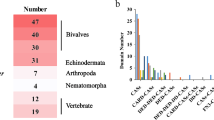
Abstract
Caspase-8, first classified as a pro-apoptotic caspase, is considered to have arisen from duplication with caspase-10 and involves multiple immune and inflammatory responses in mammals. However, few are known on the phylogeny and function of caspase-8 in molluscs, one of the largest phyla in marine invertebrates. In this study, we conducted phylogenetic and functional analysis on molluscan caspase-8-like genes. Results indicate that duplication occurred in molluscan caspase-8-like genes, resulting in at least two caspase-8 copies in some groups of bivalves. Additional studies in Pacific oyster Crassostrea gigas showed different spatio-temporal expression patterns and subcellular localizations of CgCaspase-8-1 and CgCaspase-8-2. While no interaction was observed between CgCaspase-8-2 and CgFADD, the adaptor molecule in apoptosis, yeast two-hybrid and co-immunoprecipitation assays suggested the interaction between CgCaspase-8-1 and CgFADD, indicating its pro-apoptotic function. In addition, CgCaspase-8-1 showed interaction with the CARD domain of CgRIG-I. Together with two NF-κB subunits (Cgp105 and CgRel), their transcripts were up-regulated in response to poly(I:C) stimuli, supporting the immune function of both pro, and anti-inflammation. The results provide insight into the evolution and functional diversification of Bivalvia caspase-8 genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Data Availability Statement
The data used and analyzed during this study are included in this article or available from the corresponding author on reasonable request.
References
Amé J C, Rolli V, Schreiber V et al. 1999. PARP-2, A novel mammalian DNA damage-dependent poly(ADP-ribose) polymerase. Journal of Biological Chemistry, 274(25): 17860–17868, https://doi.org/10.1074/jbc.274.25.17860.
Ashkenazi A, Dixit V M. 1998. Death receptors: signaling and modulation. Science, 281(5381): 1305–1308, https://doi.org/10.1126/science.281.5381.1305.
Benchoua A, Couriaud C, Gueágan C et al. 2002. Active caspase-8 translocates into the nucleus of apoptotic cells to inactivate poly(ADP-ribose) polymerase-2. Journal of Biological Chemistry, 277(37): 34217–34222, https://doi.org/10.1074/jbc.M203941200.
Besnault-Mascard L, Leprince C, Auffredou M T et al. 2005. Caspase-8 sumoylation is associated with nuclear localization. Oncogene, 24(20): 3268–3273, https://doi.org/10.1038/sj.onc.1208448.
Bjellqvist B, Basse B, Olsen E et al. 1994. Reference points for comparisons of two-dimensional maps of proteins from different human cell types defined in a pH scale where isoelectric points correlate with polypeptide compositions. Electrophoresis, 15(1): 529–539, https://doi.org/10.1002/elps.1150150171.
Bjellqvist B, Hughes G J, Pasquali C et al. 1993. The focusing positions of polypeptides in immobilized pH gradients can be predicted from their amino acid sequences. Electrophoresis, 14(1): 1023–1031, https://doi.org/10.1002/elps.11501401163.
Bladon J, Taylor P C. 2006. Extracorporeal photopheresis: a focus on apoptosis and cytokines. Journal of Dermatological Science, 43(2): 85–94, https://doi.org/10.1016/j.jdermsci.2006.05.004.
Boldin M P, Mett I L, Varfolomeev E E et al. 1995. Self-association of the “death domains” of the p55 tumor necrosis factor (TNF) receptor and Fas/APO1 prompts signaling for TNF and Fas/APO1 effects. Journal of Biological Chemistry, 270(1): 387–391, https://doi.org/10.1074/jbc.270.1.387.
Capella-Gutiérrez S, Silla-Martinez J M, Gabaldon T. 2009. trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics, 25(15): 1972–1973, https://doi.org/10.1093/bioinformatics/btp348.
Chaudhary P M, Eby M T, Jasmin A et al. 2000. Activation of the NF-κB pathway by caspase 8 and its homologs. Oncogene, 19(39): 4451–4460, https://doi.org/10.1038/sj.onc.1203812.
Coon S L, Bonar D B, Weiner R M. 1986. Chemical production of cultchless oyster spat using epinephrine and norepinephrine. Aquaculture, 58(3–4): 255–262, https://doi.org/10.1016/0044-8486(86)90090-6.
de Castro E, Sigrist C J A, Gattiker A et al. 2006. ScanProsite: detection of PROSITE signature matches and ProRule-associated functional and structural residues in proteins. Nucleic Acids Research, 34(S2): W362–W365, https://doi.org/10.1093/nar/gkl124.
Fan T J, Han L H, Cong R S et al. 2005. Caspase family proteases and apoptosis. Acta Biochimica et Biophysica Sinica, 37(11): 719–727, https://doi.org/10.1111/j.1745-7270.2005.00108.x.
Fortier M E, Kent S, Ashdown H et al. 2004. The viral mimic, polyinosinic: polycytidylic acid, induces fever in rats via an interleukin-1-dependent mechanism. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 287(4): R759–R766, https://doi.org/10.1152/ajpregu.00293.2004.
Galluzzi L, Maiuri M C, Vitale I et al. 2007. Cell death modalities: classification and pathophysiological implications. Cell Death & Differentiation, 14(7): 1237–1243, https://doi.org/10.1038/sj.cdd.4402148.
Galluzzi L, Vitale I, Abrams J M et al. 2012. Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death & Differentiation, 19(1): 107–120, https://doi.org/10.1038/cdd.2011.96.
Gasteiger E, Hoogland C, Gattiker A et al. 2005. Protein identification and analysis tools on the ExPASy server. In: Walker J M ed. The Proteomics Protocols Handbook. Humana, Totowa. p.571–607, https://doi.org/10.1385/1-59259-890-0:571.
Green D R, Llambi F. 2015. Cell death signaling. Cold Spring Harbor Perspectives in Biology, 7(12): a006080, https://doi.org/10.1101/cshperspect.a006080.
Hedgecock D, Gaffney P M, Goulletquer P et al. 2005. The case for sequencing the Pacific oyster genome. Journal of Shellfish Research, 24(2): 429–441, https://doi.org/10.2983/0730-8000(2005)24[429:Tcfstp]2.0.Co;2.
Henry C M, Martin S J. 2017. Caspase-8 acts in a non-enzymatic role as a scaffold for assembly of a Pro-inflammatory “FADDosome” complex upon TRAIL stimulation. Molecular Cell, 65(4): 715–729.E5, https://doi.org/10.1016/j.molcel.2017.01.022.
Hu W H, Johnson H, Shu H B. 2000. Activation of NF-κB by FADD, Casper, and caspase-8. Journal of Biological Chemistry, 275(15): 10838–10844, https://doi.org/10.1074/jbc.275.15.10838.
Huang B Y, Zhang L L, Xu F et al. 2019. Oyster versatile IKKα/βs are involved in toll-like receptor and RIG-I-like receptor signaling for innate immune response. Frontiers in Immunology, 10: 1826, https://doi.org/10.3389/fimmu.2019.01826.
Katoh K, Standley D M. 2013. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molecular Biology and Evolution, 30(4): 772–780, https://doi.org/10.1093/molbev/mst010.
Kerr J F R, Wyllie A H, Currie A R. 1972. Apoptosis: a basic biological phenomenon with wideranging implications in tissue kinetics. British Journal of Cancer, 26(4): 239–257, https://doi.org/10.1038/bjc.1972.33.
Kruidering M, Evan G I. 2000. Caspase-8 in apoptosis: the beginning of “the end”? IUBMB Life, 50(2): 85–90, https://doi.org/10.1080/713803693.
Lacoste A, Cueff A, Poulet S A. 2002. P35-sensitive caspases, MAP kinases and Rho modulate β-adrenergic induction of apoptosis in mollusc immune cells. Journal of Cell Science, 115(4): 761–768, https://doi.org/10.1242/jcs.115-4.761.
Launay S, Hermine O, Fontenay M et al. 2005. Vital functions for lethal caspases. Oncogene, 24(33): 5137–5148, https://doi.org/10.1038/sj.onc.1208524.
Lavrik I, Golks A, Krammer P H. 2005. Death receptor signaling. Journal of Cell Science, 118(2): 265–267, https://doi.org/10.1242/jcs.01610.
Letunic I, Khedkar S, Bork P. 2021. SMART: recent updates, new developments and status in 2020. Nucleic Acids Research, 49(D1): D458–D460, https://doi.org/10.1093/nar/gkaa937.
Li C Y, Qu T, Huang B Y et al. 2015. Cloning and characterization of a novel caspase-8-like gene in Crassostrea gigas. Fish & Shellfish Immunology, 46(2): 486–492, https://doi.org/10.1016/jfisi.2015.06.035.
Li H L, Zhu H, Xu C J et al. 1998. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the fas pathway of apoptosis. Cell, 94(4): 491–501, https://doi.org/10.1016/s0092-8674(00)81590-1.
Li Z X, Wang C, Jiang F J et al. 2016. Characterization and expression of a novel caspase gene: evidence of the expansion of caspases in Crassostrea gigas. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 201: 37–45, https://doi.org/10.1016/j.cbpb.2016.07.001.
Liu F Y, Li Y L, Yu H W et al. 2021. MolluscDB: an integrated functional and evolutionary genomics database for the hyper-diverse animal phylum Mollusca. Nucleic Acids Research, 49(D1): D988–D997, https://doi.org/10.1093/nar/gkaa918.
Liu W Z, Xie Y B, Ma J Y et al. 2015. IBS: an illustrator for the presentation and visualization of biological sequences. Bioinformatics, 31(20): 3359–3361, https://doi.org/10.1093/bioinformatics/btv362.
Lu G X, Yu Z C, Lu M M et al. 2017. The self-activation and LPS binding activity of executioner caspase-1 in oyster Crassostrea gigas. Developmental & Comparative Immunology, 77: 330–339, https://doi.org/10.1016/j.dci.2017.09.002.
Matsuda I, Matsuo K, Matsushita Y et al. 2014. The C-terminal domain of the long form of cellular FLICE-inhibitory protein (c-FLIPL) inhibits the interaction of the caspase 8 prodomain with the receptor-interacting protein 1 (RIP1) death domain and regulates caspase 8-dependent nuclear factor κB (NF-κB) activation. Journal of Biological Chemistry, 289(7): 3876–3887, https://doi.org/10.1074/jbc.M113.506485.
Micheau O, Tschopp J. 2003. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell, 114(2): 181–190, https://doi.org/10.1016/s0092-8674(03)00521-x.
Philip N H, DeLaney A, Peterson L W et al. 2016. Activity of uncleaved caspase-8 controls anti-bacterial immune defense and TLR-induced cytokine production independent of cell death. PLoS Pathogens, 12(10): e1005910, https://doi.org/10.1371/journal.ppat.1005910.
Puente X S, Sánchez L M, Overall C M et al. 2003. Human and mouse proteases: a comparative genomic approach. Nature Reviews Genetics, 4(7): 544–558, https://doi.org/10.1038/nrg1111.
Qin Y P, Zhang Y H, Li X Y et al. 2020. Characterization and functional analysis of a caspase 3 gene: evidence that ChCas 3 participates in the regulation of apoptosis in Crassostrea hongkongensis. Fish & Shellfish Immunology, 98: 122–129, https://doi.org/10.1016/j.fsi.2020.0L007.
Qu T, Huang B Y, Zhang L L et al. 2014. Identification and functional characterization of two executioner caspases in Crassostrea gigas. PLoS One, 9(2): e89040, https://doi.org/10.1371/journal.pone.0089040.
Rajput A, Kovalenko A, Bogdanov K et al. 2011. RIG-I RNA helicase activation of IRF3 transcription factor is negatively regulated by caspase-8-mediated cleavage of the RIP1 protein. Immunity, 34(3): 340–351, https://doi.org/10.1016/j.immuni.2010.12.018.
Ren J, Wen L P, Gao X J et al. 2009. DOG 1.0: illustrator of protein domain structures. Cell Research, 19(2): 271–273, https://doi.org/10.1038/cr.2009.6.
Romero A, Novoa B, Figueras A. 2015. The complexity of apoptotic cell death in mollusks: an update. Fish & Shellfish Immunology, 46(1): 79–87, https://doi.org/10.1016/j.fsi.2015.03.038.
Ronquist F, Teslenko M, van der Mark P et al. 2012. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Systematic Biology, 61(3): 539–542, https://doi.org/10.1093/sysbio/sys029.
Roth S, Ruland J. 2011. Caspase-8: clipping off RIG-I signaling. Immunity, 34(3): 283–285, https://doi.org/10.1016/j.immuni.201L03.011
Salvesen G S, Dixit V M. 1997. Caspases: intracellular signaling by proteolysis. Cell, 91(4): 443–446, https://doi.org/10.1016/s0092-8674(00)80430-4.
Schütze S, Tchikov V, Schneider-Brachert W. 2008. Regulation of TNFR1 and CD95 signalling by receptor compartmentalization. Nature Reviews Molecular Cell Biology, 9(8): 655–662, https://doi.org/10.1038/nrm2430.
Subramanian B, Gao S H, Lercher M J et al. 2019. Evolview v3: a webserver for visualization, annotation, and management of phylogenetic trees. Nucleic Acids Research, 47(W1): W270–W275, https://doi.org/10.1093/nar/gkz357.
Takeuchi O, Akira S. 2010. Pattern recognition receptors and inflammation. Cell, 140(6): 805–820, https://doi.org/10.1016/j.cell.2010.0L022.
Vogeler S, Carboni S, Li X X et al. 2021. Phylogenetic analysis of the caspase family in bivalves: implications for programmed cell death, immune response and development. BMC Genomics, 22(1): 80, https://doi.org/10.1186/s12864-021-07380-0.
Wajant H. 2002. The Fas signaling pathway: more than a paradigm. Science, 296(5573): 1635–1636, https://doi.org/10.1126/science.1071553.
Wang J, Chun H J, Wong W et al. 2001. Caspase-10 is an initiator caspase in death receptor signaling. Proceedings of the National Academy of Sciences of the United States of America, 98(24): 13884–13888, https://doi.org/10.1073/pnas.241358198.
Wang K, Yin X M, Chao D T et al. 1996. BID: a novel BH3 domain-only death agonist. Genes & Development, 10(22): 2859–2869, https://doi.org/10.1101/gad.10.22.2859.
Xiang Z M, Qu F F, Qi L et al. 2013. Cloning, characterization and expression analysis of a caspase-8 like gene from the Hong Kong oyster, Crassostrea hongkongensis. Fish & Shellfish Immunology, 35(6): 1797–1803, https://doi.org/10.1016/j.fsi.2013.08.026.
Yang B Y, Li L L, Pu F et al. 2015. Molecular cloning of two molluscan caspases and gene functional analysis during Crassostrea angulata (Fujian oyster) larval metamorphosis. Molecular Biology Reports, 42(5): 963–975, https://doi.org/10.1007/s11033-014-3833-y.
Yoneyama M, Fujita T. 2009. RNA recognition and signal transduction by RIG-I-like receptors. Immunological Reviews, 227(1): 54–65, https://doi.org/10.1111/j.1600-065X.2008.00727.x.
Zhang G F, Fang X D, Guo X M et al. 2012. The oyster genome reveals stress adaptation and complexity of shell formation. Nature, 490(7418): 49–54, https://doi.org/10.1038/nature11413.
Zhang L L, Li L, Zhang G F. 2011. Gene discovery, comparative analysis and expression profile reveal the complexity of the Crassostrea gigas apoptosis system. Developmental & Comparative Immunology, 35(5): 603–610, https://doi.org/10.1016/j.dci.2011.01.005.
Acknowledgment
We thank Jie MENG for her valuable suggestions and comments on this manuscript, and Oceanographic Data Center, IOCAS, for data support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Science & Technology Innovation Project of Laoshan Laboratory (No. LSKJ202203001), the Center for Ocean Mega-Research of Science, Chinese Academy of Sciences (No. COMS2019Q11), the GHfund B (No. 20210702), and the Taishan Scholars Program
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Deng, S., Qu, T., Zhang, G. et al. Oyster studies reveal the duplication and functional diversification of Bivalvia caspase-8 genes. J. Ocean. Limnol. 41, 1109–1121 (2023). https://doi.org/10.1007/s00343-022-2129-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-022-2129-7