Abstract
Quinoa (Chenopodium quinoa) agriculture has been a cornerstone of highland Andean diets for thousands of years, but it has received relatively little attention from archaeologists studying diet through stable isotope analysis. In this study, we present the largest sample published to date (n = 49) of archaeological carbon (δ13C) and nitrogen (δ15N) stable isotope ratios in quinoa, as well as single samples of cactus (Opuntia spp.), wild bean (Trifolium amabile), and potato (Solanum tuberosum) from a Late Intermediate Period (cal ad 1250–1450) hillfort town in the western Lake Titicaca basin. Quinoa δ15N averages + 8.83‰±2.17, indicating that agricultural fields at this site were fertilized with camelid manure, but values were significantly higher in samples recovered from high-status compounds than low-status ones. This suggests that high-status groups within the community had larger camelid herds and/or older fields that had been improved with fertilizer for longer periods of time, possibly allowing their plants to be more productive than those of lower-status groups. Mean quinoa δ13C was − 23.95‰±0.72, which indicates that plants were not significantly more water-stressed than modern or historic comparative samples grown with similar methods. This concurs with paleoclimate data suggesting that the environment surrounding this settlement was in a period of drought recovery during the occupation. Alternatively, fertilizer may have allowed plants to combat the effects of drought without recycling sub-stomatal CO2. This study is an important contribution to research on foodways in the ancient Andes and non-cereal grain-dependent societies more broadly.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Quinoa (Chenopodium quinoa Willd.) is a nutritious and hardy staple crop that has sustained communities of the Peruvian and Bolivian Altiplano through millennia of climatic fluctuation and socio-political change (Miller et al. 2021). Nutritionally, this pseudo-cereal (non-grass seed or grain) ‘superfood’ has high protein content, contains all eight essential amino acids, is gluten-free, and has a high micronutrient content (Repo–Carrasco et al. 2003). Moreover, because it was domesticated in an arid, high-altitude environment, it is productive in a wide range of climates and regions. Yet, to date, it has received relatively little attention in archaeological literature relative to maize agriculture (Bruno and Whitehead 2003; López and Recalde 2016; Langlie 2020; Miller et al. 2021). Fortunately, its nutritional and ecological benefits have attracted the interest of food scientists, leading to a growing body of research on the physiology of modern quinoa (Stikić et al. 2015; Alandia et al. 2016; Gámez et al. 2019). This study synthesizes plant science of modern quinoa and archaeological chemistry of cereal grains to interpret new isotopic data on ancient quinoa.
The past two decades have seen considerable advancements in archaeological plant chemistry, with an emphasis on the role of animal manure fertilizers, detected using nitrogen stable isotopes, in ancient agricultural systems (Ferrio et al. 2005; Bogaard et al. 2007; Szpak et al. 2012; Szpak 2014; Nitsch et al. 2015; Treasure et al. 2016). This is a particularly important line of inquiry in the South American Andes, where manure, particularly seabird guano, has historically been an important source of wealth (Szpak et al. 2012; Gootenberg 2014; Santana–Sagredo et al. 2021). Additionally, archaeologists have begun to leverage stable carbon isotopes in C3 plants as an indicator of ancient water availability (Wallace et al. 2013; Styring et al. 2016; Nitsch et al. 2017). To date, these efforts have been primarily focused on cereal grains such as wheat and maize (DeNiro and Hastorf 1985; Boyd et al. 2008; Szpak et al. 2012, 2013). Thus, non-cereal staples such as tubers, legumes, and pseudo-cereals represent a prominent gap in the current literature.
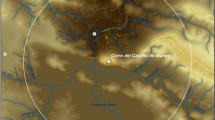
Samples analysed in this paper come from a site known as Ayawiri (Fig. 1) and predominantly date to the Andean Late Intermediate Period (LIP; ad 1100–1450). The LIP coincides with the global Medieval Climate Anomaly, during which, according to numerous lines of evidence, precipitation in the Central Andes was lower than average from approximately ad 800 to 1300 (Abbott et al. 1997; Binford et al. 1997; Mélice and Roucou 1998; Thompson et al. 1998, 2013; Calaway 2005; Arnold et al. 2021a; Guédron et al. 2023). Additionally, the LIP in much of the Andean highlands saw political balkanization, endemic warfare (Covey 2008; Arkush and Tung 2013), and a shift away from maize agriculture (Schreiber 1987; Williams 2002; Miller et al. 2021). In place of maize, communities emphasized camelid herding as an economic base, which was better-suited to drier conditions and socio-political decentralisation (Stanish 2007; Kellett 2016; Arnold et al. 2021b).
With this political and economic re-organization, communities relocated from valley bottoms to defensible hillforts called pukaras - of which Ayawiri is an example (Fig. 2). This settlement style became common in the latter half of the LIP on the Altiplano of Peru and Bolivia, and seems to have been designed for defensibility against potential invaders and to facilitate pastoral access to high-altitude grazing lands (Arkush 2011, 2020). Subsistence at Ayawiri relied heavily on camelid herding and the cultivation of tubers and chenopods, all of which were able to thrive at these higher elevations (Bruno 2014; Nielsen 2018; Langlie and Capriles 2021; Miller et al. 2021), in rain-fed terraces (Langlie and Arkush 2016; Langlie 2020).
Using carbon and nitrogen stable isotopes from charred plant remains, we address four important gaps in scientific understanding:
-
1.
We characterize the isotopic range of ancient quinoa grains with the largest data set published to date.
-
2.
We test for the presence of manuring and intra-site differences in its practice.
-
3.
We assess the possibility of detecting water stress in ancient quinoa plants through δ13C.
-
4.
We discuss how these new insights should impact the interpretation of dietary isotopes in humans and animals in the ancient Andes.
Background
Nitrogen isotopes
The two stable isotopes of nitrogen 15N and 14N, are distributed differentially in organisms based upon their abundance in the natural environment, the trophic level of consumers, and the metabolic processes of producers. The ratio of the heavier to lighter isotope is measured in parts per mil (‰) relative to a standard (most commonly Ambient Inhalable Reservoir, or AIR) and represented as δ15N. δ15N is positively correlated with trophic level in animals, with each trophic level typically representing roughly 3–4‰ (DeNiro and Epstein 1981). δ15N in bone collagen, which reflects the isotopic composition of dietary protein, is commonly used to identify the staple protein sources of archaeological populations (Froehle et al. 2010; Nitsch et al. 2017; Turner et al. 2018). The underlying assumptions of this approach are that terrestrial plant protein has low δ15N values relative to animal protein, and that the high protein content of animal products has a strong influence over bone collagen values.
Yet δ15N values in plants vary greatly, both due to the mycorrhizal (fungal) symbionts of different plant taxa (Szpak 2014; Hussain et al. 2018), and the nitrogen content of the soil in which they grow. In particular, nitrogen-rich fertilizers, including animal manures that were used in ancient societies, are high in 15N and can enrich plant values by one trophic level or more (Bogaard et al. 2007; Christensen et al. 2022). Moreover, quinoa has a high protein content relative to other staple crops cultivated in the Andes (e.g. maize, potatoes), at approximately 15% (Abugoch et al. 2008). If fertilized with animal manure, quinoa theoretically presents a similar isotopic profile to terrestrial meat in human bone collagen, confounding simple dietary models based on δ15N. Though no controlled studies to date have specifically evaluated the effects of camelid manure on quinoa plants, plant science studies of quinoa physiology have reported the fertilization protocols used in controlled experiments (Alandia et al. 2016; Rezzouk et al. 2020a). Separately, archaeologists have begun to study the influence of camelid manure on cereal grains (Szpak et al. 2012). Together, we use the research on quinoa fertilization and camelid manure to inform the interpretation of this study.
The impact of manuring on the δ15N values of crops depends upon the intensity and duration of manuring, as well as the species of animal manure used and the physiological processes of the crop plant. Szpak et al. (2012) found that six samples of maize grains fertilized with camelid manure were on average 1.8‰ higher, at +8.1‰±1.6, than the control group, which averaged +6.3‰±0.3. Additionally, one meta-analysis of manuring studies found that camelid dung had slightly higher δ15N values and greater enrichment in the values of crop plants than did cattle dung, which is a more common choice for modern farmers in the Andes (Alandia et al. 2016; Rezzouk et al. 2020a). Separately, archaeologists have begun to study the influence of camelid manure on cereal grains (Szpak et al. 2012).
Quinoa grown in subsistence farming conditions in the Andean region is likely to provide the most accurate reference data for ancient chenopods. Modern quinoa grains (n = 3 bulk samples) purchased at markets in northern Peru had an average value of +7.88‰±1.35Footnote 1 (Szpak et al. 2013, p 9). It is likely that these crops were fertilized in some capacity, though the type, quantity, and duration is unknown. Modern quinoa grains grown in the Cusco region of Peru (n = 5) had a value of +8.44‰±0.56 (Turner et al. 2010, p 524). The authors note that these grains were not subjected to chemical fertilizers, but do not comment on the possibility of animal fertilizers. Miller et al. (2021) provide a rare and valuable baseline data set of quinoa, fertilized with camelid manure (the intensity and duration unknown), from the Lake Poopó region of Bolivia, which averaged +8.1‰±2.29 (n = 3).
Experimental studies provide greater clarity on the influence of fertilizers. In one paper, quinoa plants fertilized with a mixture of cattle and chicken manure had a mean of +12.53‰±2.11 (n = 40) (Rezzouk et al. 2020b). A study of quinoa plant leaves grown in unmanured conditions in Northwest Argentina were notably lower, ranging from − 5.50 to +6.03‰ in different varieties, though unfortunately, neither an overall mean nor raw data was provided (González et al. 2011). Additionally, the number of years over which manure is applied to agricultural fields elevates the δ15N values of plants (Szpak 2014). Collectively, archaeological and experimental studies suggest that unmanured quinoa plants could be expected to have a mean below +7.0‰, while quinoa fertilized with camelid manure, at an intensity and duration significant enough to be detected, could produce a wide range of values above +7.0‰. This proposed value may need to be raised in the case of sustained drought, as aridity has a well-documented positive correlation with δ15N (Heaton 1987; Aranibar et al. 2004), though, as discussed below, aridity may also be detected independently through the analysis of stable carbon isotopes (Styring et al. 2016).
Carbon isotopes
Plants absorb the two stable isotopes of carbon, 12C and 13C, in different proportions based on their photosynthetic pathway. The ratio of the heavier to the lighter isotope is typically expressed as δ13C and measured in parts per mil relative to a standard (most commonly Vienna Pee Dee Belemnite, abbreviated VPDB). Most plants in the Andes follow the C3 (Calvin–Benson) pathway, though maize and kiwicha (Amaranthus caudatus L.) follow the C4 (Hatch-Slack) pathway. Plants following the C3 photosynthetic pathway discriminate strongly against the heavier, less abundant 13C isotope, resulting in δ13C values around −25.5‰. C4 plants discriminate less against the heavier isotope, resulting in higher (less negative) δ13-C values, with an average of −12.5‰ (Tykot 2004, p 435). Cacti and succulents follow the CAM photosynthetic pathway, which results in a wide range of δ13C values. Under sub-optimal water availability, plant stomata close in order to limit water loss and plants recycle intercellular CO2, discriminating less against the 13C isotope and leading to higher (less negative) δ13C values (Stroud et al. 2021). The application of nitrogen-rich fertilizers such as animal manure seems to increase average stomatal conductance in crops, including quinoa (Hati et al. 2006; Alandia et al. 2016), thus lowering δ13C values (Busari et al. 2016). Interestingly, this suggests that δ13C and δ15N could be negatively correlated in fertilized plants.
The standard measure for carbon discrimination in plants is represented as Δ13C (Farquhar et al. 1982), calculated using the atmospheric δ13CO2 value for the date of the site cal bp (Ferrio et al. 2005). This is similar to a typical δ13C measure, but allows for the direct comparison of ancient and modern plant carbon use, and converts the original data to an absolute value (thus, water stress is associated with lower Δ13C values, but higher δ13C values). For ancient samples, which all dated from 3,800—500 cal bp, we used δ13Cair= −6.4‰, based on the Monte Carlo average from Antarctic ice cores from this time period (Eggleston et al. 2016). For modern samples, we used the value δ13Cair= −8.0 to account for the Suess Effect. Δ13C values reported were calculated using the following formula:
Additionally, there is some evidence that different varieties of quinoa respond differently to drought conditions (Sun et al. 2014; Stikić et al. 2015; Hussain et al. 2018; Gámez et al. 2019). In one study, the leaves of a quinoa variety grown on the hyper-arid coast of Chile discriminated less against 13C under water stress - typically considered a sign of greater water use efficiency - while a variety grown on the Altiplano displayed no overall difference in Δ13C under water-stressed conditions as compared to well-watered ones (Gámez et al. 2019). In sum, lower discrimination against 13C should not yet be interpreted as evidence of well-watered conditions based on δ13C alone.
Notably, most previous studies of this topic in both plant science and archaeology have focused on irrigated, rather than rain-fed, agriculture (Wallace et al. 2013; Sun et al. 2014; Styring et al. 2016; but see González et al. 2011). This study is based upon the same scientific principles, but δ13C is instead used as a proxy for water stress from low precipitation. Due to the currently limited scientific understanding of these complex interactions between environment, plant taxa, and agricultural practices, the results from Ayawiri will be evaluated qualitatively against baseline values from comparable studies and read in concert with climate data.
Published ancient and modern baseline data for chenopod seeds is summarized in Table 2. While there are a number of plant science studies of Δ13C in modern quinoa, standard practice is to sample the foliar tissue, which limits this set of comparable data (e.g. González et al. 2011; Gámez et al. 2019). Leaves are only rarely available in ancient plant assemblages, and Δ13C in leaves is not directly comparable to values in seeds (Szpak et al. 2013). Szpak and colleagues (2013, p 9) also found that modern quinoa seeds collected in northern Peru had average δ13C values of −25.57‰ (Δ13C=18.03‰), while Turner et al. (2010) reported that seeds collected in markets in the Cusco region averaged −25.48 (Δ13C=17.94‰). Modern quinoa cultivar seeds from the Lake Poopó region of Bolivia averaged −25.60‰ (Δ13C=18.06‰), while archaeological chenopods dating from the Formative to Tiwanaku periods in this same region averaged −22.97‰ (Δ13C=17.13‰), corrected for charring (Miller et al. 2021). In a foundational study by DeNiro and Hastorf (1985) of plants from the Upper Mantaro Valley of Peru, modern seeds averaged −25.20‰ (Δ13C = 17.64‰), and ancient chenopods dating from the Initial through Late Horizon periods averaged − 24.24‰ (Δ13C=18.45‰), when adjusted for charring. The mean Δ13C of this comparative dataset (n = 21) is 17.23‰, so if Ayawiri falls substantially below this value, it could cautiously be interpreted as evidence of water stress. Conversely, though, high or average Δ13C relative to the comparative dataset should not be interpreted as definitive evidence of well-watered conditions until carbon isotope discrimination in quinoa varieties is better understood.
Use of charred plant remains
Charred seeds, like the ones analysed in this study, are the most commonly preserved form of plant remains in high-altitude regions of the Andes (DeNiro and Hastorf 1985). According to methodological research, charred plant remains largely retain the isotopic values that plants possessed in life when charred at the low temperatures (~ 200–300 °C) that allow for preservation and taxonomic identification of ancient grains (DeNiro and Hastorf 1985; Charles et al. 2015; Stroud et al. 2023). However, charring slightly enriches measured δ13C and δ15N values, so whenever it was necessary to directly compare carbonized plant remains to uncarbonized ones, we corrected for the charring effect by subtracting 0.16‰ from δ13C and 0.32‰ from δ15N (Stroud et al. 2023).
Site overview and environmental context
Ayawiri, also known as Machu Llaqta, is a dense residential complex located at approximately 4,100 m above sea level (m a.s.l.) atop a high, flat mesa in the western Lake Titicaca Basin (Fig. 1). Proyecto Machu Llaqta conducted investigations at Ayawiri between 2009 and 2014. Radiocarbon dates and ceramics place the first occupation in the Upper Formative Period (200 cal bc–cal ad 25) and the second occupation in the Late Intermediate Period (cal ad 1250–1450). Three stone defensive walls protect the fortress from the north, and the site is bounded on all other sides by cliffs. Within the fortress, additional walls separate approximately 100 residential compounds (Arkush 2018). At maximum occupation, it could have housed between one to two thousand residents. Around 1450, the settlement was rapidly abandoned, perhaps as the result of an attack by enemies (Arkush 2017).
Ayawiri falls within the broader environmental setting of the Andean Altiplano, which stretches from the northern Lake Titicaca Basin in Peru into Central Bolivia, making it the second-largest plateau in the world, with an average altitude above 3,800 m a.s.l. Notably, the site is also situated approximately 8 km from the southern shore of Lake Umayo, a small lake that drains into Lake Titicaca, which could also have provided lacustrine resources, such as reeds and lake fish, for residents of Ayawiri (Baker et al. 2009). Holocene palaeoclimate records from these two nearby lakes allow for a relatively high-resolution reconstruction of the Ayawiri’s environmental history. Two recent studies of sediment cores in the Lago Menor at Lake Titicaca reconstruct lake levels (Bruno et al. 2021; Guédron et al. 2023), and stable oxygen isotopes (δ18O) from carbonate sediments were previously used to model precipitation at Lake Umayo (Baker et al. 2009). According to these indicators, the Formative Period occupation of Ayawiri saw roughly equivalent - though marginally lower - precipitation in the region of Lake Umayo than the LIP (Baker et al. 2009, p 316), and this is corroborated by a multi-proxy study of lake levels in the Lago Menor of Lake Titicaca (Guédron et al. 2023). Diatom reconstruction of the Lago Menor also suggests somewhat shallow lake levels in the Lago Menor of Lake Titicaca during the second part of this period (Bruno et al. 2021, p 139), but this does not appear to be part of a larger region-wide trend (Arnold 2016; Guédron et al. 2023). According to all three climate reconstructions, precipitation decreased sharply from approximately ad 800–1000 and remained low throughout the early LIP. The period of lowest precipitation varies by study; it is found at approximately ad 1100 according to Baker et al. (2009), around ad 1200 according to Bruno et al. (2021, p 139), and at ad 1000, with an additional dip around ad 1300, according to Guédron et al. (2023). From approximately 1300–1500, according to all three studies, precipitation and lake levels increased steadily. Thus, during the primary time period addressed by this study, it is likely that the region was in a period of recovery from this sustained drought.
Subsistence at Ayawiri
Ayawiri is located in the tundra above 4,000 m a.s.l., known as the puna, where camelids (llamas and alpacas) are herded and crops are rarely grown, while its terraces are located in the suni, 3,500–4,000 m a.s.l. (though this zone can be extended up to 4,050 m a.s.l. with adaptations such as terracing), where tubers, chenopods, and high-altitude legumes (e.g. tarwi, Lupinus mutabilis Sweet) are cultivated (Vidal 2014). These agricultural terraces, built during the LIP, wrapped around three sides of the hillfort (Langlie 2018). Because of its high-altitude, semi-arid environment and nutrient-poor soils, only a narrow range of plants and animals can survive in the Altiplano. Despite this precarity, residents of the Altiplano have developed agricultural systems capable of supporting dense populations for thousands of years. We infer the probable subsistence system at Ayawiri based on previously published archaeological evidence and comparative ethnographic data from modern communities in the south-central highlands.
Based on an abundance of macrobotanical and zooarchaeological remains, diet at Ayawiri seems to have been dominated by C3 plants (chenopods and tubers) and terrestrial mammals (camelids). Notably, no maize (Zea mays L.) was detected at the site (Langlie 2020). Maize agriculture is challenging above 3,500 m a.s.l., requiring large quantities of water and specialized infrastructure to protect against nightly frosts, and evidently the residents of Ayawiri did not prioritize growing or trading for the crop. In contrast, tubers and chenopods are adapted to high altitudes and require less water, and thus were well-adapted to the high, arid Altiplano in the LIP (Vacher 1998; Bruno 2014; Miller et al. 2021). Still, water stress has been associated with lower grain weights in quinoa plants, so the potential impact of drought on chenopod cultivation should not be dismissed (Gámez et al. 2019).
Analysis of camelid dung samples indicates they are rich in companion weeds, demonstrating that llama herds were grazed in agricultural fields, making it likely that their dung was, at least to some extent, deposited on the crops (Langlie and Capriles 2021). This practice is widely documented in modern ethnographic accounts of sectoral fallowing systems, which were common in the Central Andes until at least the end of the 20th century. Such fallowing systems designate most fields as communal pastureland for 2–10 years following the cropping cycle, leaving ample opportunity for natural fertilization (Orlove and Godoy 1986, p 170; Zimmerer 1997). It is likely that these fallow pasturelands were used communally in the ancient past, though both private and communal grazing lands are present in modern comparative contexts (Orlove and Godoy 1986, p 173). Fertilizer, typically animal manure, is added to potatoes, which are the first crop to be planted following a period of fallowing (Winterhalder et al. 1974, p 95; Orlove and Godoy 1986, p 170; Bruno 2011, p 225). Quinoa is typically planted 1–3 years after potatoes, and does not receive additional fertilizer (Winterhalder et al. 1974, p 95; Bruno 2011). Manure is a particularly valuable resource in this ecological region because it is an efficient means of transforming the biomass of the Altiplano into accessible nutrients for crops (Winterhalder et al. 1974). Manure may be especially crucial for quinoa, which has high nitrogen demands (Zimmerer 1997, p 140).
Perhaps more crucially, manuring adds mass to the soil, thus improving water retention by preventing erosion (Winterhalder et al. 1974; Arriaga and Lowery 2003; Blanco-Canqui et al. 2015). Like many of their modern counterparts, agricultural fields at Ayawiri were rain-fed (unirrigated) terraces, meaning that residents would have managed soil moisture and quality based on their knowledge of seasonality and geology, as well as through practices such as crop rotation and fertilization (Bruno 2011, p 218). Thus, it is likely that the residents of Ayawiri used crops and camelid herds in tandem, allowing fallowed fields to feed their herds and manure from the camelids to nourish their soils, which created a highly efficient system for ensuring sufficient nitrogen and water content for the potato and quinoa crops.
Materials and methods
Sampling
Charred macrobotanical remains were recovered from flotation of soil samples during the 2011 and 2012 seasons of Proyecto Machu Llacta. Excavators collected on average 10 L soil samples from floors, and point samples were collected from smaller, dense contexts (hearths and pits). These soil samples were processed with a SMAP-style flotation device, and original counts and identifications are reported by Langlie (2018, 2020). Samples used in this study (n = 52) were selected to reflect a variety of domestic contexts within the site, representing five compounds of high, middle, and low status (Fig. 3, summarized in ESM 1), and depositions in hearths, pits, and floors. The majority (n = 49) were quinoa (Fig. 4), as these were by far the most abundant taxon at the site, but potato (Solanum tuberosum), cactus (Opuntia spp.) seed, and wild bean (Trifolium amabile) seeds are represented by one sample each. All samples date to the LIP (cal ad 1250–1450), with the exception of three quinoa seeds from the Upper Formative (200 cal bc–cal ad 25).
Preparation and analysis
At the Cornell Stable Isotope Lab, samples were crushed, homogenised, and weighed into tin capsules. Each sample in this study represents 2–9 individual seeds, with the exception of a piece of whole potato, in order to ideally reach a weight of 2–3 mg. The samples were washed with water during flotation, then dried and stored in sealed plastic bags or microcentrifuge vials until analysis. Upon visual examination, no soil adhered to individual grains, and while it was not feasible to examine the internal microstructure of each individual quinoa grain, the samples analysed by Langlie (2016, ESM 1) using SEM imagery revealed intact microstructure. We chose to forgo chemical pre-treatment for several reasons. There is currently limited research on appropriate pre-treatments for ancient charred botanical samples, and all documented protocols lead to significant mass loss in the sample, particularly for small seeds like quinoa (Vaiglova et al. 2014; Brinkkemper et al. 2018). In particular, strong acidic treatments can alter the δ13C and δ15N of the sample, rather than removing contaminants (Vaiglova et al. 2014). Since no known pre-treatment consistently removes the necessary contaminants from carbonized plant remains, and the impact of contaminants, such as carbonates, humic and fulvic acids, and nitrates, has relatively little impact on the resulting isotopic values, we determined that the risks outweighed the benefits (Brinkkemper et al. 2018).
Samples were analysed on a Thermo Delta V isotope ratio mass spectrometer interfaced to a NC2500 elemental analyser. Isotope corrections were conducted using two-point normalization based on in-house standards of KCRN (δ13C=−13.02‰±0.10, δ15N=1.28‰±0.33) and CBT (δ13C=−25.58‰±0.06, δ15N=17.48‰±0.38). In order to verify that the instrument could accurately measure values with different amplitudes, a chemical methionine standard (δ13C=−26.85‰±0.27, δ15N=−0.09‰±0.69) was analysed, and found that amplitudes between 200 mV and 15,000 mV have an error associated with a linearity of 0.69‰ for δ15N and amplitudes between 200 mV and 10,000 mV for δ13C have an error with linearity of 0.27‰. Precision was verified using the in-house standard RICE (δ13C=−29.11‰±0.10, δ15N=0.90‰±0.18), run every 10 samples, and was reported to have standard deviations of 0.18‰ for δ15N and 0.10‰ for δ13C.
Ratios are reported as delta-values (δ) unless otherwise noted and measured in parts per mil (‰). δ13C was measured relative to VPDB, and δ15N was measured relative to AIR. %C, %N, and C:N atomic ratio are reported in ESM 2. Raw isotope data is reported to one decimal place value, while calculations based on these figures are reported to two place values.
Results
The full data set can be found in ESM 2 and is visualized in Fig. 5. All statistical analyses and data visualization were conducted in R with RStudio version 1.4.1103. The full data set of quinoa samples (n = 49) had a δ13C range of −25.3‰ to −22.0‰ and a mean of −23.95‰±0.72. Quinoa from the LIP (n = 46) had the same range and a mean of −23.98‰±0.73, and the formative sample (n = 3) ranged from − 23.1‰ to − 23.6‰ with a mean of −23.37‰±0.25. In the full quinoa data set, δ15N ranged from +2.8‰ to +13.55‰ with a mean of +8.83‰±2.17. For the LIP, values ranged from +4.7‰ to +13.6‰ with a mean of + 8.94‰±2.04, and for the formative sample, the range was +2.8‰ to +9.4‰ with a mean of +7.07‰±3.70. The potato sample had a δ13C value of −24.9‰ and δ15N of +5.5‰, while wild bean was measured at −24.3‰ and +4.0‰, respectively. The cactus seeds, representing the only non-C3 plant in the sample, a δ13C value of − 10.7‰, and δ15N of +− 7.9‰.
Scatterplot of all plant results from Ayawiri. Axis breaks were achieved with R code from Xu et al. (2021)
Following recommendations from Szpak and Chiou (2020), we tested for a correlation between C:N atomic ratio and δ15N, and found a weak positive correlation in the full dataset (Spearman’s rho = 0.29, p = 0.04) and the chenopod dataset (Spearman’s rho = 0.35, p = 0.01). According to Szpak and Chiou (2020), a strong positive correlation in C:N atomic ratio and δ15N may be evidence of diagenetic nitrogen loss in the sample, which preferentially retains the heavier 15N isotope. Since this topic requires more research (Szpak and Chiou 2020, p 533), and the correlation is weak to moderate, we do not currently interpret this as evidence of diagenetic alteration. Additionally, δ13C and δ15N are negatively, but weakly, correlated in the full dataset (Spearman’s rho = −0.26, p = 0.06) and the chenopod dataset (Spearman’s rho = −0.33, p = 0.02). The possible implications of this are discussed in section ‘Aridity and drought tolerance’.
Table 1 presents δ13C and δ15N summary statistics for each compound. Summary statistics for excavation loci and use contexts are reported in Tables S1-S2 in ESM 1. We used a one-way ANOVA to test for differences in δ15N between compounds, and found that at least one pair was significantly different (F statistic = 5.349, p < 0.01). Tukey-Kramer HSD intervals showed that compound 118 had significantly lower δ15N than compounds 59 and 6. Variances were not equal among δ13C between compounds, so we tested for difference using a Kruskal–Wallis test, and found that at least one pair was significantly different (chi-squared = 11.691, p = 0.02). According to a post-hoc Wilcoxon rank sum test with a Bonferroni correction, compound 118 had significantly higher δ13C than compounds 59 and 72.
The same analyses were conducted to test for differences between LIP chenopod values in different types of depositional contexts, which were designated as hearths, pits, and floors. No significant differences were detected for δ13C or δ15N.
Discussion
Patterns in manuring of crops
It is highly likely that the chenopods at Ayawiri were fertilized, at least to some extent, with camelid manure, based on other lines of evidence from the site and comparison to previously published data. The mean δ15N value observed at Ayawiri falls above our proposed threshold of + 7.0‰, and was the second-highest among datasets of quinoa grain grown on small farms in the highland Andes (Table 2) - including the value reported in Miller et al. (2021) for modern quinoa known to have been fertilized with camelid manure. Interestingly, the potato sample had low δ15N relative to other plants, at 5.5‰, running contrary to Bruno’s (Bruno 2011, p 225) modern observation that potatoes are the most intensively fertilized crop. This could be an anomaly due to the small sample size of potato, or reflect different priorities in fertilization between past and present farmers.
Higher δ15N values are also associated with higher-status (i.e. larger) compounds (Fig. 6). This relationship is statistically significant for high-status compounds 6 and 59 and low-status compound 118. This suggests that some higher-status patio groups practiced more intensive manuring than the broader community, perhaps as a result of possessing larger camelid herds. Alternatively, residents of the higher-status compounds may have planted their crops in older fields that had been improved with manure for a longer period of time, since, in ethnographically documented communities, fallow pasture lands included in the sectoral rotation system were accessible to all households in the community, not just those who owned them for cultivation (Orlove and Godoy 1986, p 176; Zimmerer 1997, p 123). Lower mean δ15N values in the formative period quinoa sample - prior to the construction of the agricultural terraces - also support this interpretation. These two explanations are not mutually exclusive, and the latter points to the probable importance of “first families” (Arkush 2018, p 16) at Ayawiri. In other words, economic privilege was associated with more nutrient-rich fields, likely resulting in greater crop yields. The economic importance of manure, particularly for agropastoralists, has been observed ethnographically in numerous cultures, including in the ancient and modern Andes (Winterhalder et al. 1974; Jones 2013; Allen 2016; Langlie and Capriles 2021).
Though notable, this status trend is not a strict difference, as compound 44 is a high-status patio group with low mean δ15N. This suggests that there was variation in manuring practices and/or herd size among higher-status groups, which is consistent with previous findings that heterarchical corporate divisions were the main drivers of social organization at Ayawiri (Arkush 2018; Velasco 2022). In light of the very small sample of low-status compounds, and the exception provided by compound 44, we emphasize that there are other possible explanations for this intra-site variation. While manure is, to some extent, a function of the size of herds possessed by a landholding group, long fallow periods could reduce this impact while also preserving soil quality. It is also possible that some farmers chose not to spread manure evenly across the field. Camelids defecate communally, leading to build-up of manure in centralized areas (Winterhalder et al. 1974, p 102), and since it is likely that camelids at Ayawiri were grazed in fallow agricultural fields (ESM 1 Langlie 2016, p 303), it is possible that some of this original patterning was retained, leading to uneven soil δ15N values across the field. Therefore, it is likely that some of the variability visible at Ayawiri is due to agricultural processes without clearly patterned relationships to status or depositional contexts.
Aridity and drought tolerance
These results provide robust baseline data for researchers interested in using δ13C to infer water stress in quinoa plants. Perhaps most significantly, this study establishes that quinoa grown in traditional, unirrigated highland conditions can be expected to have δ13C values of approximately − 24‰ (Δ13C=18‰, Suess corrected). Carbon isotope discrimination from the present study was similar to (though relatively high among) comparative datasets (Table 2) from a variety of time periods and regions of the Andes.
Note that, though δ13C in crop plants has typically been used to infer irrigation practices (Wallace et al. 2013), because the terraces at Ayawiri were rain-fed, the findings presented here address local precipitation. Comparative interpretation suggests that the quinoa cultivated at Ayawiri did not employ sub-stomatal CO2 recycling to a significant degree as a water-saving mechanism, despite the lack of irrigation in the agricultural fields. It is possible that these plants employed other drought-resistant adaptations, including developing deep, dense root systems or employing epidermal cell bladders as water reservoirs (Jensen et al. 2000; Jacobsen et al. 2009; Sun et al. 2014; Gámez et al. 2019). Or, because the occupation at Ayawiri occurred during a period of drought recovery, the majority of these samples did not experience substantial water stress.
Alternatively, the negative correlation observed between δ13C and δ15N runs counter to the expectation that aridity leads to higher δ13C and higher δ15N. As discussed above, manuring increases the stomatal conductance of plants, lowering δ13C (Alandia et al. 2016), and improves soil water retention (Arriaga and Lowery 2003; Blanco-Canqui et al. 2015). Szpak and Chiou noted a weak, non-statistically significant negative correlation in δ13C and δ15N in plants from Northern Peru (not including quinoa), which they interpreted as evidence that the high observed δ15N values were the result of fertilizer, rather than water stress (2020, p 534). This pattern can also be observed in comparative datasets from the Andes (Fig. 7) and experimental studies of quinoa plants treated with organic fertilizer (Hussain et al. 2018, p 416; Rezzouk et al. 2020b, p 8). Thus, we believe that the significant difference in δ13C between compound 118 and compounds 59 and 72 is a result of the differences in manuring levels among these groups.
Use as dietary baseline data
δ13C and δ15N in bone collagen have most commonly been used to identify the relative consumption of meat and maize in Andean diets (Hastorf 1991; Tung 2021). The findings presented here add to recent cautions against using δ15N from human bone alone to identify dietary protein source (Szpak et al. 2012; Santana-Sagredo et al. 2021) by demonstrating that manured chenopods, with high protein content and δ15N values easily as high as + 9‰, could be mistaken for the meat of C3-feeding camelids when inferred through human bone collagen. For example, in Miller et al.‘s (2021) study of diet on the Altiplano, archaeological chenopods samples (n = 3) had higher δ15N values (+10.37‰±2.21) than archaeological camelids (+ 8.53‰±1.67). Researchers should consider the possibility of quinoa as a dietary staple, deferring to additional lines of evidence, such as palaeoethnobotany, zooarchaeology and compound-specific stable isotope analysis before forwarding human bone collagen results as evidence of meat consumption (Szpak et al. 2012; e.g. Miller et al. 2021). In particular, this study calls into question interpretations of meat consumption in cases where animals were foddered or grazed on the same crops that their manure fertilized. In the case of Ayawiri, human bone collagen δ15N is more than 4‰ higher than the mean for quinoa, indicating that both manured plants and terrestrial animals likely contributed to dietary protein (Whittemore 2022 and unpubl. data). Enamel apatite δ13C confirms that C4 plants were not significant components of diet (Whittemore et al. 2022).
Conclusions
Our research indicates that the residents of Ayawiri were growing quinoa plants without extreme drought and nourishing their crops with fertilizer to increase yields. Some members of the community, who lived in higher-status compounds, seem to have had access to crops grown on fields steeped in significantly more manure. This may have been a result of larger camelid herds or older agricultural fields in this subset of the community. Still, this does not neatly follow a pronounced status division, in keeping with our understanding of Ayawiri as a society without extreme differences in material wealth.
To our knowledge, this is the largest study to date of the isotopic composition of ancient quinoa remains. Our findings offer a large sample of δ13C and δ15N values for quinoa farmed with pre-colonial methods and emphasize what many researchers have suspected: that manuring may have a significant impact on δ15N in human tissues in the ancient Andes, and should therefore be considered in dietary mixing models. Additionally, these results indicate that, at least in the case of quinoa, a negative correlation between δ13C and δ15N may be associated with manuring. Further research on this topic will allow for more accurate interpretations of ancient quinoa production and a better understanding of the numerous ancient societies that relied on C3 crops.
Data availability
All original data used in this article are available in the text and supplementary information.
Notes
Note that, although we report standard deviations, the number of seeds included in each data point varies substantially between studies, and are not consistently reported; i.e. a sample size of n = 1 could reflect anything from one to a several hundred seeds, so these standard deviations should not be directly compared between publications.
References
Abbott MB, Binford MW, Brenner M, Kelts KR (1997) A 3500 14C yr high-resolution record of water-level changes in Lake Titicaca, Bolivia/Peru. Quat Res 47:169–180. https://doi.org/10.1006/qres.1997.1881
Abugoch LE, Romero N, Tapia CA, Silva J, Rivera M (2008) Study of some physicochemical and functional properties of quinoa (Chenopodium quinoa Willd) protein isolates. J Agric Food Chem 56:4745–4750. https://doi.org/10.1021/jf703689u
Alandia G, Jacobsen S-E, Kyvsgaard NC, Condori B, Liu F (2016) Nitrogen sustains seed yield of Quinoa under intermediate drought. J Agron Crop Sci 202:281–291. https://doi.org/10.1111/jac.12155
Allen CJ (2016) Stones who love me: dimensionality, enclosure and petrification in Andean Culture. Arch Sci Soc Relig 174:327–346. https://doi.org/10.4000/assr.27854
Aranibar JN, Otter L, Macko SA et al (2004) Nitrogen cycling in the soil-plant system along a precipitation gradient in the Kalahari sands. Glob Chang Biol 10:359–373. https://doi.org/10.1111/j.1365-2486.2003.00698.x
Arkush EN (2011) Hillforts of the ancient Andes: Colla warfare, society, and landscape. University Press of Florida, Gainesville
Arkush E (2017) The end of Ayawiri: abandonment at an Andean hillfort town of the late intermediate period. J Field Archaeol 42:241–257. https://doi.org/10.1080/00934690.2017.1308748
Arkush E (2018) Coalescence and defensive communities: insights from an Andean hillfort town. Camb Archaeol J 28:1–22. https://doi.org/10.1017/S0959774317000440
Arkush E (2020) Land use, settlement patterns, and collective defense in the Titicaca Basin: the constitution of defensive community. Andean Past 13:15
Arkush E, Tung TA (2013) Patterns of war in the Andes from the archaic to the late horizon: insights from settlement patterns and cranial trauma. J Archaeol Res 21:307–369. https://doi.org/10.1007/s10814-013-9065-1
Arnold B (2016) Belts vs. blades: the binary bind in Iron Age mortuary contexts in Southwest Germany. J Archaeol Method Theory 23:832–853. https://doi.org/10.1007/s10816-016-9289-8
Arnold TE, Hillman AL, Abbott MB, Werne JP, McGrath SJ, Arkush EN (2021a) Drought and the collapse of the Tiwanaku civilization: new evidence from Lake Orurillo, Peru. Quat Sci Rev 251:106693. https://doi.org/10.1016/j.quascirev.2020.106693
Arnold TE, Hillman AL, McGrath SJ, Abbott MB, Werne JP, Hutchings J, Arkush EN (2021b) Fecal stanol ratios indicate shifts in camelid pastoralism in the highlands of Peru across a 4,000-year lacustrine sequence. Quat Sci Rev 270:107193. https://doi.org/10.1016/j.quascirev.2021.107193
Arriaga FJ, Lowery B (2003) Soil physical properties and crop productivity of an eroded soil amended with cattle manure. Soil Sci 168:888–899. https://doi.org/10.1097/01.ss.0000106403.84926.7e
Baker PA, Fritz SC, Burns SJ, Ekdahl E, Rigsby CA (2009) The Nature and Origin of Decadal to Millennial Scale Climate variability in the Southern Tropics of South America: the Holocene Record of Lago Umayo, Peru. In: Vimeux F, Sylvestre F, Khodri M (eds) Past climate variability in South America and surrounding regions: from the last glacial Maximum to the Holocene. Developments in Paleoenvironmental Research 14. Springer, Dordrecht, pp 301–322. https://doi.org/10.1007/978-90-481-2672-9_13
Binford MW, Kolata AL, Brenner M, Janusek JW, Seddon MT, Abbott M, Curtis JH (1997) Climate variation and the rise and fall of an Andean civilization. Quat Res 47:235–248. https://doi.org/10.1006/qres.1997.1882
Blanco-Canqui H, Hergert GW, Nielsen RA (2015) Cattle manure application reduces soil compactibility and increases water retention after 71 years. Soil Sci Soc Am J 79:212–223. https://doi.org/10.2136/sssaj2014.06.0252
Bogaard A, Heaton THE, Poulton P, Merbach I (2007) The impact of manuring on nitrogen isotope ratios in cereals: archaeological implications for reconstruction of diet and crop management practices. J Archaeol Sci 34:335–343. https://doi.org/10.1016/j.jas.2006.04.009
Boyd M, Varney T, Surette C, Surette J (2008) Reassessing the northern limit of maize consumption in North America: stable isotope, plant microfossil, and trace element content of carbonized food residue. J Archaeol Sci 35:2:545–2556. https://doi.org/10.1016/j.jas.2008.04.008
Brinkkemper O, Braadbaart F, van Os B, van Hoesel A, van Brussel AAN, Fernandes R (2018) Effectiveness of different pre-treatments in recovering pre-burial isotopic ratios of charred plants. Rapid Commun Mass Spectrom 32:251–261. https://doi.org/10.1002/rcm.8033
Bruno MC (2011) 8. Farmers’ experience and knowledge: utilizing soil diversity to Mitigate Rainfall variability on the Taraco Peninsula, Bolivia. In: Miller NF, Moore KM, Ryan K (eds) Sustainable Lifeways: Cultural Persistence in an ever-changing environment. University of Pennsylvania Museum of Archaeology and Anthropology, Philadelphia, pp 212–243. https://doi.org/10.9783/9781934536322.212
Bruno MC (2014) Beyond raised fields: exploring farming practices and processes of agricultural change in the Ancient Lake Titicaca Basin of the Andes. Am Anthropol 116:130–145. https://doi.org/10.1111/aman.12066
Bruno MC, Whitehead WT (2003) Chenopodium cultivation and formative period agriculture at Chiripa, Bolivia. Lat Am Antiq 14:339–355. https://doi.org/10.2307/3557565
Bruno MC, Capriles JM, Hastorf CA, Fritz SC, Weide DM, Domic AI, Baker PA (2021) The rise and fall of Wiñaymarka: rethinking cultural and environmental interactions in the Southern Basin of Lake Titicaca. Hum Ecol 49:131–145. https://doi.org/10.1007/s10745-021-00222-3
Busari MA, Salako FK, Tuniz C (2016) Stable isotope technique in the evaluation of tillage and fertilizer effects on soil carbon and nitrogen sequestration and water use efficiency. Eur J Agron 73:98–106. https://doi.org/10.1016/j.eja.2015.11.002
Calaway MJ (2005) Ice-cores, sediments and civilisation collapse: a cautionary tale from Lake Titicaca. Antiquity 79:778–790. https://doi.org/10.1017/S0003598X00114929
Charles M, Forster E, Wallace M, Jones G (2015) Nor ever lightning char thy grain 1: establishing archaeologically relevant charring conditions and their effect on glume wheat grain morphology. STAR: Sci Technol Archaeol Res. https://doi.org/10.1179/2054892315Y.0000000008
Christensen BT, Jensen JL, Dong Y, Bogaard A (2022) Manure for millet: grain δ15N values as indicators of prehistoric cropping intensity of Panicum miliaceum and Setaria italica. J Archaeol Sci 139:105554. https://doi.org/10.1016/j.jas.2022.105554
Covey RA (2008) Multiregional perspectives on the archaeology of the Andes during the late intermediate period (c. A.D. 1000–1400). J Archaeol Res 16:287–338. https://doi.org/10.1007/s10814-008-9021-7
DeNiro MJ, Epstein S (1981) Influence of diet on the distribution of nitrogen isotopes in animals. Geochim Cosmochim Acta 45:341–351. https://doi.org/10.1016/0016-7037(81)90244-1
DeNiro MJ, Hastorf CA (1985) Alteration of 15N14N and 13C12C ratios of plant matter during the initial stages of diagenesis: studies utilizing archaeological specimens from Peru. Geochim Cosmochim Acta 49:97–115. https://doi.org/10.1016/0016-7037(85)90194-2
Eggleston S, Schmitt J, Bereiter B, Schneider R, Fischer H (2016) NOAA/WDS Paleoclimatology - Antarctic Ice Core 155,000 Year CO2 and Gas Stable Isotope Data, δ13CO2. NOAA National Centers for Environmental Information, Mississippi
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137. https://doi.org/10.1071/pp9820121
Ferrio JP, Araus JL, Buxó R, Voltas J, Bort J (2005) Water management practices and climate in ancient agriculture: inferences from the stable isotope composition of archaeobotanical remains. Veget Hist Archaeobot 14:510–517. https://doi.org/10.1007/s00334-005-0062-2
Froehle AW, Kellner CM, Schoeninger MJ (2010) FOCUS: effect of diet and protein source on carbon stable isotope ratios in collagen: follow up to Warinner and, Tuross (2009). J Archaeol Sci 37(2):2662–2670. https://doi.org/10.1016/j.jas.2010.06.003
Gámez AL, Soba D, Zamarreño ÁM, García-Mina JM, Aranjuelo I, Morales F (2019) Effect of water stress during Grain Filling on Yield, Quality and physiological traits of Illpa and rainbow quinoa (Chenopodium quinoa Willd.) cultivars. Plants 8:173. https://doi.org/10.3390/plants8060173
González JA, Bruno M, Valoy M, Prado FE (2011) Genotypic variation of gas exchange parameters and leaf stable carbon and nitrogen isotopes in ten quinoa cultivars grown under drought. J Agron Crop Sci 197:81–93. https://doi.org/10.1111/j.1439-037X.2010.00446.x
Gootenberg PE (2014) Between silver and guano: Commercial Policy and the state in Postindependence Peru. Princeton University Press, Princeton
Guédron S, Delaere C, Fritz SC et al (2023) Holocene variations in Lake Titicaca water level and their implications for sociopolitical developments in the Central Andes. Proc Natl Acad Sci USA 120:e2215882120. https://doi.org/10.1073/pnas.2215882120
Hastorf CA (1991) Gender, space, and food in prehistory. In: Gero JM, Conkey MW (eds) Engendering Archaeology: women and Prehistory. Blackwell Publishers, Oxford, pp 132–159
Hati KM, Mandal KG, Misra AK, Ghosh PK, Bandyopadhyay KK (2006) Effect of inorganic fertilizer and farmyard manure on soil physical properties, root distribution, and water-use efficiency of soybean in vertisols of Central India. Biores Technol 97(2):182–2188. https://doi.org/10.1016/j.biortech.2005.09.033
Heaton THE (1987) The 15N/14N ratios of plants in South Africa and Namibia: relationship to climate and coastal/saline environments. Oecologia 74:236–246. https://doi.org/10.1007/BF00379365
Hussain MI, Al- Dakheel AJ, Reigosa MJ (2018) Genotypic differences in agro-physiological, biochemical and isotopic responses to salinity stress in quinoa (Chenopodium quinoa Willd.) plants: prospects for salinity tolerance and yield stability. Plant Physiol Biochem 129:411–420. https://doi.org/10.1016/j.plaphy.2018.06.023
Jacobsen S-E, Liu F, Jensen CR (2009) Does root-sourced ABA play a role for regulation of stomata under drought in quinoa (Chenopodium quinoa Willd). Sci Hortic 122:281–287. https://doi.org/10.1016/j.scienta.2009.05.019
Jensen CR, Jacobsen S-E, Andersen MN, Núñez N, Andersen SD, Rasmussen L, Mogensen VO (2000) Leaf gas exchange and water relation characteristics of field quinoa (Chenopodium quinoa Willd.) during soil drying. Eur J Agron 13:11–25. https://doi.org/10.1016/S1161-0301(00)00055-1
Jones DR (2013) Manure matters: historical, Archaeological and Ethnographic Perspectives. Ashgate Publishing, Farnham
Kellett LC (2016) Chanka settlement ecology: disentangling settlement decision-making during a time of risk in the Andean highlands. In: Kellett LC, Jones E (eds) Settlement Ecology of the ancient Americas. Routledge, London, pp 239–265
Langlie BS (2016) Farming through the Auca Runa: Agricultural Strategies and Terraces during the Late Intermediate Period, Altiplano, Peru. PhD thesis, Washington University in St. Louis, St. Louis, MO
Langlie BS (2018) Building ecological resistance: late intermediate period farming in the south-central highland Andes (CE 1100–1450). J Anthropol Archaeol 52:167–179. https://doi.org/10.1016/j.jaa.2018.06.005
Langlie BS (2020) Late Intermediate period plant use at a Colla hillfort, Puno, Peru (AD 1300–1450). Lat Am Antiq 31:702–719. https://doi.org/10.1017/laq.2020.28
Langlie BS, Capriles JM (2021) Paleoethnobotanical evidence points to agricultural mutualism among early camelid pastoralists of the Andean Central Altiplano. Archaeol Anthropol Sci 13:107. https://doi.org/10.1007/s12520-021-01343-y
López ML, Recalde MA (2016) The first quinoa (Chenopodium quinoa Willd) macrobotanical remains at Sierras del Norte (Central Argentina) and their implications in pre-hispanic subsistence practices. J Archaeol Sci Rep 8:426–433. https://doi.org/10.1016/j.jasrep.2016.06.053
Mélice JL, Roucou P (1998) Decadal time scale variability recorded in the Quelccaya summit ice core δ18O isotopic ratio series and its relation with the sea surface temperature. Clim Dyn 14:117–132. https://doi.org/10.1007/s003820050213
Miller MJ, Kendall I, Capriles JM, Bruno MC, Evershed RP, Hastorf CA (2021) Quinoa, potatoes, and llamas fueled emergent social complexity in the Lake Titicaca Basin of the Andes. Proc Natl Acad Sci USA 118:e2113395118. https://doi.org/10.1073/pnas.2113395118
Nielsen AE (2018) Agropastoral Taskscapes and Seasonal Warfare in the Southern Andes during the Regional Developments Period (Thirteenth–Fifteenth Centuries). In: A, C (eds) Political landscapes of the late intermediate period in the Southern Andes: the Pukaras and their hinterlands, the latin American Studies Book Series. Springer, Cham, pp 247–268. https://doi.org/10.1007/978-3-319-76729-1_10
Nitsch EK, Charles M, Bogaard A (2015) Calculating a statistically robust δ13C and δ15N offset for charred cereal and pulse seeds. STAR: Sci Technol Archaeol Res 1:1–8. https://doi.org/10.1179/2054892315Y.0000000001
Nitsch E, Andreou S, Creuzieux A et al (2017) A bottom-up view of food surplus: using stable carbon and nitrogen isotope analysis to investigate agricultural strategies and diet at bronze age Archontiko and Thessaloniki Toumba, northern Greece. World Archaeol 49:105–137. https://doi.org/10.1080/00438243.2016.1271745
Orlove BS, Godoy R (1986) Sectoral fallowing systems in the Central Andes. J Ethnobiol 6:169–204
Repo-Carrasco R, Espinoza C, Jacobsen S-E (2003) Nutritional value and use of the andean crops quinoa (Chenopodium quinoa) and Kañiwa (Chenopodium pallidicaule). Food Rev Int 19:179–189. https://doi.org/10.1081/FRI-120018884
Rezzouk FZ, Shahid MA, Elouafi IA, Zhou B, Araus JL, Serret MD (2020a) Agronomic performance of irrigated quinoa in desert areas: comparing different approaches for early assessment of salinity stress. Agric Water Manag 240:106205. https://doi.org/10.1016/j.agwat.2020.106205
Rezzouk FZ, Shahid MA, Elouafi IA, Zhou B, Araus JL, Serret MD (2020b) Agronomical and analytical trait data assessed in a set of quinoa genotypes growing in the UAE under different irrigation salinity conditions. Data Brief 31:105758. https://doi.org/10.1016/j.dib.2020.105758
Santana-Sagredo F, Schulting RJ, Méndez-Quiros P et al (2021) White gold’ guano fertilizer drove agricultural intensification in the Atacama Desert from AD 1000. Nat Plants 7:152–158. https://doi.org/10.1038/s41477-020-00835-4
Schreiber KJ (1987) Conquest and consolidation: a comparison of the Wari and Inka occupations of a highland Peruvian Valley. Am Antiq 52:266–284. https://doi.org/10.2307/281780
Stanish C (2007) Agricultural intensification in the Titicaca Basin. In: Thurston TL, Fisher CT (eds) Seeking a richer Harvest: the Archaeology of Subsistence Intensification, Innovation, and change. Studies in Human Ecology and Adaptation 3. Springer, New York, NY, pp 125–139. https://doi.org/10.1007/978-0-387-32762-4_6
Stikić R, Jovanović Z, Marjanović M, Đorđević S (2015) The effect of drought on water regime and growth of quinoa (Chenopodium quinoa Willd). Ratar Povrt 52:80–84. https://doi.org/10.5937/ratpov52-8000
Stroud E, Bogaard A, Charles M (2021) A stable isotope and functional weed ecology investigation into chalcolithic cultivation practices in Central Anatolia: Çatalhöyük, Çamlıbel Tarlası and Kuruçay. J Archaeol Sci Rep 38:103010. https://doi.org/10.1016/j.jasrep.2021.103010
Stroud E, Charles M, Bogaard A, Hamerow H (2023) Turning up the heat: assessing the impact of charring regime on the morphology and stable isotopic values of cereal grains. J Archaeol Sci 153:105754. https://doi.org/10.1016/j.jas.2023.105754
Styring AK, Ater M, Hmimsa Y et al (2016) Disentangling the effect of farming practice from aridity on crop stable isotope values: a present-day model from Morocco and its application to early farming sites in the eastern Mediterranean. Anthropocene Rev 3:2–22. https://doi.org/10.1177/2053019616630762
Sun Y, Liu F, Bendevis M, Shabala S, Jacobsen S-E (2014) Sensitivity of two quinoa (Chenopodium quinoa Willd.) varieties to progressive drought stress. J Agron Crop Sci 200:12–23. https://doi.org/10.1111/jac.12042
Szpak P (2014) Complexities of nitrogen isotope biogeochemistry in plant-soil systems: implications for the study of ancient agricultural and animal management practices. Front Plant Sci 5:288. https://doi.org/10.3389/fpls.2014.00288
Szpak P, Chiou KL (2020) A comparison of nitrogen isotope compositions of charred and desiccated botanical remains from northern Peru. Veget Hist Archaeobot 29:527–538. https://doi.org/10.1007/s00334-019-00761-2
Szpak P, Millaire J-F, White CD, Longstaffe FJ (2012) Influence of seabird guano and camelid dung fertilization on the nitrogen isotopic composition of field-grown maize (Zea mays). J Archaeol Sci 39:3:721–3740. https://doi.org/10.1016/j.jas.2012.06.035
Szpak P, White CD, Longstaffe FJ, Millaire J-F, Vásquez Sánchez VF (2013) Carbon and nitrogen isotopic survey of Northern Peruvian plants: baselines for paleodietary and paleoecological studies. PLoS ONE 8:e53763. https://doi.org/10.1371/journal.pone.0053763
Thompson LG, Davis ME, Mosley-Thompson E et al (1998) A 25,000-Year tropical climate history from bolivian ice cores. Science 282:1:858–1864. https://doi.org/10.1126/science.282.5395.1858
Thompson LG, Mosley-Thompson E, Davis ME, Zagorodnov VS, Howat IM, Mikhalenko VN, Lin P-N (2013) Annually resolved ice core records of tropical climate variability over the past ~ 1800 years. Science 340:945–950. https://doi.org/10.1126/science.1234210
Treasure ER, Church MJ, Gröcke DR (2016) The influence of manuring on stable isotopes (δ13C and δ15N) in celtic bean (Vicia faba L.): archaeobotanical and palaeodietary implications. Archaeol Anthropol Sci 8:555–562. https://doi.org/10.1007/s12520-015-0243-6
Tung TA (2021) Making and marking maleness and valorizing violence: a bioarchaeological analysis of embodiment in the Andean past. Curr Anthropol 62:125–144. https://doi.org/10.1086/712305
Turner BL, Kingston JD, Armelagos GJ (2010) Variation in dietary histories among the immigrants of Machu Picchu: carbon and nitrogen isotope evidence. Chungará (Arica) 42:515–534. https://doi.org/10.4067/S0717-73562010000200012
Turner BL, Bélisle V, Davis AR et al (2018) Diet and foodways across five millennia in the Cusco region of Peru. J Archaeol Sci 98:137–148. https://doi.org/10.1016/j.jas.2018.07.013
Tykot RH (2004) Stable isotopes and diet: You are what you eat. In: Martini M, Milazzo M, Piacentini M (eds) Physics Methods in Archaeometry 154. IOS Press, Amsterdam, pp 433–444
Vacher JJ (1998) Responses of two main Andean crops, quinoa (Chenopodium quinoa Willd) and papa amarga (Solanum juzepczukii Buk.) to drought on the Bolivian Altiplano: significance of local adaptation. Agric Ecosyst Environ 68:99–108. https://doi.org/10.1016/S0167-8809(97)00140-0
Vaiglova P, Snoeck C, Nitsch E, Bogaard A, Lee-Thorp J (2014) Impact of contamination and pre-treatment on stable carbon and nitrogen isotopic composition of charred plant remains. Rapid Commun Mass Spectrom 28(2):497–2510. https://doi.org/10.1002/rcm.7044
Velasco MC (2022) Burying the dead at Ayawiri: mortuary diversity and postmortem manipulation at an Andean hillfort (AD 1100–1450). Lat Am Antiq. https://doi.org/10.1017/laq.2022.50
Vidal JP (2014) Las ocho regiones naturales del Perú. Terra Brasilis (Nova Série): Revista da Rede Brasileira de História da Geografia e Geografia Histórica 3:1–20. https://doi.org/10.4000/terrabrasilis.1027
Wallace M, Jones G, Charles M, Fraser R, Halstead P, Heaton THE, Bogaard A (2013) Stable carbon isotope analysis as a direct means of inferring crop water status and water management practices. World Archaeol 45:388–409. https://doi.org/10.1080/00438243.2013.821671
Whittemore AF, Tung TA, Langlie BS, Arkush E, Velasco MC (2022) Growing up with warfare: Childhood diet and mobility at a hillfort in the Lake Titicaca Basin (1275–1450 CE). In: Navigating Culture Change: Studies in the (Re)Creation of New Worlds. Presented at the Society for American Archaeology 87th Annual Meeting
Williams PR (2002) Rethinking disaster-induced collapse in the demise of the Andean highland states: Wari and Tiwanaku. World Archaeol 33:361–374. https://doi.org/10.1080/00438240120107422
Winterhalder B, Larsen R, Thomas RB (1974) Dung as an essential resource in a highland peruvian community. Hum Ecol 2:89–104. https://doi.org/10.1007/BF01558115
Xu S, Chen M, Feng T, Zhan L, Zhou L, Yu G (2021) Use ggbreak to effectively utilize plotting space to deal with large datasets and outliers. Front Genet 12:774846. https://doi.org/10.3389/fgene.2021.774846
Zimmerer KS (1997) Changing Fortunes: Biodiversity and Peasant Livelihood in the peruvian Andes. University of California Press, Berkeley
Acknowledgements
Excavations at Ayawiri were authorized by the Peruvian Ministry of Culture (RD N° 023-2011-DGPC-VMPCIC/MC). Many thanks to Kim and Jed Sparks of the Cornell Stable Isotope Lab for providing bench space and technical support for this project. This article was greatly improved by stimulating discussions at the Frontiers in Archaeological Sciences 3: Rethinking the Paradigm conference, hosted at Cornell University in October 2022. We thank two anonymous reviewers whose comments particularly strengthened our discussion of land use and engagement with the literature on δ15N.
Funding
Excavations at Ayawiri were funded by a National Science Foundation grant (BCS-0849094 / BCS-1101148) to Elizabeth Arkush and Aimee Plourde. Archaeobotanical analysis was supported by a National Science Foundation Doctoral Dissertation Improvement Grant (1305140) awarded to BrieAnna Langlie, and Washington University in Saint Louis Graduate School. Anna Fancher Whittemore was supported by an NSF-GRFP during the second phase of analyses and writing of this article.
Author information
Authors and Affiliations
Contributions
AFW: Conceptualization, investigation, methodology, visualization, Writing—Original draft; BASL: Funding acquisition, data curation, Writing—Review & editing; EA: funding acquisition, Writing—Review & editing; MCV: Supervision, Writing—Review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Communicated by L. A. Newsom.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Whittemore, A.F., Langlie, B.S., Arkush, E. et al. Isotopic insights into quinoa agriculture at an Andean hillfort town (cal ad 1250–1450). Veget Hist Archaeobot 33, 393–406 (2024). https://doi.org/10.1007/s00334-023-00952-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-023-00952-y