Abstract
To provide a theoretical basis for further research on improving nitrogen absorption and utilization efficiency, in this study we investigated the effects of nitrogen fertilization on the yield, nitrogen metabolism, and antioxidant response of different rice genotypes. A pot experiment was conducted with three different rice genotypes (NIL-7, NIL-8, and 02428) grown under two different nitrogen application levels (HN, 1.88 g N/10 kg soil, and LN, 0.94 g N/10 kg soil). The grain yield, growth, antioxidant attributes, and N metabolism of different rice genotypes were assessed. We found that HN treatment significantly improved effective panicles per pot, total dry weight, and yield, but reduced harvest index of NIL-7, NIL-8, and 02428 compared with LN treatment. Compared with LN, HN treatment increased grain yield in NIL-7, NIL-8, and 02428 by 9.05%, 16.75%, and 48.35%, respectively. The activities of the enzymes involved in the nitrogen metabolism were changed and the regulation of antioxidant attributes was observed in HN treatment as compared to LN. The content of malondialdehyde (MDA) was decreased for HN relative to LN. Moreover, the efficiency of the N application was different among genotypes. In conclusion, nitrogen application regulated the grain yield, nitrogen metabolism, and antioxidant response of different rice genotypes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rice is a staple food globally and the main food for more than 65% of China’s population (Jiang et al. 2019a; Muthayya et al. 2014). Increasing rice yield is an important contribution to meeting the food needs of an ever-increasing global population (Deng et al. 2019). Nitrogen (N) is an essential macronutrient for obtaining high rice yield and thus N fertilization has been a key factor for improved rice productivity over the last 50 years (Peng et al. 2010; Vijayalakshmi et al. 2013). However, high-N input and low nitrogen use efficiency (NUE) have been serious problems in rice production (Yang et al. 2021). Unreasonable N fertilizer application has led to a higher fertilizer loss, lower returns for farmers, and serious environmental issues (Singh et al. 2014). Therefore, sustainable strategies for N fertilizer management are needed to improve crop yield and quality while reducing environmental risks (Ye et al. 2007; Trost et al. 2016; Cheng et al. 2021; Zhou et al. 2021). Approximately 20% of the world’s rice is grown in China, with a relatively high average N application rate (Liu et al. 2004). Therefore, research on the optimization of the response of rice to N fertilization is important and an effective strategy for the improvement of N fertilizer management. Moreover, understanding the effects of N fertilization on the productivity of specific rice genotypes would have particular practical significance.
N is the most essential limiting factor for crop growth and yield (Liang et al. 2015). The N fertilizer rate was found to be positively correlated with rice yield but negatively correlated with NUE (Zhao and Sha 2014). Excessive N fertilization of rice plants caused lodging, pest damage, yield reduction, and NUE decline (Liang et al. 2015; Sun et al. 2019). Other results showed that N application increased the photosynthetic capacity, dry matter accumulation, and grain yield in rice (Zhou et al. 2017, 2018; Nakhjiri et al. 2021). Moreover, N application influenced the number of panicles and seed setting rate, eventually increasing grain yield (Zhou et al. 2021). Thus, appropriate N management improves crop growth and raises grain yield (Zhang et al. 2021). Previous studies have found that the effectiveness of N has a relationship with genotypes (Pan et al. 2016; Ladha et al. 1998). The N fertilizer effect on increasing rice yield varied considerably among different rice genotypes (Wang et al. 2019). Hence, further research needs to be conducted to elucidate the physiological response mechanism of specific genotypes to different N fertilizer applications.
An earlier study revealed that the use of N fertilizer stimulated plant N metabolism (Ding et al. 2014). Nitrate reductase (NR) plays a key role in the absorption and utilization of N in plants (Sun et al. 2015). NR activity is positively correlated with the N fertilizer rate and thus an increase in N fertilizer application promoted NR activity (Andrews et al. 2013). Glutamate synthetase (GOGAT) is critically involved in carbon and N metabolism and is indispensable for efficient N assimilation in rice (Lu et al. 2011). Higher N application increased carbon and N content and NR and glutamine synthase (GS) activities in rice leaves, whereas it decreased GOGAT activity (Ding et al. 2014). Some recent studies have also shown that a high level of N enhanced the antioxidant defense system and some nitrogen assimilation enzymes, such as GS and GOGAT (Umnajkitikorn et al. 2021).
The reactive oxygen species (ROS) level represents the metabolic status, and excessive increase in the ROS level implies the occurrence or existence of damage in the plant cell (Mittler et al. 2004; Ahmad et al. 2010; Miller et al. 2010; Vighi et al. 2017). The concentration of ROS is regulated by antioxidant systems of enzymes, such as superoxide dismutase (SOD), peroxidase (POD), and catalase (CAT), as well as by antioxidant molecules, including ascorbate, carotenoids, flavonoids, and glutathione (Tewari et al. 2007). Split N fertilizer application was reported to significantly enhance the antioxidant enzyme activity and reduce the accumulation of malondialdehyde (MDA) in wheat (Zhang et al. 2021). In recent years the effect of nitrogen deficiency on antioxidant status in rice has attracted wide attention (Lin et al. 2011). N supply could delay leaf senescence in rice due to an increase in the activities of antioxidant enzymes and a decrease in the MDA content (Zhou et al. 2017). Recently, Xu et al. (2021) found that N supplementation improved the activities of N-related metabolic enzymes and reduced the MDA content in the leaves of two rice genotypes.
Previous reports have been published on the effects of N application on the yield, N metabolism, and antioxidant response of different rice genotypes with close genetic backgrounds. Therefore, we conducted the present study to explore the influence of different N dosages on the yield, N metabolism, and antioxidant response of three rice genotypes (NIL-7, NIL-8, and 02428). Our findings would provide a theoretical basis for N fertilizer management in rice production.
Materials and Methods
Experimental Description
Seeds from three rice genotypes (NIL-7, NIL-8, and 02428), provided by the College of Agriculture, South China Agricultural University (Guangzhou, China), were used as experimental materials in the present study. This experiment was conducted in the net house of Experimental Farm of College of Agriculture, South China Agricultural University, from March to July 2018.
Experimental Design
A pot experiment was conducted with two N fertilizer levels: low-N treatment with 0.94 g N per 10 kg of soil (LN) and high-N treatment with 1.88 g N per 10 kg of soil (HN), whereas the N treatments were monitored according to Mo et al. (2019). N fertilizer was applied as a base fertilizer, together with calcium phosphate (3.2 g per 10 kg of soil) and potassium chloride (1.11 g per 10 kg of soil). The fertilizer treatment was performed 2 days before rice transplantation. Each rice seedling was raised under wet seedling raising conditions. The three-leaf seedlings were transplanted into the pots (31 cm in diameter and 29 cm in height), with three seedlings per hill and five hills in each pot and 6 pots per treatment. After the transplanting, about 2–4 cm water layer was maintained in the pot and the water was drained off a week before harvest. Insects, weeds, and disease management were performed in accordance with the local rice growing practices.
Sampling and Measurements
Grain Yield and Biomass
At rice maturity stage, the grain yield and biomass were measured. The grain yield was measured following the method of Mo et al. (2015). Three representative plants were randomly selected and transported to the laboratory. The plant samples were separated into leaves, stems, and panicles and oven-dried at 80 °C to a constant weight for dry weight determination. The grain number per panicle, seed setting rate, grain weight, and the effective panicles per pot were measured. The harvest index is calculated according to the following formula:
Determination of the Physiological Attributes
At both the heading stage (66 days after transplanting) and maturity stage (91 days after transplanting), 15 fresh leaves were harvested from each treatment, washed with distilled water, immediately frozen in liquid nitrogen, and stored at − 80 °C refrigerators for biochemical analysis.
Nitrogen Metabolism Enzymes
The activity of NR was measured according to the method of Chen and Wang (2015). Fresh samples (0.50 g) were homogenized with 4 mL phosphate-buffered solution (PBS) in an ice bath, followed by centrifugation at 4000 rpm and 4 °C for 15 min; the supernatant was the crude enzyme extract. The reaction mixture contained 0.4 mL of crude enzyme solution, 1.2 mL of 0.1 M KNO3, and 0.4 mL of 0.25 mM NADH. The absorbance was measured at a wavelength of 540 nm. One NR active unit (U) was defined as the absorbance reduction and was expressed in U/g fresh weight (FW).
The GOGAT and GS activities were determined using the methods reported by Wang et al. (2005). Fresh samples (1.0 g) were homogenized in extraction buffer and was then centrifuged at 8000 rpm and 4 °C for 20 min. The supernatants were used for the determination of GOGAT activity and GS activity. For measurement of the GOGAT activity, mix the reaction solution, buffer, and crude enzyme solution and then measure the absorbance at 340 nm for 5 min at intervals of 30 s. One GOGAT active unit (U) was defined as the absorbance reduction of 0.01 per minute and was expressed in U/g FW. For GS activity measurement, the reaction mixture was admixed to the crude enzyme solution and then the absorbance at a wavelength of 540 nm was measured. One GS active unit (U) was defined as the absorbance reduction of 1 per hour and was expressed in U/mg FW.
Antioxidant Response Parameters
Fresh samples (0.30 g) were homogenized with 3 mL PBS in an ice bath and were centrifuged at 8000 rpm and 4 °C for 15 min; the supernatant was used for the determination of SOD, POD, and CAT activities and MDA content (Li et al. 2019).
The SOD activity was measured by the nitro-blue tetrazolium (NBT) method. The absorbance was determined at 560 nm. One unit of SOD activity was defined as 50% inhibition of the color reaction and was expressed in U/g FW.
For POD activity, the enzyme extract (50 μL) was added to the reaction solution containing 1 mL of 0.3% H2O2, 0.95 mL of 0.2% guaiacol, and 1 mL of 50 mM sodium phosphate buffer (pH 7.0). The absorbance at 470 nm was measured. One unit of POD activity was defined as the absorbance increase of 1 per minute and was expressed in U/g FW.
For the CAT activity, an aliquot of enzyme extract (50 μL) was added to the reaction solution containing 1 mL of 0.3% H2O2 and 1.95 mL of sodium phosphate buffer, and the absorbance was measured at 240 nm. One unit of CAT activity was defined as the absorbance increase of 0.01 per minute and was expressed in U/g FW.
The MDA content was measured using the thiobarbituric acid (TBA) method. MDA was reacted with TBA, and the absorbance of the reaction solution was recorded at 532, 600, and 450 nm. The MDA content was expressed in μmol/g FW.
Statistical Analyses
Experimental data were analyzed by Statistix version 8 (Analytical Software, Tallahassee, FL, USA), including variance analysis, correlation analysis, and principal component analysis. The difference among means was assessed by the least significant difference (LSD) test at P < 0.05. Figures were created by Microsoft Office 2016.
Results
Grain Yield and Yield Components of Different Rice Genotypes
The effective panicles per pot for NIL-7, NIL-8, and 02428 in HN treatment were higher than those in LN treatment by 32.04%, 49.26%, and 41.82%, respectively. The grain number per panicle in NIL-7 was significantly increased (by 21.72%), whereas no significant difference was detected between NIL-8 and 02428. In addition, HN treatment significantly decreased the seed setting rate in NIL-7 (by 28.8%). HN treatment caused also a reduction in grain weight. HN treatment significantly increased the grain yield in NIL-8 and 02428, which was 16.75% and 48.35% higher, correspondingly, than that in LN treatment. The genotypes significantly affected the grain yield and yield components apart from the grain weight. The interaction of rice genotypes and N treatment (G × N) significantly influenced the seed setting rate and grain yield. The N treatment effect on rice yield and yield components varied among the rice genotypes was studied (Table 1).
Total Dry Weight and Harvest Index of Different Rice Genotypes
HN treatment increased significantly the total dry weight in NIL-7, NIL-8, and 02428, which was 29.84%, 27.91%, and 49.15% higher, respectively, than that measured in LN. HN treatment significantly decreased the harvest index in NIL-7, NIL-8, and 02428 (by16.21%, 7.55%, and 0.84%, correspondingly). The differences in total dry weight and harvest index among the rice genotypes were statistically significant (Table 2).
NR, GS, and GOGAT Activities of Different Rice Genotypes
The examined rice genotypes had different NR activities at the heading and maturity stages. At the two stages, HN treatment led to NR activities higher than those in the LN treatment. The NR activity of NIL-8 at the heading stage was significantly higher (26.23%); at the maturity stage the NR activity in NIL-7 and NIL-8 was also significantly increased, by 60.95% and 64.73%, respectively. The interaction of rice genotypes and N treatment (G × N) significantly affected the NR activity at the maturity stage. Additionally, significant differences among the rice genotypes were established in the NR activity (Table 3).
Appreciable differences were found in the GS activity among different rice genotypes. At the maturity stage, the GS activity of NIL-8 was the highest, followed by those in 02428 and NIL-7. The difference in the enzyme activities among the rice genotypes reached a significant level. At the heading stage, the difference between NIL-8 and 02428 was statistically significant. HN treatment increased the GS activity in NIL-8 by 35.49% as compared to that of LN at the heading stage and by 201.50% that of LN at the maturity stage. The interaction of rice genotypes and N treatment (G × N) significantly influenced the GS activity at the heading stage and maturity stage. Different GS activities were observed between the heading and maturity stages (Table 4).
HN treatment in the studied rice genotypes promoted more GOGAT activity at the heading and maturity stages than LN treatment. As compared with LN, HN treatment more substantially enhanced the GOGAT activity in NIL-7 (by 128.83%) at the heading stage and in NIL-7 and 02428 by 66.05% and 38.27%, respectively, at the maturity stage. The interaction of rice genotypes and N treatment (G × N) significantly affected the GOGAT activity at the handing stage. However, the GOGAT activity varied significantly at the maturity stage among different rice genotypes (Table 5).
The SOD, POD, and CAT Activity and the MDA Content in Different Rice Genotypes
The SOD activity varied greatly among different rice genotypes. Under LN treatment, the SOD activity of NIL-7 was the highest, followed in a descending order by those of 02428 and NIL-8 at the heading stage. At the maturity stage, the SOD activity of NIL-7 was the highest, followed by those of NIL-8 and 02428. Under HN treatment, the SOD activities of NIL-7 and NIL-8 were significantly higher than that of 02428 at the heading stage. At the maturity stage, the SOD activity of NIL-8 was the highest, followed by those of NIL-7 and 02428. Furthermore, compared with LN, HN treatment remarkably increased the SOD activity in NIL-8 by 58.85% at the heading stage. The interaction of rice genotypes and N treatment (G × N) significantly influenced the SOD activity at the heading stage and maturity stage. Rice genotypes showed a remarkable influence on the SOD activity at the heading stage and maturity stage (Table 6).
Large differences were established in the effect of N treatment on the POD activity of the experimental rice genotypes. The POD activities in NIL-7 and NIL-8 were higher than that in LN, but that in the HN treatment in 02428 was lower at the heading stage. At the maturity stage, the POD activities in NIL-8 and 02428 were higher, whereas that in NIL-7 was lower in HN treatment than in LN. Compared with LN, HN treatment significantly increased the POD activity in NIL-7 (by 48.48%) at the heading stage; increases of 37.29% and 26.12%, respectively, were observed in NIL-8 and 02428 at the maturity stage. The interaction of rice genotypes and N treatment (G × N) significantly influenced the POD activity at the heading stage and maturity stage (Table 7).
There were significant differences in the effect of the N fertilizer application on the CAT activity among different rice genotypes. The CAT activity in NIL-8 in HN treatment was significantly increased (by 196.22%) at the maturity stage than that recorded in LN treatment. HN treatment led to higher CAT activities in NIL-7 and NIL-8 at the heading stage than those in LN, but that parameter had a lower value in 02428; the CAT activities in NIL-8 and 02428 at the maturity stage were higher, but that in NIL-7 was lower than the respective value of that parameter in LN. The interaction of rice genotypes and N treatment (G × N) significantly affected the CAT activity at the maturity stage (Table 8).
Under LN treatment, the MDA content of NIL-8 at the heading stage was significantly higher than those in NIL-7 and 02428. At the maturity stage, the MDA content of NIL-7 was the highest, followed by those of NIL-8 and 02428. The differences in the MDA content among the rice genotypes reached significant levels. Besides, the MDA content in NIL-8 in HN treatment was significantly lower (by 30.82%) than that in LN at the heading stage; at the maturity stage in NIL-8 and 02428, it was lower by 12.31% and 18.62%, respectively. Briefly, significant differences among the rice genotypes were observed in the effect of N fertilizer application on MDA content; the higher N fertilizer application reduced the MDA content. N treatments showed a remarkable impact on the MDA content at the heading stage and maturity stage. The interaction of rice genotypes and N treatment (G × N) significantly influenced the MDA content at the heading stage (Table 9).
Correlation Analysis
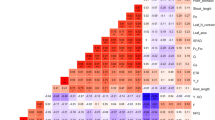
Grain yield had a significant positive correlation with total dry weight (r = 0.8867, P < 0.05). Besides, grain yield showed a remarkable negative correlation with grain weight (r = − 0.8698, P < 0.05) (Fig. 1).
PCA Analysis
PCA revealed five principal components (PCs) with a cumulative percentage of the variance of 100%, and variance percentages for PC1, PC2, PC3, PC4, and PC5 were 83.3%, 7.9%, 5.6%, 2.4%, and 0.3%, respectively (Fig. 2). For PC1, the top three parameters (CAT activity at the maturity stage, CAT activity at the heading stage, and grain number per panicle) were detected with high loading values. For PC2, the top three parameters (CAT activity at the heading stage, CAT activity at the maturity stage, and NR activity at the maturity stage), for PC3, top 3 parameters (grain number per panicle, CAT activity at the heading stage, and total dry weight), for PC4, top three parameters (total dry weight, NR activity at maturity stage, and effective panicles per hole) and PC5, top 3 parameters (SOD activity at the heading stage, POD activity at the heading stage, and grain number per panicle) were investigated with high loading value (Table 10).
Discussion
N is a key limiting factor for agricultural productivity. It is well known that N fertilization increases grain yield (Makino, 2011). A similar result was observed in the present study (Table 1). Besides, the application of N fertilization affected the grain yield as a consequence of the changes in N metabolism and antioxidant response; the effects of N application varied greatly among the studied rice genotypes (Tables 1, 2, 3, 4, 5, 6, 7, 8 and 9).
Many studies have shown that rice yield and N fertilizer application are positively correlated (Tang et al. 2020; Jiang et al. 2019b). Here, we reached the same conclusion. Previous studies of yield components have revealed that the effective panicles per pot and the grain number per panicle increased with the rise in the applied N fertilizer quantity (Zhang et al. 2013; Li et al. 2020; Liu et al. 2019). In this study, significantly more effective panicles per pot were obtained in HN treatment than in LN treatment, but no significant difference was detected in the grain number per panicle between the two N fertilization treatments (Table 1). However, the seed setting rate and the grain weight were lower in HN treatment (Table 1), and grain yield showed a significant negative correlation with grain weight (Fig. 1a). Similar results have also been reported previously (Zhang et al. 2013). In another investigation, N treatments influenced more considerably the number of panicles and the seed setting rate, eventually affecting grain yield (Zhou et al. 2021). In addition, N fertilization had an impact on dry weight and the increase in grain yield (Yamuangmorn et al. 2018). In this study, correlation analysis indicated that grain yield had a significant positive correlation with total dry weight (Fig. 1b). The harvest index in HN treatment was significantly lower than that in LN treatment (Table 2). Moreover, Haefele et al. (2008) indicated that, within a range of genotypes tested, considerable differences exist in the efficiency of nitrogen acquisition and intimal N use efficiency. Distinct genotypic differences exhibit in rice plant-associated N2 fixation and the efficiency of N absorption and transport (Cheng et al. 2011; Broadbent et al. 1987). Thus, it is necessary to improve grain yield and grain N by the management of N fertilizer in specific rice genotypes (Fongfon et al. 2021). In this study, the genotypes significantly affected the grain yield and yield components apart from the grain weight. Therefore, the grain yield rise in HN treatment resulted from the increase in the number of effective panicles and total dry weight, as well as the change in the seed setting rate and the grain weight of different rice genotypes.
Furthermore, rice plants absorb and utilize inorganic N such as NH4+and NO3− by the GS/GOGAT cycle (Krapp 2015; Xu et al. 2012). GOGAT and GS are key enzymes involved in N assimilation and translocation (Lu et al. 2011; Xu et al. 2013). The application of high levels of nitrogen enhances the assimilation of NO3− and NH4+ and increases the activity of enzymes related to nitrogen metabolism (Zhong et al. 2017). The effect of N application on GS activity in functional rice leaves was different among different varieties and growth stages (Gang et al. 2010; Ning et al. 2009). The GS activity in the leaves was found to increase with the rise in N level (Chen et al. 2008). In this study, considerable differences were established in the GS activity between different rice genotypes and different stages (Table 4). The GOGAT activity at the maturity stage in HN treatment was significantly higher than that in LN treatment; the GOGAT activity at the heading stage was higher than that at the maturity stage (Table 5). Studies have shown that NR is a rate-limiting factor in the nitrate reduction pathway and a direct response to the level of N metabolism (Cong et al. 2019; Corpas et al. 2011). Chen et al. (2008) revealed the existence of a complex relationship between the NR activity and the N level. The NR activity in the leaves gradually increased with the rise in the amount of the N applied (Ye et al. 2005). The findings of this investigation showed that the NR activities at the heading and maturity stages in HN treatment were higher than those in LN treatment and varied significantly among different rice genotypes (Table 3). In conclusion, the improvement of the grain yield in HN treatment was also related to the increase in the NR and GOGAT activities. N treatment affected differently N metabolism among the studied rice genotypes.
In addition, plant resistance was closely related to the antioxidant enzyme activity. POD, CAT, and SOD activities in plants reflect their ability to eliminate ROS (Shi et al. 2021). A study has investigated the effect on the antioxidant system in rice of different nitrogen sources and they found that glycine markedly reduced the increase in the level of MDA and significantly increased the activities of POD and SOD, which might be attributed to the decreased levels of ROS as a consequence of N stimulating effect on the antioxidant system (Gao et al. 2017). Exogenous nitrogen might be affecting the expression of genes involved in metabolism, growth, and defense responses in rice (Asgher et al. 2022). An earlier study showed that the application of a suitable N fertilizer rate promoted the POD, CAT, and SOD activities in the roots and leaves and reduced the MDA content (Yu et al. 2020). In another investigation, the application of an appropriate N fertilizer amount significantly improved the activities of antioxidant enzymes (SOD, CAT, and POD) at the seedling period of rice (Jiang et al. 2005). The MDA content in HN treatment was significantly lower than that in LN treatment, which was consistent with previous research results (Table 9). However, we found no significant differences in the SOD, POD, and CAT activities between HN and LN treatments (Tables 6, 7 and 8). ROS concentration is regulated by antioxidant systems, such as antioxidative enzymes (SOD, POD, CAT, glutathione peroxidase, and enzymes involved in the ascorbate glutathione cycle) and antioxidant compounds (ascorbate, tocopherol, carotenoids, flavonoids, and glutathione) (Tewari et al. 2007). Therefore, further in-depth research is needed to evaluate the association between ROS regulation and N metabolism under N application. In a word, increasing N fertilizer regulated the activity of SOD, POD, and CAT and decreased the MDA content, finally influencing the grain yield of different rice genotypes.
Moreover, some researchers had found that there were significant interaction effects among nitrogen applications, rice cultivars, and shading treatments for yield and yield related in rice (Pan et al. 2016; Feng et al. 2020). In the present study, genotypes significantly affected the investigated parameters apart from grain weight, the GS activity and GOGAT activity at the maturity stage, and the POD activity and MDA content at the heading stage. N treatment showed a remarkable influence on the grain yield and yield components (except for grain numbers per panicle). The interaction of genotype and N treatment (G × N) significantly influenced the grain yield, seed setting rate, NR activity, and CAT activity at the maturity stage, the GOGAT activity and MDA content at the heading stage, and the GS activity, SOD activity, and POD activity at the heading stage and maturity stage (Tables 1, 2, 3, 4, 5, 6, 7, 8 and 9). Besides the PCA analysis indicated that some core parameters could be used to explain the effect of N application on grain yield, nitrogen metabolism, and antioxidant response in different rice genotypes. The total cumulative percentage of variance of 100% includes PC1 (83.3%), PC2 (7.9%), PC3 (5.6%), PC4 (2.4%), and PC5 (0.3%) (Fig. 2). Within the 5 principal components, the grain numbers per panicle, the total dry weight, NR activity at the maturity stage, and CAT activity at the heading stage and maturity stage (Table 10). Those results showed that the N fertilizer application regulated rice yield and influenced the nitrogen metabolism and antioxidant response.
Conclusion
Altogether, increasing the N fertilizer rate significantly increased the number of effective panicles per pot and the total dry weight of the studied rice genotypes. The application of a higher N amount also improved the activities of nitrate reductase and glutamate synthetase, which subsequently increased rice yield. On the other hand, the rise in the applied N fertilizer amount changed the activity of the superoxide dismutase, peroxidase, and catalase and reduced the MDA content in the rice plants. In conclusion, N fertilizer application regulates rice yield by affecting the yield formation and biomass accumulation and modulating the activities of enzymes involved in N metabolism, the antioxidant enzyme activity, and MDA content. Distinct genotype internal efficiency of N and recovery efficiency of applied N were significantly different, so the specific values of these parameters vary among different rice genotypes. Thus, it is essential to elucidate the physiological response mechanism of specific genotypes to different N fertilizer applications.
Data Availability
The data sets supporting the results of this article are included within the article.
Code Availability
No code.
References
Andrews M, Raven JA, Lea PJ (2013) Do plants need nitrate? the mechanisms by which nitrogen form affects plants. Ann Appl Biol 163(2):174–199. https://doi.org/10.1111/aab.12045
Ahmad P, Jaleel CA, Salem MA, Nabi G, Sharma S (2010) Roles of enzymatic and non-enzymatic antioxidants in plants during abiotic stress. Crit Rev Biotechnol 30(3):161–175. https://doi.org/10.3109/07388550903524243
Asgher M, Sehar Z, Rehaman A, Rashid S, Ahmed S, Per TS, Alyemeni MN, Khan NA (2022) Exogenously-applied L-glutamic acid protects photosynthetic functions and enhances arsenic tolerance through increased nitrogen assimilation and antioxidant capacity in rice (Oryza sativa L.). Environ Pollut 301:119–151. https://doi.org/10.1016/j.envpol.2022.119008
Broadbent FE, Datta SKD, Laureles EV (1987) Measurement of nitrogen utilization efficiency in rice genotypes. Agron J 79(5):786–791. https://doi.org/10.2134/agronj1987.00021962007900050006x
Cong XH, Shi FZ, Ruan XM, Luo YX, Wang YL, Xu YZ, Luo ZX (2019) Effect of nitrogen application rate on key enzymes of nitrogen utilization and carbon and nitrogen metabolism of different rice varieties. J Henan Agric Univ 53(3):325–330 (in Chinese with English Abstract)
Corpas FJ, Leterrier M, Valderrama R, Airaki M, Chaki M, Palma JM, Barroso JB (2011) Nitric oxide imbalance provokes a nitrosative response in plants under abiotic stress. Plant Sci 181(5):604–611. https://doi.org/10.1016/j.plantsci.2011.04.005
Cheng J, Jiang H, Liu Y, Dai T, Cao W (2011) Methods on identification and screening of rice genotypes with high nitrogen efficiency. Rice Sci 18(2):127–135
Cheng B, Jiang Y, Cao CG (2021) Balance rice yield and eating quality by changing the traditional nitrogen management for sustainable production in China. J Clean Prod 312(4):127–793. https://doi.org/10.1016/j.jclepro.2021.127793
Chen JX, Wang XF (2015) Laboratory physiology experimental guidance. South China University of Technology Press, Guangzhou
Chen XF, Ning SG, Wei DZ, Zhang HP (2008) Effect of nitrogen level on nitrogen metabolism in rice seedlings. Chin J Eco-Agric 16(3):571–575 (in Chinese with English Abstract)
Deng NY, Grassini P, Yang HS, Huang JL, Cassman KG, Peng SB (2019) Closing yield gaps for rice self-sufficiency in China. Nat Commun 10(1):17–25. https://doi.org/10.1038/s41467-019-09447-9
Ding CQ, You J, Chen L, Wang SH, Ding YF (2014) Nitrogen fertilizer increases spikelet number per panicle by enhancing cytokinin synthesis in rice. Plant Cell Rep 33(2):363–371. https://doi.org/10.1007/s00299-013-1536-9
Fongfon S, Prom-u-thai C, Pusadee T, Jamjod S (2021) Responses of purple rice genotypes to nitrogen and zinc fertilizer application on grain yield, nitrogen, zinc, and anthocyanin concentration. Plant Physiol Ecophysiol 10(8):17–17. https://doi.org/10.3390/plants10081717
Feng H, Li Y, Yan Y, Wei X, Yang Y, Zhang L, Lin M, Tang X, Mo Z (2020) Nitrogen regulates the grain yield, antioxidant attributes, and nitrogen metabolism in fragrant rice grown under lead-contaminated soil. J Soil Sci Plant Nutr 20(4):1–13. https://doi.org/10.1007/s42729-020-00278-y
Gao X, Chu Z, Zhu L, Zhang L, Hussain S, Wu L, Jin Q (2017) Glycine increases cold tolerance in rice via the regulation of n uptake, physiological characteristics, and photosynthesis. Plant Physiol Biochem 112:251–260. https://doi.org/10.1016/j.plaphy.2017.01.008
Gang S, Wang JG, Yang L, Gao Y, Zang JX, Zhao HW (2010) Effects of nitrogen level on key enzyme to nitrogen metabolism of rice in cold region. Res Agric Mod 31(2):224–227 (in Chinese with English Abstract)
Haefele SM, Jabbar SMA, Siopongco JDLC, Tirol-Padre A, Amarante ST, Cruz PCS, Cosico WC (2008) Nitrogen use efficiency in selected rice (Oryza sativa L.) genotypes under different water regimes and nitrogen levels. Field Crops Res 107(2):137–146. https://doi.org/10.1016/j.fcr.2008.01.007
Jiang ZW, Lin WX, Li YZ, Zhuo CY, Yang HJ, Xie HA (2005) Effects of nitrogen fertilizer rates on some physiological characteristics of ratoon rice. Fujian J Agric Sci 20(3):168–171 (in Chinese with English Abstract)
Jiang M, Li X, Xin L, Tan M (2019a) Paddy rice multiple cropping index changes in southern China. J Geogr Sci 29(11):1773–1787. https://doi.org/10.1007/s11442-019-1689-8
Jiang Z, Zhong Y, Yang J, Wu Y, Li H, Zheng L (2019b) Effect of nitrogen fertilizer rates on carbon footprint and ecosystem service of carbon sequestration in rice production. Sci Total Environ 670(20):210–217. https://doi.org/10.1016/j.scitotenv.2019.03.188
Krapp A (2015) Plant nitrogen assimilation and its regulation: a complex puzzle with missing pieces. Curr Opin Plant Biol 25:115–122. https://doi.org/10.1016/j.pbi.2015.05.010
Ladha JK, Tirolpadre A, Punzalan GC, Castillo E, Singh U, Reddy CK (1998) Nondestructive estimation of shoot nitrogen in different rice genotypes. J Agron 90(1):33–40. https://doi.org/10.2134/agronj1998.00021962009000010007x
Liang Z, Bao A, Li H, Cai H (2015) The effect of nitrogen level on rice growth, carbon-nitrogen metabolism, and gene expression. Biologia 70(10):1340–1350. https://doi.org/10.1515/biolog-2015-0148
Li S, Jiang H, Wang J, Wang Y, Pan S, Tian H, Duan M, Wang S, Tang X, Mo Z (2019) Responses of plant growth, physiological, gas exchange parameters of super and non-super rice to rhizosphere temperature at the tillering stage. Sci Rep 9:1–17. https://doi.org/10.1038/s41598-019-47031-9
Li Y, Lai R, Li W, Liu J, Huang M, Tang Y, Tang X, Pan S, Duan M, Tian H, Wu L, Wang S, Mo Z (2020) γ-Aminobutyric acid regulates grain yield formation in different fragrant rice genotypes under different nitrogen levels. J Plant Growth Regul 39:738–750. https://doi.org/10.1007/s00344-019-10016-z
Lin YL, Chao YY, Huang WD, Kao CH (2011) Effect of nitrogen deficiency on antioxidant status and cd toxicity in rice seedlings. Plant Growth Regul 64(3):263–273. https://doi.org/10.1007/s10725-011-9567-0
Liu LJ, Sang DZ, Liu CL, Wang ZQ, Yang JC, Zhu QS (2004) Effects of real-time and site-specific nitrogen managements on rice yield and nitrogen use efficiency. J Agric Sci 03:23–29
Liu K, Deng J, Lu J, Wang X, Lu B, Tian X, Zhang Y (2019) High nitrogen levels alleviate yield loss of super hybrid rice caused by high temperatures during the flowering stage. Front Plant Sci 10:357. https://doi.org/10.3389/fpls.2019.00357
Lu YE, Luo F, Yang M, Li XH, Lian XM (2011) Suppression of glutamate synthase genes significantly affects carbon and nitrogen metabolism in rice (Oryza sativa L.). Sci China Life Sci 54:651–663. https://doi.org/10.1007/s11427-011-4191-9
Makino A (2011) Photosynthesis, grain yield, and nitrogen utilization in rice and wheat. Plant Physiol 155(1):125–129. https://doi.org/10.1104/pp.110.165076
Mittler R, Vanderauwera S, Gollery M, Breusegem FV (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9(10):490–498. https://doi.org/10.1016/j.tplants.2004.08.009
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ 33(4):453–467. https://doi.org/10.1111/j.1365-3040.2009.02041.x
Mo Z, Li W, Pan S, Fitzgerald TL, Xiao F, Tang Y, Wang Y, Duan M, Tian H, Tang X (2015) Shading during the grain filling period increases 2-acetyl-1-pyrroline content in fragrant rice. Rice 8:1–10. https://doi.org/10.1186/s12284-015-0040-y
Mo Z, Li Y, Nie J, He L, Pan S, Duan M, Tian H, Xiao L, Zhong K, Tang X (2019) Nitrogen application and different water regimes at booting stage improved yield and 2-acetyl-1-pyrroline (2AP) formation in fragrant rice. Rice 12:74
Muthayya S, Sugimoto JD, Montgomery S, Maberly GF (2014) An overview of global rice production, supply, trade, and consumption. Ann N Y Acad Sci 1324(1):7–14. https://doi.org/10.1111/nyas.12540
Ning SJ, Dou HJ, Chen XF, Lin WX, Wei DZ (2009) Dynamics of nitrogen metabolism activity in rice root at the late development stage. Chin J Eco-Agric 17(3):506–511 (in Chinese with English Abstract)
Nakhjiri SE, Ashouri M, Sadeghi SM, Roshan NM, Rezai M (2021) The effect of irrigation management and nitrogen fertilizer on grain yield and water-use efficiency of rice cultivars in northern Iran. Gesunde Pflanz 73:359–366. https://doi.org/10.1007/s10343-021-00562-6
Pan S, Liu H, Mo Z, Patterson B, Duan M, Tian H, Hu S, Tang X (2016) Effects of nitrogen and shading on root morphologies, nutrient accumulation, and photosynthetic parameters in different rice genotypes. Sci Rep 6(1):1–14. https://doi.org/10.1038/srep32148
Peng S, Buresh RJ, Huang J, Zhong X, Zou Y, Yang J, Wang G, Liu Y, Hu R, Tang Q, Cui K, Zhang F, Dobermann A (2010) Improving nitrogen fertilization in rice by site-specific N management. A review. Agron Sustain Dev 30(3):649–656. https://doi.org/10.1051/agro/2010002
Singh H, Verma A, Ansari MW, Shukla A (2014) Physiological response of rice (Oryza sativa L.) genotypes to elevated nitrogen applied under field conditions. Plant Signal Behav 9(7):29015–32923. https://doi.org/10.4161/psb.29015
Sun CQ, Yang YJ, Guo ZL, Qu F (2015) Effects of fertilization and density on soluble sugar proteins and nitrate reductase in hybrid foxtail millet. J Plant Nutr Fertil 21(05):1169–1177 (in Chinese with English Abstract)
Sun ZG, Wang BX, Yang B, Xu B, Xing YG, Kazeem BB, Xu DY (2019) Effects of nitrogen application levels on nitrogen use efficiency and agronomic traits of rice cultivars. Acta Agric Jiangxi 31(12):23–28 (in Chinese with English Abstract)
Shi LH, Li C, Tang HM, Wen L, Li WY, Xiao XP, Cheng KK, Liu Q, Cheng AW (2021) Effects of different long-term fertilizer management on physiological characteristic of root and yield of rice double-cropping rice paddy field. J China Agric Univ 26(09):18–29 (in Chinese with English Abstract)
Tang J, Tang C, Guo BW, Zhang CX, Zhang ZZ, Wang K, Zhang HC, Chen H, Sun MZ (2020) Effect of nitrogen application on yield and rice quality of mechanical transplanting high quality late rice. Acta Agron Sin 46(01):117–130 (in Chinese with English Abstract)
Tewari RK, Kumar P, Sharma PN (2007) Oxidative stress and antioxidant responses in young leaves of mulberry plants grown under nitrogen, phosphorus or potassium deficiency. J Integr Plant Biol 49(3):313–322. https://doi.org/10.1111/j.1744-7909.2007.00358.x
Trost B, Prochnow A, Meyer-Aurich A, Drastig K, Baumecker M, Ellmer F (2016) Effects of irrigation and nitrogen fertilization on the greenhouse gas emissions of a cropping system on a sandy soil in northeast Germany. Eur J Agron 81:117–128. https://doi.org/10.1016/j.eja.2016.09.008
Umnajkitikorn K, Fukudome M, Uchiumi T, Teaumroong N (2021) Elevated nitrogen priming induced oxinitro-responses and water deficit tolerance in rice. Plants 10(2):381
Vighi IL, Benitez LC, Amaral MN, Moraes GP, Auler PA, Rodrigues GS, Deuner S, Mais LC, Braga BJ (2017) Functional characterization of the antioxidant enzymes in rice plants exposed to salinity stress. Biol Plant 61:540–550. https://doi.org/10.1007/s10535-017-0727-6
Vijayalakshmi P, Kiran TV, Rao YV, Srikanth B, Rao IS, Sailaja B, Rao PR, Subrahmanyam D, Neeraja CN, Voleti SR (2013) Physiological approaches for increasing nitrogen use efficiency in rice. Plant Physiol Rep 18:208–222. https://doi.org/10.1007/s40502-013-0042-y
Wang XC, Xiong SP, Ma XM, Zhang JJ, Wang ZQ (2005) Effects of different nitrogen forms on key enzyme activity involved in nitrogen metabolism and grain protein content in speciality wheat cultivars. Acta Ecol Sin 25:802–807
Wang W, Xu X, Zhu C, Gu J, Zhang W, Liu G, Zhu J (2019) Elevated CO2-induced changes in cytokinin and nitrogen metabolism are associated with different responses in the panicle architecture of two contrasting rice genotypes. Plant Growth Regul 89:119–129. https://doi.org/10.1007/s10725-019-00511-4
Xu G, Fan X, Miller AJ (2012) Plant nitrogen assimilation and use efficiency. Annu Rev Plant Biol 63(1):153–182. https://doi.org/10.1146/annurev-arplant-042811-105532
Xu C, Wang D, Chen S, Zhang X (2013) Effects of aeration on root physiology and nitrogen metabolism in rice. Rice Sci 20(2):148–153. https://doi.org/10.1016/S1672-6308(13)60118-3
Xu Y, Li K, Zhu K, Tian Y, Yu Q, Zhang W, Wang Z (2021) Effect of nitrogen rates on the growth and grain yield of a leaf early-senescent rice mutant. Agron J 113:1503–1522. https://doi.org/10.1002/agj2.20587
Yu XF, Xian HY, Qiu XL, Zou BL, Zhang SH, Liu GS, Zhang YF, Zeng M, Zeng LC, Yang SM (2020) Effect of low temperature and nitrogen fertilizer coupling on physiological indexes of rice. Southwest China J Agric Sci 33(10):2190–2197 (in Chinese with English Abstract)
Yang X, Geng J, Liu Q, Zhang H, Hao X, Sun Y, Lu X (2021) Controlled-release urea improved rice yields by providing nitrogen in synchrony with the nitrogen requirements of plants. J Sci Food Agric 101(10):4183–4192. https://doi.org/10.1002/jsfa.11056
Yamuangmorn S, Dell B, Rerkasem B, Prom-U-Thai C (2018) Applying nitrogen fertilizer increased anthocyanin in vegetative shoots but not in grain of purple rice genotypes. J Sci Food Agric 98(12):4527–4532. https://doi.org/10.1002/jsfa.8978
Ye QB, Zhang HC, Dai QG, Li H, Huo ZY, Xu K, Tang J (2005) Effects of nitrogen amount applied and planting density on nitrate reductase activity of rice during middle-late growth stages. Plant Physiol J 41(1):41–44 (in Chinese with English Abstract)
Ye Q, Zhang H, Wei H, Zhang Y, Wang B, Xia K, Hou Z, Dai Q, Xu K (2007) Effects of nitrogen fertilizer on nitrogen use efficiency and yield of rice under different soil conditions. Front Mech Eng China 1:30–36. https://doi.org/10.1007/s11703-007-0005-z
Zhao HW, Sha HJ (2014) Recent research of fertilizer- nitrogen use efficiency in paddy flied of China. J Northeast Agric Univ 45(2):116–122 (in Chinese with English Abstract)
Zhang XL, Su XJ, Chen CH, Tang MY, Wang Q, Chen L, Lang TF (2013) Effects of different nitrogen application levels on population structure, yield and nitrogen use efficiency of Liangfengyou 339. J South Agric 44(10):1667–16670 (in Chinese with English Abstract)
Zhang Z, Yu Z, Zhang Y, Shi Y (2021) Split nitrogen fertilizer application improved grain yield in winter wheat (Triticum aestivum L.) via modulating antioxidant capacity and 13C photosynthate mobilization under water-saving irrigation conditions. Ecol Process 10(1):2–13. https://doi.org/10.1186/s13717-021-00290-9
Zhou W, Lv T, Yang Z, Wang T, Fu Y, Chen Y, Hu B, Ren W (2017) Morphophysiological mechanism of rice yield increase in response to optimized nitrogen management. Sci Rep 7(1):17226. https://doi.org/10.1038/s41598-017-17491-y
Zhou C, Huang Y, Jia B, Wang Y, Wang Y, Xu Q, Li R, Wang S, Dou F (2018) Effects of cultivar, nitrogen rate, and planting density on rice-grain quality. Agronomy 8(11):246–246. https://doi.org/10.3390/agronomy8110246
Zhou C, Jia B, Wang S, Huang Y, Wang Y, Han K, Wang W (2021) Effects of nitrogen fertilizer applications on photosynthetic production and yield of Japonica rice. Int J Plant Prod 15:599–613. https://doi.org/10.1007/s42106-021-00156-2
Zhong C, Cao X, Hu J, Zhu L, Zhang J, Huang J, Jin Q (2017) Nitrogen metabolism in adaptation of photosynthesis to water stress in rice grown under different nitrogen levels. Front Plant 8:1079. https://doi.org/10.3389/fpls.2017.01079
Acknowledgements
We acknowledgement the funding supported by the Guangdong Provincial key field research and development plan project (2019B020221003) and the Guangdong Basic and Applied Basic Research Foundation (2021A1515010410).
Funding
This project was financially supported by the Guangdong Provincial key field research and development plan project (2019B020221003) and the Guangdong Basic and Applied Basic Research Foundation (2021A1515010410).
Author information
Authors and Affiliations
Contributions
ZM designed the experiments, YY performed the traits investigation, GL, YY, and ZM analyzed the data and wrote the manuscript, and WX, and ZM revised and edited the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Additional information
Handling Editor: Mikihisa Umehara.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liao, G., Yang, Y., Xiao, W. et al. Nitrogen Modulates Grain Yield, Nitrogen Metabolism, and Antioxidant Response in Different Rice Genotypes. J Plant Growth Regul 42, 2103–2114 (2023). https://doi.org/10.1007/s00344-022-10684-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-022-10684-4