Abstract
Marine habitats vary widely in structure, from incredibly complex coral reefs to simpler deep water and open ocean habitats. Hydromechanical models of swimming kinematics and microevolutionary studies suggest that these habitats select for different body shape characteristics. Fishes living in simple habitats are predicted to experience selection for energy-efficient sustained swimming, which can be achieved by fusiform body shapes. In contrast, fishes living in complex habitats are predicted to be under selection for maneuverability, which can be enhanced by deep-bodied and laterally compressed forms. To look for a signature of these processes at a broad macroevolutionary scale, we quantified the body shapes of 3322 species of marine teleostean fishes using a series of linear measurements. We scored each species for whether they were reef-associated or not and tested for morphological differences using a phylogenetic framework. Our results confirmed significant overall shape differences between reef-associated teleosts and those occupying structurally simpler marine habitats. Reef-associated species have, on average, deeper bodies and higher depth-to-width ratios, while non-reef species are more streamlined with narrower and shallower caudal peduncles. Despite the numerous evolutionary forces that may influence body shapes on a broad macroevolutionary scale, our results reveal differences in body shapes between reef-associated and non-reef species that are consistent with hydromechanical models of swimming kinematics as well as with microevolutionary patterns.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reefs are among the most diverse and biologically productive environments on Earth (Sargent and Austin 1949, 1954; Odum and Odum 1955; Kohn and Helfrich 1957; Connell 1978; Hatcher 1988; Ferreira et al. 2001; Steneck et al. 2002; Monismith 2007). Despite representing only a small percentage of total oceanic surface area, reefs are home to a disproportionately large fraction of marine species (Spalding and Grenfell 1997; Spalding et al. 2001; Roberts et al. 2002; Spalding and Brown 2015). Among fishes, an estimated 4000 to 8000 species (i.e., ~ 22–44% of marine fishes) are associated with coral and temperate reef habitats (Roberts and Ormond 1987; Choat and Bellwood 1991; Spalding et al. 2001; Aguilar-Medrano and Arias-González 2018). One of the reasons reefs can support such diverse communities is that they are structurally heterogeneous and generally far more complex than other marine habitats (Emery 1978; Choat and Bellwood 1991; Sale 1991; Spalding et al. 2001; Gratwicke and Speight 2005a, b; Lingo and Szedlmayer 2006). These characteristics increase the number of available ecological niches as well as overall productivity (Talbot 1965; Risk 1972; Luckhurst and Luckhurst 1978; Bellwood and Wainwright 2002; Alfaro et al. 2007). Consequently, reefs can support both ecologically and functionally diverse fish communities (Gratwicke and Speight 2005a; Farré et al. 2015; Aguilar-Medrano and Arias-González 2018; Evans et al. 2019).
Habitat structural complexity can also have major implications for fish morphology by influencing the direction and/or the magnitude of evolutionary change. Complex habitats, such as coral and temperate reefs, contain numerous physical obstacles that fishes must avoid or use to their advantage and are therefore predicted to select for morphologies that improve maneuverability, i.e., unsteady swimming (Webb 1994; Blake 2004; Langerhans and Reznick 2010). Structurally simpler habitats, for example the pelagic zone where obstacles and refuges from predators are limited, are expected instead to select for morphologies that improve energy-efficient swimming over long distances, i.e., steady swimming (Webb 1994; Blake 2004; Langerhans and Reznick 2010). It has been argued that there are trade-offs related to body shape associated with increasing performance for either steady or unsteady swimming (Webb 1982; Blake 1983; Webb 1994; Langerhans 2009; Domenici 2010; Langerhans and Reznick 2010). Hydromechanical models predict that steady swimming is facilitated by a fusiform or streamlined body shape and a narrow caudal peduncle, as these characteristics help to minimize energy loss from drag (Breder 1926; Keast and Webb 1966; Webb 1984; Webb and Weihs 1986; Blake et al. 1995; Walker 1997; Blake 2004; Langerhans 2009; Langerhans and Reznick 2010). Conversely, unsteady swimming performance can be improved by deeper, laterally compressed bodies and deeper caudal peduncles, traits that are thought to facilitate sharp turns and rapid acceleration (Alexander 1967; Webb 1984; Webb and Weihs 1986; Webb et al. 1996; Walker 1997; Schrank et al. 1999; Langerhans 2009). In structurally complex reefs, lateral compression may also facilitate predator evasion by allowing fishes to seek refuge in narrow spaces created between rocks or corals (Hixon and Beets 1993; Walker 2000; Lingo and Szedlmayer 2006).
The predictions concerning body shapes that improve performance and fitness in simple versus complex habitats have become pervasive in the fish biology literature and are cited in ichthyology textbooks as a classic example of how habitat affects body shape (e.g., Moyle and Cech 2004; Helfman et al. 2009). These predictions have so far been well supported both by interspecific comparisons of swimming kinematics within an experimental framework (Webb et al. 1996; Schrank et al. 1999; Webb and Fairchild 2001) and by microevolutionary ecological studies comparing body shapes between populations occupying habitats of differing structural complexity (Ehlinger and Wilson 1988; Walker 1997). Support for some of these predictions has also been shown in species-level comparisons across specific taxonomical groups (Davis and Birdsong 1973; Frédérich et al. 2016). However, it remains to be seen if there is a macroevolutionary signature for these patterns at a very broad taxonomic scale. Additionally, given the exceptional ecological and functional diversity of coral and temperate reef fish communities (Gratwicke and Speight 2005a; Farré et al. 2015; Aguilar-Medrano and Arias-González 2018; Evans et al. 2019), we also expect to find evidence for increased morphological disparity among the reef-associated species (Choat and Bellwood 1991; Claverie and Wainwright 2014). In order to test these hypotheses, we compiled a large morphometric dataset of linear traits measured on teleostean fishes with a sufficient phylogenetic scope to incorporate multiple independent evolutionary transitions between reef and non-reef habitats (Supplementary Figure S1). We then applied phylogenetic comparative methods to compare body and caudal peduncle shapes as well as disparities between reef-associated teleosts (complex habitats) and those occupying other types of habitats (considered as less complex).
Methods
Morphological measurements
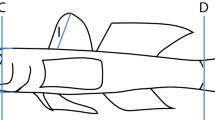
For this study, we used a subset of 3322 marine teleosts that are part of a larger morphometric dataset assembled from the Smithsonian National Museum of Natural History preserved fish collections [see Price et al. (2019) for a more comprehensive description of the full dataset]. The subset includes species that can occupy both marine and brackish habitats but excludes those that are found exclusively in brackish salinities. We restricted our analyses to six body and caudal peduncle linear measurements for which we had clear hydromechanical predictions about how they would differ as a function of habitat structural complexity (Table 1). These original variables were also used to calculate six additional composite variables that add information about the overall shape of the body and caudal peduncle (Table 2). Multivariate analyses (e.g., morphological disparity, phylogenetic PCAs in Supplementary Material) were performed only on the original variables as the ratios contain redundant information.
We size-standardized the original variables using a modification of the log-shape ratios method (Mosimann 1970; Mosimann and James 1979; Darroch and Mosimann 1985). Briefly, we divided each morphological variable by the geometric mean of three linear measurements indicative of the overall size of our specimens (i.e., standard length, maximum body depth and maximum fish width) and log-transformed the resulting ratios. This method was preferred to using residuals of a phylogenetic regression on standard length for two reasons: (1) it preserves information about evolutionary allometry in the data, and (2) it allows us to conserve standard length as a shape variable instead of using it only as a size proxy. Composite variables were not size-standardized as they are ratios of one or more of the original variables and therefore already account for relative size.
Habitat complexity
To compare body shapes between species occupying complex habitats and those occupying simpler ones, we used a binary coding scheme, as coral and temperate reefs are unquestionably the most structurally complex among aquatic habitats (Emery 1978; Monismith 2007) and detailed quantitative data on habitat structural complexity is non-existent for most species. Therefore, we scored each of the species in our dataset as either reef-associated (i.e., complex habitats, n = 1496) or not (i.e., simpler habitats, n = 1826). Although teleosts living in non-reef habitats may experience a wide variety of structural complexities, these are unlikely to match the complexity of coral and temperate reefs. Habitat information was extracted from Fishbase (Froese and Pauly 2020) using the species function from the rfishbase package (Boettiger et al. 2012) in R. To ensure data quality, we performed random spot-checks across the dataset, verifying the Fishbase data against other sources, including scientific literature and other online data repositories.
Analyses
The phylogenetic context for all analyses was provided by a tree published by Rabosky et al. (2018), pruned to the species contained in our dataset. Phylogenetic ANOVAs (Garland et al. 1993) and pgls ANOVAs (Freckleton et al. 2002) were performed on both the size-standardized variables and the composite variables to determine whether there were significant differences in body shape between reef-associated and non-reef teleosts. The phylogenetic and pgls ANOVAs were implemented using the R packages geiger (Pennell et al. 2014) and caper (Orme et al. 2018), respectively. For the phylogenetic ANOVAs sensu Garland et al. (1993), the statistical significance of the F value was determined by generating a null distribution based on 1000 simulations of the dependent variable on the phylogenetic tree under a Brownian motion model of evolution.
Although our hypotheses explicitly predict differences in body shapes across habitats and not differences in body size, we also ran the ANOVAs on the log-transformed geometric mean to see if reef-associated and non-reef species differed in overall size. Moreover, we looked for differences in allometric trajectories between reef-associated and non-reef teleosts. Given that we predicted that reef and non-reef habitats would select for specific body shape characteristics, we tested for differences in allometric trajectories within an adaptive framework. For this purpose, we used the method implemented in the R package slouch (Kopperud et al. 2020) that can be used to determine whether traits under differing selective regimes are evolving toward different primary optima (Hansen et al. 2008). For each of the morphological traits, we compared the fit of four models: (1) a grand mean (or intercept-only) model including only the dependent variable, (2) a more complex grand mean model estimating separate intercepts for the reef-associated and non-reef taxa, (3) a regression model of the dependent variable as a function of size and (4) a regression model that estimates separate coefficients for the reef-associated and non-reef taxa. In the regression models, the primary optimum is modeled as a linear function between the trait of interest and size. The fit of each of the models can be compared using the Akaike information criterion (AICc) modified for small sample sizes (Hurvich and Tsai 1989; Burnham and Anderson 2004). These models allow us to determine whether the trait is approaching an optimum and how fast it is doing so, as well as the proportion of variation in the trait that can be explained by adaptation toward that optimum. The rate at which the trait value approaches the optimum is obtained by interpreting the phylogenetic half-life, which is defined as the “time it takes for the expected trait value to move half the distance from the ancestral state to the primary optimum” (Hansen 1997; Hansen et al. 2008). The half-life is interpreted relative to the length of the phylogeny: a relatively short phylogenetic half-life implies rapid adaptation toward the primary optimum, whereas a longer half-life implies a persistence of the ancestral influence (a half-life of infinity corresponds to Brownian motion) (Hansen et al. 2008). The regression models also include estimated slopes for both an optimal regression and an evolutionary regression. The optimal regression can be interpreted as the expected relationship between shape variables and size in the absence of constraints on the evolution of the traits toward their respective optima. The evolutionary regression is the observed relationship between the shape variables and size and is expected to have a shallower slope compared to the optimal regression if there is a lag in adaptation. The comparisons of allometric trajectories were performed on the log-transformed original variables prior to size standardization.
Finally, to test for differences in morphological disparities between reef-associated and non-reef teleosts, we estimated and compared group-wise disparities using the morphol.disparity function in geomorph (Adams et al. 2020). Briefly, the function calculates disparity as the sum of the diagonal elements from the group covariance matrix scaled by the number of observations in each group (Zelditch et al. 2012). Disparity was estimated first on the log-transformed original variables and then on the log-shape ratios (size-standardized variables).
Results
The results of phylogenetic ANOVAs (sensu Garland et al. 1993) on the original size-standardized variables indicate significant differences between reef-associated and non-reef teleostean fishes in standard length, maximum body depth, head depth and minimum caudal peduncle depth and width (Table 3; Fig. 1). Reef-associated species were found to be less elongate than non-reef species, to have deeper bodies and heads, and to possess both deeper and wider caudal peduncles. We did not find a significant difference in maximum fish width between reef-associated and non-reef species. Similar results were obtained for the composite variables (Table 3, Fig. 2): we found significant differences between reef-associated and non-reef teleosts in fineness and depth-to-width ratios. Reef-associated species have, on average, lower fineness ratios and greater depth-to-width ratios than non-reef species. We did not find significant differences in caudal peduncle compression index or in either of the two caudal tapering indices between reef-associated and non-reef teleosts. We focus here on the results from the Garland et al. (1993) method because the phylogenetic structure of habitat transitions within teleosts (see Supplementary Fig. S1) indicates that taking into account the phylogenetic pattern of the discrete trait may also remove much of the ecological signal as well (i.e., we expect adaptation toward phylogenetically structured optima). The results from the pgls ANOVAs are available in Supplementary Table S5. There were some key differences between these two methods with the pgls ANOVAs uncovering significant differences in maximum fish width and both caudal tapering indices, while significant differences in standard length and fineness ratio between reef-associated and non-reef species were lost.
The comparison of geometric means between habitats showed no significant difference in overall body size between reef-associated and non-reef teleosts (Table 3). As for differences in allometric trajectories, the results from our analyses using slouch (Table 4) revealed that with the exception of maximum fish width, the models with separate estimates of the regression coefficients for reef-associated and non-reef species were always preferred over those with combined estimates of the parameters, as indicated by their lower AICc values. For maximum fish width, a model with a single estimate of the regression coefficients for reef-associated and non-reef teleosts was equally well supported. The best supported models explained large proportions of the variation with R2 values ranging from 0.62 to 0.89. Compared to the regression models, the intercept-only models explain only a fraction of the variation in the data as demonstrated by their low R2 values. Moreover, the amount of variation explained by the two-intercept models is only slightly larger than that of the single-intercept models and the differences in the estimated intercepts between the reef-associated and non-reef species are quite low. These results indicate that size variation has a strong effect on body shape and that whether a fish lives in a reef or a non-reef habitat has a measurable but negligible impact on this allometric relationship. Furthermore, for all variables, the estimated regression slopes are close to one, which suggests that the impressive variation in body shapes that can be observed among teleostean fishes does not require large deviations from isometry to be produced. Finally, for all of the best-fitting models, the phylogenetic half-lives are far shorter than the depth of the tree, suggesting that all traits are rapidly adapting toward their respective primary optima.
Prior to size standardization, we did not find a significant difference in disparities between the reef-associated and non-reef teleosts. However, the comparison of Procrustes variances estimated from the size-standardized variables revealed that disparities were significantly greater in non-reef species (Table 5).
Discussion
Our results reveal clear body shape differences between teleostean lineages inhabiting the structurally complex reefs and those that occupy relatively simpler habitats, which cannot be explained by differences in evolutionary allometry. The directions of the shape differences are consistent with predictions from hydromechanical models of swimming kinematics and observations from micro- and macroevolutionary studies. Contrary to our expectation, we also found that non-reef teleostean fishes are more morphologically disparate than reef-associated ones.
We predicted that across teleostean fishes, performance-based selection for either sustained swimming or greater maneuverability would lead to body shape differences between species associated with complex reef habitats and those living in structurally simpler habitats. We found that, on average, reef-associated species had deeper bodies relative to their overall size and width, which supports our prediction that they should be deeper and more laterally compressed than species in relatively simpler habitats, as these traits are thought to improve accelerating and turning maneuvers (Alexander 1967; Webb 1983, 1994; Domenici 2010). Reef-associated species were also found to have deeper and wider caudal peduncles relative to their size compared to the non-reef species. Thicker caudal peduncles may be advantageous for fast starts in reef environments by contributing to the thrust generated by the caudal fin during quick bursts of acceleration (Webb 1994; Walker 1997; Domenici 2010; Bellwood et al. 2014). In contrast, non-reef teleosts were more elongate, had greater fineness ratios and were not as deep-bodied as the reef-associated species. Furthermore, the caudal peduncles of non-reef teleosts were narrower (width) and shallower (depth) than those of the reef-associated species. These results match our predictions for a more streamlined morphology with a tapering tail in structurally simpler habitats, characteristics which are thought to facilitate steady swimming by maximizing thrust and minimizing energy loss from drag and recoil (Fisher and Hogan 2007; Domenici 2010; Langerhans and Reznick 2010). These shape differences may also be explained in part by the recurrence of elongated morphologies with reduced/tapering tails in deep-sea habitats (e.g., Notacanthiformes, Macrouridae, Ophidiiformes).
Given the results mentioned above, it may seem surprising that neither depth nor width caudal peduncle tapering indices were found to be significantly different between the reef-associated and non-reef teleosts, despite that they are both, on average, lower for the non-reef species. This result could reflect an organismal design constraint: the shape of the caudal peduncle is most likely influenced by the overall shape of the body such that deeper bodied species will also have a relatively deeper caudal peduncle. However, the significant results for the size-standardized minimum caudal peduncle depth and width variables indicate that reef-associated species have a comparatively deeper and wider caudal peduncle relative to their overall size than non-reef species.
Although our predictions based on hydromechanical models were well supported by the data, this is not to say that all sustained swimmers should exhibit a streamlined morphology, nor that high maneuverability can only be achieved with a deep and laterally compressed body shape. Indeed, many of these models are predicated on the assumption that the main thrust producing appendage is the caudal fin. However, fish swimming can be broadly divided in two main categories: (1) body–caudal fin (BCF) swimmers generate thrust using undulations along the body ultimately transmitting force to the caudal fin, and (2) median-paired fin (MPF) swimmers generate thrust using combinations of their other median and paired fins (Alexander 1967; Webb 1975; Webb 1994; Sfakiotakis et al. 1999). Therefore, hydromechanical constraints that apply for BCF swimming kinematics may not always be the same as those that influence MPF swimming kinematics. For example, Walker et al. (2013) found differences in the strength and the direction of the relationship between fineness ratio and endurance swimming performance when comparing BCF and MPF swimmers. Likewise, high maneuverability can be achieved with body shapes other than deep and laterally compressed forms. Many species occupying complex habitats predominantly use MPF swimming gaits (Gerstner 1999; Blake 2004), and Blake (2004) argued that there may be multiple optimal body forms for MPF swimmers. Boxfishes provide one such example where high maneuverability is achieved without a deep and laterally compressed body (Blake 1977; Walker 2000; Bartol et al. 2005). Another way to increase maneuverability is to have a more flexible body (e.g., Fish 2002). Anguilliformes provide a great example of this strategy, as eels are arguably just as efficient as laterally compressed deep-bodied fishes in navigating structurally complex habitats owing to their flexible elongated bodies and reduced lateral appendages (Webb 1994; Pfaff et al. 2016). These alternative strategies may help explain why we did not find a significant decrease in body width in reef teleosts compared to non-reef species. Our results may indicate that a laterally compressed body shape is merely the most prevalent strategy for occupants of complex habitats, despite other strategies also being quite common.
The results from our slouch analyses indicate that allometry is a major source of shape variation across teleostean fishes, as the intercept-only models were a poor fit to the data. For all traits except maximum fish width, the best-fitting model allowed the allometric intercept to vary between reef and non-reef habitats, and the next best-fitting model was a single allometric slope and intercept (ΔAICc: 46–70), which suggests that there is a measurable difference between the evolutionary allometries in reef-associated and non-reef lineages. The phylogenetic half-life estimates for the best-fitting models reveal that while adaptation is not immediate (half-life = 0), it is rapid relative to the depth of the phylogeny, which is again indicative of the strength of the allometric relationship. Similarly, the difference between the optimal and evolutionary regression slopes indicates few constraints on optimal shape evolution, although the evolutionary slope is slightly shallower suggesting some minor lag in adaptation. These results are consistent with the idea that changes in body proportions can provide a path of least resistance for evolutionary change (Marroig and Cheverud 2005; Friedman et al. 2019). However, when we consider the additional variation explained by the two-intercept models compared to the single-intercept ones, the amount is minuscule (< 0.6%) and the estimated intercepts for reef-associated and non-reef fishes are very similar. Therefore, habitat, in the form of either living in a complex reef or a simpler non-reef environment, has an identifiable but negligible impact on evolutionary allometries within marine teleosts. Thus, the strong shape differences we observed between reef-associated and non-reef fishes were not primarily driven by differences in evolutionary allometries.
The comparison of morphological disparity estimates between reef-associated and non-reef teleosts revealed that once shape variation correlated to size was removed from the data, structurally simpler habitats contained greater body shape disparity (Table 5). This finding contradicts our prediction, stemming from several lines of evidence, that the structurally complex reef habitats should provide more axes of niche diversification than the simpler ones (Willis et al. 2005). First, reefs are well known for supporting fish communities that are highly ecologically and functionally diverse (Gratwicke and Speight 2005a; Farré et al. 2015; Aguilar-Medrano and Arias-González 2018; Evans et al. 2019). Second, higher rates of morphological evolution have been recorded for reef-associated species compared to their non-reef counterparts in both the Haemulidae (Price et al. 2013) and the Labridae (Price et al. 2011), two families that are prevalent in reef environments [although see Frédérich et al. (2016) who found higher phenotypic rates and disparities in non-reef Carangoidei]. Finally, MPF swimming is thought to be more common in structurally complex habitats (Gerstner 1999), and one might expect higher disparities among the MPF swimmers compared to the BCF swimmers. This is because most MPF swimmers rely on the caudal fin at faster gaits (i.e., steady swimming) but primarily use combinations of their other median and paired fins at slower gaits (i.e., unsteady swimming) (Webb 1994; Blake 2004). This can facilitate a decoupling between the body regions that are engaged at faster versus slower gaits, leading to a greater number of possibilities to simultaneously optimize morphology for these specific gaits (Blake et al. 1995; Blake 2004). Higher disparities in the non-reef species may be explained by the fact that in our binary scoring of habitats, the “structurally simpler” category incorporates several different types of habitats (e.g., benthic, pelagic, deep-sea) that may each involve additional selective pressures. Additionally, differences in disparities could also be explained by the greater amount of time that has been spent outside of reef environments during the evolutionary history of teleosts. Indeed, results from stochastic character mapping suggest that teleosts have spent, on average, 16% more time outside of reef habitats during their evolutionary history, thus providing increased opportunity for non-reef lineages to accumulate morphological disparity (Supplementary Material).
Our results support predictions based on long-standing hydromechanical models. However, the R2 values obtained from the phylogenetic ANOVAs indicate that habitat complexity is by no means the only factor that explains variation in size-standardized body shape across marine teleostean fishes, and thus, some caution is necessary when interpreting these results. For several reasons, it is perhaps not too surprising that the amount of variation explained only by differences in habitat complexity is somewhat low. We are examining evolutionary patterns across more than 3000 species, and many other factors are known to influence fish body shape [e.g., trophic ecology (Keast and Webb 1966; Lavin and McPhail 1985; Bellwood et al. 2006; Clabaut et al. 2007), predation pressure (Lavin and McPhail 1985; Brönmark and Miner 1992; Langerhans et al. 2004; Domenici et al. 2008; Langerhans 2009; Price et al. 2015), depth (Zimmerman et al. 2006; Clabaut et al. 2007), water flow (Langerhans et al. 2003; McGuigan et al. 2003; Kerfoot and Schaefer 2006; Leal et al. 2011)]. Moreover, due to the lack of quantitative data on structural complexity, we were only able to compare species with two very broadly defined habitat categories. We also did not distinguish between BCF and MPF swimmers in our analyses, and fishes that use these different forms of locomotion may show different pattern strengths between shape and swimming performance (Walker et al. 2013). Finally, we have focused largely on body shapes, yet fin traits are also important factors in swimming kinematics (Weihs 1989; Gerstner 1999; Fulton et al. 2001; Colgate and Lynch 2004; Bartol et al. 2005). Despite these caveats, the strong support for our predictions concerning the specific body shapes expected to increase swimming performance in complex versus less complex environments provides evidence that habitat complexity has driven divergent body shape evolution in reef-associated and non-reef marine teleosts. Our results therefore confirm reef habitats have made an important contribution to shaping and maintaining present-day patterns of teleostean morphological diversity (Bellwood et al. 2015).
Change history
14 August 2020
This erratum has been initiated as several formatting errors were overlooked during proofing stage by vendor.
References
Adams D, Collyer M, Kaliontzopoulou A (2020) Geomorph: software for geometric morphometric analyses. Version 3.2.1. https://cran.r-project.org/package=geomorph
Aguilar-Medrano R, Arias-González JE (2018) Functional reef fish groups of the Mexican Caribbean: implications of habitat complexity. Rev Mex Biodivers 89:1138–1153
Alexander RM (1967) Functional design in fishes. Hutchinson & Co Ltd., London
Alfaro ME, Santini F, Brock CD (2007) Do reefs drive diversification in marine teleosts? Evidence from the pufferfish and their allies (order Tetraodontiformes). Evolution 61:2104–2126
Bainbridge R (1960) Speed and stamina in three fish. J Exp Biol 37:129–153
Bartol IK, Gharib M, Webb PW, Weihs D, Gordon MS (2005) Body-induced vortical flows: a common mechanism for self-corrective trimming control in boxfishes. J Exp Biol 208:327–344
Bellwood DR, Wainwright PC (2002) The history and biogeography of fishes on coral reefs. In: Sale PF (ed) Coral reef fishes: dynamics and diversity in a complex ecosystem. Academic Press, San Diego, pp 5–32
Bellwood DR, Wainwright PC, Fulton CJ, Hoey AS (2006) Functional versatility supports coral reef biodiversity. Proc R Soc B-Biol Sci 273:101–107
Bellwood DR, Goatley CHR, Brandl SJ, Bellwood O (2014) Fifty million years of herbivory on coral reefs: fossils, fish and functional innovations. Proc R Soc B-Biol Sci 281:20133046
Bellwood DR, Goatley CHR, Cowman PF, Bellwood O (2015) The evolution of fishes on coral reefs: fossils, phylogenies, and functions. In: Mora C (ed) Ecology of fishes on coral reefs. Cambridge University Press, Cambridge, pp 55–63
Blake RW (1977) On ostraciiform locomotion. J Mar Biol Assoc UK 57:1047–1055
Blake RW (1983) Functional design and burst-and-coast swimming in fishes. Can J Zool-Rev Can Zool 61:2491–2494
Blake RW (2004) Fish functional design and swimming performance. J Fish Biol 65:1193–1222
Blake RW, Chatters LM, Domenici P (1995) Turning radius of yellowfin tuna (Thunnus albacares) in unsteady swimming maneuvers. J Fish Biol 46:536–538
Blasina G, Molina J, Cazorla AL, de Astarloa JD (2016) Relationship between ecomorphology and trophic segregation in four closely related sympatric fish species (Teleostei, Sciaenidae). C R Biol 339:498–506
Boettiger C, Lang DT, Wainwright PC (2012) rfishbase: exploring, manipulating and visualizing fishbase data from R. J Fish Biol 81:2030–2039
Breder CM (1926) The locomotion of fishes. Zoologica 4:159–297
Brönmark C, Miner JG (1992) Predator-induced phenotypical change in body morphology in Crucian carp. Science 258:1348–1350
Burnham KP, Anderson DR (2004) Multimodel inference-understanding AIC and BIC in model selection. Soc Methods Res 33:261–304
Casatti L, Castro RMC (2006) Testing the ecomorphological hypothesis in a headwater riffles fish assemblage of the rio São Francisco, southeastern Brazil. Neotrop Ichthyol 4:203–214
Choat JH, Bellwood DR (1991) Reef fishes: their history and evolution. In: Sale PF (ed) The ecology of fishes on coral reefs. Academic Press, San Diego, pp 39–66
Clabaut C, Bunje PME, Salzburger W, Meyer A (2007) Geometric morphometric analyses provide evidence for the adaptive character of the Tanganyikan cichlid fish radiations. Evolution 61:560–578
Claverie T, Wainwright PC (2014) A morphospace for reef fishes: elongation is the dominant axis of body shape evolution. PLoS One 9:e112732
Colgate JE, Lynch KM (2004) Mechanics and control of swimming: a review. IEEE J Ocean Eng 29:660–673
Connell JH (1978) Diversity in tropical rain forests and coral reefs-high diversity of trees and corals is maintained only in a non-equilibrium state. Science 199:1302–1310
Darroch JN, Mosimann JE (1985) Canonical and principal components of shape. Biometrika 72:241–252
Davis WP, Birdsong RS (1973) Coral reef fishes which forage in the water column-a review of their morphology, behavior, ecology and evolutionary implications. Helgol Wiss Meer 24:292–306
de Assumpção L, Makrakis MC, Makrakis S, Wagner RL, da Silva PS, de Lima AF, Kashiwaqui EAL (2012) The use of morphometric analysis to predict the swimming efficiency of two Neotropical long-distance migratory species in fish passage. Neotrop Ichthyol 10:797–804
de Queiroz AC, Vallinoto M, Sakai Y, Giarrizzo T, Barros B (2018) Scaling mimesis: morphometric and ecomorphological similarities in three sympatric plant-mimetic fish of the family Carangidae (Teleostei). PLoS One 13:e0194437
Domenici P (2010) Escape responses in fish: kinematics, performance and behavior. In: Domenici P, Kapoor BG (eds) Fish locomotion: an eco-ethological perspective. Science Publishers, Enfield, pp 123–170
Domenici P, Turesson H, Brodersen J, Brönmark C (2008) Predator-induced morphology enhances escape locomotion in Crucian carp. Proc R Soc B-Biol Sci 275:195–201
Ehlinger TJ, Wilson DS (1988) Complex foraging polymorphism in bluegill sunfish. Proc Natl Acad Sci USA 85:1878–1882
Emery AR (1978) The basis of fish community structure: marine and freshwater comparisons. Environ Biol Fishes 3:33–47
Evans KM, Williams KL, Westneat MW (2019) Do coral reefs promote morphological diversification? Exploration of habitat effects on labrid pharyngeal jaw evolution in the era of big data. Integr Comp Biol 59:696–704
Farré M, Lombarte A, Recasens L, Maynou F, Tuset VM (2015) Habitat influence in the morphological diversity of coastal fish assemblages. J Sea Res 99:107–117
Ferreira CEL, Gonçalves JEA, Coutinho R (2001) Community structure of fishes and habitat complexity on a tropical rocky shore. Environ Biol Fishes 61:353–369
Fish FE (2002) Balancing requirements for stability and maneuverability in cetaceans. Integr Comp Biol 42:85–93
Fisher R, Hogan JD (2007) Morphological predictors of swimming speed: a case study of pre-settlement juvenile coral reef fishes. J Exp Biol 210:2436–2443
Freckleton RP, Harvey PH, Pagel M (2002) Phylogenetic analysis and comparative data: a test and review of evidence. Am Nat 160:712–726
Frédérich B, Marramà G, Carnevale G, Santini F (2016) Non-reef environments impact the diversification of extant jacks, remoras and allies (Carangoidei, Percomorpha). Proc R Soc B-Biol Sci 283:20161556
Freitas CEC, Costa EL, Soares MGM (2005) Ecomorphological correlates of thirteen dominant fish species of Amazonian floodplain lakes. Acta Limnol Bras 17:339–347
Friedman ST, Martinez CM, Price SA, Wainwright PC (2019) The influence of size on body shape diversification across Indo-Pacific shore fishes. Evolution 73:1873–1884
Froese R, Pauly D (2020) Fishbase. www.fishbase.org
Fulton CJ, Bellwood DR, Wainwright PC (2001) The relationship between swimming ability and habitat use in wrasses (Labridae). Mar Biol 139:25–33
Garland T, Dickerman AW, Janis CM, Jones JA (1993) Phylogenetic analysis of covariance by computer simulation. Syst Biol 42:265–292
Gatz AJ (1979) Ecological morphology of fresh water stream fishes. Tulane Stud Zool Bot 21:91–124
Gerstner CL (1999) Maneuverability of four species of coral-reef fish that differ in body and pectoral-fin morphology. Can J Zool-Rev Can Zool 77:1102–1110
Gratwicke B, Speight MR (2005a) The relationship between fish species richness, abundance and habitat complexity in a range of shallow tropical marine habitats. J Fish Biol 66:650–667
Gratwicke B, Speight MR (2005b) Effects of habitat complexity on Caribbean marine fish assemblages. Mar Ecol-Prog Ser 292:301–310
Hansen TF (1997) Stabilizing selection and the comparative analysis of adaptation. Evolution 51:1341–1351
Hansen TF, Pienaar J, Orzack SH (2008) A comparative method for studying adaptation to a randomly evolving environment. Evolution 62:1965–1977
Hatcher BG (1988) Coral-reef primary productivity: a beggar’s banquet. Trends Ecol Evol 3:106–111
Helfman G, Collette BB, Facey DE, Bowen BW (2009) The diversity of fishes: biology, evolution, and ecology, 2nd edn. Wiley-Blackwell, Oxford
Hixon MA, Beets JP (1993) Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol Monogr 63:77–101
Hurvich CM, Tsai CL (1989) Regression and time-series model selection in small samples. Biometrika 76:297–307
Keast A, Webb D (1966) Mouth and body form relative to feeding ecology in the fish fauna of a small lake, Lake Opinicon, Ontario. Fish Res Board Can J 23:1845–1874
Kerfoot JR, Schaefer JF (2006) Ecomorphology and habitat utilization of Cottus species. Environ Biol Fishes 76:1–13
Kohn AJ, Helfrich P (1957) Primary organic productivity of a Hawaiian coral reef. Limnol Oceanogr 2:241–251
Kopperud BT, Pienaar J, Voje KL, Orzack SH, Hansen TF (2020) SLOUCH: stochastic linear Ornstein-Uhlenbeck comparative hypotheses. Version 2.1.4. https://CRAN.R-project.org/package=slouch
Langerhans RB (2009) Trade-off between steady and unsteady swimming underlies predator-driven divergence in Gambusia affinis. J Evol Biol 22:1057–1075
Langerhans RB, Reznick DN (2010) Ecology and evolution of swimming performance in fishes: predicting evolution with biomechanics. In: Domenici P, Kapoor BG (eds) Fish locomotion: an eco-ethological perspective. Science Publishers, Enfield, pp 200–248
Langerhans RB, Layman CA, Langerhans AK, Dewitt TJ (2003) Habitat-associated morphological divergence in two Neotropical fish species. Biol J Linnean Soc 80:689–698
Langerhans RB, Layman CA, Shokrollahi AM, DeWitt TJ (2004) Predator-driven phenotypic diversification in Gambusia affinis. Evolution 58:2305–2318
Lavin PA, McPhail JD (1985) The evolution of fresh-water diversity in the threespine stickleback (Gasterosteus aculeatus) - Site-specific differentiation of trophic morphology. Can J Zool-Rev Can Zool 63:2632–2638
Leal CG, Junqueira NT, Pompeu PS (2011) Morphology and habitat use by fishes of the Rio das Velhas basin in southeastern Brazil. Environ Biol Fishes 90:143–157
Lingo ME, Szedlmayer ST (2006) The influence of habitat complexity on reef fish communities in the northeastern Gulf of Mexico. Environ Biol Fishes 76:71–80
Luckhurst BE, Luckhurst K (1978) Analysis of influence of substrate variables on coral-reef fish communities. Mar Biol 49:317–323
Marroig G, Cheverud JM (2005) Size as a line of least evolutionary resistance: diet and adaptive morphological radiation in new world monkeys. Evolution 59:1128–1142
McGuigan K, Franklin CE, Moritz C, Blows MW (2003) Adaptation of rainbow fish to lake and stream habitats. Evolution 57:104–118
Monismith SG (2007) Hydrodynamics of coral reefs. Annu Rev Fluid Mech 39:37–55
Mosimann JE (1970) Size allometry: size and shape variables with characterizations of the lognormal and generalized gamma distributions. J Am Stat Assoc 65:930–945
Mosimann JE, James FC (1979) New statistical methods for allometry with application to Florida red-winged blackbirds. Evolution 33:444–459
Moyle PB, Cech JJ (2004) Fishes: an introduction to ichthyology, 5th edn. Pearson Benjamin Cummings, San Francisco
Odum HT, Odum EP (1955) Trophic structure and productivity of a windward coral reef community on Eniwetok Atoll. Ecol Monogr 25:291–320
Ohlberger J, Staaks G, Hölker F (2006) Swimming efficiency and the influence of morphology on swimming costs in fishes. J Comp Physiol B 176:17–25
Oliveira EF, Goulart E, Breda L, Minte-Vera CV, Paiva LRD, Vismara MR (2010) Ecomorphological patterns of the fish assemblage in a tropical floodplain: effects of trophic, spatial and phylogenetic structures. Neotrop Ichthyol 8:569–586
Orme D, Freckleton RP, Thomas G, Petzoldt T, Fritz S, Isaac N, Pearse W (2018) caper: comparative analysis of phylogenetics and evolution in R. Version 1.0.1. https://CRAN.R-project.org/package=caper
Pennell MW, Eastman JM, Slater GJ, Brown JW, Uyeda JC, FitzJohn RG, Alfaro ME, Harmon LJ (2014) geiger v2.0: An expanded suite of methods for fitting macroevolutionary models to phylogenetic trees. Bioinformatics 30:2216–2218
Pfaff C, Zorzin R, Kriwet J (2016) Evolution of the locomotory system in eels (Teleostei: Elopomorpha). BMC Evol Biol 16:159
Prado AVR, Goulart E, Pagotto JPA (2016) Ecomorphology and use of food resources: inter- and intraspecific relationships of fish fauna associated with macrophyte stands. Neotrop Ichthyol 14:e150140
Price SA, Friedman ST, Wainwright PC (2015) How predation shaped fish: the impact of fin spines on body form evolution across teleosts. Proc R Soc B-Biol Sci 282:20151428
Price SA, Holzman R, Near TJ, Wainwright PC (2011) Coral reefs promote the evolution of morphological diversity and ecological novelty in labrid fishes. Ecol Lett 14:462–469
Price SA, Tavera JJ, Near TJ, Wainwright PC (2013) Elevated rates of of morphological and functional diversification in reef-dwelling haemulid fishes. Evolution 67:417–428
Price SA, Friedman ST, Corn KA, Martinez CM, Larouche O, Wainwright PC (2019) Building a body shape morphospace of teleostean fishes. Integr Comp Biol 59:716–730
Rabosky DL, Chang J, Title PO, Cowman PF, Sallan L, Friedman M, Kaschner K, Garilao C, Near TJ, Coll M, Alfaro ME (2018) An inverse latitudinal gradient in speciation rate for marine fishes. Nature 559:392–398
Risk MJ (1972) Fish diversity on a coral reef in the Virgin Islands. Atoll Res Bull 153:1–4
Roberts CM, Ormond RFG (1987) Habitat complexity and coral-reef fish diversity and abundance on Red-Sea fringing reefs. Mar Ecol-Prog Ser 41:1–8
Roberts CM, McClean CJ, Veron JEN, Hawkins JP, Allen GR, McAllister DE, Mittermeier CG, Schueler FW, Spalding M, Wells F, Vynne C, Werner TB (2002) Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295:1280–1284
Sale PF (1991) Introduction. In: Sale PF (ed) The ecology of fishes on coral reefs. Academic Press, San Diego, pp 3–15
Sargent MC, Austin TS (1949) Organic productivity of an atoll. Trans Am Geophys Union 30:245–249
Sargent MC, Austin TS (1954) Bikini and nearby Atolls. Part 2. Oceanography (Biologic). Biologic economy of coral reefs. US Geological survey professional paper 260-E:293–300
Schrank AJ, Webb PW, Mayberry S (1999) How do body and paired-fin positions affect the ability of three teleost fishes to maneuver around bends? Can J Zool-Rev Can Zool 77:203–210
Sfakiotakis M, Lane DM, Davies JBC (1999) Review of fish swimming modes for aquatic locomotion. IEEE J Ocean Eng 24:237–252
Spalding MD, Grenfell AM (1997) New estimates of global and regional coral reef areas. Coral Reefs 16:225–230
Spalding MD, Brown BE (2015) Warm-water coral reefs and climate change. Science 350:769–771
Spalding MD, Ravilious C, Green EP (2001) World atlas of coral reefs. University of California Press, Berkeley
Steneck RS, Graham MH, Bourque BJ, Corbett D, Erlandson JM, Estes JA, Tegner MJ (2002) Kelp forest ecosystems: biodiversity, stability, resilience and future. Environ Conserv 29:436–459
Talbot FH (1965) A description of the coral structure of Tutia Reef (Tanganyika territory, East Africa), and its fish fauna. Proc Zool Soc Lond 145:431–470
Walker JA (1997) Ecological morphology of lacustrine threespine stickleback Gasterosteus aculeatus L. (Gasterosteidae) body shape. Biol J Linnean Soc 61:3–50
Walker JA (2000) Does a rigid body limit meneuverability? J Exp Biol 203:3391–3396
Walker JA, Alfaro ME, Noble MM, Fulton CJ (2013) Body fineness ratio as a predictor of maximum prolonged-swimming speed in coral reef fishes. PLoS One 8:e75422
Watson DJ, Balon EK (1984) Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo. J Fish Biol 25:371–384
Webb PW (1975) Hydrodynamics and energetics of fish propulsion. B Fish Res Board Can 190:1–158
Webb PW (1982) Locomotor patterns in the evolution of actinopterygian fishes. Am Zool 22:329–342
Webb PW (1983) Speed, acceleration and manoeuvrability of two teleost species. J Exp Biol 102:115–122
Webb PW (1984) Body form, locomotion and foraging in aquatic vertebrates. Am Zool 24:107–120
Webb PW (1994) The biology of fish swimming. In: Maddock L, Bone Q, Rayner JMV (eds) Mechanics and physiology of animal swimming. Cambridge University Press, Cambridge, pp 45–62
Webb PW, Weihs D (1986) Functional locomotor morphology of early life-history stages of fishes. Trans Am Fish Soc 115:115–127
Webb PW, Fairchild AG (2001) Performance and maneuverability of three species of teleostean fishes. Can J Zool-Rev Can Zool 79:1866–1877
Webb PW, LaLiberte GD, Schrank AJ (1996) Does body and fin form affect the maneuverability of fish traversing vertical and horizontal slits? Environ Biol Fishes 46:7–14
Weihs D (1989) Design-features and mechanics of axial locomotion in fish. Am Zool 29:151–160
Willis SC, Winemiller KO, Lopez-Fernandez H (2005) Habitat structural complexity and morphological diversity of fish assemblages in a Neotropical floodplain river. Oecologia 142:284–295
Zelditch ML, Swiderski DL, Sheets HD (2012) Geometric morphometrics for biologists: a primer, 2nd edn. Elsevier Academic Press, London
Zimmerman MS, Krueger CC, Eshenroder RL (2006) Phenotypic diversity of lake trout in Great Slave Lake: differences in morphology, buoyancy, and habitat depth. Trans Am Fish Soc 135:1056–1067
Acknowledgements
Support for the research was provided by National Science Foundation grant DEB-1830127 to SAP & PCW, with additional support by Clemson University’s Creative Inquiry program for undergraduate research. We wish to thank Nicholas Hix, Carley McGlinn, Hannah Wells and Lucas McCutcheon for their participation in developing the research question and generating preliminary results. We are also grateful to the curators and staff of the Smithsonian National Museum of Natural History Division of Fishes for their support during the three summers of data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Topic Editor Andrew Hoey
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Larouche, O., Benton, B., Corn, K.A. et al. Reef-associated fishes have more maneuverable body shapes at a macroevolutionary scale. Coral Reefs 39, 1427–1439 (2020). https://doi.org/10.1007/s00338-020-01976-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-020-01976-w