/K+ ratio was highest in the roots of GLN-22 at ECe:SAR (20:40) (Fig. 5c).
Biochemical Attributes
The biochemical responses of quinoa showed improvements under saline-sodic conditions, indicating an enhanced antioxidant activity with increasing ECe:SAR levels. The highest % age increase in SOD activity was observed in genotype GLN-22, showing a 60% rise compared to its respective control at ECe:SAR (20:20). This was followed by increases of 54, 43, and 36% at control and ECe:SAR (10:40, 20:40, and 10:20). (Fig. 6a).
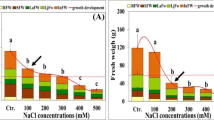
Maximum % age enhancement in POD activity was recorded in GLN-33 at ECe:SAR (20:40), which exhibited a remarkable 584% increase compared to its respective control. This was followed by increases of 281, 258, and 105% at ECe:SAR (20:20, 10:40, and 10:20) (Fig. 6b). Regarding CAT activity, the highest increase was observed in GLN-22 (37%) at ECe:SAR (20:40), followed by 21, 20, and 12% increases at ECe:SAR (10:40, 20:20, and 10:20) respectively (Fig. 6c). In terms of organic osmolyte proline concentration, the maximum % age increase was observed in UAF-Q7 (46% increase compared to the control) at ECe:SAR (20:20), followed by 44, 19, and 13% increases at ECe:SAR (10:20, 20:40, and 10:40). Interestingly, proline concentration decreased to 9% at ECe:SAR (20:20) in GLN-33 (Fig. 1a). But generally, among all the quinoa genotypes, GLN-29 showed a remarkable increase in activity of antioxidant enzymes, and proline contents at highest ECe:SAR level (Figs. 1a, 6a–c).
Yield and Yield-Related Attributes
Data regarding yield and yield-related attributes is provided in Table 3. Salinity and sodicity levels had a relatively less significant influence on plant height, main panicle lengths, and width for all quinoa genotypes (UAF-Q7, GLN-22, GLN-29, and GLN-33). Notably, even the ECe:SAR (10:20) stress had a significant negative effect on plant height, main panicle length, and panicle width across all genotypes. Furthermore, increased levels had even more pronounced negative effects. The most substantial decrease in plant height and main panicle length was observed in UAF-Q7 (15 and 39%) at the highest salinity and sodicity levels, i.e., ECe:SAR (20:40), followed by decreases of 11, 8, and 2% in plant height and 20, 18 and 7% in panicle length at ECe:SAR (20:20, 10:40, and 10:20). Plant height of GLN-33 increased by up to 2% at ECe:SAR (10:20). Additionally, the maximum decrease in panicle width was recorded in GLN-22 (18%) at ECe:SAR (20:40) compared to the control. Total dry biomass (TDB) of quinoa genotypes was also negatively influenced by increasing salinity and sodicity levels. But even at all salinity and sodicity levels, GLN-22 had attained the highest TDB. Meanwhile, the maximum decrease in TDB was noted in UAF-Q7 (40%) at ECe:SAR (20:40) compared to the control, followed by reductions of 14, 10, and 4% at ECe:SAR (10:40, 20:20, and 10:20). Seed yield, 1000 seeds weight, and harvest index (HI) were also adversely affected by increasing salinity and sodicity levels, with GLN-29 showing the minimum effects of ECe:SAR. The most significant decrease in seed yield, 1000 seeds weight, and HI was observed in GLN-22 (78, 76, and 58% compared to the control) at ECe:SAR (20:20, 10:20, and 10:40), respectively. The highest decrease in number of branches per plant were recorded in GLN-33, at 22% at ECe:SAR (20:40), while an increase of 2% was observed in UAF-Q7 at ECe:SAR (10:20). Leaf area reduced with increasing salinity and sodicity, with UAF-Q7 experiencing the highest reduction at the highest ECe:SAR level (69% compared to the control), followed by decreases of 41, 17, and 7% at ECe:SAR (20:20, 10:40, and 10:20) according to percentage decrease but in general, GLN-29's leaf area was reduced significantly (Table 3).
Soil Analysis After Harvest
The ECe, pH, and SAR levels exhibited significant decreases in the soil where quinoa genotype GLN-22 was cultivated, whereas the smallest reduction was observed in the soil where GLN-29 was grown. The highest pH was noted in the soil with ECe:SAR (20:40) where GLN-29 was cultivated, while the maximum ECe was observed in the soil with ECe:SAR (20:40) where GLN-33 was grown. The highest SAR value was recorded at ECe:SAR (10:40) where GLN-29 was grown, while the lowest ECe, pH, and SAR values were noted in the soil where GLN-22 was cultivated at all salinity and sodicity levels (Table 4).
Correlation Analysis
A strong negative correlation was observed between yield attributes and increasing salt stress. Conversely, a positive correlation was noted in the accumulation of Na+ ions in older leaves under stress. Moreover, there was a significant positive correlation observed between biochemical attributes (SOD, POD, CAT, Proline) and salt stress. Seed yield and harvest index exhibited a strong correlation with all physiological, biochemical parameters, and yield-related attributes, except for plant height, potassium levels in older leaves, sodium levels in younger leaves, and roots (Fig. 7).
Correlation analysis between the plant attributes under study. RWC relative water contents, MSI membrane stability index, SPAD chlorophyll content taken by SPAD meter, RDW root dry weight, RFW root fresh weight, SFW shoot fresh weight, SDW shoot dry weight, PH plant height, PL panicle length, PW panicle width, SY seed yield, TDB total dry biomass, GP germination percentage, KO K in older leaves, KY K in younger leaves, KR K in roots, NaO Na in older leaves, NaY Na in younger leaves, NaR Na in roots, Photosyn photosynthesis, Trans transpiration, SOD super oxide dismutase, POD peroxidase, CAT catalase
Discussion
All genotypes exhibited a typical halophytic nature, displaying higher growth and biomass and a shorter time to complete each phenological stage under moderate salinity and sodicity levels (ECe10:SAR20). However, high salinity and sodicity levels led to a significant decrease in biomass compared to the control treatment (Table 3). These results differ from the findings of Hariadi et al. (2011), who reported an increase in growth of different quinoa genotypes under 150 mM salinity level. Survival of quinoa even at 500 mM salinity level was also observed in a Peruvian variety (Koyro and Eisa 2008). Salinity and sodicity induced physiological drought and hydrolysis of stored foods, resulting in a slight loss in dry biomass yield. Overall, GLN-29 recorded less yield reduction under all saline-sodic conditions (Table 3). ECe:SAR level (10:20) was considered optimum for the growth of tested quinoa cultivars. These results are consistent with previous studies (Koyro and Eisa 2008; Hariadi et al. 2011; Iqbal et al. 2019) that reported decreased biomass production and yield of quinoa due to higher levels of soil sodicity. The lower 1000-seed weight of all genotypes at high levels of salinity (30 and 40 dS m−1) could be attributed to higher protein concentration in seeds than carbohydrates (Koyro and Eisa 2008). Panicle length and width of all tested genotypes were affected by increasing salinity and sodicity, while the maximum reduction in panicle width and number of branches per plant might be due to the maximum plant height of quinoa plants. Our findings support the study of Beyrami et al. (2020) who reported that soil salinity has adverse effects on different phenological and yield-related attributes such as dry biomass yield, seed yield and harvest index, plant height, panicle length, panicle width, number of branches, number of panicles per plant, and 1000-seed weight. GLN-22 prolonged its vegetative stage and utilized all nutrients from the soil in gaining maximum height and total dry biomass, thus reaching maturity late with a drastic reduction in seed yield.
The physiological traits, such as relative water contents (RWC) of leaves, were not significantly affected by salinity stress in UAF-Q7, GLN-29, and GLN-33, confirming their salt-tolerant nature. However, in saline-sodic soil with ECe:SAR level (20:40), RWC was reduced, validating prior research on quinoa under salt stress conditions (Takagi and Yamada 2013; Amjad et al. 2015). Significant declines in RWC, proline, and membrane stability index were found in genotype GLN-22 because of oxidative stress (Fig. 1). Similar results were also reported by Abbas et al. (2021) who concluded that soil salinity and sodicity decreased the membrane stability index of quinoa. Leaf chlorophyll contents (SPAD value) were decreased at the lower salinity and sodicity level ECe:SAR (10:20), but increased in GLN-33 at ECe:SAR (10:40), confirming the salt tolerance nature of quinoa genotype GLN-33 (Fig. 2c). Reduction in chlorophyll contents of quinoa at higher salinity levels might be due to degradation of chlorophyll structure (Rangani et al. 2016). Riaz et al. (2020) also reported the decrease in chlorophyll contents and RWC to a greater extent in genotype Puno than A-1, indicating higher stability of chlorophyll structure in A-1 than Puno. In our study, the reduction in transpiration and photosynthesis was observed with the increasing salinity and sodicity levels in all studied genotypes, especially in GLN-22 (Fig. 2a, b), which might be due to reduced leaf area because of which the plant decreased transpiration to avoid water loss from its leaf surface. Leaf area of the plant is an imperative morphological attribute that illustrates the photo-assimilation in plants. Higher photosynthesis rate in salt-tolerant quinoa genotypes was due to increased chlorophyll contents and stomatal closure under salt stress (Qureshi and Daba 2020).
An increasing trend of Na+ concentration in quinoa plant tissues of all cultivars was recorded with increasing ECe:SAR levels in the medium (Fig. 3). Shabala et al. (2013) explored the genotypic differences regarding salt tolerance among fourteen quinoa genotypes based on shoot Na+ uptake and found that exclusion as well as sequestration of Na+ into vacuoles are important traits for salt tolerance of crop plants. Similar to this, in our study, we observed that the tolerant genotypes i.e., GLN-29 and GLN-33 accumulated higher Na+ in shoots than roots, so they probably sequestered the excessive Na+ into their leaf vacuoles. It suggests that the most prominent mechanism of salinity tolerance in quinoa is vacuolar Na+ compartmentalization rather than root exclusion (Maughan et al. 2009; Shabala et al. 2013). We found that K+:Na+ ratio was decreased in genotypes GLN-29 and UAF-Q7 with increasing levels of salinity only and in GLN-29 and GLN-33 with increasing sodicity. This ratio is an important element elucidating the salt tolerance potential of quinoa (Adolf et al. 2013). The higher cytoplasmic concentration of Na+ leads to a lower K+:Na+ ratio, which ultimately affects plant metabolism. Moreover, loss of K+ from leaf mesophyll cells under salinity causes the activation of many proteases which initiate the programmed cell death (Shabala et al. 2005; Shabala 2009). Hence, the capability of the plants to limit K+ loss and maintenance of high ionic ratio (K+:Na+) in cytoplasm is an indication of their salt tolerance potential (Adolf et al. 2012) which was proved in the current study.
A lower degree of Na+ accumulation was observed in younger leaves of GLN-29 and UAF-Q7 at ECe:SAR (10:20 and 20:20) and in GLN-29 and GLN-33 at ECe:SAR (10:40 and 20:40). These cultivars responded to salt by translocating Na+ in older leaves (Fig. 3a). This strategy of low accumulation of Na+ might be linked with preferential K+ uptake at root parenchyma and translocation to leaf, as leaf K+ concentration was also higher in UAF-Q7 and GLN-33 along with GLN-29. Quinoa plants accumulate more K+ in leaves under salt stress (Adolf et al. 2013) which was confirmed in this study. Both osmotically induced stomatal closure and excessive Na+ accumulation in the cytosol under saline-sodic regimes decrease the plant’s capability to utilize light absorbed by photosynthetic pigments and lead to the generation of reactive ROS (Tavakkoli et al. 2011; Shabala et al. 2012, 2013). The antioxidant defense system operates in plant cells to limit the excessive accumulation of ROS in cell (Adolf et al. 2013; Waqas et al. 2017; Iqbal et al. 2018). Such a defensive system was found operative in leaves of all tested quinoa cultivars (Figs. 1a, 6).
Quinoa, like other halophytes, appears to have an unusual ability to use superoxide dismutase (SOD) to protect cell machinery (Ismail et al. 2015). The end product of SOD activity is H2O2 that can either operate as a signal to stimulate adaptive reaction in response to adversative environmental conditions (Bose et al. 2014) or cause cell membrane damage, therefore over-produced H2O2 is detoxified by catalase (CAT) and scavenged by peroxidase (POD) (Mittler 2002). In the present study, both CAT and POD remained stable under normal and salt stress regimes in all quinoa cultivars and found functioning to safeguard from ROS. Moreover, the strong correlation between all antioxidative enzymes and sodium uptake in older leaves is an indication of the protective role of antioxidant enzymes against salt stress (Fig. 7). The increased activities of SOD in quinoa are necessary for rapid induction of H2O2 “signature” for the sake of triggering adaptive responses cascade and role of other antioxidant enzymes might be for decreasing H2O2 basal levels, once triggering signals of H2O2. Furthermore, the role of non-enzymatic antioxidants seems to be osmo-protection, ROS scavenging or quenching and preventing K+ loss, an indirect role. In the non-enzymatic strategy proline production and accumulation in response to various abiotic stimuli, particularly salt stress, is generally recognized as a biochemical sign of tolerance to stress; their function as an osmoprotectant, suitable solute for adjustment of osmolytes and antioxidants or ROS suppressor is well established. According to the results of the current study, the level of proline was increased under salinity and sodicity stress in all the genotypes especially in GLN-29 (Fig. 1a). Moreover, there is a strong correlation between proline and antioxidant enzymes (Fig. 7).
According to post-quinoa harvest soil results, salinity and sodicity exhibited significant influence on soil properties (Table 4). At the soil ECe:SAR (10:20 and 20:20) levels, along with GLN-29 grown soil, UAF-Q7 grown soil and at ECe:SAR (10:40 and 20:40) GLN-33 grown soil had higher pHs, ECe and SAR values which might be due to their resistivity to saline-sodic conditions so they uptake less amount of salt in their cells thus leaving a higher quantity of salts in the root zone. While the soil where GLN-22 was grown had the lowest values of pHs, ECe, and SAR which might be due to its sensitivity to saline-sodic condition as GLN-22 accumulated a higher concentration of salts in it thus absorbing much quantity of salts from the root zone that’s why its yield was reduced. However, quinoa post-harvest soil characteristics are not well documented, therefore further studies must be conducted for conclusive findings.
Conclusion
The findings of this study provide compelling evidence that quinoa not only survive in saline-sodic conditions but also produce significant seed yield that confirms the halophytic nature of quinoa. The tested quinoa genotypes demonstrated significant potential to withstand salinity and sodicity stress. Among all the tested quinoa genotypes, UAF-Q7 interestingly exhibited its best performance under moderately saline-sodic conditions, while GLN-33 excelled at the highest salinity-sodicity levels. Notably, GLN-29 consistently outperformed at all salinity and sodicity levels. On the other hand, GLN-22 showed poor performance with a substantial 78% yield reduction. The salt tolerance in quinoa genotype GLN-29 appears to be associated with maintaining low Na+ levels and high K+ concentrations, along with elevated activities of antioxidant enzymes. These findings have significant implications for utilizing quinoa genotypes in the sustainable management of salt-affected soils across a wide range of salinity and sodicity levels.
References
Abbas G, Amjad M, Saqib M, Murtaza B, Asif NM, Shabbir A (2021) Soil sodicity is more detrimental than salinity for quinoa (Chenopodium quinoa Willd.): A multivariate comparison of physiological, biochemical and nutritional quality attributes. J Agron Crop Sci 207:59–73
Abbas G, Saqib M, Akhtar J, Haq MAU (2015) Interactive effects of salinity and iron deficiency on different rice genotypes. J Plant Nutr Soil Sci 178:306–311. https://doi.org/10.1002/jpln.201400358
Abbas G, Saqib M, Akhtar J, Murtaza G (2017) Physiological and biochemical characterization of Acacia stenophylla and Acacia albida exposed to salinity under hydroponic conditions. Can J For Res 47:1293–1301. https://doi.org/10.1139/cjfr-2016-0499
Adolf VI, Jacobsen SE, Shabala S (2013) Salt tolerance mechanisms in quinoa (Chenopodium quinoa Willd.). Environ Exp Bot 92:43–54. https://doi.org/10.1016/j.envexpbot.2012.07.004
Adolf VI, Shabala S, Andersen MN, Razzaghi F, Jacobsen SE (2012) Varietal differences of quinoa’s tolerance to saline conditions. Plant Soil 357:117–129
Afzal I, Haq MZU, Ahmed S, Hirich A, Bazile D (2023) Challenges and perspectives for integrating quinoa into the agri-Food system. Plants 12:3361. https://doi.org/10.3390/plants12193361
Akram NA, Shafiq F, Ashraf M, Iqbal M, Ahmad P (2021) Advances in salt tolerance of some major fiber crops through classical and advanced biotechnological tools: a review. J Plant Growth Regul 40:891–905. https://doi.org/10.1007/s00344-020-10158-5
Amjad M, Akhtar S, Yang A, Akhtar J, Jacobsen SE (2015) Antioxidative response of quinoa exposed to iso-osmotic, ionic and non-ionic salt stress. J Agron Crop Sci 201:452–460. https://doi.org/10.1111/jac.12140
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil Environ 39:205–207. https://doi.org/10.1007/BF00018060
Bertero D, Medan D, Hall A (1996) Changes in apical morphology during floral initiation and reproductive development in quinoa (Chenopodium quinoa Willd.). Ann Bot 78:317–324. https://academic.oup.com/aob/article/78/3/317/2587489
Beyrami H, Rahimian MH, Salehi M, Yazdani-Biouki R (2020) Effect of different levels of irrigation water salinity on quinoa (Chenopodium quinoa) yield and yield components in spring planting. J Crop Prod 12:111–120
Bose J, Rodrigo-Moreno A, Shabala S (2014) ROS homeostasis in halophytes in the context of salinity stress tolerance. J Exp Bot 65:1241–1257. https://doi.org/10.1093/jxb/ert430
Bottomley PJ, Angle JS, Weaver R (2020) Methods of soil analysis, Part 2: microbiological and biochemical properties, vol 12. Wiley, New York
Chance, B. and Maehly, A.C. (1955) Assay of catalases and peroxidases. Methods Enzymol 2:764-817. https://doi.org/10.1016/S0076-6879(55)02300-8
Flowers TJ, Colmer TD (2015) Plant salt tolerance: adaptations in halophytes. Ann Bot 115:327–331. https://doi.org/10.1093/aob/mcu267
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Haider G, Ghafoor A (1992) Manual of salinity research methods. International Water Logging and Salinity Research Institute, Lahore
Hariadi Y, Marandon K, Tian Y, Jacobsen SE, Shabala S (2011) Ionic and osmotic relations in quinoa (Chenopodium quinoa Willd.) plants grown at various salinity levels. J Exp Bot 62:185–193. https://doi.org/10.1093/jxb/erq257
Hasanuzzaman M, Nahar K, Alam M, Bhowmik PC, Hossain M, Rahman MM, Fujita M (2014) Potential use of halophytes to remediate saline soils. Biomed Res Int. https://doi.org/10.1155/2014/589341
Hunt R (1978) Plant Growth Analysis. Edward Arnold, London, 26–38
Hussain MI, Farooq M, Muscolo A, Rehman A (2020) Crop diversification and saline water irrigation as potential strategies to save freshwater resources and reclamation of marginal soils—review. Environ Sci Pollut Res 27:28695–28729. https://doi.org/10.1007/s11356-020-09111-6
Iqbal H, Yaning C, Waqas M, Rehman H, Shareef M, Iqbal S (2018) Hydrogen peroxide application improves quinoa performance by affecting physiological and biochemical mechanisms under water-deficit conditions. J Agron Crop Sci 204:541–553. https://doi.org/10.1111/jac.12284
Iqbal S, Basra SMA, Afzal I, Wahid A, Saddiq MS, Hafeez MB, Jacobsen SE (2019) Yield potential and salt tolerance of quinoa on salt-degraded soils of Pakistan. J Agron Crop Sci 205:13–21. https://doi.org/10.1111/jac.12290
Ismail H, Maksimović JD, Maksimović V, Shabala L, Živanović BD, Tian Y, Shabala S (2015) Rutin, a flavonoid with antioxidant activity, improves plant salinity tolerance by regulating K+ retention and Na+ exclusion from leaf mesophyll in quinoa and broad beans. Funct Plant Biol 43:75–86. https://doi.org/10.1071/FP15312
Jacobsen SE (2017) The scope for adaptation of quinoa in Northern Latitudes of Europe. J Agron Crop Sci 203:603–613. https://doi.org/10.1111/jac.12228
Koyro HW, Eisa SS (2008) Effect of salinity on composition, viability and germination of seeds of Chenopodium quinoa Willd. Plant Soil 302:79–90. https://doi.org/10.1007/s11104-007-9457-4
Maughan PJ, Turner TB, Coleman CE, Elzinga DB, Jellen EN, Morales JA, Udall JA, Fairbanks DJ, Bonifacio A (2009) Characterization of Salt Overly Sensitive 1 (SOS1) gene homoeologs in quinoa (Chenopodium quinoa Willd.). Genome 52:647–657. https://doi.org/10.1139/G09-041
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trend Plant Sci 7:405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Ann Rev Plant Biol 59:651–681. https://doi.org/10.1146/annvrev.arplant.59.032607.092911
Negrao S, Schmöckel S, Tester M (2017) Evaluating physiological responses of plants to salinity stress. Ann Bot 119:1–11. https://doi.org/10.1093/aob/mcw191
Panta S, Flowers T, Lane P, Doyle R, Haros G, Shabala S (2014) Halophyte agriculture: success stories. Environ Exp Bot 107:71–83. https://doi.org/10.1016/j.envexpbot.2014.05.006
Parvez S, Abbas G, Shahid M, Amjad M, Hussain M, Asad SA, Imran M, Naeem MA (2020) Effect of salinity on physiological, biochemical and photostabilizing attributes of two genotypes of quinoa (Chenopodium quinoa Willd.) exposed to arsenic stress. Ecotoxicol Environ Saf 15:109814. https://doi.org/10.1016/j.ecoenv.2019.109814
Qureshi AS, Daba AW (2020) Evaluating growth and yield parameters of five quinoa (Chenopodium quinoa W.) genotypes under different salt stress conditions. J Agric Sci 12:128–140. https://doi.org/10.5539/jas.v12n3p128
Rangani J, Parida AK, Panda A, Kumari A (2016) Coordinated changes in antioxidative enzymes protect the photosynthetic machinery from salinity induced oxidative damage and confer salt tolerance in an extreme halophyte Salvadora persica L. Front Plant Sci 7:50–68. https://doi.org/10.3389/fpls.2016.00050
Rehman S, Abbas G, Shahid M, Saqib M, Farooq AB, Hussain M, Murtaza B, Amjad M, Naeem MA, Farooq A (2019) Effect of salinity on cadmium tolerance, ionic homeostasis and oxidative stress responses in conocarpus exposed to cadmium stress: Implications for phytoremediation. Ecotoxicol Environ Saf 171:146–153. https://doi.org/10.1016/j.ecoenv.2018.12.077
Riaz F, Abbas G, Saqib M, Amjad M, Farooq A, Ahmad S, Naeem MA, Umer M, Khalid MS, Ahmed K, Ahmad N (2020) Comparative effect of salinity on growth, ionic and physiological attributes of two quinoa genotypes. Pak J Agric Sci 57:115–122. https://doi.org/10.21162/PAKJAS/20.9018
Ruiz K, Biondi S, Martínez E, Orsini F, Antognoni F, Jacobsen SE (2016) Quinoa—a model crop for understanding salt-tolerance mechanisms in halophytes. Plant Biosyst 150:357–371. https://doi.org/10.1080/11263504.2015.1027317
Sairam RK, Rao KV, Srivastava G (2002) Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci 163:1037–1046. https://doi.org/10.1016/S0168-9452(02)00278-9
Salinity US (1954) Laboratory staff. “Diagnosis and improvement of saline and alkali soils.” Agric Handb 60:83–100. https://doi.org/10.20710/dojo.71.1_18
Shabala S (2009) Salinity and programmed cell death: unravelling mechanisms for ion specific signalling. J Exp Bot 60:709–712. https://doi.org/10.1093/jxb/erp013
Shabala S, Shabala L, Van Volkenburgh E, Newman I (2005) Effect of divalent cations on ion fluxes and leaf photochemistry in salinized barley leaves. J Exp Bot 56:1369–1378. https://doi.org/10.1093/jxb/eri138
Shabala L, Mackay A, Tian Y, Jacobsen SE, Zhou D, Shabala S (2012) Oxidative stress protection and stomatal patterning as components of salinity tolerance mechanism in quinoa (Chenopodium quinoa). Physiol Plant 146:26–38. https://doi.org/10.1111/j.1399-3054.2012.01599.x
Shabala S, Hariadi Y, Jacobsen SE (2013) Genotypic difference in salinity tolerance in quinoa is determined by differential control of xylem Na+ loading and stomatal density. J Plant Physiol 170:906–914. https://doi.org/10.1016/j.jplph.2013.01.014
Shavrukov Y, Langridge P, Tester M (2009) Salinity tolerance and sodium exclusion in genus Triticum. Breed Sci 59:671–678. https://doi.org/10.1270/jsbbs.59.671
Stanschewski CS, Rey E, Fiene G, Craine EB, Wellman G, Melino VJ, Patiranage DSR, Johansen K, Schmöckel SM, Bertero D, Oakey H (2021) Quinoa phenotyping methodologies: an international consensus. Plants 10:1759. https://doi.org/10.3390/plants10091759
Steel RGD, Torrie JH and Dicky DA (1997) Principles and procedures of statistics : a biometrical approach, 3rd Edn. McGraw Hill, Inc. Book Co., New York, p 352–358
Syed A, Sarwar G, Shah SH, Muhammad S (2021) Soil salinity research in 21st century in Pakistan: its impact on availability of plant nutrients, growth and yield of crops. Commun Soil Sci Plant Anal 52:183–200. https://doi.org/10.1080/00103624.2020.1854294
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trend Plant Sci 15:89–97. https://doi.org/10.1016/j.tplants.2009.11.009
Tahjib-Ul-Arif M, Siddiqui MN, Sohag AA, Sakil MA, Rahman MM, Polash MA, Mostofa MG, Tran LS (2018) Salicylic acid-mediated enhancement of photosynthesis attributes and antioxidant capacity contributes to yield improvement of maize plants under salt stress. J Plant Growth Regul 37:1318–1330
Takagi H, Yamada S (2013) Roles of enzymes in anti-oxidative response system on three species of chenopodiaceous halophytes under NaCl-stress condition. Soil Sci Plant Nutr 59:603–611. https://doi.org/10.1080/00380768.2013.809600
Tavakkoli E, Fatehi F, Coventry S, Rengasamy P, McDonald GK (2011) Additive effects of Na+ and Cl– ions on barley growth under salinity stress. J Exp Bot 62:2189–2203. https://doi.org/10.1093/jxb/erq422
Waqas M, Yaning C, Iqbal H, Shareef M, Rehman H, Yang Y (2017) Paclobutrazol improves salt tolerance in quinoa: beyond the stomatal and biochemical interventions. J Agron Crop Sci 203:315–322. https://doi.org/10.1111/jac.12217
Acknowledgements
The authors are highly thankful to Professor Mark Tester, KAUST, Saudi Arabia for the provision of quinoa accessions and Department of Agronomy, UAF for the provision of quinoa cultivar UAF-Q7.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
IA and GM: Conception and Design of the work, provision of resources, supervision. AZ and AR: Conduct experiment, acquisition, analysis, and interpretation of data, draft the work. WS and ZURF: Data analysis and revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that he/she has no conflict of interest.
Additional information
Handling Author: Vijay Pratap Singh.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zafar, A., Murtaza, G., Afzal, I. et al. Comparative Physiological and Biochemical Mechanisms of Salt Tolerance in Four Quinoa Cultivars Under Varying Salinity and Sodicity Levels. J Plant Growth Regul 43, 2888–2904 (2024). https://doi.org/10.1007/s00344-024-11315-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-024-11315-w