Abstract
Reproduction and recruitment are essential processes for the continued success of coral communities. Islas Marias Archipelago is considered a connectivity node among coral communities distributed along the Northeastern Tropical Pacific (NTP); as such, sexual reproduction of scleractinian corals affects the maintenance of the local populations and the long-distance connectivity of reefs across the region. In this study, successful sexual reproduction in the species of the three most abundant scleractinian corals genera was demonstrated, in part by documenting gamete presence and maturation in tissues and spatio-temporal variability in juvenile coral settlement, which were quantified across substrate type, habitat quality, and environmental factors. Only 12 larvae recruited (ten Porites, two Pavona) to artificial substrates, and monthly recruitment density of 1.82 ± 0.23 recruits m−2 (n = 383) was recorded on natural substrates. There were significant differences between genera, with Porites producing the highest density of recruits (0.60 ± 0.45 recruits m−2, followed by Pavona (0.52 ± 0.24 recruits m−2) and Pocillopora (0.28 ± 0.06 recruits m−2). The highest coral recruitment was observed at Baby Reef (2.57 ± 0.46 recruits m−2), followed by Cleofas II (1.81 ± 0.21 recruits m−2) and Japanese Garden (1.24 ± 0.46 recruits m−2); the former site was characterized by the highest cover of crustose coralline algae. We found successful recruitment of the main reef-forming species in the Mexican Pacific and suggest that Islas Marias is a region of critical importance in terms of its function as a source of genetic variability, and, the generation of, new individuals that will be key to sustaining coral reef ecosystems in the NTP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reproduction (sexual and asexual) is critical for the maintenance of coral reef ecosystems, as well as their repopulation and recovery following disturbance (Harrison 2011; Glynn et al. 2017a, b). Sexual reproduction involves gamete production, fertilization, the transport, settlement, and survival of the resulting larvae (Fadlallah 1983; Harrison and Wallace 1990; Harrison 2011; Glynn et al. 2017a, b). Although corals can reproduce asexually (Fautin 2003; Baums et al. 2006), recruitment resulting from sexual reproduction can modulate marine population dynamics (Gaines and Roughgarden 1985; Doherty and Fowler 1994; Palma et al. 1999).
Sexual recruitment can be classified as self-recruitment when the larvae settle near the natal population contributing and favoring the maintenance of the natal population. In contrast, subsidiary recruitment refers to larval recruitment at a distinct enough location to such that they are effectively a different population (Harrison and Wallace 1990; Black et al. 1991; Cowen 2002); this strengthens genetic links between regions and enhances connectivity. It is important to notice that both types of recruitment promote genetic diversity, which favors adaptation to environmental change (Fadlallah 1983; Harrison and Wallace 1990; Harrison 2011). Biophysical models tend to favor local recruitment (Steneck 2006, Wood et al. 2014; Lequeux et al. 2018) because of a combination of oceanographic conditions, larval behavior, and larvae high mortality rates associated with planktonic conditions (Cowen et al. 2000; Strathmann et al. 2002; Levin 2006). However, coral larvae under favorable conditions (e.g., low predation pressure and high water flow) have the potential (energy reserves) for long-distance dispersal (Fabricius and Metzner 2004). Seasonal cycles of sexual reproduction and recruitment depend not only on environmental factors such as sea surface temperature (SST), light, and nutrients (Harrison and Wallace 1990; Mendes and Woodley 2002; Carpizo-Ituarte et al. 2011; Rodríguez-Troncoso et al. 2011; Santiago-Valentín et al. 2018), but also biotic conditions, namely larval (1) availability (which represents a culmination of gamete production and fertilization), (2) dispersal capacity, and (3) settlement cues (Pawlik 1992; Hadfield and Paul 2001); the latter are chemicals that signal to larvae that suitable substrate is nearby and can also induce metamorphosis. Conspecific corals, crustose coralline algae (Morse and Morse 1991; Morse et al. 1994; Heyward and Negri 1999), and even rubble (Negri et al. 2001; Golbuu and Richmond 2007) have been known to have associated biofilms that emit settlement cues, whereas turf algae, macroalgae, and cyanobacteria generally inhibit larval settlement (Kuffner and Paul 2004; Birrell et al. 2005, 2008; Kuffner et al. 2006; Doropoulos et al. 2016, 2017).
The Tropical Eastern Pacific (TEP) is characterized by marginal environmental conditions that limit the development of coral reefs (Glynn 1976; Cortés 1997; Glynn and Ault 2000). In this region, coral communities have experienced mass coral bleaching and reduction of live coral cover by 50 to 90% since the 1980s (Glynn and D’Croz 1990; Reyes-Bonilla et al. 2002). These losses are often attributed to anomalous temperatures, such as those brought upon by El Niño–Southern Oscillation (ENSO) events (Glynn and Ault 2000), but anthropogenic disturbance at local scales has clearly shaped these ecosystems as well (López-Pérez et al. 2012). The Islas Marias Biosphere Reserve, which is located in the Northeastern Mexican Pacific (NMP), represents an important source of larval for the entire west coast of Mexico and into the Gulf of California (Glynn et al. 1996, 2011; Reyes-Bonilla and López-Pérez 1998; Pérez-Vivar et al. 2006; López-Pérez et al. 2015). Thus, sexual reproduction of scleractinian corals at this site is essential for the maintenance of not only the local population, but also other reefs of the NMP. It is unfortunate, then, that prior studies have not documented adult coral reproduction in this region. Herein, we provide evidence of successful sexual reproduction in species of the three most common genera of scleractinian corals (Porites, Pocillopora, and Pavona) by tracking gametogenesis over space and time, as well as by documenting recruitment to both natural and artificial substrates across a variety of temperature and light regimes at Islas Marias.
Methods
Study area
Islas Marias Biosphere Reserve (Fig. 1) is an archipelago located on the western coast of Mexico that is characterized by a dry season from November to April and a rainy season from May to October (Bullock 1986). Northwesterly winds prevail most of the year, and seasonal upwelling occurs in the Central Mexican Pacific (CMP) from December to April coinciding with the dry season (Fiedler and Talley 2006; Palacios-Hernández et al. 2010). The islands are located in an oceanographic transitional zone with highly variable, mixed-water conditions resulting from the convergence of three ocean currents: the California current, the Mexican coastal current, and the Gulf of California current (Fig. 1A); promoting a highly variable annual temperature range from 18 to 35 °C (Wyrtki 1965; Pennington et al. 2006; Kessler 2006; Palacios-Hernández et al. 2010; Pantoja et al. 2011). This includes inter-annual thermal anomalies driven by ENSO events that have a detrimental effect on coral communities (Glynn et al. 2000; Glynn 2001; Reyes-Bonilla et al. 2002).
Study area. A General ocean circulation pattern of the Central Mexican Pacific and location of the Islas Marias Archipelago. B Sample site location around Isla Maria Cleofas. CL Cleofas II, JG Japanese Garden, BR Baby Reef, GCC Gulf of California current, CCM Mexican coastal current, CC California current, 1—San Juanito, 2—Maria Madre, 3—Maria Magdalena, 4—Maria Cleofas
The archipelago is composed of four islands—Maria Madre, Maria Magdalena, Maria Cleofas, and San Juanito (Fig. 1); 16 scleractinian coral species can be found there (39% of the total coral species inhabiting the Mexican Pacific; López-Pérez et al. 2015). The four most abundant genera are Pocillopora (31.5% cover across all five species), Porites (27% cover across two species), Pavona (5% cover across four species), and Psammocora (5% cover across two species; Reyes-Bonilla 2003; López-Pérez et al. 2015). Maria Cleofas (Fig. 1B) is the closest to the coast (~ 100 km from the state of Nayarit, Mexico) and is characterized by the highest live coral cover (38.5%; López-Pérez et al. 2016).
Gamete development
Histological studies were conducted to confirm the presence of female and male gametes; for this purpose, we collected five fragments from each coral species Porites panamensis, Porites lobata, Pavona gigantea, Pavona clavus and the three most abundant of species of pocilloporid (Pocillopora cf. eydouxi, Pocillopora cf. effusus, and Pocillopora cf. verrucosa; Schmidt-Roach et al. 2014) distributed in a depth range between 6 and 12 m, in both June and July of 2016 coinciding with the high temperature and light levels documented during these months. These have been hypothesized to trigger gametogenesis by others working in this region (Glynn et al. 1991, 1994, 1996; Carpizo-Ituarte et al. 2011; Rodríguez-Troncoso et al. 2011; Santiago-Valentín et al. 2018). Each of the fragments was fixed in formalin and decalcified in a solution of 10% acetic acid for 24 h. Coral tissues were dehydrated in a graded ethanol series, cleared in xylene, and embedded in Paraplast® using a Leica EG1160 tissue embedding system following the protocols of Santiago-Valentín et al. (2015). The samples were cut to 6 µm thickness with a Leica RM2125RT semiautomatic rotary microtome, stained using the Masson trichrome technique (Lynch et al. 1972), and finally mounted with synthetic resin onto glass slides. All samples were examined and photo-documented using a Carl Zeiss AxioScope® optical microscope. The presence of gametes and the reproductive stages in the coral tissues were determined according to the criteria proposed by Glynn et al. (1994), Carpizo-Ituarte et al. (2011), and Rodríguez-Troncoso et al. (2011).
Coral recruitment
Larval recruitment was evaluated on both artificial and natural substrates. The former consisted of ten terracotta settlement tiles (25 × 28 × 2 cm; 0.856 cm2 each), which were secured with cable ties to a steel bar embedded in the substrate near healthy colonies within the reef, with a distance of approximately three meters between each one, in a range depth of 7–9 m at both Cleofas II and Japanese Garden. The tiles were immersed for 15 months (June 2016–August 2017) and monitored in situ using a UV-FL-1 Dive Light (Nightsea®) torch with a yellow filter to detect the presence of recruits, which were identified in situ at genera level; recruit size was measured using a plastic gauge (Foy®) with an accuracy of 0.01 mm.
For the evaluation of recruitment on natural substrates, five quadrants (1 m2) were spaced at 5-m intervals on three 25 m belt transects run parallel to the coast. In each quadrant, taxonomic classification, and assessment, was made of the presence and size of the recruits as described above in June 2016, July 2016, and April, May, and August 2017 (n = 5 survey times) at Baby Reef, Cleofas II, and Japanese Garden (Fig. 1B). Seawater temperature (SWT) was measured in situ every 25 min during the study period using HOBO® Pendant temperature loggers placed near the sample colonies. Day length was calculated as the time difference (in h) between sunrise and sunset as described by Stull (2000).
The variation in recruitment density (recruits m−2) between genera (Porites, Pavona, and Pocillopora), site (Cleofas II, Japanese Garden, and Baby Reef), and month (see above.) was tested with permutational analysis of variance (PERMANOVA) since the data did not meet the assumptions of parametric statistical analyses. A Euclidean distance matrix of the recruitment density data was constructed. A type I, fixed-factor, sum of squares (type III) model was used, and 10,000 permutations were generated using PRIMER ver. 6.1.11 + PERMANOVA ver. 1.0.1 (Anderson et al. 2008; Clarke and Gorley 2006). Pairwise tests were performed when overall model factors were significant (p < 0.05). A multiple linear regression analysis was performed to examine the relationship between recruit density and temperature and day length with SigmaPlot ver. 1.1.
Benthic characterization
The benthos was characterized by evaluating six evenly distributed 1 m2 quadrants at each of five transects (30 m2 per site) at the following sites: Baby Reef (from 5 to 12 m deep), Japanese Garden (5–15 m deep), and Cleofas II (3–6 m deep) allowing the characterization of different reef conditions along the study area. The percent cover data were square-root transformed, and a Bray–Curtis similarity matrix was constructed. To test for differences between sites, a one-way analysis of similarity (ANOSIM) was conducted with PRIMER ver. 6.1.11. Linear regression analysis was performed with SigmaPlot to examine the relationship between recruit density and the cover of the other benthic groups, though coral and rubble cover were excluded since their variance inflation factors were high; they also tended to be multicollinear.
Molecular systematics
Due to the difficulties in taxonomically classifying recruits, species-level genotyping assays were carried out in 13 recruits randomly collected during sampling (five in Cleofas II and eight in Baby Reef). Genomic DNA from adults (n = 2 fragments per coral species) and recruits was extracted using the Wizard® SV genomic DNA purification system (Promega). Partial sequences of the internal transcribed spacer ITS (ITS1-5.8-ITS2) and cytochrome c oxidase subunit 1 gene (cox1) were amplified with PCR using the following primers: ITS4 (5′-TCCTCCGCTTATTGATATGC-3′) and ITS5 (3′-GGAAGTAAAAGTCGTAACAAGG-5′; Cruz-Barraza et al. 2012; ~ 600 bp) and the degenerate primers LCOI490 (5′-GGTCAACAAATCATAAAGAYATYGG-3′) and HCOI21908 (5′-TAAACTTCAGGGTGACCAAARAAYCA-3′; Folmer et al. 1994; ~ 600 bp). Please see Santiago-Valentín et al. (2019) for PCR details. PCR products were visualized on Tris–acetate–EDTA–agarose (2%) gels, purified using the Wizard SV gel and PCR clean-up system (Promega) and sequenced at Macrogen Inc® (Seoul, Korea). Sequences were manually edited to obtain a consensus sequence using Geneious (ver.4.8.5) and then analyzed using BLAST. Each gene sequence was submitted to the National Center for Biotechnology Information (NCBI): cox1 (MN005652–MN005661) and ITS (MK946633–MK946642). In order to determine the relationship among samples (adults and recruits), a maximum likelihood (ML) tree of K2P distances was created (Kimura 1980) with MEGA7. To corroborate the taxonomic identity of the samples using the cox1 gene, sequences were downloaded from the GenBank, according to the access numbers: P. panamensis NC024182; P. lobata LT558153; P. clavus DQ643836; and P. damicornis LC331996). As outgroups to root the tree, Gorgonia flabellum (GQ342418.1) and Dendronephthya gigantea (AF320104.1) were used for the cox1 and ITS trees, respectively.
Results
Gamete development
Gametogenic development was observed in coral species sampled with the same patterns in species of the same genera. (Full detail of reproductive activity by species is included in Table 1). Immature Porites oocytes (stages I, II, and III) were detected in June while mature oocytes (stage IV) were observed in July; spermiaries in stages III and IV were observed in June and July. Pavona colonies showed stages II–III oocytes and stages III–IV spermiaries were observed in July. Immature morphotypes Pocillopora spp. oocytes (stages I–II) were detected in June and July (Table 1). The species with the highest percentage of reproductive activity (gamete presence) were P. panamensis (72%), followed by P. clavus (50%) and Pocillopora cf. verrucosa (30%).
Recruitment
On the artificial substrates, ten Porites and two Pavona recruits were recorded, and the highest number of recruits was observed in April 2017 (5 Porites + 1 Pavona), 11 months after tile installation. The recruits varied in size: Porites recruits ranged from 2 to 7 mm, while Pavona recruits averaged only 0.4 mm. A mortality rate of 42% recruits was reported in May and August of 2017. Cleofas II had the highest recruitment (eight Porites and two Pavona), while only two Porites recruits were recorded at Japanese Garden.
On natural substrata, a total of 383 coral recruits throughout the period of study were recorded, with a mean monthly density of 1.82 ± 0.23 recruits m−2 (± SD for this and all following recruit data). Three coral genera were identified among recruits Porites, Pavona, and Pocillopora (Fig. 2), and recruitment density varied across genera (Table 2). Porites was most dominant (0.60 ± 0.45 recruits m−2), followed by Pavona (0.52 ± 0.24) and Pocillopora (0.28 ± 0.06; Table 1 and Fig. 3). Recruitment density also differed significantly across the three sites (Table 2); highest recruitment was observed at Baby Reef (2.57 ± 0.46 recruits m2), followed by Cleofas II (1.81 ± 0.21) and Japanese Garden (1.24 ± 0.46; Fig. 3 and Table 2). No significant differences in recruit density were detected over time (Table 2). However, significant differences within genera over time were observed (Table 2); highest Porites recruitment was documented in June (1.40 ± 0.05 recruits m−2), while for Pavona it was in August (0.87 ± 0.2 recruits m−2). Although Pocillopora showed less variation, the highest density of recruits was found in July (0.36 ± 0.03 recruits m−2). The results show that the peak of recruits at Cleofas Island was recorded during the period with the highest value of daylight hours (13.24 h) and SWT (27.67 °C). However, the monthly recruitment data did not correlate with SWT or day length (R = 0.5659, N = 5, p = 0.68; Fig. 4).
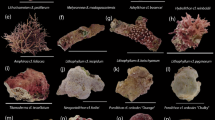
Coral genera studied at Isla Maria Cleofas. Adult colonies (scale bars = 20 cm) of Porites (A), Pavona (B), and Pocillopora (C). Recruits of Porites (D) and Pavona (E) attached to coral rubble colonized by crustose coralline algae and a Pocillopora recruit (F) attached to rocks colonized by crustose coralline algae (scale bars = 1 cm). Recruits of Porites (G), Pavona (H), and Pocillopora (I) under ultraviolet fluorescence (scale bars = 0.5 mm)
Benthic characterization
A total of 13 species were recorded across the genera Pocillopora (seven species), Porites (two), Pavona (three), and Psammocora (one), with an overall coral cover of 11.56%. The genus that contributed most to cover was Pocillopora (8.52%), followed by Pavona (2.18%) and Porites (1.05%; see the contribution to coverage by species in Table 3). The benthic composition differed across sites (ANOSIM; R = 0.185, p = 0.01). Pocillopora was most abundant at Baby Reef (12.22%); its cover at Cleofas and Japanese Garden was very similar (7.15 and 7.56%, respectively). The relative dominance of the various pocilloporid species differed across sites; at Cleofas II, Pocillopora verrucosa was most abundant (4.61%), whereas at Japanese Garden and Baby Reef Pocillopora eydouxi (2.33%) and Pocillopora effusus (6.89%), respectively, were most commonly observed. The genus Pavona was most abundant at Japanese Garden (5.22%), and P. clavus (3.83%) was the most dominant pavonid species at all three sites (See Table 3). Of the three target genera, Porites has the lowest coverage across the three sites, though its cover differed significantly across sites; this was driven by a difference between Baby Reef and Cleofas II (0.97 and 0.90%, respectively) over Japanese Garden (0.58%). P. panamensis (0.39%) was the most commonly documented poritid at Japanese Garden and Baby Reef, while P. lobata (0.57%) dominated Cleofas II; in contrast, P. lobata was not present at Baby Reef (Table 3).
There was a positive correlation between the density of recruits and the benthic groups (R = 0.49, n = 18, p = 0.27). Meanwhile, recruit density correlated negatively with sediment (R = − 0.09; p = 0.027), rubble (R = − 0.07; p = 0.03), and the “other” group, which featured organisms such as barnacles, octocorals, and encrusting organisms (R = − 0.23; p = 0.019). Coral recruit density was not affected by macroalgae (R = − 0.009; p = 0.601) or turf algae (R = − 0.046; p = 0.133) cover. There was a positive correlation between cover of crustose coralline algae and coral recruitment (R = 0.057; p = 0.019); Baby Reef was characterized as the site with both highest larval recruitment and cover of crustose coralline algae (CCA; 26.8%), and the low presence of sediments (1.8%). In contrast, the lowest recruitment was recorded at Japanese Garden, which presented the highest sediment coverage compared to the other sites (9.4%; Fig. 4).
Molecular analysis
ML trees of K2P distances (Fig. 5) were reconstructed for two genes, cox1 mtDNA (n = 7 670-bp sequences) and ITS nDNA (n = 5 474-bp sequences), and the recruit sequences were clustered with those of the sampled adults. Sequence comparisons revealed 100% nucleotide similarity between adults (identified morphologically) and recruits for cox1 (Fig. 5A) and ITS (Fig. 5B). The molecular analysis revealed that, independent of the loci used, recruits belonged to five species: P. panamensis, P. lobata, P. gigantea, P. clavus, and Pocillopora damicornis.
Neighbor-joining tree of DNA sequences from adults and recruits of Porites panamensis, Porites lobata, Pavona gigantea, Pavona clavus, and PocilloporadamicornisA Partial fragment of the cytochrome oxidase subunit 1 gene (cox1). B Internal transcribed spacer (ITS). Numbers on internal nodes represent bootstrap values (100 replicates)
Discussion
Successful sexual reproduction in corals depends on the sequential processes of gamete production, fertilization, larval transport, and larval settlement (Harrison 2011; Glynn et al. 2017a, b); the former and latter processes were evaluated here for scleractinian corals of the Northeastern Mexican Pacific (NMP; Islas Marias). The gametogenic period of Central Mexican Pacific (CMP) corals is shorter in duration, compared to other regions of the Tropical Eastern Pacific (TEP; Santiago-Valentín et al. 2018). This observation has been attributed to variation in temperature and day length (Carpizo-Ituarte et al. 2011; Santiago-Valentín et al. 2018). To delimit the sampling time of gametogenic development, previous records from the CMP, which conclude that during April to August and/or temperatures above 26 °C, promote gamete development (see Carpizo-Ituarte et al. 2011). In this study, it was not possible to relate gametogenic development with temperature; however, mature eggs were observed in colonies of Porites, and mature sperm in Pavona and Pocillopora colonies, during June and July which presented temperatures higher than 27 °C. That being said, coral reproduction was not assessed during the cold season when gametic development is absent, and so the relationship between temperature and gametogenesis should be corroborated by studies in which corals are tracked at other points in the year. Regardless, the presence of gametes in advanced stages of maturation is direct evidence of the reproductive ability of scleractinian coral colonies in the Islas Marias region.
Coral settlement has been evaluated on artificial substrates (e.g., plexiglass and terracotta; Richmond 1985; Reyes-Bonilla and Calderón-Aguilera 1994; Medina-Rosas et al. 2005; López-Pérez et al. 2007; Cabral-Tena et al. 2018), as well as natural ones (Smith 1991; Reyes-Bonilla and Calderón-Aguilera 1994; Glynn and Leyte-Morales 1997; Glynn et al. 2000, 2011; Guzmán and Cortés 2007; Glynn et al. 2011), and, as documented herein, recruitment to the former tends to be low (or not at all; Birkeland 1977; Wellington 1982). This has been attributed to the use of inappropriate substrate material (Muñoz et al. 2018), as well as to competition and predation exerted by organisms such as macroalgae, barnacles, and sponges (Sammarco 1982; Richmond 1987).
The density of recruits on natural substrates was the hierarchy of the target genera followed according to their life history traits (Ritson-Williams et al. 2009). Porites panamensis are gonochoric and release sperm into the water column. Sperm is uptaken by females, egg are fertilized internally (Carpizo-Ituarte et al. 2011; Glynn et al. 1994; Rodríguez-Troncoso et al. 2011; Santiago-Valentín et al. 2019), and the larvae are then released in a pre-competence stage. Porites lobata is a gonochoric spawner (Glynn et al. 1994, 2017a, b). Pocillopora synchronously spawn and are hermaphroditic (Carpizo-Ituarte et al. 2011; Chávez-Romo and Reyes-Bonilla 2007; Glynn et al. 1991; Rodríguez-Troncoso et al. 2011), whereas Pavona are sequential hermaphrodites or gonochoric transmission spawners (Carpizo-Ituarte et al. 2011; Glynn et al. 1996; Rodríguez-Troncoso et al. 2011; Santiago-Valentín et al. 2015). Species representing these reproductive modes differ in colony size, gametic cycles, larval competency, dispersal distance, and dinoflagellate symbiont transmission (Richmond and Hunter 1990). Brooders are typically smaller than spawning corals and have multiple planulating cycles per year, as opposed to one or two cycles in broadcast spawners (Szmant 1986).
Also, brooding and spawning corals present differences in their dispersal potential (Baird et al. 2009); as larvae of brooders are released in an advanced stage of development, they tend to settle more quickly (Miller and Mundy 2003) and can recruit at high densities, as was observed herein and elsewhere in the TEP for P. panamensis (Szmant 1986; Glynn et al. 2017a, b). Indeed, Smith (1991) and Glynn et al. (2000) reported even higher values (12.92 ind m−2 yr−1; Table 4) at Isla Uva, Panama, than those reported here (and throughout text). In contrast, the Pavona recruitment rates at Islas Marias were higher than reported elsewhere in the TEP (Table 4). Pocillopora was characterized by the lowest settlement rates at the study sites, and even the rates at the site with the highest pocilloporid recruitment, Baby Reef (1.57 ind m−2 yr−1; Table 4), were lower than those documented at Isla Caño, Costa Rica (1.81 ind m−2 yr−1). Although no significant differences in recruit density were observed over time, peak recruitment months differed for Porites (June), Pavona (August), and Pocillopora (July). These temporal differences may reflect physiological differences (e.g., energy stores, maturation time), and/or external reproduction cues (Carpizo-Ituarte et al. 2011; Rodríguez-Troncoso et al. 2011, 2014; Santiago-Valentín et al. 2018).
Differences in recruitment density were observed across sites, and such differences could be attributed to depth, sedimentation, or any number of other abiotic factors (Harrison 2011; Glynn et al. 2017a, b). Environmental factors can directly influence the physiology of larvae or gametes (Wittenberg and Hunte 1992) or else change their substrate settlement behavior (Gilmour 1999). Herein, inter-site differences appear to be linked to CCA cover, as has been documented previously (Morse et al. 1994; Raimondi and Morse 2000), though it is unclear whether the biofilms present on these algae, or the algae themselves, are responsible for the observed settlement trends (Johnson et al. 1991; Webster et al. 2004). Coral larvae tend to show preference for settlement on CCA with the least potent antifouling defenses (Carleton and Sammarco 1987). Cleofas II had the highest amount of dead coral rubble, which would presumably make it conducive for larval settlement (Heyward and Negri 1999). However, there was in inverse relationship between rubble cover and recruitment herein, possibly due to the generally unconsolidated nature of the rubble and its tendency to be coated in sediments (which would smother coral recruits). Also, larval interactions with the biological inhabitants of reef communities can influence larval survival and settlement, and encrusting organisms (e.g., barnacles, sponges), algal turfs, macroalgae, and sediment can negatively impact the settlement of coral larvae (Kuffner and Paul 2004; Birrell et al. 2005, 2008; Kuffner et al. 2006). The high cover of these non-coral organisms at both Cleofas II and Japanese Garden could explain their low recruitment rate compared to Baby Reef. However, it is also important to characterize the impact of the environmental variables of each site that are promoting a change in the components of the habitat, and how the synergy of abiotic and biotic variables modifies coral recruitment.
In general, the taxonomic identification of scleractinian corals is based on the morphology of adult colonies, as well as their corallites (Veron 2000); however, in the early stages of development (i.e., larvae and recruits), these characteristics are not distinguishable between species (Hillis et al. 1996). Therefore, molecular markers were used herein to identify recruits, and at Isla Maria Cleofas. There was successful sexual recruitment of the five most abundant coral species in the study area: P. panamensis, P. lobata, P. gigantea, P. clavus, and Pocillopora sp; whether or not these recruitment levels are sufficient to maintain the current populations remains to be determined.
The five adult species present at the study sites differed in distribution, as has been documented elsewhere in the TEP (Cortés and Guzmán 1998; Glynn and Ault 2000; Maté 2003; Reyes-Bonilla et al. 2005). P. panamensis is a widely distributed endemic species from the TEP (Glynn 2003) whose reproductive biology has been documented previously (Smith 1991; Carpizo-Ituarte et al. 2011; Glynn et al. 1994; Rodríguez-Troncoso et al. 2011; Santiago-Valentín et al. 2018, 2019), while P. lobata has a more limited distribution in the Mexican Pacific (Carriquiry and Reyes-Bonilla 1997) and Central America (Glynn and Ault 2000). In contrast to these species, successful recruitment of P. gigantea and P. clavus along the Mexican coastline had not previously been reported. Regarding the pocilloporids, they are generally thought to predominantly maintain their population sizes by asexual reproduction; although histological studies have clearly shown gametogenesis (Carpizo-Ituarte et al. 2011; Chávez-Romo and Reyes-Bonilla 2007; Glynn et al. 1991; Rodríguez-Troncoso et al. 2011), the amount of recruitment documented by others in this region was relatively low (Glynn et al. 1994; López-Pérez et al. 2007; Medina-Rosas et al. 2005). Our data show that Islas Marias might be an important bastion of genetic variation for the genus Pocillopora.
The Islas Marias Archipelago is an essential stop for coral larvae migrating between the Gulf of California and the Mexican Pacific (Glynn et al. 1996; Reyes-Bonilla and López-Pérez 1998) via Revillagigedo (Pérez-Vivar et al. 2006), as suggested by biophysical connectivity models (López-Pérez et al. 2016). The current study provides empirical data on the reproductive ability of adult colonies, and, despite having only documented recruitment at local scales, there is the potential for gametes and/or larvae spawned from corals of Islas Marias to travel toward the Gulf of California or other sites along the Mexican Pacific. To date, the connectivity models have been built; using generalized data, however, they provide us with knowledge of a time line that a larva need to connect between populations; as such, a larva from Islas Marias could travel to the North Mexican Pacific and the CMP coastal reefs if is transported during at least 40 days. If the time is extended to 120 days, the larvae can reach insular sites such as Revillagigedo Island and the South Mexican Pacific (López-Pérez et al. 2016; Lequeux et al. 2018). Nevertheless, the lack of data regarding larval competency time generates a gap to accurately model coral population dynamics along the Mexican Pacific. Therefore, in order to build a specific model about connectivity future research should examine the relative contribution of self-recruitment and subsidiary recruitment, as well as their role in the recovery and maintenance of coral populations at Isla Maria Cleofas.
References
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA + for PRIMER: guide to software and statistical methods. UK, PRIMER-E, Plymouth, p 214
Baird AH, Guest JR, Willis BL (2009) Systematic and biogeographical patterns in the reproductive biology of scleractinian corals. Annu Rev Ecol Evol Syst 40:551–571. https://doi.org/10.1146/annurev.ecolsys.110308.120220
Baums IB, Miller MW, Hellberg ME (2006) Geographic Variation in Clonal Structure in a Reef Building Caribbean Coral, Acropora palmata. Ecol Monogr 76:503– 519. https://doi.org/10.1890/0012-9615(2006)076%5b0503:GVICSI%5d2.0.CO;2
Birkeland C (1977) The importance of rate of biomass accumulation in early successional stages of benthic communities to the survival of coral recruits. In: Proceedings of the 3rd International Coral Reef Symposium, Miami 1: 15-21
Birrell CL, McCook LJ, Willis BL, Diaz-Pulido GA (2008) Effects of benthic algae on the replenishment of corals and the implications for the resilience of coral reefs. Oceanogr Mar Biol: An Annual Review 46:25–63
Birrell CL, McCook LJ, Willis BL (2005) Effects of algal turfs and sediment on coral settlement. Mar Pollut Bull 51:408–414. https://doi.org/10.1016/j.marpolbul.2004.10.022
Black KP, Moran PJ, Hammond LS (1991) Numerical models show coral reefs can be self-seeding. Mar Ecol Prog Ser 74:1–11
Bullock SH (1986) Climate of Chamela, Jalisco, and trends in the south coastal region of Mexico. Archives for Meteorology, Geophysics, and Bioclimatology, Series B 36(3–4):297–316. https://doi.org/10.1007/BF02263135
Cabral-Tena RA, Paz-García DA, Reyes-Bonilla H, González-Peláez SS, Balart EF (2018) Spatiotemporal variability in coral (Anthozoa: Scleractinia) larval recruitment in the Southern Gulf of California. Pac Sci 72(4):435–448. https://doi.org/10.2984/72.4.4
Carleton JH, Sammarco PW (1987) Effects of substratum irregularity on success of coral settlement quantification by comparative geomorphological techniques. Bull Mar Sci 40:85–98
Carpizo-Ituarte E, Vizcaíno-Ochoa V, Chi-Barragán G, Tapia-Vázquez O, Cupul-Magaña AL, Medina-Rosas P (2011) Evidence of sexual reproduction in the hermatypic corals Pocillopora damicornis, Porites panamensis, and Pavona gigantea in Banderas Bay, Mexican Pacific. Cienc Mar 37(1): 97-112. https://doi.org/10.7773/cm.v37i1.1773
Carriquiry JD, Reyes-Bonilla H (1997) Community structure and geographic distribution of the coral reefs of Nayarit, Mexican Pacific. Cienc Mar 23(2): 77-96. https://doi.org/10.7773/cm.v23i2.793
Chávez-Romo HE, Reyes-Bonilla H (2007) Reproducción sexual del coral Pocillopora damicornis al sur del Golfo de California, México. Cienc Mar 33(4): 495-501
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. Primer-E Ltd, Plymouth, UK, p 192
Cortés J (1997) Biology and geology of eastern Pacific coral reefs. Coral Reefs 16(1):S39–S46. https://doi.org/10.1007/s003380050240
Cortés J, Guzmán H (1998) Organismos de los arrecifes coralinos de Costa Rica: Descripción, distribución geográfica e historia natural de los corales zooxantelados (Anthozoa: Scleractinia) del Pacífico. Rev Biol Trop 46(1):55–92 (in Spanish)
Cowen RK, Lwiza KM, Sponaugle S, Paris CB, Olson DB (2000) Connectivity of marine populations: open or closed? Science 287(5454):857–859. https://doi.org/10.1126/science.287.5454.857
Cowen RK (2002) Larval Dispersal and Retention and Consequences for population connectivity. In: Sale PF (ed) Coral reef fishes: dynamics and diversity in a complex ecosystem. Academic press, Elsevier Science, p 149
Cruz-Barraza JA, Carballo JL, Rocha-Olivares A, Ehrlich H, Hog M (2012) Integrative taxonomy and molecular phylogeny of genus Aplysina (Demospongiae: Verongida) from Mexican Pacific. PLoS One 7(8):e42049. https://doi.org/10.1371/journal.pone.0042049
Doherty P, Fowler T (1994) An empirical test of recruitment limitation in a coral reef fish. Science 263(5149):935–939. https://doi.org/10.1126/science.263.5149.935
Doropoulos C, Roff G, Bozec YM, Zupan M, Werminghausen J, Mumby PJ (2016) Characterizing the ecological trade-offs throughout the early ontogeny of coral recruitment. Ecol Monogr 86(1):20–44. https://doi.org/10.1890/15-0668.1
Fabricius KE, Metzner J (2004) Scleractinian walls of mouths: Predation on coral larvae by corals. Coral Reefs 23:245–248. https://doi.org/10.1007/s00338-004-0386-x
Fadlallah YH (1983) Sexual reproduction, development and larval biology in scleractinian corals. Coral reefs 2(3):129–150. https://doi.org/10.1007/BF00336720
Fautin DG (2003) Reproduction of Cnidaria. Can J Zool 80:1735–1754. https://doi.org/10.1139/z02-133
Fiedler PC, Talley LD (2006) Hydrography of the eastern tropical Pacific: A review. Prog Oceanogr 69(2–4):143–180. https://doi.org/10.1016/j.pocean.2006.03.008
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299
Gaines S, Roughgarden J (1985) Larval settlement rate: a leading determinant of structure in an ecological community of the marine intertidal zone. Proc. Natl. Acad. Sci. U.S.A. 82:3707–3711. https://doi.org/10.1073/pnas.82.11.3707
Gilmour J (1999) Experimental investigation into the effects of suspended sediment on fertilization, larval survival and settlement in a scleractinian coral. Mar Biol 135:451–462. https://doi.org/10.1007/s002270050645
Glynn PW (1976) Some physical and biological determinants of coral community structure in the eastern Pacific. Ecol Monogr 46(4):431–456. https://doi.org/10.2307/1942565
Glynn PW (2001) Eastern Pacific coral reef ecosystems. In: Seeliger, U., Kjerfve, B. (eds.). Coastal Marine Ecosystems of Latin America, Ecology Study Springer-Verlag Berlin Heidelberg 144: 281-305. https://doi.org/10.1007/978-3-662-04482-7_20
Glynn PW (2003) Coral communities and coral reefs of Ecuador. Latin American coral reefs. Elsevier, Amsterdam 449-472. https://doi.org/10.1016/B978-044451388-5/50020-5
Glynn PW, Ault JS (2000) A biogeographic analysis and review of the far eastern Pacific coral reef region. Coral reefs 19(1):1–23. https://doi.org/10.1007/s003380050220
Glynn PW, Colley SB, Carpizo-Ituarte E, Richmond RH (2017a) Coral reproduction in the eastern Pacific. In: Glynn PW, Manzello DP, Enochs IC (eds) Coral reefs of the eastern tropical Pacific: persistence and loss in a dynamic environment, Coral Reefs of the World 8. Springer, Dordrecht
Glynn PW, Colley SB, Eakin CM, Smith DB, Córtes J, Gassman NJ, Del-Rosario JB, Feingold JS (1994) Reef coral reproduction in the eastern Pacific: Costa Rica, Panamá, and Galápagos Islands (Ecuador). II. Poritidae. Mar Biol 118(2):191–208. https://doi.org/10.1007/BF01313501
Glynn PW, Colley SB, Gassman NJ, Black K, Cortés J, Maté JL (1996) Reef coral reproduction in the eastern Pacific: Costa Rica, Panamá, and Galapagos Islands (Ecuador). III. Agariciidae (Pavona gigantea and Gardineroseris planulata). Mar Biol 125(3): 579-601. https://doi.org/10.1007/BF00353270
Glynn PW, Colley SB, Guzmán HM, Enochs IC, Cortés J, Maté JL, Feingold J (2011) Reef coral reproduction in the eastern Pacific: Costa Rica, Panamá and the Galápagos Islands (Ecuador). VI. Agariciidae. Pavona clavus. Mar Biol 158:1601–1617. https://doi.org/10.1007/s00227-011-1673-z
Glynn PW, Colley SB, Ting JH, Maté JL, Guzman HM (2000) Reef coral reproduction in the eastern Pacific: Costa Rica, Panamá and Galapagos Islands (Ecuador). IV. Agariciidae, recruitment and recovery of Pavona varians and Pavona sp. a. Mar Biol 136(5): 785-805. https://doi.org/10.1007/s002270000286
Glynn PW, D’Croz L (1990) Experimental evidence for high temperature stress as the cause of El Nino-coincident coral mortality. Coral reefs 8(4):181–191. https://doi.org/10.1007/BF00265009
Glynn PW, Gassman NJ, Eakin CM, Córtes J, Smith DB, Guzman HM (1991) Reef coral reproduction in the Eastern Pacific: Costa Rica, Panamá, and Galapagos islands (Ecuador). Mar Biol 109(3):355–368. https://doi.org/10.1007/BF01313501
Glynn PW, Mones AB, Podestá GP, Colbert A, Colgan MW (2017b) El Niño-Southern Oscillation: Effects on Eastern Pacific Coral Reefs and Associated Biota. In: Glynn P, Manzello D, Enochs I (eds) Coral Reefs of the Eastern Tropical Pacific. Springer, Netherlands, pp 251–290. https://doi.org/10.1007/978-94-017-7499-4_8
Glynn PW, Morales-Leyte GE (1997) Coral reefs of Huatulco, West Mexico: reef development in upwelling Gulf of Tehuantepec. Rev Biol Trop 45(3):1033–1047
Golbuu Y, Richmond RH (2007) Substratum preferences in planula larvae of two species of scleractinian corals, Goniastrea retiformis and Stylaraea punctata. Mar Biol 152:639–644. https://doi.org/10.1007/s00227-007-0717-x)
Guzmán HM, Cortés J (2001) Changes in reef community structure after fifteen years of natural disturbances in the eastern Pacific (Costa Rica). Bull Mar Sci 69(1):133–149
Guzmán HM, Cortés J (2007) Reef recovery 20 years after the 1982–1983 El Niño massive mortality. Mar Biol 151:401–411. https://doi.org/10.1007/s00227-006-0495-x
Hadfield MG, Paul VJ (2001) Natural chemical cues for settlement and metamorphosis of marine invertebrate larvae. In: Ecology Boca (ed) McClintock J, Baker B Marine Chemical. CRC Press, Raton, pp 431–462
Harrison PL (2011) Sexual reproduction of scleractinian corals. In: Dubinsky Z, Stambler N (eds.), Coral Reefs: An Ecosystem in Transition. Springer, New York, pp 59–85. https://doi.org/10.1007/978-94-007-0114-4_6
Harrison PL, Wallace CC (1990) Reproduction, dispersal and recruitment of scleractinian corals. In: Dubinsky Z (ed) Coral reefs, Ecosystems of the world; 25. Elsevier, Amsterdam, Netherlands, pp 133–207
Heyward AJ, Negri AP (1999) Natural inducers for coral larval metamorphosis. Coral Reefs 18:273–279. https://doi.org/10.1007/s003380050193
Hillis D, Moritz C, Mable B (1996) Molecular Systematics. Massachusetts, Sinauer Associates, Sunderland, p 655
Johnson CR, Muir CD, Reysenback AL (1991) Characteristic bacteria associated with the surfaces of coralline algae: a hypothesis for bacterial induction of marine invertebrate larvae. Mar Ecol Prog Ser 4:281–294
Kessler WS (2006) The circulation of the Eastern Tropical Pacific: a review. Prog Oceanogr 69:181–217. https://doi.org/10.1016/j.pocean.2006.03.009
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120. https://doi.org/10.1007/BF01731581
Kuffner IB, Paul VJ (2004) Effects of the benthic cyanobacterium Lyngbya majuscula on the larval settlement of the reef corals Acropora surculosa and Pocillopora damicornis. Coral Reefs 23:455–458. https://doi.org/10.1007/s00338-004-0416-8
Kuffner IB, Walters LJ, Becerro MA, Paul VJ, Ritson-Williams R, Beach K (2006) Inhibition of coral recruitment by macroalgae and cyanobacteria. Mar Ecol Prog Ser 323:107–117. https://doi.org/10.3354/meps323107
Lequeux BD, Ahumada-Sempoal MA, López-Pérez A, Reyes-Hernández C (2018) Coral connectivity between equatorial eastern Pacific marine protected areas: A biophysical modeling approach. PLoS One 13(8):e0202995. https://doi.org/10.1371/journal.pone.0202995
Levin LA (2006) Recent Progress in Understanding Larval Dispersal: New Directions and Digressions. Integr Comp Biol 46:282–297
López-Pérez RA, Calderón-Aguilera LE, Reyes-Bonilla H, Carriquiry JD, Medina-Rosas P, Cupul-Magaña AL, Herrero-Pérezrul MD, Hernández-Ramírez HA Ahumada-Sempoal MA, Luna-Salguero BM (2012) Coral communities and reefs from Guerrero, southern Mexican Pacific. Marine Ecology 33(4):407–416. https://doi.org/10.1111/j.1439-0485.2011.00505.x
López-Pérez A, Cupul-Magaña A, Ahumada-Sempoal MA, Medina-Rosas P, Reyes-Bonilla H, Herrero-Pérezrul MD, Reyes-Hernández Lara-Hernández J (2015) The coral communities of the Islas Marias archipelago, Mexico: structure and biogeographic relevance to the Eastern Pacific. Marine Ecology 37(3):679–690. https://doi.org/10.1111/maec.12337
López-Pérez A, Guendulain-García S, Granja-Fernández R, Hernández-Urraca V, Galván-Rowland L, Zepeta-Vilchis R, López-López D (2016) Reef community changes associated with the 2009–2010 El Niño in the southern Mexican Pacific 1. Pac Sci 70(2):175–190. https://doi.org/10.2984/70.2.4
López-Pérez RA, Mora-Pérez MG, Leyte-Morales GE (2007) Coral (Anthozoa: Scleractinia) recruitment at Bahías de Huatulco, Western Mexico: implications for coral community structure and dynamics. Pac Sci 61: 355-369. https://doi.org/10.2984/1534-6188(2007)61%5b355:CASRAB%5d2.0.CO;2
Lynch M, Raphael S, Mellor L, Spare P, Inwood M (1972) Métodos de Laboratorio. Nueva Editorial Interamericana S.A. de C.V. Segunda edición. México, pp 1522
Maté JL (2003) Corals and coral reefs of the Pacific coast of Panamá. In: Cortés J (ed) Latin American coral reefs. Elsevier, Amsterdam, pp 387-417. https://doi.org/10.1016/B978-044451388-5/50018-7
Medina-Rosas P, Carriquiry J, Cupul-Magaña A (2005) Recruitment of Porites (Scleractinia) on artificial substrata in reefs affected by the 1997–98 El Niño in Banderas Bay, Mexican Pacific. Cienc Mar 31: 103–109. https://doi.org/10.7773/cm.v31i11.75
Mendes JM, Woodley JD (2002) Timing of reproduction in Montastraea annularis: relationship to environmental variables. Mar Ecol Prog Ser 227:241–251. https://doi.org/10.3354/meps227241
Miller K, Mundy C (2003) Rapid settlement in broadcast spawning corals: implications for larval dispersal. Coral Reefs 22:99–106. https://doi.org/10.1007/s00338-003-0290-9
Morse DE, Morse ANC (1991) Enzymatic Characterization of the Morphogen Recognized by Agaricia humilis (Scleractinian Coral) Larvae. Biol Bull 181:104–122. https://doi.org/10.2307/1542493
Morse DE, Morse ANC, Raimondi PT, Hooker N (1994) Morphogen-based chemical flypaper for Agaricia humilis coral larvae. Biol Bull 186:172–181. https://doi.org/10.2307/1542051
Muñoz CG, Jaramillo-González J, Zapata FA (2018) Evidence of sexually-produced coral recruitment at Gorgona Island, Eastern Tropical Pacific. Bol Investig Mar Costeras 47(2): 97-110. http://dx.doi.org/10.25268/bimc.invemar.2018.47.2.749
Negri AP, Webster NS, Hill RT, Heyward AJ (2001) Metamorphosis of broadcast spawning corals in response to bacteria isolated from crustose algae. Mar Ecol Prog Ser 223:121–131. https://doi.org/10.3354/meps223121
Palacios-Hernández E, Carrillo LE, Filonov A, Brito-Castillo L, Cabrera-Ramos CE (2010) Seasonality and anomalies of sea surface temperature off the coast of Nayarit. Mexico. Ocean Dyn 60(1):81–91. https://doi.org/10.1007/s10236-009-0244-z
Palma AT, Steneck RS, Wilson CJ (1999) Settlement-driven, multiscale demographic patterns of large benthic decapods in the Gulf of Maine. J. Exp. Mar. Biol. Ecol. 241:107–136. https://doi.org/10.1016/S0022-0981(99)00069-6
Pantoja DA, Marinone S, Parés-Sierra A, Gómez-Valdivia F (2011) Numerical modeling of seasonal and mesoscale hydrography and circulation in the Mexican Central Pacific Modelación numérica de la hidrografía y circulación estacional y de mesoescala en el Pacífico central mexicano. Cienc Mar 38: 363-379. https://doi.org/10.7773/cm.v38i2.2007
Pawlik JR (1992) Chemical ecology of the settlement of benthic marine invertebrates. Oceanogr Mar Biol: An Annual Review 30:273–335
Pennington JT, Mahoney KL, Kuwahara VS, Kolber DD, Calienes R, Chávez FP (2006) Primary production in the eastern tropical Pacific: A review. Prog Oceanogr 69(2):285–317. https://doi.org/10.1016/j.pocean.2006.03.012
Pérez-Vivar TL, Reyes-Bonilla H, Padilla C (2006) Corales pétreos (Scleractinia) de las Islas Marías, Pacífico de México Stony corals (Scleractinia) from the Marías Islands, Mexican Pacific. Cienc Mar 32(2): 259-270
Raimondi PT, Morse ANC (2000) The Consequences of Complex Larval Behavior in a Coral. Ecology 81:3193–3211. https://doi.org/10.1890/0012-658(2000)081%5b3193:TCOCLB%5d2.0.CO;2
Reyes-Bonilla H (2003) Coral reefs of the Pacific coast of Mexico. In: Cortés J (ed), Latin American Coral Reefs. pp 331–349. https://doi.org/10.1016/B978-044451388-5/50015-1
Reyes-Bonilla H, Calderón-Aguilera LE, Cruz-Piñon G, Medina-Rosas P, López-Pérez RA, Herrero-Pérez MD, Leyte-Morales GE, Cupul-Magaña AL, Carriquiry JD (2005) Atlas de corales pétreos (Anthozoa: Scleractinia) del Pacífico mexicano. Centro de Investigación Científica y Educación Superior de Ensenada pp 124
Reyes-Bonilla H, Carriquiry J, Leyte-Morales G, Cupul-Magana A (2002) Effects of the El Nino-Southern Oscillation and the anti-El Nino event (1997-1999) on coral reefs of the western coast of Mexico. Coral Reefs 21(4):368–372. https://doi.org/10.1007/s00338-002-0255-4
Reyes-Bonilla H, López-Pérez A (1998) Biogeografía de los corales pétreos (Scleractinia) del Pacifico de México. Cienc Mar 24(2):211–224. https://doi.org/10.7773/cm.v24i2.744
Reyes-Bonilla HR, Calderón-Aguilera LE (1994) Parámetros poblacionales de Porites panamensis (Anthozoa: Scleractinia), en el arrecife de Cabo Pulmo, México. Rev Biol Trop 42:121–128
Richmond RH (1985) Reversible metamorphosis in coral planula larvae. Mar Ecol Prog Ser 22(1):181–185
Richmond RH (1987) Energetics, competency, and long-distance dispersal of planula larvae of the coral Pocillopora damicornis. Mar Biol 93:527–533. https://doi.org/10.1007/BF00392790
Richmond RH, Hunter CL (1990) Reproduction and recruitment of corals: comparisons among the Caribbean, the Tropical Pacific, and the Red Sea. Mar Ecol Prog Ser 60:185–203. https://doi.org/10.3354/meps060185
Ritson-Williams R, Arnold SN, Fogarty ND, Steneck RS, Vermeij MJ, Paul VJ (2009) New perspectives on ecological mechanisms affecting coral recruitment on reefs. Smithson Contrib Mar Sci 38:437
Rodríguez-Troncoso AP, Carpizo-Ituarte E, Leyte-Morales GE, Chi-Barragán G, Tapia-Vázquez O (2011) Sexual reproduction of three coral species from the Mexican South Pacific. Mar Biol 158(12):2673–2683. https://doi.org/10.1007/s00227-011-1765-9
Rodríguez-Troncoso AP, Carpizo-Ituarte E, Pettay DT, Warner ME, Cupul-Magaña AL (2014) The effects of an abnormal decrease in temperature on the Eastern Pacific reef-building coral Pocillopora verrucosa. Mar Biol 161(1):131–139. https://doi.org/10.1007/s00227-013-2322-5
Sammarco PW (1982) Polyp bail-out: an escape response to environmental stress and a new means of reproduction in corals. Mar Ecol Prog Ser, Oldendorf 10:57–65. https://doi.org/10.3354/meps010057
Schmidt-Roach S, Miller KJ, Lundgren P, Andreakis N (2014) With eyes wide open: a revision of species within and closely related to the Pocillopora damicornis species complex (Scleractinia; Pocilloporidae) using morphology and genetics. Zoological Journal of the Linnean Society 170:1–33. https://doi.org/10.1111/zoj.12092
Santiago-Valentín JD, Colley SB, Glynn PW, Cupul-Magaña AL, López-Pérez RA, Rodríguez-Zaragoza FA, Benítez-Villalobos F, Bautista-Guerrero E, Zavala-Casas DA, Rodríguez-Troncoso AP (2018) Regional and species specific sexual reproductive patterns of three zooxanthellate scleractinian corals across the Eastern Tropical Pacific. Mar Ecol 39(2):e12497. https://doi.org/10.1111/maec.12497
Santiago-Valentín JD, Rodríguez-Troncoso AP, Bautista-Guerrero E, López-Pérez A, Cupul-Magaña AL (2019) Successful sexual reproduction of the scleractinian coral Porites panamensis: evidence of planktonic larvae and recruitment. Inv Biol 138(1):29–39. https://doi.org/10.1111/ivb.12235
Santiago-Valentín JD, Rodríguez-Troncoso AP, Carpizo-Ituarte E, Benitez-Villalobos F, Torres-Hernandez P, López-Pérez A (2015) Reproductive pattern of the reef-building coral Pavona gigantea (Scleractinia: Agariciidae) off southwestern Mexico. Cien Mar 41(3):233–246. https://doi.org/10.7773/cm.v41i3.2482
Smith DB (1991) The reproduction and recruitment of Porites panamensis Verrill at Uva Island, Pacific Panamá. MS thesis, Univ Miami, Coral Gables, Florida, p 64
Steneck RS (2006) Staying connected in a turbulent world. Science 311(5760):480–481. https://doi.org/10.1126/science.1123541
Strathmann RR, Hughes TR, Kuris AM, Lindeman KC, Morgan SG, Pandolfi JM, Warner RR (2002) Evolution of Local Recruitment and Its Consequences for Marine Populations. Bull of Mar Sci 70:377–396
Stull R (2000) Meteorology for scientist and engineers, 2nd edn. California, Pacific Grove, p 502
Szmant MA (1986) Reproductive ecology of Caribbean reef corals. Coral Reefs 5:43–53. https://doi.org/10.1007/BF00302170
Veron JEN (2000) Corals of the World. New Litho, Surrey Hills, Melbourne, Australia 1:490
Webster NS, Smith LD, Heyward AJ, Watts JEM, Webb RI, Blackall LL, Negri AP (2004) Metamorphosis of a scleractinian coral in response to microbial biofilms. Appl Environ Microbiol 70:1213–1221. https://doi.org/10.1128/AEM.70.2.1213-1221.2004
Wellington GM (1982) Depth zonation of corals in the Gulf of Panamá: control and facilitation by resident reef fishes. Ecol Monogr 52(3):223–241. https://doi.org/10.2307/2937329
Wittenberg M, Hunte W (1992) Effects of eutrophication and sedimentation on juvenile corals I. abundance, mortality and community structure. Mar Biol 112:131–138. https://doi.org/10.1007/BF00349736
Wood S, Paris CB, Ridgwell A, Hendy EJ (2014) Modelling dispersal and connectivity of broadcast spawning corals at the global scale. Glob Ecol Biogeogr 23:1–11
Wyrtki K (1965) Surface currents of the Eastern Tropical Pacific Ocean. Inter-American Tropical Tuna Commission Bulletin 9:271–303
Acknowledgements
JDSV was funded by a doctoral fellowship from Mexico’s CONACYT (National Council of Science and Technology Grant 262538). Research support was also provided by the National Geographic Society (W405-15 and NGS-55349R-19) to APRT, the “Program to Improve Teaching” (PRODEP-UdeG-239013) to EBC, and the “Integrated program for institutional growth” from the University of Guadalajara (P/PIFI-2010-14MSU0010Z-10) to ACM. We thank the authorities of CONANP and PROZONA, and especially those based at Islas Marias. Finally, the authors thank Dr. Anderson Mayfield for providing critical comments on the article, as well as proofreading it.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Anastazia Banaszak
Rights and permissions
About this article
Cite this article
Santiago-Valentín, J.D., Rodríguez-Troncoso, A.P., Bautista-Guerrero, E. et al. Settlement ecology of scleractinian corals of the Northeastern Tropical Pacific. Coral Reefs 39, 133–146 (2020). https://doi.org/10.1007/s00338-019-01872-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-019-01872-y