Abstract
Objective
Body composition assessment derived from cross-sectional imaging has shown promising results as a prognostic biomarker in several tumor entities. Our aim was to analyze the role of low skeletal muscle mass (LSMM) and fat areas for prognosis of dose-limiting toxicity (DLT) and treatment response in patients with primary central nervous system lymphoma (PCNSL).
Methods
Overall, 61 patients (29 female patients, 47.5%) with a mean age of 63.8 ± 12.2 years, range 23–81 years, were identified in the data base between 2012 and 2020 with sufficient clinical and imaging data. Body composition assessment, comprising LSMM and visceral and subcutaneous fat areas, was performed on one axial slice on L3-height derived from staging computed tomography (CT) images. DLT was assessed during chemotherapy in clinical routine. Objective response rate (ORR) was measured on following magnetic resonance images of the head accordingly to the Cheson criteria.
Results
Twenty-eight patients had DLT (45.9%). Regression analysis revealed that LSMM was associated with objective response, OR = 5.19 (95% CI 1.35–19.94, p = 0.02) (univariable regression), and OR = 4.23 (95% CI 1.03- 17.38, p = 0.046) (multivariable regression). None of the body composition parameters could predict DLT. Patients with normal visceral to subcutaneous ratio (VSR) could be treated with more chemotherapy cycles compared to patients with high VSR (mean, 4.25 vs 2.94, p = 0.03). Patients with ORR had higher muscle density values compared to patients with stable and/or progressive disease (34.46 ± vs 28.18 ± HU, p = 0.02).
Conclusions
LSMM is strongly associated with objective response in patients with PCNSL. Body composition parameters cannot predict DLT.
Clinical relevance statement
Low skeletal muscle mass on computed tomography (CT) is an independent prognostic factor of poor treatment response in central nervous system lymphoma. Analysis of the skeletal musculature on staging CT should be implemented into the clinical routine in this tumor entity.
Key Points
• Low skeletal muscle mass is strongly associated with the objective response rate.
• No body composition parameters could predict dose-limiting toxicity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Primary central nervous system (CNS) lymphoma (PCNSL) is an aggressive extranodal lymphoma exclusively involving the brain, spinal cord, cranial nerves, leptomeninges, and eyes [1]. Diffuse large B-cell lymphoma (DLBCL) is the most common histological subtype of this entity [1]. The incidence of PCNSL is increasing in recent years with a reported age-adjusted incidence of PCNSL of 0.16 per 100,000 [2]. The highest incidence rate was observed among older patients over 65 years. To exclude other lymphoma types with a secondary CNS infiltration, computed tomography (CT) is performed for staging purposes among other diagnostic modalities [1]. PCNSL has different diagnostic implications, treatment regimes, and outcomes compared to lymphoma occurring in the other part of the body, which results in separate observations and studies of this entity [1, 2].
Nowadays, assessment of body composition represents an emergent research field in general medicine, in particular in radiology and oncology [3,4,5,6,7]. So far, the skeletal muscle area and fat areas, most commonly subcutaneous and visceral fat areas, can be quantified on radiological images. Parameters of body composition can predict relevant outcomes in oncology [3,4,5,6,7,8]. For instance, low skeletal muscle mass (LSMM) is an important factor for occurrence of treatment toxicity [8]. However, data regarding body composition in PCNSL are still scarce to date.
Therefore, the aim of the present study was to elucidate the prognostic role of CT-defined body composition parameters on dose-limiting toxicity (DLT) and treatment response in patients with PCNSL.
Methods
Patient acquisition
This retrospective study was approved by the institutional review board (Nr. 145/21, Ethics Committee, Otto-von-Guericke-University Magdeburg, Magdeburg, Germany).
All patients with PCNSL were retrospectively assessed within the time period 2012 to 2020 in one university hospital. Overall, 61 patients (29 female patients, 47.5%) with a mean age of 63.8 ± 12.2 years, range 23–81 years, were identified in the data base with sufficient clinical and imaging data. In every case, the diagnosis of PCNSL was confirmed by histopathological examination after stereotactic biopsy before admission of steroids.
Inclusion criteria for the present study were as follows:
-
-contrast-enhanced staging CT at baseline diagnosis
-
-histopathological diagnosis after biopsy or surgery
-
-first-line standard treatment with radio-chemotherapy
Exclusion criteria were as follows:
-
-no available staging CT or no contrast-enhanced CT
-
-secondary CNS involvement lymphoma
-
-second- or third-line treatment.
Objective response rate
All patients were initially evaluated with magnetic resonance imaging (MRI) of the brain during clinical routine. Response was assessed based on serial brain MRI, beginning within 4 weeks from initiation of induction treatment and reassessed every month thereafter for 4–6 months when possible. Patients then underwent CNS imaging with MRI every 3 months for the first year and followed clinically after stable findings on imaging. No MRIs were performed during steroid therapy. Measurable lesions were required to be at least 2 cm in diameter. Treatment response was defined as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). As such, complete response (CR) was defined as complete disappearance of contrast enhancement on MRI. Partial response (PR) is defined as a 50% decrease in enhancing tumor diameters. Progressive disease (PD) is defined as 25% increase in the enhancing lesions and appearance of any new CNS or non-CNS site of disease. Any other situation was characterized as stable disease (SD). Objective response was defined as CR or PR, as previously described [10].
Treatment
Conventional therapy for PCNSL is divided into induction and consolidation phase. In 2020, the National Comprehensive Cancer Network guidelines recommend systemic therapy for patients suitable for or capable of tolerating high-dose chemotherapy, while for unfit patients, 24–36 Gy of whole-brain radiotherapy (WBRT) with a boost to gross disease for a total of 45 Gy is indicated, as first recommendation is a high-dose methotrexate at 8 g/m2 with rituximab and temozolomide or a reduced dose of 3.5 g/m2 methotrexate with rituximab, vincristine, and procarbazine as well as WBRT [1,2,3].
Dose-limiting toxicity
DLT was defined as any treatment-related grade 3 or 4 hepatic toxicity (i.e., aminotransferase levels exceeding 5.1 or 10 times the normal reference values) or any grade ≥ 4 hematologic toxicity (neutrophils < 500/mm3, hemoglobin < 5 g/dL, platelets < 25 × 109/L) during the first treatment course, according to the Radiation Therapy Oncology Group criteria [11]. The DLT was evaluated following 3 months of post-treatment follow-up.
Imaging technique
All CT scans were obtained on a clinical multidetector CT scanner of different vendors (Siemens Somatom Definition AS + , Siemens Healthcare or Canon Aquilion Prime, Canon Medical Systems). The analyzed CT images were obtained before any form of treatment at the baseline staging CT. The CT protocol was as follows: acquisition slice thickness 1 mm with 5-mm reconstructions, tube voltage 120 kV, automatic tube current modulation, pitch factor 1.2, collimation 0.6 mm. In all cases, contrast media was given and the CT scan was acquired in portal venous phase after 40 s after intravenous injection.
All images were assessed in consensus by two experienced radiologists (V.F. and A.S.) who were blinded to the clinical course of the patients. Measurements were performed on axial images in the soft tissue window (window of 45 to 250 HU) on a dedicated workstation (Infinitt PACS, version 3.0, Infinitt Healthcare).
Body composition quantification
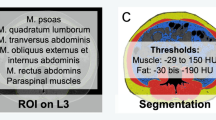
Parameters of body composition were semiautomatically measured with the freely available ImageJ software 1.48v (National Institutes of Health Image Program). One axial slice on the mid of the third lumbar vertebra (L3) was used according to the previous descriptions [4, 6]. Skeletal muscle area (SMA) was calculated with the threshold values of − 29 and 150 HU [6]. SMA was divided by the height squared to calculate the skeletal muscle index (SMI). For sarcopenia definition, the SMI threshold proposed by Prado et al was used: 52.4 cm2/m2 for male and 38.5 cm2/m2 for female patients [7].
Fat areas were measured using the HU threshold levels of − 190 and − 30 HU [6]. Visceral adipose tissue (VAT) was measured as the intra-abdominal fat and subcutaneous adipose tissue (SAT) as the fat area located subcutaneously. Visceral to subcutaneous ratio (VSR) was calculated as a ratio of visceral to subcutaneous fat.
The proposed threshold value of 100 cm2 was utilized as a cut-off value to determine visceral and/or subcutaneous obesity [12]. High VSR was defined as 1.1. Figures 1a and b display two representative patients for illustrative purposes.
Representative cases of the patient sample. Red, skeletal muscle area; blue, subcutaneous adipose tissue area; yellow, visceral adipose tissue area; green, intramuscular adipose tissue area. a Case with high skeletal muscle area and normal adipose tissue areas. b Case with high visceral and subcutaneous fat areas and low skeletal muscle area
Statistical analysis
The statistical analysis and graphics creation were performed using SPSS (IBM SPSS Statistics for Windows, version 25.0: IBM corporation). Collected data were evaluated by means of descriptive statistics (means, absolute and relative frequencies). Group differences were calculated with the Mann–Whitney-U- test and Fisher exact test, when suitable. Uni- and multivariable regression analysis was used to elucidate possible associations. In all instances, p values were interpretated exploratorely.
Results
The patients received in median 4 cycles of chemotherapy, range 1–8 cycles.
Overall, 19 patients (31.1%) had progressive disease, 23 patients showed complete response (37.7% of all patients and 65.7% of patients with treatment response), and 12 patients had partial response (19.7% of all patients and 34.2% of patients with treatment response), and 3 patients had stable disease (4.9%). For 4 patients (6.6%), no treatment was possible, and therefore no follow-up was performed.
The mean values of the analyzed body composition parameters were as follows: VAT, 165 ± 101.5 cm2; SAT, 190.8 ± 82.5 cm2; VSR, 0.96 ± 0.71; SMI, 45.8 ± 9.7 cm2/m2. Visceral obesity was identified in 35 patients (57.4%).
Associations between body composition and DLT
DLT occurred in 28 patients (45.9%). No body composition parameter was associated with occurrence of DLT (Tables 1 and 2).
However, patients with normal VSR could receive more chemotherapy cycles compared to patients with high VSR (mean 4.25 vs 2.94, p = 0.03). There were no strong associations between dichotomized body composition parameters and metric parameters on DLT (Table 3).
Associations between body composition and ORR
Patients with objective response had higher muscle density values compared to patients with SD and/or PD (Table 4). Moreover, muscle gauge was also higher in patients with objective response. Other body composition parameters were not associated with objective response.
Regression analysis revealed that sarcopenia predicted OR, OR = 5.19 (95% CI 1.35–19.94) p = 0.02 (univariable regression), and OR = 4.23 (95% CI 1.03, 17.38), p = 0.046 (multivariable regression) (Table 5).
Discussion
The present study used CT-defined body composition parameters for prediction DLT and treatment response in patients with PCNSL. As a key finding, body composition parameters were not able to predict DLT but sarcopenia was strongly associated with objective response. Therefore, assessment of body composition in PCNSL can be recommended for prediction of treatment course.
The emerging field of body composition utilized cross-sectional imaging modalities acquired for diagnostic purposes and can provide novel quantitative biomarkers of the constitution of the body [3,4,5,6,7,8,9]. A plethora of studies were published investigating the prognostic relevance of LSMM and adipose tissue throughout medicine, predominantly in oncology [3,4,5,6,7,8,9].
Notably, most studies tested the prognostic implications of body composition parameters but not their predictive role [3]. However, for direct treatment guidance, the predictive role of an imaging biomarker is of great importance. As a novel finding of the present study, sarcopenia is strongly associated with objective response. There are no published data regarding possible associations between body composition and objective response.
According to the literature, established independent prognostic factors in PNCSL are age, Karnofsky performance index, sex, and response to induction chemotherapy [13]. Regarding predictive imaging markers, a recent study could highlight the important role of dynamic contrast-enhanced MRI, which was correlated with objective response [14]. However, assessment of sarcopenia is a by-product of existing imaging data, whereas dynamic contrast-enhanced MRI must be obtained additionally with a further time consumption.
Several reasons could cause the identified association between sarcopenia and objective response. It was clearly shown that skeletal muscle area as a surrogate parameter for overall body constitution is an important factor for chemotherapy tolerance and effectiveness. As such, in a recent meta-analysis, sarcopenia was associated with overall therapy toxicity, OR = 2.19, 95% CI 1.78–2.68 [8]. This was clearly shown as well as in curative as palliative setting. Furthermore, the present study identified that also muscle quality expressed by muscle density and muscle gauge was associated with objective response. Finally, an important finding of the present study is that VSR might be an interesting factor to predict the number of chemotherapy cycles in patients with PCNSL.
Previously, only few studies analyzed the prognostic role of body composition in PCNSL. In a recent study, it has been shown that LSMM/sarcopenia predicted progression-free survival (HR = 4.40, 95% CI 1.66–11.61, p = 0.003) and overall survival (HR = 3.16, 95% CI 1.09–9.11, p = 0.034) [15]. Similar results were reported also by other authors [16].
According to the literature, measure of the muscle status on CT at the L3 level represents a standardized method to quantify the skeletal musculature [17]. The employed method, also used in the present study, provides reliable and validated results [6]. In PCNSL, previous investigations used different measurements, levels, and values for estimation of sarcopenia. So far, Leone et al measured skeletal muscle status on the L3 level as well as temporal muscle thickness derived by brain MRI [15]. Furtner et al used only the temporal muscle thickness [16]. It was shown that temporal muscle thickness was an independent predictor of mortality (HR = 2.5, 95% CI 1.6–3.9, p < 0.001) [16]. Clearly, further studies are needed to compare the prognostic and predictive role of temporal muscle thickness as a surrogate parameter of muscle status in comparison to the standardized approach on the level of L3. Importantly, every patient with PCNSL needs a staging CT to rule out extracranial manifestations [2]; analysis of body composition can be performed in clinical routine without the need for new scans.
Interestingly, as reported previously, reduced muscle quality and increased intermuscular fat were associated with poor prognosis in DLBCL [18]. However, in the present study, no associations between DLT and/or objective response and intramuscular adipose tissue could be found.
The present study is not free from limitations. First, it is a retrospective analysis of one center. This fact might result in a selection bias. Second, the time delay between CT imaging and treatment differed slightly due to clinical routine. However, the effect of treatment on body composition might be neglected in a short time frame. Third, treatment regime differed between the patients. However, this reflects the daily clinical routine. Fourth, contrast media phase could have an influence on HU measurement of the muscle and fat density. However, all patients were investigated in portal venous phase, which should reduce possible bias. Beyond that, the absolute HU differences between different contrast media phases are relatively small [19].
In conclusion, sarcopenia is strongly associated with objective response in patients with PCNSL. None of body composition parameters can predict DLT in PCNSL.
Abbreviations
- CI:
-
Confidence interval
- CR:
-
Complete response
- CT:
-
Computed tomography
- DLBCL:
-
Diffuse large B-cell lymphoma
- DLT:
-
Dose-limiting toxicity
- FFM:
-
Fat-free mass
- FM:
-
Fat mass
- HU:
-
Hounsfield unit
- IMAT:
-
Intramuscular adipose tissue
- L:
-
Lumbar
- LSMM:
-
Low skeletal muscle mass
- MRI:
-
Magnetic resonance imaging
- OR:
-
Odds ratio
- ORR:
-
Objective response rate
- PCNSL:
-
Primary central nervous system lymphoma
- PD:
-
Progressive disease
- SAT:
-
Subcutaneous adipose tissue
- SMA:
-
Skeletal muscle area
- SMI:
-
Skeletal muscle index
- TAT:
-
Total adipose tissue
- VAT:
-
Visceral adipose tissue
- VSR:
-
Visceral to subcutaneous ratio
- WBRT:
-
Whole-brain radiotherapy
References
Schaff LR, Grommes C (2022) Primary central nervous system lymphoma. 140(9):971–979
Yuan Y, Ding T, Wang S, Chen H, Mao Y, Chen T (2021) Current and emerging therapies for primary central nervous system lymphoma. Biomark Res 9(1):32
Xia L, Zhao R, Wan Q et al (2020) Sarcopenia and adverse health-related outcomes: an umbrella review of meta-analyses of observational studies. Cancer Med 9(21):7964–7978
Borggreve AS, den Boer RB, van Boxel GI et al (2020) The predictive value of low muscle mass as measured on CT scans for postoperative complications and mortality in gastric cancer patients: a systematic review and meta-analysis. J Clin Med 9(1):199
Yang Z, Zhou X, Ma B et al (2018) Predictive value of preoperative sarcopenia in patients with gastric cancer: a meta-analysis and systematic review. J Gastrointest Surg 22(11):1890–1902
Albano D, Messina C, Vitale J, Sconfienza LM (2020) Imaging of sarcopenia: old evidence and new insights. Eur Radiol 30(4):2199–2208
Prado CM, Lieffers JR, McCargar LJ (2008) Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol 9(7):629–635
Surov A, Pech M, Gessner D et al (2021) Low skeletal muscle mass is a predictor of treatment related toxicity in oncologic patients. A meta-analysis Clin Nutr 40(10):5298–5310
Jia S, Qiao R, Xiao Y et al (2020) Prognostic value of sarcopenia in survivors of hematological malignances undergoing a hematopoietic stem cell transplantation: a systematic review and meta-analysis. Support Care Cancer 28(8):3533–3542
Abrey LE, Batchelor TT, Ferreri AJ et al (2005) Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol 23(22):5034–43
Cox JD, Stetz J, Pajak TF (1995) Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys 31(5):1341–1346
Saravana-Bawan B, Goplen M, Alghamdi M, Khadaroo RG (2021) The relationship between visceral obesity and post-operative complications: a meta-analysis. J Surg Res 267:71–81
Houillier C, Soussain C, Ghesquières H et al (2020) Management and outcome of primary CNS lymphoma in the modern era: an LOC network study. Neurology 94(10):e1027–e1039
Fu F, Sun X, Li Y et al (2021) Dynamic contrast-enhanced magnetic resonance imaging biomarkers predict chemotherapeutic responses and survival in primary central-nervous-system lymphoma. Eur Radiol 31(4):1863–1871
Leone R, Sferruzza G, Calimeri T et al (2021) Quantitative muscle mass biomarkers are independent prognosis factors in primary central nervous system lymphoma: the role of L3-skeletal muscle index and temporal muscle thickness. Eur J Radiol 143:109945
Furtner J, Nenning KH, Roetzer T et al (2021) Evaluation of the temporal muscle thickness as an independent prognostic biomarker in patients with primary central nervous system lymphoma. Cancers (Basel) 13(3):566
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(4):601
Besutti G, Massaro F, Bonelli E et al (2021) Prognostic impact of muscle quantity and quality and fat distribution in diffuse large B-cell lymphoma patients. Front Nutr 8:620696
Gohmann RF, Temiz B, Seitz P et al (2021) Segmentation and characterization of visceral and abdominal subcutaneous adipose tissue on CT with and without contrast medium: influence of 2D- and 3D-segmentation. Quant Imaging Med Surg 11(10):4258–4268
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Alexey Surov.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the institutional review board.
Ethical approval
Institutional review board approval was obtained.
Study subjects or cohorts overlap
No study subjects or cohorts have been previously reported.
Methodology
• retrospective
• observational
• performed at one institution
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Surov, A., Meyer, H.J., Hinnerichs, M. et al. CT-defined sarcopenia predicts treatment response in primary central nervous system lymphomas. Eur Radiol 34, 790–796 (2024). https://doi.org/10.1007/s00330-023-09712-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-09712-y