Abstract
Objective
To investigate the diagnostic contribution of T2-w nerve lesions and of muscle denervation in peripheral motor neuropathies by magnetic resonance neurography (MRN).
Methods
Fifty-one patients with peripheral motor neuropathies underwent high-resolution MRN by large coverage axial T2-w sequences of the upper arm, elbow, and forearm. Images were evaluated by two blinded readers for T2-w signal alterations of median, ulnar, and radial nerves, and for denervation in respective target muscle groups.
Results
All 51 patients displayed nerve lesions in at least one of three nerves, and 43 out of 51 patients showed denervation in at least one target muscle group of these nerves. In 21 out of 51 patients, the number of affected nerves matched the number of affected target muscle groups. In the remaining 30 patients, T2-w lesions were encountered more frequently than target muscle group denervation. In 153 nerve-muscle pairs, 72 showed denervation, but only one had increased muscle signal without a lesion in the corresponding nerve.
Conclusions
MRN-based diagnosis of peripheral motor neuropathies is more likely by visualization of peripheral nerve lesions than by denervation in corresponding target muscles. Increased muscular T2-w signal without concomitant nerve lesions should raise suspicion of an etiology other than peripheral neuropathy.
Key Points
• In peripheral neuropathy, T2-w nerve lesions are more frequent than muscle denervation.
• Muscle denervation almost never occurs without detectable lesions in corresponding nerves.
• MRN-aided diagnosis of peripheral motor neuropathy should focus primarily on nerve lesions.
• Increased muscular T2-w signal intensity without concomitant nerve lesions indicates other aetiology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In peripheral neuropathies with involvement of motor fascicles, denervation of affected muscles is displayed by a pathological increase in the T2-weighted (T2-w) signal intensity [1], which is attributed to increased levels of extracellular fluid [2, 3]. The pattern of muscle denervation has long been used as an indirect sign for the diagnosis of peripheral motor neuropathy by magnetic resonance imaging (MRI) [4–7]. High-resolution magnetic resonance neurography (MRN) now allows for direct depiction of nerve lesions [8–12]. These lesions of different aetiologies all present with an increased T2-w signal as a sensitive albeit unspecific sign of neuropathy, and can be detected with high precision by visual assessment of T2-w images [8, 10, 13–16].
Patients with an initial diagnosis of peripheral neuropathy with involvement of motor fascicles and muscle paresis are frequently referred for MRN examination for diagnostic confirmation by imaging. This procedure can be a useful complement to clinical examination and electrophysiological testing, as MRN may better determine the localization and extent of nerve lesions [16] and may also detect lesions in clinically unaffected nerves [17]. Further, cross-sections of forearm musculature contain the majority of muscles innervated by median, ulnar, and radial nerves, whereas electromyography (EMG) is invasive and can examine only a limited number of muscles.
If muscle denervation is due to peripheral neuropathy, lesions should be expected to occur in a nerve before the resulting denervation in its respective target muscles. Therefore, nerve lesions on MRN might be expected to be a more sensitive sign of peripheral neuropathy with motor involvement. In electrophysiological studies, however, the contrary is the case: EMG is often a more sensitive parameter than nerve conduction studies (NCS) [18–21]. In denervated muscle, EMG findings closely correspond to the increased T2-w signal on MRI [22]. By analogy, it may also be possible in MRN that muscle denervation represents a more sensitive sign of motor neuropathy than the presence of nerve lesions.
In this study, we aimed to address this question by testing whether nerve lesions or muscle denervation patterns were more frequently detected in patients with peripheral neuropathy with involvement of motor axons.
Materials and methods
Clinical and demographic patient data
The study was approved by the institutional ethics board (University of Heidelberg Ethics Committee; S-057/2009), and written informed consent was obtained from all participants. Patients were examined at the Department of Neuroradiology of Heidelberg University Hospital in Germany between January 2012 and April 2013, at least four weeks after onset of symptoms. Patients were included in our study based on a clinical and electrophysiological diagnosis of peripheral motor neuropathy, i.e., clinically diagnosed weakness, atrophy, and/or positive electrophysiological findings suspicious of motor neuropathy. Exclusion criteria were (previous or subsequent) diagnosis of a disease other than peripheral neuropathy as a possible cause of muscle weakness, e.g., spinal canal stenosis, myopathy, or motor neuron disease. Furthermore, the typical compressive neuropathies of carpal tunnel syndrome and ulnar neuropathy at the elbow were excluded, as the protocol did not systematically include coverage of hand muscles. Sensory co-involvement was not an exclusion criterion, since most peripheral neuropathies affect both motor and sensory fascicles. A total of 51 patients fulfilled the criteria and were included in the study.
MRN imaging
Examinations were conducted using a 3 Tesla unit (MAGNETOM Verio, Siemens AG, Erlangen, Germany). Subjects were examined in the prone position with the arm extended at the upper arm, the elbow, and the forearm, placed in a knee 8-channel phased array coil. Coverage comprised the upper arm, the elbow region, and the forearm in two image slabs. A third image slab was acquired in 24 of 51 cases because the forearm musculature was not sufficiently covered by the second image slab. If the brachial plexus, distal forearm, or wrist were also imaged as part of the clinical exam protocol, these additional images were not included in our analysis. To avoid any significant artificial signal increase in a T2-w sequence related to the so-called magic angle effect at the elbow [23], the longitudinal axis of the upper arm was aligned at an angle of ≤10° relative to the B0 field direction.
The sequence parameters were as follows: transversal T2-w turbo spin-echo TR/TE 7,020/52 ms, spectral fat saturation, parallel imaging (GRAPPA 2, reference lines PE 32), slice thickness 3.0 mm, number of slices 45, interslice gap 0.3 mm, FoV 130 × 130 mm2, acquisition matrix 512 × 358, pixel spacing 0.254 × 0.254 mm2, number of excitations = 3, acquisition time 7:17 min each sequence, resulting in a total acquisition time of 21:51 min.
Image analysis
Qualitative evaluation in a proximodistal direction of nerve and muscle T2-w signal from patients was performed independently by two neuroradiologists (DS, PB), who were blinded to the patients’ clinical data. Standardized rating was performed in a dichotomous fashion, either as “affected” or as “non-affected,” with regard to the following items:
-
1.
Presence of peripheral nerve lesions in the a) median nerve, b) ulnar nerve, and c) radial nerve at the upper arm and elbow level.
-
2.
Presence of muscle denervation in any muscle within the target muscle groups (according to [4]) of a) median nerve (Mm. pronator teres, flexor carpi radialis, palmaris longus, flexor digitorum superficialis, flexor digitorum profundus 2nd and 3rd, flexor pollicis longus and pronator quadratus), b) ulnar nerve (Mm. flexor carpi ulnaris and flexor digitorum profundus 4th and 5th), and c) radial nerve (Mm. triceps brachii, anconeus, brachioradialis, extensor carpi radialis longus and brevis, supinator, extensor carpi ulnaris, extensor digitorum, extensor digiti minimi, abductor pollicis longus, extensor pollicis longus and brevis, and extensor indicis).
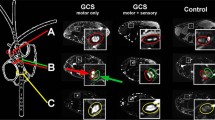
Rating was performed from proximal to distal (Fig. 1). Cohen’s κ, a statistical measure of inter-rater agreement, was calculated for the evaluation of inter-rater agreement. For further statistical analysis of the results of the ratings, final classification in the case of disagreement between the raters was achieved by a second, consensus-based evaluation.
Schematic of the imaging rationale. The left column shows a representative section of each image slab and the three assessed peripheral nerves of the upper extremity, coloured according to their identity (blue = median, red = ulnar, green = radial). Similarly, the right column shows the target muscle groups of each of the three nerves (same colour code). The location of the displayed images is indicated on the schematic of the upper extremity and the three nerves in the middle column
Additional quantitative analysis
In order to objectify the qualitative ratings of nerves, quantitative analysis of T2-w signals from nerves was performed by evaluation of the mean T2-w signal from a representative intraneural region-of-interest (nROI) of the three nerves (median, ulnar, radial). For nerves with qualitatively detectable nerve lesions, this was placed at a location representative for the lesion. For normal-appearing nerves, it was placed in a standard position of the unaffected nerve in the mid-portion of the upper arm (12 cm proximal to the humeroradial joint). Additional regions of interest were placed within adjacent non-denervated muscle (mROI), and the mean signal intensities were read out in order to calculate the normalized T2-w signal intensity, abbreviated normT2 (normT2 = normROI /mROI).
Statistical analysis
Based on qualitative ratings, two different categorizations were performed.
-
1.
Patients were grouped into one of the following three categories (Fig. 2):
-
1.
Patients with a greater number of affected nerves than target muscle groups
-
2.
Patients with the same number of affected nerves and target muscle groups
-
3.
Patients with a greater number of affected target muscle groups than nerves
Fig. 2 Classification of MRN findings. The upper panel shows a representative finding of an imaging case from group 1 exhibiting pathologic lesions in all three nerves (blue = median, red = ulnar, green = radial) while denervation only becomes evident in the median innervation territory (M. flexor digitorum superficialis). In the middle panel, a typical case from group 2 with pathologic T2-weighted signal elevation in the median nerve (blue) and the median-dependent target muscle group (Mm. flexor digitorum profundus and superficialis, flexor pollicis longus, flexor carpi radialis and palmaris longus). A case exhibiting affection of more target muscle groups than nerves (group 3) is shown in the lower panel. Here, pathologic T2-weigthed signal alterations are visible in all three target muscle groups, while nerve lesions can only be detected in the ulnar nerve. This patient was excluded, however, because he was later diagnosed with spinal canal stenosis
-
1.
-
2.
Classification of all three evaluated arm nerves and their corresponding target muscle groups in all 51 patients, resulting in 153 nerve-muscle pairs. Every nerve-muscle pair was likewise grouped into one of three categories:
-
1)
Nerve lesion without concomitant muscle denervation in its target muscle group
-
2)
Nerve lesion with concomitant muscle denervation in its target muscle group
-
3)
No nerve lesion present but presence of muscle denervation in its target muscle group
-
1)
Results of the quantitative analysis were plotted by graphs mapping normT2 values comparing affected and non-affected using MATLAB (MathWorks, release 2013a).
Results
All 51 patients exhibited nerve T2-w lesions in at least one of the three nerves evaluated. In contrast, muscle denervation at the upper arm or forearm level was detected in only 43 of 51 patients (84.3 %). Inter-rater agreement was high, with an overall Cohen’s κ of 0.92 (Table 1).
In 30 of 51 patients (58.8 %), T2-w signal alterations were detectable in a greater number of nerves than target muscle groups. In the other 21 patients (41.2 %), the number of affected nerves matched the number of target muscle groups with denervation (Fig. 3). There were no cases of a lower number of affected nerves than target muscle groups containing denervated muscles.
Qualitative assessment of pathological MRN findings. Diagram illustrating the number of patients in each of the three groups. In the majority of cases, affection of nerves was encountered more frequently than denervation in the corresponding target muscle groups. There were no cases showing affection of target muscles without an appreciable lesion in the nerve
Similar results were obtained when comparing individual nerves and their respective muscular innervation territory. Lesions of the median, ulnar, and radial nerves were found in 44, 45, and 29 cases, respectively, while muscle denervation in target muscle groups could only be detected in 30, 21, and 20 cases (Fig. 4). Of these total 153 nerve-muscle pairs, 47 (30.7 %) showed nerve lesions but no concomitant muscle denervation, 71 (46.4 %) exhibited both nerve lesions and corresponding muscle denervation, and only one pair (0.7 %) was found to have an increased T2-w muscular signal without a lesion in the corresponding nerve. In this case, a more proximal nerve lesion was found in the plexus, outside the study protocol. The remaining 34 pairs (22.2 %) had both normal nerve and muscle appearance.
Qualitative assessment of individual nerves and their respective target muscle groups. Separate assessment of the three nerves (N) and their respective target muscle groups (M) shows that detection of nerve lesions exceeds positive image findings in the target muscle groups. Among the three nerves of the upper extremity, the discrepancy between findings in nerve and target muscles is most pronounced for the ulnar nerve
To strengthen the qualitative assessment of nerve lesions, normalized T2-w signal intensity values from representative regions of interest of “affected” and “non-affected” arm nerves were compared (Fig. 5). A separation between pathological and normal nerves was observed at normT2 values of 1.69 for the median nerve, 1.79 for the ulnar nerve, and 1.80 for the radial nerve.
Quantitative evaluation of qualitatively assessed nerve lesions. Normalized T2-weighted signal values (normT2) were calculated for each of the three nerves and plotted in a percentage-wise fashion from lowest to highest signal intensity. Consistent with our qualitative assessment, normT2 values differed between “affected” and “non-affected” nerves. Most interestingly, we found similar cutoff values distinguishing the “affected” and “non-affected” categories for all three nerves: median = 1.69, ulnar = 1.79, radial = 1.8
Discussion
Diagnosis of peripheral neuropathy on MRN is based on signal alterations in affected nerves and muscles. In the present study, we showed that in patients with a clinical diagnosis of peripheral motor neuropathy, abnormal signal intensity on T2-w imaging was almost always found in at least one peripheral nerve, whereas corresponding muscle denervation was found less frequently.
It has long been the general perception that specific patterns of muscular denervation are the major hallmark for MRI diagnosis of peripheral motor neuropathies [4–7, 24]. Similarly, in electrophysiology, EMG is often more sensitive than NCS in demonstrating abnormalities in peripheral motor disease, due to technical reasons [18–21]. Our results for MR image analysis help to shift the diagnostic focus from muscular denervation to nerve lesions, and thereby have relevant implications for both MRN protocols and image evaluation.
As a consequence, for sequence protocols, MRN exams for clinically suspected peripheral neuropathy should focus primarily on direct detection of nerve lesions. Detection is more likely in regions where nerve trunks still contain all or most of their fascicles, i.e., the upper arm rather than the forearm [16, 17]. Determining the extent of peripheral nerves affected by a neuropathy is highly relevant in the classification and subsequent treatment of neuropathic disorders [25]. Visualizing denervation in target muscle groups confirms clinical motor affection but may not necessarily reflect the actual extent of peripheral nerve involvement.
A fundamental implication for image evaluation is that increased muscle T2-w signal intensity indicative of denervation is almost always associated with T2-w nerve lesions in peripheral neuropathies with involvement of motor fascicles. If such corresponding peripheral nerve lesions cannot be detected but pathologically increased T2-w signal in the musculature is present, underlying pathologies other than a peripheral neuropathic aetiology should be considered and carefully excluded.
How can the observed discrepancy between neural and muscular T2-w signal alterations be explained? One important aspect is that MR morphologic evidence of muscular denervation may represent a threshold phenomenon that is dependent on sufficiently severe axonal damage. In contrast, in the early stages of motor dominant neuropathy, T2-w hyperintense nerve signals may be caused simply by demyelination, and not necessarily accompanied by axonal damage. Further, the development of T2-w signal alterations is likely to follow a different time course in nerves versus muscles. While MRN exams in our study were all performed after clinical and electrophysiological examination, it may still have been too early in some cases for the development of muscular denervation in the innervation territory of all pathologic nerves.
A previous study examined the time course of muscular T2-w signal patterns after traumatic nerve lesions, and found good correlation with clinical recovery [26]. For less acute neuropathies, further investigation is needed with regard to the exact onset of symptoms and correlation with electrophysiology, although a study in the lower extremity found that the sensitivity of increased muscular T2-w signal intensity for denervation remained high regardless of duration, location, or severity of the neuropathy [27].
A number of limitations apply to this study. Image acquisition was not systematically paralleled by electrophysiological testing objectifying MRN images. However, studies have abundantly demonstrated the correlation of T2-w nerve lesions and pathological findings in electrophysiological exams. Second, the majority of patients included in the study were diagnosed with an inflammatory neuropathy, so the group is not completely representative of all patients examined by MRN. However, this is the group for whom these findings are most relevant, since assessment of the extent of disease is more difficult here than in neuropathies of compressive or neoplastic aetiology. Another limiting factor was that the hand region containing the distal target muscles of the median and ulnar nerves [4] was not routinely included in the standard analysis in our protocol. To minimize the effect of systematically underestimating the proportion of affected target muscle groups, we explicitly excluded cases with compressive neuropathies, as their denervation occurs in distal target muscles (e.g., carpal tunnel syndrome and ulnar neuropathy at the elbow). More importantly, for the radial nerve, the entire muscular innervation territory was systematically covered by our examination protocol, and radial nerve lesions were still found more frequently than radial nerve denervation. Finally, we used transversal 2D T2-w sequences for large coverage. The use of 3D T2-w, with its three-dimensional reconstruction possibilities, may further improve diagnostic accuracy for both nerves and muscles [28].
Taken together, our results suggest that peripheral neuropathy with involvement of motor fascicles is more frequently confirmed by detection of peripheral nerve lesions than denervation in the corresponding target muscle groups. Further, pathological muscle signals without concomitant nerve lesions should raise suspicion of an aetiology other than peripheral neuropathy.
References
Briani C, Cacciavillani M, Lucchetta M, Cecchin D, Gasparotti R (2013) MR neurography findings in axonal multifocal motor neuropathy. J Neurol 260:2420–2422
Kikuchi Y, Nakamura T, Takayama S, Horiuchi Y, Toyama Y (2003) MR imaging in the diagnosis of denervated and reinnervated skeletal muscles: experimental study in rats. Radiology 229:861–867
Polak JF, Jolesz FA, Adams DF (1988) Magnetic resonance imaging of skeletal muscle. Prolongation of T1 and T2 subsequent to denervation. Invest Radiol 23:365–369
Kim SJ, Hong SH, Jun WS et al (2011) MR imaging mapping of skeletal muscle denervation in entrapment and compressive neuropathies. Radiographics 31:319–332
Andreisek G, Crook DW, Burg D, Marincek B, Weishaupt D (2006) Peripheral neuropathies of the median, radial, and ulnar nerves: MR imaging features. Radiographics 26:1267–1287
Yamabe E, Nakamura T, Oshio K, Kikuchi Y, Ikegami H, Toyama Y (2008) Peripheral nerve injury: diagnosis with MR imaging of denervated skeletal muscle —experimental study in rats. Radiology 247:409–417
Lisle DA, Johnstone SA (2007) Usefulness of muscle denervation as an MRI sign of peripheral nerve pathology. Australas Radiol 51:516–526
Thawait SK, Chaudhry V, Thawait GK et al (2011) High-resolution MR neurography of diffuse peripheral nerve lesions. Am J Neuroradiol 32:1365–1372
Stoll G, Bendszus M, Perez J, Pham M (2009) Magnetic resonance imaging of the peripheral nervous system. J Neurol 256:1043–1051
Andreisek G, Burg D, Studer A, Weishaupt D (2008) Upper extremity peripheral neuropathies: role and impact of MR imaging on patient management. Eur Radiol 18:1953–1961
Bendszus M, Stoll G (2005) Technology insight: visualizing peripheral nerve injury using MRI. Nat Clin Pract Neurol 1:45–53
Koltzenburg M, Bendszus M (2004) Imaging of peripheral nerve lesions. Curr Opin Neurol 17:621–626
Baumer P, Mautner VF, Baumer T et al (2013) Accumulation of non-compressive fascicular lesions underlies NF2 polyneuropathy. J Neurol 260:38–46
Bäumer P, Dombert T, Staub F, Kaestel T, Bartsch AJ, Heiland S, Bendszus M, Pham M (2011) Ulnar neuropathy at the elbow MR neurography–nerve T2 signal increase and caliber. Radiology 260
Pham M, Oikonomou D, Baumer P et al (2011) Proximal neuropathic lesions in distal symmetric diabetic polyneuropathy: findings of high-resolution magnetic resonance neurography. Diabetes Care 34:721–723
Pham M, Baumer P, Meinck HM et al (2014) Anterior interosseous nerve syndrome: fascicular motor lesions of median nerve trunk. Neurology 82:598–606
Baumer P, Weiler M, Ruetters M et al (2012) MR neurography in ulnar neuropathy as surrogate parameter for the presence of disseminated neuropathy. PLoS One 7:e49742
Vucic S, Black K, Chong PS, Cros D (2007) Multifocal motor neuropathy with conduction block: distribution of demyelination and axonal degeneration. Clin Neurophysiol 118:124–130
Suarez GA (2005) Chapter 102 - Immune Brachial Plexus Neuropathy. In: Dyck PJ, Thomas PK (eds) Peripheral Neuropathy (Fourth Edition). W.B. Saunders, Philadelphia, pp 2299–2308
Wilbourn AJ, Aminoff MJ (1998) AAEM minimonograph 32: the electrodiagnostic examination in patients with radiculopathies. Muscle Nerve 21:1612–1631
Aminoff MJ, Goodin DS, Parry GJ, Barbaro NM, Weinstein PR, Rosenblum ML (1985) Electrophysiologic evaluation of lumbosacral radiculopathies: electromyography, late responses and somatosensory evoked potentials. Neurology 35:1514–1518
McDonald CM, Carter GT, Fritz RC, Anderson MW, Abresch RT, Kilmer DD (2000) Magnetic resonance imaging of denervated muscle: comparison to electromyography. Muscle Nerve 23:1431–1434
Kastel T, Heiland S, Baumer P, Bartsch AJ, Bendszus M, Pham M (2011) Magic angle effect: a relevant artifact in MR neurography at 3 T? AJNR Am J Neuroradiol 32:821–827
Ferdinand BD, Rosenberg ZS, Schweitzer ME et al (2006) MR imaging features of radial tunnel syndrome: initial experience. Radiology 240:161–168
England JD, Asbury AK (2004) Peripheral neuropathy. Lancet 363:2151–2161
Viddeleer AR, Sijens PE, van Ooyen PM, Kuypers PD, Hovius SE, Oudkerk M (2012) Sequential MR imaging of denervated and reinnervated skeletal muscle as correlated to functional outcome. Radiology 264:522–530
Deroide N, Bousson V, Daguet E et al (2012) Muscle magnetic resonance imaging sensitivity does not decrease in chronic, mild, or proximal lower limb neuropathies. Muscle Nerve 45:659–667
Viallon M, Vargas MI, Jlassi H, Lovblad KO, Delavelle J (2008) High-resolution and functional magnetic resonance imaging of the brachial plexus using an isotropic 3D T2 STIR (Short Term Inversion Recovery) SPACE sequence and diffusion tensor imaging. Eur Radiol 18:1018–1023
Acknowledgments
The scientific guarantor of this publication is Dr. med. Philipp Bäumer, Department of Neuroradiology, Heidelberg University Hospital, Germany. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Methodology: prospective, diagnostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Clinical data for all patients. Detailed overview of clinical and demographic data of the patient collective. The functional specification of the symptom is followed by the respective MRC grading in brackets. Symptoms are listed in ascending MRC grading order. Muscles are abbreviated as follows: M. brachioradialis (BR), extensor carpi radialis (longus and brevis) (ECR), extensor digitorum communis (EDC), extensor indicis (EI), extensor carpi ulnaris (ECU), pronator teres (PT), flexor carpi ulnaris (FCU), flexor digitorum profundus (FDP), flexor carpi radialis (FCR), pronator quadratus (PQ), flexor pollicis longus (FPL), flexor pollicis brevis (FPB), abductor pollicis brevis (APB), opponens pollicis (OP), abductor digiti minimi (ADM), interosseous dorsalis (IDM). Electrophysiological abbreviations are: motor unit action potential (MUAP), compound muscle action potential (CMAP), sensory nerve action potential (SNAP), distal motor latency (dml), proximal motor latency (pml), sensory evoked potentials (SEP). The following (sixth) column states the corresponding diagnosis, and the seventh column lists the respective interval of time between onset of clinical symptoms and MR neurography. The penultimate column informs about additional MR sequences performed that were not routinely included in the protocol of the study. The last column shows the assigned group of every patient after evaluation of acquired images. At the bottom, the total numbers of male and female patients, the mean age, and the median and mean time delay between onset of clinical symptoms and MR neurography are given. (JPEG 4850 kb)
Rights and permissions
About this article
Cite this article
Schwarz, D., Weiler, M., Pham, M. et al. Diagnostic Signs of Motor Neuropathy in MR Neurography: Nerve Lesions and Muscle Denervation. Eur Radiol 25, 1497–1503 (2015). https://doi.org/10.1007/s00330-014-3498-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3498-x