Abstract
Objectives
To evaluate the clinical efficacy of magnetic resonance-guided focused ultrasound surgery in a Mexican mestizo population.
Methods
This retrospective study included 159 women (mean age 37 ± 6.4 years, range 22–53 years) from 2008 to 2010. Two hundred sixty-eight symptomatic uterine fibroids were treated using MR-guided focused ultrasound surgery. Parameters included initial perfused volume, final perfused volume, non-perfused volume (NPV), and treated volume ratio (TVR). Follow-up up to 15 months assessed treatment efficacy and symptomatic relief. Non-parametric statistics and the Kaplan-Meier method were performed.
Results
T2-weighted hypointense fibroids showed a frequency of 93.6 %; isointense and hyperintense fibroids had frequencies of 5.60 and 1.1 %. There was a negative correlation between NPV and age (r = -0.083, p = 0.307) and treatment time (r = -0.253, p = 0.001). Median TVR was 96.0 % in small fibroids and 76.5 % in large fibroids. Involution of 50 % and 80 % was achieved at months 6–7 and month 11, respectively. Relief of symptoms was significant (p < 0.05).
Conclusions
Our data show that higher TVR attained immediately post-treatment of MRgFUS favours higher involution percentages at follow-up; however, careful patient selection and use of pretreatment imaging are important components for predicting success using MR-guided focused ultrasound surgery.
Key Points
• Type 1 fibroids were the most common (93.2 %).
• Age and treated volume were not correlated (r s = -0.215, p = 0.165).
• Small fibroids achieved a higher treated volume than large (96.0 % vs. 76.5 %).
• A 50 % involution was achieved at 6-month follow-up for type-1 fibroid.
• A decrease of 80 % was reached at 11 months for type-1 fibroids.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Uterine fibroids (also known as leiomyomas or myomas) are a major cause of morbidity in women of reproductive age (and sometimes even after menopause) [1]. The incidence of uterine fibroids among African-American women has been reported as high as 60 % by age 35, increasing to 80 % by age 50, whereas 40 % has been reported for white women to age 35, rising to almost 70 % by age 50 [2]. The incidence of fibroids in infertile patients is estimated at 1–2.4 %, and the resultant endometrial distortion is associated with impaired implantation and increased risk of miscarriage [3]. Symptoms include menorrhagia, dysmenorrhoea, pelvic pain and bulk-related symptoms, leg and back pain, pain during sexual intercourse, increased urinary frequency, constipation, abnormally enlarged abdomen, and infertility [4, 5].
At present, there are several alternatives for the treatment of symptomatic fibroids, including medical, surgical, and minimally invasive techniques (MIT) [1]. MIT is a group of uterus-sparing alternatives [6] that includes the following: uterine artery embolization (UAE); magnetic resonance-guided focused ultrasound surgery (MRgFUS); ultrasound-guided ablation (VizAblate®; Gynesonics, Redwood City, CA, USA), a new device that combines real-time intrauterine sonography with radiofrequency (RF) ablation; and the Acessa™ (Halt Medical, Inc., Brentwood, CA, USA), a radiofrequency ablation device used under laparoscopic and intra-abdominal ultrasound guidance. Thus far, there have been no formal comparisons among these techniques [1].
MRgFUS is the application of high-intensity focused ultrasound with the -guidance of magnetic resonance imaging (MRI) [5]. It is a method of thermal ablation for treating fibroids that uses high-intensity focused ultrasound, which passes through the anterior abdominal wall and converges into a precise target point within the fibroid, causing a rise in temperature (55–90 °C) sufficient to induce coagulative necrosis within a few seconds. Concurrent MRI allows accurate tissue targeting and real-time temperature feedback, thereby achieving controlled localized thermal ablation [7–9].
MRgFUS is a Food and Drug Administration (FDA)-approved therapy for uterine leiomyomas; it has been accepted as a safe and feasible alternative to surgical resection for the treatment of uterine fibroids, demonstrating sustained treatment efficacy [7–12].
Because there are no available studies with this modality of treatment involving Mexican mestizo subjects, the purpose of this study was to determine the clinical efficacy of this non-invasive modality for the treatment of symptomatic uterine fibroids and to calculate the percentage of involution in this group of patients.
Patients and methods
Patients
This retrospective study included 159 consecutive women with 268 symptomatic uterine fibroids referred by the gynaecology clinic of our hospital from January 2008 to October 2010. They were first clinically examined by a gynaecologist, followed by an ultrasound examination of the pelvis. Based on their findings, they were subjected to screening MRI in order to assess the feasibility for MRgFUS treatment. The screening MRI consisted of multiplanar T2-weighted images, as previously described [13]; it was not used as the baseline MRI due to health insurance constrains. The time span between the baseline MRI and the MRgFUS was 3–4 weeks.
Clinical symptoms and health-related quality of life were recorded at the time of recruitment for the study and after treatment at 1-, 3-, and 6-month follow-up using a symptom severity score (SSS) and a uterine fibroid symptom and quality of life (UFS–QoL) questionnaire, as previously described [14]. The study was approved by the institutional review board, and written informed consent was obtained, according to the principles of good clinical practice as defined by the Declaration of Helsinki.
The eligibility criteria have been previously described [7, 15]. The inclusion criteria were as follows: age above 18 years, weight less than 110 kg, not more than four fibroids, fibroid size not more than 10 cm, and fibroid location not more than 12 cm from the anterior abdominal wall and at least 4 cm away from bone and nerve bundles. The exclusion criteria included pregnancy, massive scarring over the lower abdomen, pelvic or systemic disease, contraindications for MRI such as cardiac pacemakers, calcified fibroids, and pedunculated fibroids.
Equipment, patient preparation and treatment
The MRgFUS system (ExAblate 2000; InSightec, Haifa, Israel) integrates a standard 1.5 Tesla MRI system (GE Medical Systems, Milwaukee, WI, USA) with the focused ultrasound treatment transducer in the patient table. The treatment protocol has been previously described [16]. Selected patients reported to the MRI unit in a fasting state. An intravenous line and Foley catheter were inserted. Conscious sedation was administered with fentanyl citrate.
Pretreatment MRI sequences included multiplanar T2-weighted imaging (T2-w), and axial and sagittal liver acquisition with volume acquisition (LAVA; a three-dimensional spoiled gradient-echo sequence that includes an optimized inversion pulse and fat suppression). The post-treatment protocol comprised multiplanar T1-weighted (T1-w) post gadolinium imaging, and follow-up included: multiplanar T2-w imaging and axial LAVA. MRgFUS was performed with patients in a prone position on a modified MR gantry. The transducer was situated within a bath of degassed water in the midsection of the table. This allowed a direct acoustic pathway from the transducer into the target fibroid, which was positioned directly above. To safely carry out MRgFUS, an adequate acoustic window was created for the sound wave pathway that did not traverse any abdominal wall scars or bowel loops. The goal of the therapy was the ablation of a maximum fibroid volume without damage to surrounding structures [5]. At the end of the treatment, all patients underwent contrast-enhanced T1-w MRI with intravenous administration of gadolinium (gadopentetate dimeglumine). The volume of treated fibroids was calculated from MRI images immediately post-treatment and compared with baseline T2-w images [15] (Figs. 1 and 2). Patients were scheduled for follow-up visits 3, 6, 9, 12, and 15 months after treatment. A more detailed description of the procedures involved in MRgFUS has recently been published [15].
The MRgFUS method. A temperature-sensitive phase-difference fast spoiled gradient-echo MR images acquired during sonications. B T2-w anatomic guidance. C magnitude (A) and anatomic (B) images paired with a temperature chart provide the physician information about the clinical orientation of the targeted fibroid and the thermal doses
Measured volumes during MRgFUS treatment (different patients) A sagittal T2-weighted image used for planning of treatment and measuring initial fibroid volume at T2-w (T2-V). B post-gadolinium perfused regions seen pre-treatment on T1-weighted image (initial perfused volume, INP). C T1-weighted post-gadolinium image depicts the non-perfused regions seen post-treatment (non-perfused volume, NPV)
Data collection and volume data analysis
Patient age, body mass index (BMI; kg/m2), race, total number of fibroids, total fibroid volume (cm3), and volume of the treated fibroid(s) (cm3) were all recorded. Fibroids were classified according to their dominant signal intensity on T2-w images into three types distinguished by fibroid composition [4]: type 1, hypointense to myometrium; type 2, isointense to myometrium; and type 3, hyperintense relative to skeletal muscle [4]. We obtained measures of four different volumes: the T2-weighted imaging volume (T2-V) of the fibroid represented the initial volume in a non-enhanced sequence; the T1-weighted post-gadolinium-enhanced fibroid (initial perfused volume before treatment, IPV); the non-perfused volume of each fibroid after MRgFUS treatment, which was considered the actual treated volume (non-perfused volume, NPV); and the residual T1-weighted post-gadolinium-enhanced tissue after MRgFUS treatment (final perfused volume, FPV). We calculated the treated volume ratio (TVR) by dividing (NPV/ IPV) × 100, and presented it as a percentage. In cases of patients who received ablation in more than one fibroid, the NPV in the study was the total NPV achieved following the additional MRgFUS treatments. Volumes were calculated using FuncTool Performance software (release 4.3, GE Medical Systems). Random combinations of two radiologists from a set of three (Y.F.L., I.V.B., and C.R.T.) evaluated the dataset pre- and post-treatment; kappa statistics using pairwise comparisons were higher than 80 %, which meant substantial, almost perfect agreement. Radiologists who performed the MRgFUS treatment were blinded to supplementary data such as improvement in symptoms.
Statistical analysis
Data are presented as mean ± SD for normally distributed variables, and median and interquartile range (IQR) for non-normally distributed variables. After descriptive statistics, in order to evince significant association in the fibroids features, the entire group of patients was first divided into two subgroups based on the median value of the T2-V. A chi-square test evaluated the associations between the fibroid’s size and the location, treatment time and TVR. Comparison of fibroid volumes was performed with the Wilcoxon signed-rank test. A Pearson or Spearman correlation coefficient was used to assess the degree of association between variables. NPV and minutes of treatment were compared with age; r values close to or above 0.75 signified very good to excellent agreement [17]. Fibroid volumes were globally compared using the Friedman test; the Mann-Whitney U test evaluated the TVR within the independent groups of patients presenting small or large fibroids [18].
We assessed the involution of fibroids to evaluate the percentage of change in fibroid size between MRgFUS and follow-up using the Kaplan-Meier method and the log-rank test to compare the involution curves between subgroups [19]. Survival curves were drawn after grouping the patients based on fibroid signal intensity (types 1 and 2) and location (submucosal, intramural, subserosal). This method calculated the probability of involution based on fibroid size in each subgroup. The Wilcoxon signed-rank test was also used for statistical comparison of mean SSS values between baseline and follow-up visits. All analyses were carried out using IBM SPSS Statistics software (Version 22.0.0.0; IBM Corp., Armonk, NY, USA). Statistical significance was indicated by p < 0.05 (two-tailed).
Results
Demographics and fibroid features
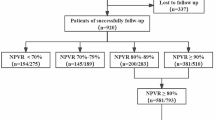
The study was conducted in 159 patients (mean age 37 ± 6.4 years, range 22–53 years), with a total of 268 evaluated fibroids. The average treatment time was 263.78 minutes (4.3 hours). No serious adverse events were reported. Most women presented only one fibroid (58.2 %). Two fibroids were observed in 19.6 %, and three and four fibroids were found in 8.5 and 5.9 % of patients, respectively. Five and six fibroids were found in 3.3 % of cases (Table 1).
Based on their signal intensity on T2-w imaging type 1 fibroids were the most common (250). The majority depicted an intramural location; their subserosal and submucosal locations were less frequent. Type 2 and 3 fibroids were rare (15 and 3 fibroids, respectively). The Chi-square test for independence indicated no significant association between the location and signal intensity of the fibroids, χ2 (4, n = 268) = 1.985, p = 0.738.
A median fibroid size of 31.0 cm3 on the initial T2-w allowed us to classify them as small or large. There was no significant association between size (small vs. large) and location, χ2 = 0.074, p = 0.964; and treatment time, χ2 = 39.873, p = 0.476. However comparison of size (small vs. large) with the TVR (thermal ablation percentage) showed an almost significant trend, χ2 = 35.897, p = 0.073.
The relationship between NPV and age was investigated using Spearman’s correlation coefficient, and a weak negative correlation was found between the two variables, r = –0.083, p = 0.307. For type 1 fibroids, we found a weak positive but no significant correlation between age and T2-v (r s = 0.137, p = 0.382), and a weak negative correlation between age and treated volume ratio (r s = –0.215, p = 0.165). In type 2 fibroids, there was a moderate correlation between age and T2-V (r s = –0.675, p = 0.066) and a weak positive association between age and TVR (r s = 0.048, p = 0.910). The relationship between NPV and treatment time showed a small but significant correlation between the variables, r = –0.253, p = 0.001.
Fibroid volumes before and after MRgFUS and treated volume ratio
The results of the Friedman test proved a statistically significant difference among the fibroid volumes across the four volumetric measurements (T2-V, IPV, NPV, and FPV), χ2 (3) = 407.844, p < 0.001. Comparison of the median volumes also showed significant difference between T2-V (Md = 31) and IPV (Md = 31), with decreased volume in the NPV (Md = 25.5) and FPV (Md = 3.0) after the thermal ablation procedure.
The Mann–Whitney U test revealed a significant difference in the TVR of small fibroids (Md = 96.0 %) compared with large fibroids (Md = 76.5 %), with a medium effect size, U = 184.0, z = –2.463, p = .014, r = 0.34. We also compared the change in TVR from immediately after MRgFUS to the follow-up, grouped by fibroid size; the Wilcoxon signed-rank test revealed a statistically significant reduction in TVR after MRgFUS in both small fibroids z = –4.705, p < .001, and large fibroids z = –3.621, p < .001, with a large effect size (r = 0.84 and r = 0.85, respectively). Table 2 depicts fibroid volumes before and after MRgFUS treatment.
Relationship between total fibroid volume per patient and duration of treatment
The total fibroid volume per patient and treatment time (minutes) showed a normal distribution; the correlation between treatment time and size was approximately 1 cm3 to 1.84 minutes. Fibroid location, region of treatment, length of sonication, and homogeneity of signal intensity were among the major factors determining treatment time. However, multivariate analysis of the weighted effect for each factor was not calculated, as this was beyond the scope of this study.
Percentage of involution and correlation of TVR with follow-up size
Based on the initial size measured on T2-w, type 1 fibroids attained a 50 % involution between the sixth and seventh months of follow-up, while type 2 fibroids achieved involution earlier, at months 5 to 6. A decrease of 80 % (fibroid involution up to 20 % of the original size) was reached at 11 and 13 months for fibroids type 1 and type 2, respectively. There was no statistical difference between the involution curves, χ2 (1) = 0.044, p = 0.834; Fig. 3.
Percentage of involution in fibroid size grouped by signal intensity. Type 1 fibroids attained 50 % involution at 6–7 months of follow-up, while involution was earlier for type 2 fibroids, at months 5 and 6. A decrease of 80 % was reached at 11 and 13 months for fibroids type 1 and type 2, respectively
When assessed by location, a target involution of 50 % was achieved between 6 and 8 months for the three locations; involution of intramural and subserosal fibroids occurred earlier than the submucosal type. A 20 % involution was reached at 11, 12, and 13 months for intramural, subserosal, and submucosal fibroids, respectively; these involution curves were not statistically different, χ2 (2) = 2.006, p = 0.367; Fig. 4.
Percentage of involution in fibroid size grouped by location. Target involution of 50 % was observed at 6–8 months for the three locations; involution was earlier for intramural and subserosal fibroids than the submucosal type. A 20 % involution was reached at 11, 12, and 13 months for intramural, subserosal, and submucosal fibroids respectively
There was a significant negative correlation between the TVR at the end of the MRgFUS and the fibroid size at the follow-up, Spearman’s rho = -0.348, p = 0.012. Approximately 31.4 % of patients completed a 15-month follow-up; 54.7 % completed 9–12 months of follow-up. There was an average of three studies per patient.
Symptom improvement
Most of the symptoms associated with the fibroids were relieved after MRgFUS (dysmenorrhoea, number of sanitary pads, abdominal pressure, urinary frequency, abdominal pain, sexual life affectation, and constipation), with a significant difference versus the baseline scale. Grading of fatigue, social life affectation, and abdominal bulk did not reflect significant differences when compared with baseline (Table 3 and Fig. 5). The TVR showed significant correlation in only three of ten relieved symptoms during the follow-up surveys—number of sanitary pads (p = 0.018), abdominal pressure (p = 0.011), and urinary frequency (p = 0.026) —and an almost significant correlation in dysmenorrhoea (p = 0.056).
Discussion
MRgFUS has been a contributor to declining hysterectomy rates in recent years as part of a growing array of minimally invasive alternatives to hysterectomy for treatment of fibroids (other procedures consider laparoscopic, hysteroscopic, and robotic myomectomy, endometrial ablation, uterine artery embolization, and medical treatment) [12, 13]. A recent study found that hysterectomy in women under the age of 50 years substantially increased the risk of cardiovascular disease later in life [20].
Magnetic resonance imaging, while more costly than other imaging methods such ultrasound, has been touted as the most sensitive modality for evaluating uterine myomas, particularly for the detection of small fibroids [21]. MRI has been shown to be accurate in the diagnosis of leiomyomas, with sensitivity of 88–93 % and specificity of 6–91 %, and in differentiating leiomyomas from focal adenomyosis [22].
In the last five years, there have been an increasing number of groups reporting longitudinal studies focusing on fibroid shrinkage and patient symptomatic relief [23–26]. We found a weak negative correlation between age and NPV and between age and TRV; these results support the recent finding that patient age does not affect clinical success [27], implying that NPV—and consequently TRV—represents the dominant factor for clinical success. Immediately post-treatment, the TVR in our study (grouping both small and large fibroid size) was 83.1 % ± 22.22, which is higher than values stated in earlier reports [26, 28], but similar to recent results obtained in centres in Europe [27] and Asia [5]. Another interesting finding in this study was the positive correlation between the TVR and the percentage of involution of fibroids at different follow-up visits. We noticed that the higher the TVR attained immediately post-treatment with MRgFUS, the higher the involution expected at the follow-up. These findings agree with the increasing body of evidence showing that most of the fibroid volume should be ablated in order to achieve lasting results [29].
We agree with remarks by Yoon et al [26] who noted that in early studies, the strict FDA regulations at the time allowed treating physicians to target only a small portion of the fibroids within a predefined time frame, resulting in the modest NPV ratios attained during the initial stages of those studies. Regulations also limited the scope of treatment and allowed only up to 50 % of the fibroid volume to be targeted. Without these constraints in our procedures, the probability of attaining 80 % involution in fibroid size was calculated at 12 months; this figure surpassed the shrinkage rates of 9–32 % reported in previous works [23, 26, 30]. The mean TVR (up to 96 %) and percentage of involution at 6 months (50 %) in our study are similar to a recent report by Himabindu et al. [5] (70 % and 40 %, respectively) conducted outside the United States. The relevance of increased ablation volumes in recent studies is evidence of increased involution expectancy at several follow-ups; this was a missed opportunity in older studies with more stringent FDA limitations at the time.
In agreement with previous reports, our study adds to a growing body of evidence showing that high NPV levels immediately post-treatment significantly correlate with smaller involution sizes (that is, higher shrinkage rates) at follow-up visits [29]. Considering the clinical outcome for symptom relief, the evaluation of the SSS can have different statistical implications; our patients experienced a significant reduction of symptoms and underwent continuous improvement over the course of the first year. Consistent with recent reports [5, 27], improvements were observed in the categories of pain- and bleeding-related symptoms.
Some limitations in this study should be addressed. Our study design was retrospective, and our patients were not enrolled in a clinical trial. This posed difficulties in achieving the scheduled follow-up in all patients, as many patients returned earlier, while the first follow-up visit for others was after the first year. We took advantage of this situation, and used the Kaplan–Meier method to calculate the probability of involution. We did not calculate the risk of retreatment [27], as this condition was not presented in the studied sample. Because we did not include the thermal dose volume (TDV) in our analysis, we used the TVR as an estimator of treatment effectiveness. Those patients who achieved 80 % shrinkage at the 12- and 15-month follow-up visits proved that it was safe to increase ablation percentage levels. The calculated probability of involution in our data suggests that it will have a positive effect on the long-term outcome of this treatment modality. Some associations between variables would require further investigation to determine whether certain laboratory biomarkers could produce moderation. For instance, the categorization of fibroids as hyperintense or hypointense based upon T2-w signal intensity correlates with vascularization [31], and is responsive to a gonadotrophin-releasing hormone (GnRH) agonist pretreatment, potentially improving MRgFUS success [32]. Therefore, the type of fibroid influences the NPV, resultant fibroid size, and SSS, with reduced values found in patients with hyperintense fibroids [4, 30].
We justified the presentation of results in the type 2 fibroid subgroup (a small sample size of 15) by using non-parametric statistics. Readers, however, should be cautious in drawing conclusions based on this small subgroup. Further investigation is needed comparing MRgFUS with existing alternative uterus-saving methods, mainly myomectomy and uterine fibroid embolization (UFE). A prospective randomized controlled trial would help to provide clarity. Furthermore, long-term follow-up (i.e. follow-up after at least 3 years) is lacking in the current study. Thus far, 24-month follow-up studies show approximately 20 % lower recurrence rates when the treated volume was around 60 % of the fibroid volume [33, 34]. We did not include data on re-intervention and pregnancy rates in our population, as this was beyond the scope of the study; a future study by our group will address this research line.
In conclusion, MRgFUS represents an innovative technology for the minimally invasive treatment of fibroids. Our data support the ability of MRgFUS to both improve quality of life and reduce fibroid size. These data indicate that at 6 months, it is possible to achieve a 50 % reduction in fibroid size; however, careful patient selection and use of pretreatment imaging remain important components for predicting the success of MRgFUS.
References
Khan AT, Shehmar M, Gupta JK (2014) Uterine fibroids: current perspectives. Int J Womens Health 6:95–114
Baird DD, Dunson DB, Hill MC, Cousins D, Schectman JM (2003) High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol 188:100–107
Hanstede MM, Tempany CM, Stewart EA (2007) Focused ultrasound surgery of intramural leiomyomas may facilitate fertility: a case report. Fertil Steril 88:e495–e497
Funaki K, Fukunishi H, Funaki T, Sawada K, Kaji Y, Maruo T (2007) Magnetic resonance-guided focused ultrasound surgery for uterine fibroids: relationship between the therapeutic effects and signal intensity of preexisting T2-weighted magnetic resonance images. Am J Obstet Gynecol 196:e181–e186
Himabindu Y, Sriharibabu M, Nyapathy V, Mishra A (2014) Early evaluation of magnetic resonance imaging guided focused ultrasound sonication in the treatment of uterine fibroids. Indian J Med Res 139:267–272
van der Kooij SM, Ankum WM, Hehenkamp WJ (2012) Review of nonsurgical/minimally invasive treatments for uterine fibroids. Curr Opin Obstet Gynecol 24:368–375
Stewart EA, Gedroyc WM, Tempany CM et al (2003) Focused ultrasound treatment of uterine fibroid tumors: safety and feasibility of a noninvasive thermoablative technique. Am J Obstet Gynecol 189:48–54
Tempany CM, Stewart EA, McDannold N, Quade BJ, Jolesz FA, Hynynen K (2003) MR imaging-guided focused ultrasound surgery of uterine leiomyomas: a feasibility study. Radiology 226:897–905
Hindley J, Gedroyc WM, Regan L et al (2004) MRI guidance of focused ultrasound therapy of uterine fibroids: early results. AJR Am J Roentgenol 183:1713–1719
Stewart EA, Rabinovici J, Tempany CM et al (2006) Clinical outcomes of focused ultrasound surgery for the treatment of uterine fibroids. Fertil Steril 85:22–29
Fennessy FM, Tempany CM, McDannold NJ et al (2007) Uterine leiomyomas: MR imaging-guided focused ultrasound surgery–results of different treatment protocols. Radiology 243:885–893
Leslie TA, Kennedy JE (2007) High intensity focused ultrasound in the treatment of abdominal and gynaecological diseases. Int J Hyperth 23:173–182
Vazquez-Lamadrid J, Fernandez-de Lara Y, Carrasco-Choque A, Romero-Trejo C, Cosme-Labarthe J, Roldan-Valadez E (2011) Principles and technique of magnetic resonance guided focused ultrasound surgery (MRgFUS) in the treatment of uterine fibroids. Gac Med Mex 147:333–341
Spies JB, Coyne K, Guaou Guaou N, Boyle D, Skyrnarz-Murphy K, Gonzalves SM (2002) The UFS-QOL, a new disease-specific symptom and health-related quality of life questionnaire for leiomyomata. Obstet Gynecol 99:290–300
Machtinger R, Fennessy FM, Stewart EA, Missmer SA, Correia KF, Tempany CM (2013) MR-guided focused ultrasound (MRgFUS) is effective for the distinct pattern of uterine fibroids seen in African-American women: data from phase III/IV, non-randomized, multicenter clinical trials. J Ther Ultrasound 1:23
Rabinovici J, Inbar Y, Revel A et al (2007) Clinical improvement and shrinkage of uterine fibroids after thermal ablation by magnetic resonance-guided focused ultrasound surgery. Ultrasound Obstet Gynecol 30:771–777
Pallant J (2011) Correlation. In: Pallant J (ed) SPSS survival manual, 4th edn. Allen & Unwin, Crows Nest, pp 128–142
Pallant J (2011) Non-parametric statistics. In: Pallant J (ed) SPSS survival manual, 4th edn. Allen & Unwin, Crows Nest, pp 213–238
Stel VS, Dekker FW, Tripepi G, Zoccali C, Jager KJ (2011) Survival analysis I: the Kaplan-Meier method. Nephron Clin Pract 119:c83–c88
Ingelsson E, Lundholm C, Johansson AL, Altman D (2011) Hysterectomy and risk of cardiovascular disease: a population-based cohort study. Eur Heart J 32:745–750
Dueholm M, Lundorf E, Hansen ES, Ledertoug S, Olesen F (2002) Accuracy of magnetic resonance imaging and transvaginal ultrasonography in the diagnosis, mapping, and measurement of uterine myomas. Am J Obstet Gynecol 186:409–415
Byun JY, Kim SE, Choi BG, Ko GY, Jung SE, Choi KH (1999) Diffuse and focal adenomyosis: MR imaging findings. Radiographics 19:S161–170
Kim HS, Baik JH, Pham LD, Jacobs MA (2011) MR-guided high-intensity focused ultrasound treatment for symptomatic uterine leiomyomata: long-term outcomes. Acad Radiol 18:970–976
Funaki K, Fukunishi H, Sawada K (2009) Clinical outcomes of magnetic resonance-guided focused ultrasound surgery for uterine myomas: 24-month follow-up. Ultrasound Obstet Gynecol 34:584–589
Gorny KR, Woodrum DA, Brown DL et al (2011) Magnetic resonance-guided focused ultrasound of uterine leiomyomas: review of a 12-month outcome of 130 clinical patients. J Vasc Interv Radiol 22:857–864
Yoon SW, Cha SH, Ji YG, Kim HC, Lee MH, Cho JH (2013) Magnetic resonance imaging-guided focused ultrasound surgery for symptomatic uterine fibroids: estimation of treatment efficacy using thermal dose calculations. Eur J Obstet Gynecol Reprod Biol 169:304–308
Mindjuk I, Trumm CG, Herzog P, Stahl R, Matzko M (2014) MRI predictors of clinical success in MR-guided focused ultrasound (MRgFUS) treatments of uterine fibroids: results from a single centre. Eur Radiol. doi:10.1007/s00330-014-3538-6
Machtinger R, Inbar Y, Cohen-Eylon S, Admon D, Alagem-Mizrachi A, Rabinovici J (2012) MR-guided focus ultrasound (MRgFUS) for symptomatic uterine fibroids: predictors of treatment success. Hum Reprod 27:3425–3431
Okada A, Morita Y, Fukunishi H, Takeichi K, Murakami T (2009) Non-invasive magnetic resonance-guided focused ultrasound treatment of uterine fibroids in a large Japanese population: impact of the learning curve on patient outcome. Ultrasound Obstet Gynecol 34:579–583
Lenard ZM, McDannold NJ, Fennessy FM et al (2008) Uterine leiomyomas: MR imaging-guided focused ultrasound surgery–imaging predictors of success. Radiology 249:187–194
Yamashita Y, Torashima M, Takahashi M et al (1993) Hyperintense uterine leiomyoma at T2-weighted MR imaging: differentiation with dynamic enhanced MR imaging and clinical implications. Radiology 189:721–725
Smart OC, Hindley JT, Regan L, Gedroyc WG (2006) Gonadotrophin-releasing hormone and magnetic-resonance-guided ultrasound surgery for uterine leiomyomata. Obstet Gynecol 108:49–54
Hudson SB, Stewart EA (2008) Magnetic resonance-guided focused ultrasound surgery. Clin Obstet Gynecol 51:159–166
Tempany CM (2007) From the RSNA refresher courses: image-guided thermal therapy of uterine fibroids. Radiographics 27:1819–1826
Acknowledgments
The scientific guarantor of this publication is Ernesto Roldan-Valadez. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.This article was supported in part by Medica Sur Clinic and Foundation.
Ana Luz Carrasco-Choque, MD, was enrolled as clinical fellow of the whole-body MRI program during 2009-2010. Ingrid Vivas-Bonilla, MD, was attending radiologist at the MRI unit from 2001 to 2009. Cecilia Romero-Trejo, MD, was attending radiologist at the MRI unit from 2005 to 2010.
Antonio R. Villa MD, PhD, provided statistical advice for the preparation of this manuscript.
One of the authors has significant statistical expertise. Institutional Review Board approval was obtained. Methodology: retrospective.
Conflicts of interest
The authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carrasco-Choque, A.L., Lara, Y.Fd., Vivas-Bonilla, I. et al. Perfusion volume correlates, percentage of involution, and clinical efficacy at diverse follow-up survey times after MR-guided focused ultrasound surgery in uterine fibroids: first report in a Mexican mestizo population. Eur Radiol 25, 2905–2912 (2015). https://doi.org/10.1007/s00330-015-3707-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3707-2