Abstract
Among nudibranch molluscs, the family Tritoniidae gathers taxa with an uncertain phylogenetic position, such as some species of the genus Tritonia Cuvier, 1798. Currently, 37 valid species belong to this genus and only three of them are found in the Southern Ocean, namely T. challengeriana Bergh, 1884, T. dantarti Ballesteros & Avila, 2006, and T. vorax (Odhner, 1926). In this study, we shed light on the long-term discussed systematics and taxonomy of Antarctic Tritonia species using morpho-anatomical and molecular techniques. Samples from the Weddell Sea and Bouvet Island were dissected and prepared for scanning electron microscopy. The three molecular markers COI, 16S, and H3 were sequenced and analysed through maximum-likelihood and Bayesian methods. The phylogenetic analyses and species delimitation tests clearly distinguished two species, T. challengeriana widely spread in the Southern Ocean and T. dantarti endemic to Bouvet Island. Colouration seems to be an unreliable character to differentiate among species since molecular data revealed both species can either have orange or white colour morphotypes. This variability could be explained by pigment sequestration from the soft coral species they feed on. Morphological analyses reveal differences between Antarctic and Magellanic specimens of T. challengeriana. However, the relationship between T. challengeriana specimens from these two regions remains still unclear due to the lack of molecular data. Therefore, the validity of the T. antarctica Martens & Pfeffer, 1886, exclusively found in Antarctic waters requires further systematic work.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antarctic benthic fauna tend to present long life cycles, slow growth rates due to slow metabolism, and direct development, and this is particularly true for molluscs (Peck et al. 2006; Moles et al. 2017). All these common characteristics seem to be the consequence of the peculiar characteristics of the Southern Ocean (SO), e.g. low temperatures, relative stability in the frequency of physical disturbance, and pronounced seasonality (Dayton et al. 1994; Jörger et al. 2014; Chown et al. 2015) aided by the onset of the Antarctic Circumpolar Current (ACC), ca. 25 Mya (Beu et al. 1997). During the late Eocene glacial periods, shelf fauna was completely impoverished with some species migrating into shelters and deep-sea waters, these being one of the major shelters for eurybathic species during the Last Glacial Maximum (Thatje et al. 2005). Certain taxa were able to re-colonize shallow waters during interglacial periods or when iceberg scouring wrecked the benthic communities and left free space available (Thatje et al. 2005). The deeper shelf of the Antarctic continent and the periodic destruction of benthic habitat on the shelf were hypothesized as natural evolutionary drivers towards eurybathy (i.e. the capacity of species of living at a wide depth range), a widely shared feature of the Antarctic benthic fauna (Thatje et al. 2005; Allcock and Strugnell 2012). Numerous taxa present circum-Antarctic distributions due to the action of the ACC, the main responsible for the connectivity between populations due to the clockwise dispersion of larvae and/or adults around the SO (Thatje 2012; Riesgo et al. 2015). On the other hand, the Polar Front acts as a North–South barrier for water exchange above 1000 m depth (Clarke et al. 2005). The idea of the SO being isolated by the Polar Front has been challenged during the last years, revealing species connectivity and genetic flow with the adjacent areas (e.g. South Africa and the Magellanic region; Griffiths 2010; Chown et al. 2015).
Gastropods are one of the major taxa represented in the SO, with numerous species still being discovered (e.g. Moles et al. 2018, 2019; Fassio et al. 2019; Layton et al. 2019). In the SO, nudibranchs are currently represented by less than a hundred recognized species (Moles 2016; De Broyer et al. 2019), although this species richness could increase with the application of molecular techniques. Among nudibranchs, the Dendronotida gathers several taxa with unassigned or unstable phylogenetic position (Goodhear et al. 2015). One of these taxa is the family Tritoniidae, among which the genus Tritonia Cuvier, 1798, appears to be the most speciose (WoRMS Editorial Board 2018). Currently, 37 valid species belong to the genus Tritonia, and only three of them are found in the SO, with Antarctic, Sub-Antarctic, and Magellanic distributions, namely T. dantarti Ballesteros & Avila, 2006, T. vorax (Odhner, 1926), and T. challengeriana Bergh, 1884, respectively. Tritonia vorax was firstly described from South Georgia as Duvaucelia vorax by Odhner (1926) and then transferred into Tritonia by Marcus in 1959. Tritonia dantarti was described in 2006 from Bouvet Island (Ballesteros and Avila 2006). Tritonia challengeriana, instead, was described for the first time by Bergh (1884) from the Magellan Strait. Since then, the latter species has been found in South Georgia, the Falkland Islands, Tierra del Fuego, and in several Antarctic locations (Antarctic Peninsula, Ross Sea, Scotia Arc; Wägele 1995; Schrödl 2003). In Antarctica, T. antarctica Pfeffer in Martens & Pfeffer, 1886, was first described by Pfeffer (1886) from South Georgia, and later ascribed to T. challengeriana by Odhner (1926), as Duvaucelia. Years later, Wägele (1995) differentiated between Magellanic specimens which were identified as T. challengeriana and specimens occurring south of the Antarctic convergence, regarded as T. antarctica. This was based on the presence of oral lips and the absence of mantle glands in T. antarctica. However, Schrödl (1996) mentioned that oral lips may also be present in T. challengeriana from the Chilean Patagonia. Mantle glands were found in histological sections of T. antarctica from South Georgia although in much lower numbers than in T. challengeriana from the Magellan area, and this led to synonymize again T. antarctica with T. challengeriana (Schrödl 2003). According to Schrödl (2003, 2009), other described species are no longer valid and are considered synonyms of T. challengeriana, i.e. Microlophus poirieri Rochebrune & Mabille, 1889, Candiella australis Berg, 1898, and Tritonia appendiculata Eliot, 1905. Descriptions of type material were often limited to a single individual and thus, these identifications might be unreliable (Wägele 1995; Schrödl 2003, 2009; Shields 2009). Furthermore, until now, no molecular data are available for any of these species, when given the wide range of distribution that T. challengeriana seems to present. So, the implementation of molecular tools could prove helpful to solve this phylogenetic conundrum. We aim to combine molecular techniques, used here for the first time in this species complex, with detailed morpho-anatomical analysis to shed light into the long-term discussed systematics and taxonomy of Antarctic Tritonia species.
Materials and methods
Sample collection
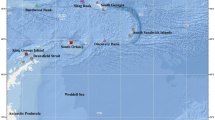
Specimens were collected by Agassiz trawl, bottom trawl, and Rauschert dredge at the Sub-Antarctic Bouvet Island and the eastern Weddell Sea in 1998 during the ANT XV/3 (Arntz and Gutt 1999) and in 2003–2004 during the ANT XXI/2 cruises (Brey 2005) of the R/V Polarstern (Alfred Wegener Institute, Bremerhaven, Germany) (Fig. 1). In order to build the maps of Fig. 1 the R packages ggplot2, marmap, and ggsn were used. The specimens of Tritonia were collected at 130–789 m depth at 17 different stations (Online Resource 1). Specimens were photographed on board and preserved in either Karnovsky, 70% ethanol, or 10% formalin in seawater for morpho-anatomical analyses, or frozen and later transferred to 96% ethanol, for molecular analyses. Specimens are deposited at the Centre of Biodiversity Resources (CRBA) in the Faculty of Biology, Universitat de Barcelona.
DNA amplification and extraction
Total genomic DNA was extracted from foot tissue with the DNeasy Tissue Kit (Qiagen, Valencia, CA, USA) following the manufacturer’s protocol. Molecular markers included two fragments of the mitochondrial genes cytochrome c oxidase I (COI) and 16S rRNA, and the nuclear gene histone 3 (H3). Partial sequences of the protein-encoding COI marker were amplified using the primers LCO1490 and HCO2198 (Folmer et al. 1994), the 16S marker was amplified using 16Sar-L and 16Sbr-H (Palumbi et al. 2002), and the H3 marker was amplified with H3AD5′3′ and H3BD5′3′ (Colgan et al. 1998). PCR amplifications were carried out in a 10 μL-reaction including 5.1 μL of Sigma dH2O, 3.3 μL REDExtract-N-Amp PCR ReadyMix (Sigma Aldrich, St. Louis, MO, USA), 0.3 μL of each primer, and 1 μL of genomic DNA, following standard protocols implemented in our lab (Moles et al. 2016). The PCR for COI consisted of an initial denaturation step at 95 °C for 3 min, 39 cycles of denaturation at 94 °C for 45 s, annealing at 48–50 °C for 30 s, extension at 72 °C for 2 min, and a final extending step at 72 °C for 10 min. The PCR program for 16S involved an initial denaturing step at 94 °C for 3 min, 39 cycles of denaturation at 94 °C for 30 s, annealing at 44–52 °C for 30 s, extension at 72 °C for 2 min, and a final extending step at 72 °C for 10 min. For H3 amplifications, we used an initial denaturation step at 94 °C for 3 min, 35 amplification cycles (94 °C for 35 s, 50 °C for 1 min, and 72 °C for 1 min and 15 s), and a final extension at 72 °C for 2 min. Amplified products were sequenced at the UB Scientific and Technological Centers (CCiT-UB) on an ABI 3730XL DNA Analyzer (Applied Biosystems, CA).
Phylogenetic analyses
Chromatograms were visualized, edited, and assembled in Geneious Pro 8.1.5 (Kearse et al. 2012). To check for contamination, sequences were compared against the GenBank database using the BLAST algorithm (Basic Local Alignment Search Tool; Altschul et al. 1990). Single-gene sequences were aligned with the MUSCLE algorithm and alignments were trimmed to a position at which more than 50% of the sequences had nucleotides. Missing positions at the ends were coded as missing data. We used Gblocks 0.91b on the final trimmed alignment for identifying and excluding blocks of ambiguous data in the single, non-codifying gene alignments of 16S, using both relaxed and stringent settings (Talavera and Castresana 2007). The total dataset contained 41 specimens of Tritonia and 17 closely related outgroup taxa (Online Resource 2). The concatenated alignment consisted of 1415 characters, including COI with 3rd codon position (ca. 601 bp), 16S unmodified (ca. 486 bp), and H3 with 3rd codon position (ca. 328 bp). The best-fit model of evolution (GTR + Г + I; Yang 1996) was chosen using the Akaike information criterion (AIC; Posada and Buckley 2004) implemented in jModelTest 2.1.7 (Posada 2008) with the selected partition for each gene (Online Resource 3).
For each gene, a maximum-likelihood (ML) analysis was conducted, the final result was given by a concatenated alignment of all three genes. ML analyses were conducted using RAxML 8.1.2 (Stamatakis 2014), a GTR model of sequence evolution with corrections for a discrete gamma distribution and invariable sites (GTR + Г + I; Yang 1996) was specified for each gene partition, and 500 independent searchers were conducted. Nodal support was estimated through a bootstrap algorithm (500 replicates) using the GTR-CAT model (Stamatakis et al. 2008). Bayesian inference (BI) was performed on the concatenated alignment of the three genes using MrBayes 3.2.5 (Ronquist et al. 2011). Two runs were conducted in MrBayes for 10 million generations, sampling every 2000 generations, using random starting trees. 25% of the runs were discarded as burn-in after checking for stationarity with Tracer 1.7 (Rambaut et al. 2018). Bootstrap support (BS) and posterior probabilities (PP) were thereafter mapped onto the optimal tree from the independent searches. Only nodal support values of > 70 and > 0.85 for BS and PP, respectively, were depicted in Fig. 2. The tree was rooted using the four selected Proctonotoidea species Curnon granulosa, Leminda millecra, and two species of Dirona, as the sister group to the rest of the Dendronotoidea species included in this study (following Goodhear et al. 2015).
Phylogenetic tree of Tritonia species and outgroup species considered using Bayesian inference (BI) and maximum likelihood (ML) on the combined COI, 16S, and H3 datasets. BI topology is shown in the main figure, and numbers on the nodes indicate posterior probability values (BI) and bootstrap support values (ML). Only values > 75 and > 0.85 depicted for ML and BI, respectively. Specimens in red are from Bouvet Island (Sub-Antarctic distribution) and the specimens in blue are from the Weddell Sea (Antarctic distribution). The specimen of T. dantarti from GenBank placed in the red cluster was registered as T. antarctica (Voucher n. CASIZ171177). The results of the ABGD (green/left) and GMYC (purple/right) analyses are represented in bars, distinguishing the two SO species groups studied
Species delimitation tests
To examine the molecular distinctiveness of the different Antarctic Tritonia morpho-species, we used the Automatic Barcode Gap Discovery (Pulliandre et al. 2011) via the web interface (ABGD; http://wwwabi.snv.jussieu.fr/public/abgd/abgdweb.html). ABGD of the COI alignment was run using the K80 calibrated index of genetic distance with transition/transversion ratio (TS/TV) equal to 2.0. We applied default values for Pmin, Pmax, and a relative gap of 1.0. Additionally, with the same alignment, a Generalized Mixed Yule Coalescent (GMYC; Fujisawa and Barraclough 2013) analysis was performed. The ultrametric tree, necessary for the GMYC, was generated with BEAST (Suchard et al. 2018), with GTR + Г + I substitution model, and a lognormal relaxed clock with a rate of 1.0, for 10 million generations. TreeAnnotator was used to discard 25% as burn-in. The GMYC was performed on the webserver (https://species.h-its.org/gmyc/) with single threshold parameters.
Morphological analyses
Photographs of whole animals were taken with a Nikon d300 Sigma 105 mm f 2.8–32. Total length (L) was measured aided by a calliper. Specimens were dissected sagittally with the aid of fine forceps under a stereomicroscope. Radula and jaws were obtained from the buccal bulb after dissolving the oral bulb’s soft tissue in a 10% NaOH solution for up to four hours and later rinsed with distilled water in ultrasound baths. The reproductive system was depicted and the penial papilla extracted. This was critical point dried before mounting on stubs with carbon sticky-tabs, as for the radulae and jaws, for scanning electron microscopy (SEM). The stubs were carbon-coated and images were taken using a J-7100F Jeol scanning electron microscope at the UB Scientific and Technological Centers (CCiT-UB).
Results
Phylogenetic analyses
ML and BI analyses recovered a tree with maximum support for T. challengeriana specimens from both the Weddell and the Ross Seas (PP = 1, BS = 100), and for T. dantarti including only the Sub-Antarctic specimens from Bouvet Island (PP = 1, BS = 98; Fig. 2). One specimen attributed to T. antarctica (voucher number CASIZ171177) clustered with our specimens of T. dantarti and thus, might be considered a misidentification. The sister taxa to both SO species sequenced were the North Pacific T. festiva, a clade composed of the type species of the genus T. hombergii, and an unidentified species of Tritonia. The SO species clustered in a clade with high support from the BI of different Tritonia species. We recovered the unidentified Tritonia sp. 3, Tritonia sp. 6, Tritonia sp. 7, and Tritonia sp. G all in a well-supported clade with all sequenced Marionia species. The relationships of the Antarctic monotypic Tritoniella belli were not found in our analyses. The relationships between Bornella, Marionia, Tritoniella, and Tritonia clades were not recovered in this study.
The ABGD analysis additionally supported the taxonomic identity of T. challengeriana and T. dantarti, with an intraspecific variation of 1.7 and 1.9% on average, respectively (Table 1). Intraspecific variation within other Tritonia species considered in this study ranged from 0 to 7%. The interspecific variation of T. challengeriana and T. dantarti ranged from 12 to 14% (Table 1). The GMYC analysis also recovered both species as different, as for the ABGD analysis (Online Resource 4).
Systematics
Class GASTROPODA Cuvier, 1795
Subclass HETEROBRANCHIA Burmeister, 1837
Order NUDIBRANCHIA Cuvier, 1817
Suborder CLADOBRANCHIA Willan & Morton, 1984
Family TRITONIIDAE Lamarck, 1809
Genus Tritonia Cuvier, 1798
Type species: Tritonia hombergii Cuvier, 1803
Tritonia challengeriana Berg, 1884
Tritonia challengeriana Bergh, 1884: 45–47, pl. 11, figs. 16–19; Eliot 1907: 354–355; 1907: 3–5; Wägele 1995: 41–45; Schrödl 2003: 97–101
Tritonia antarctica Pfeffer in Martens & Pfeffer, 1886: 112, pl. 3, figs. 6a,b; Wägele 1995: 21–46
Microlophus poirieri Mabille & Rochebrune, 1889: 11–12, pl. 6, figs. 1a,b
Tritonia poirieri (Mabille & Rochebrune, 1889): Wägele 1995: 43
Candiella australis Bergh, 1898a: 536–539, pl. 31, figs. 17–25
Tritonia appendiculata Eliot, 1905: 526, figs. 6–10
Tritonia australis (Bergh): Dall 1909: 202
Duvaucelia challengeriana (Bergh): Odhner 1926: 35–37, pl. 1, fig. 14
Duvaucelia poirieri (Rochebrune & Mabille): Odhner 1926: 38–39
Myrella poirieri (Rochebrune & Mabille): Odhner 1963: 51–52
Marionia cucullata (Couthouy in Gould): Vicente and Arnaud 1974: 539, figs. 6, 7, pl. 3, figs. 1–3
Material examined: Out of the 37 specimens collected, 32 were frozen, two were fixed in 10% formalin in seawater, two in 70% ethanol, and one in 96% ethanol. South of Vestkapp, eastern Weddell Sea, 73°36.6′S, 22°24.7′W, 736 m depth: 1 spc., dissected and sequenced, T08, voucher CRBA-92763, L = 25 mm, barcode MN651129. Halley Bay, 74°35.8′S, 26°55.0′W, 789 m depth: 1 spc., dissected and sequenced, T10, voucher CRBA-92765, L = 22 mm, barcode MN651131. North of Kapp Norvegia, 71°07.34′ S, 11°27.80′W, 146 m depth: 1 spc., dissected, T25, voucher CRBA-92800 L = 26 mm. Drescher Inlet, 72°05.18′S, 19°38.62′W, 598 m depth: 1 spc., dissected T28, L = 42 mm (Online Resource 1).
External morphology (Fig. 3a–c): Body length 22–42 mm after preservation (Table 2). Body wider dorsally than ventrally. Colour of preserved specimens milky white, beige-brownish when viscera seen by transparency; live specimens homogeneously white to orange. Dorsal mantle surface smooth, with subepithelial white knobs, found mostly in posterior region of mantle. White pigmentation seen on notal margin, gills, and margin of rhinophoral sheath (Fig. 3a). Rhinophoral sheath broad; margin and plumes smooth. Gills ramified, dichotomous, large or small, from 6 to 19 per side, situated in parallel to each other. Oral veil prominent, bilobed or not. Five to ten velar processes present. Mouth surrounded by thick lips without distinct oral tentacles. Foot perimeter smaller than notal surface (Fig. 3c). Genital papilla found on right side of body. Anal opening placed at ½ of body length (Fig. 3b).
Preserved specimen of Tritonia challengeriana from the eastern Weddell Sea (T28; voucher CRBA-92804). a Dorsal view. b Lateral view. c Ventral view. d Scanning electron microscopy (SEM) of the radula showing the tricuspidate rachidian teeth, the first and subsequent lateral teeth (T08; voucher CRBA-92763). e Detail of the jaw's denticles (SEM) (T08; voucher CRBA-92763). f Schematic drawing of the reproductive system (T28; voucher CRBA-92804). am ampulla; cgl, capsule gland; mgl, mucous gland; re, seminal receptacle; vd, vas deferens; vg vaginal duct. g Detail of the penial papilla (SEM) (T28; voucher CRBA-92804)
Digestive system (Fig. 3d, e): Oral lips smooth, large. Oral tube short. Oral bulb and pharynx thick. Radular formula 30–37 × (33–49)1.1.1.(33–49); transparent in colour. Rachidian teeth presenting median prominent cusp, one smaller cusp found per each side (Fig. 3d). Two different lateral teeth, with one short, broad cusp. Jaws yellowish, curved towards inside; several rows of conic denticles, curved, striated, as border denticles (Fig. 3e). Jaws 5–9 mm in length. Ratio jaw:body 0.21–0.3 in length. Oesophagus running dorsally from pharynx. Salivary glands thin, elongated, running laterally from first half of body, then ventrally following oesophagus. Stomach situated ventrally. Intestine generally striated, originating dorsally from stomach, turning right, ending in anal opening.
Reproductive system (Fig. 3f, g): Reproductive system androdiaulic. Gonad large, wrinkled, covering digestive gland posteriorly. Gonoduct opening in ampulla connected by spermiduct to vas deferens. Bifurcation into vas deferens and oviduct not easily detected (Fig. 3g). Penial sheath terminal, conical, thin; penial papilla conical, slightly twisted (Fig. 3f). Seminal receptacle voluminous, with short duct; going shortly into genital opening. Granulated capsule gland at bottom of wide mucous gland, preceding short oviduct.
Ecology (Fig. 4a–c): The specimens were collected from 146 to 789 m depth. Sclerites of alcyonarian octocorals were found in the gut contents of three out of the four specimens studied.
Distribution: Argentinian Patagonia (Marcus et al. 1969), Falkland Islands (Eliot 1907), Chilean Patagonia (Bergh 1884; Schrödl 1996) to Ancud Bay (Schrödl 1996), South Georgia (Odhner 1926), Adélie Land (Vincente and Arnaud 1974), Victoria Land (Ross Sea, Schiaparelli et al. 2006), and eastern Weddell Sea (Wägele 1995; this study).
Remarks: Most synonyms of genera and species to T. challengeriana were based on external morphological similarities (Wägele 1995; Schrödl 2003). For instance, Rochebrune and Mabille (1889) described Microlophus poirieri from Patagonia, Falkland Islands, and South Georgia, based only on their external morphology and colouration. Microlophus poirieri was later synonymized to T. challengeriana by Wägele (1995) based on their external morphology. Marionia cucullata was described from Adélie Land (Vicente and Arnaud 1974) but the similarity in oral lips’ shape and the low number of gills allowed Wägele (1995) to synonymize this genus and species to T. antarctica. Tritonia appendiculata Elliot, 1905 was described from Harbour, Scotia Bay, the South Orkney Islands at 16 m depth. Its body colour was greenish-yellow, this is the only character that differs from our specimens since most of the morphological characters overlap with T. challengeriana. For instance, the species presents 19 gills per side and, on the dorsal surface, subepithelial knobs organized as “warts” were present (Eliot 1905). The oral veil presents twelve simple digitate processes and the lips are projected on each side of the mouth. The relation of jaw length (10 mm) to body length (51.5 mm) is 0.19 for T. appendiculata, while Wägele (1995) found a similar ratio for T. challengeriana of 0.23, as in our study. Thus, our data support Wägele’s synonymy. Tritonia poirieri Mabille & Rochebrune, 1891, det. Odhner 1926, was found at Fitzroy Channel, at 14 m depth. The species' body shape resembles that of Doris, with the notal margin bent downward. Other than the peculiar body shape, there were not enough differences to identify T. poirieri as a distinct species from T. challengeriana (Wägele 1995).
Wägele (1995) differentiated Magellanic specimens of T. challengeriana from the specimens occurring south of the Polar Front, regarded as T. antarctica Martens & Pfeffer, 1886. The major difference was the presence of oral lips and mantle glands, exclusively found in T. antarctica. Later on, Schrödl (2003) described these two characters in T. challengeriana from Chilean Patagonia and synonymized it to T. antarctica. Our specimens are morphologically similar to the T. antarctica specimens described by Wägele (1995), with visible white knobs on the dorsal surface of the body and the presence of conspicuous oral lips. Our description of T. challengeriana overlaps with the measurements and descriptions from Pfeffer (1886) and Wägele (1995), thus highlighting a major similarity to T. antarctica than to the Magellanic T. challengeriana. On the other hand, Tritonia vorax (Odhner, 1926) is found in South Georgia (Wägele 1995), Burdwood Bank, and the Chilean Patagonia (Odhner 1926; Schrödl 1996). Living specimens present a whitish to brownish colouration, with white or opaque white reticulations on the notal surface. Preserved specimens can be whitish, yellowish or pinkish and their notum can be more or less smooth. This species differs from T. challengeriana by having less number of gills, extremely large and strong jaws, which cause an elevated mediodorsal protuberance in between the rhinophores, and the lack of oral lips, with higher jaws:body length ratio than that of T. challengeriana (Table 2). Differences between T. challengeriana and T. dantarti are discussed in the Remarks section below.
Tritonia dantarti Ballesteros & Avila, 2006
Material examined: Thirteen specimens collected at stations PS65/028-1 and PS65/029-1 in Bouvet Island. Six specimens were fixed in 70% ethanol, four were frozen, one in 96% ethanol, and two in Karnowsky. Bouvet Island, 54°30.1′S, 3°13.97′W, 260 m depth: 1 spc., dissected and sequenced, T14.3, voucher CRBA-92771, L = 18 mm, barcode MN651134; 54°22.49′S, 3°17.58′W, 130 m depth: 1 spc., dissected, T16, voucher CRBA-92782, L = 23 mm; 54°22.49′S, 3°17.58′W, 130 m depth: 1 spc., dissected, T18.1, voucher CRBA-92787, L = 20 mm, barcode (Online Resource 1).
External morphology (Fig. 5a–c): Body short, thick; 18–23 mm length. Colour beige to milky white in preserved specimens, living specimens sometimes completely white (specimens T14.3, voucher CRBA-92771 and T14.4 voucher CRBA-92772, Fig. 6a) or bright orange on dorsal surface, with warts forming a reticulation; white laterally (specimen T15.1 voucher CRBA-92777, Fig. 6b). Dorsal mantle surface smooth with subepithelial white knobs. Notal margin unpigmented. Rhinophores with large sheath; smooth margin with emerging plumes. Single small gills or largely ramified, from 15 to 29 per side (Fig. 5a). Oral veil not prominent, bilobed or not; velar processes, short, 9 to 19 in number. Lips thick, surrounding buccal bulb, without recognizable tentacles. Foot narrower than notum (Fig. 5c). Genital papilla on right side at 1/3 of body length. Anal opening at ½ of body length (Fig. 5b). Length and morphometrical data reported in Table 2.
Preserved specimen of Tritonia dantarti from Bouvet Island (T16; voucher CRBA-92782). a Dorsal view. b Lateral view. c Ventral view. d Scanning electron microscopy (SEM) of the radula showing the rachidian teeth, the first and subsequent lateral teeth. e Detail of the jaw denticles (SEM). f Detail of the penial papilla (SEM). g Schematic drawing of the reproductive system. am ampulla, cgl capsule gland, mgl mucous gland, re seminal receptacle, vd vas deferens, vg vaginal duct
Specimens of Tritonia dantarti from Bouvet Island (photographs by M. Ballesteros). a Two specimens collected a 260 m of depth from Bouvet Island displaying whitish colouration (T14.3; voucher CRBA-92771, and T14.4; voucher CRBA-92772). b Specimen from Bouvet Island with orange colouration, collected at 130 m depth (T15.1; voucher CRBA-92777)
Digestive system (Fig. 5d–e): Oral lips thick, smooth. Pharynx large, compact; with a pair of curved jaws of yellowish margin. Rachidian teeth broad, monocuspid (Fig. 5d). Radular formula: 36–37 x (43–46)1.1.1.(43–46). Oesophagus running dorsally from pharynx. Jaw denticles broad, conical, striated, hooked on top, arranged in several rows (Fig. 5e). Jaws 5–6.5 mm in length. Ratio jaw:body 0.25–0.28 mm in length. Salivary glands large, isodiametric, running laterally in first body half, then ventrally under oesophagus. Stomach situated ventrally. Intestine generally striated, originating dorsally from stomach, turning to right side, ending at anal opening.
Reproductive system (Fig. 5f–g): Reproductive system situated between buccal bulb and digestive gland. Gonad brownish, warty, covering digestive gland. Genital papilla opening in a bulb, spermiduct could not be observed (Fig. 5g). Seminal receptacle wide. Penis thin, flagellated (Fig. 5f), occasionally conical. Penial papilla with conical shape. Mucous gland well developed, situated on top of entire system; granulated capsule gland preceding short oviduct, often convoluted.
Ecology (Fig. 4d, e): Specimens of T. dantarti were collected in Bouvet Island at 130–134 m depth, in sea bottoms dominated by ophiuroids (Ophionotus victoriae), sea stars (Porania antarctica), holothuroids, sedentary polychaetes, hydroids, alcyonarians, different actinian species, amphipods, and pycnogonids. Gut contents showed that T. dantarti feeds on alcyonarians of the genus Alcyonium.
Distribution: Northwest and southeast of Bouvet Island.
Remarks: Tritonia dantarti is clearly distinguished from its counterpart T. vorax by the possession of oral lips, completely lacking in T. vorax. In T. dantarti the oral veil can be bilobed or not, while it is always bilobed in T. vorax. Moreover, T. dantarti presents lesser teeth rows and a monocuspidate rachidian tooth, while T. vorax presents a higher number of rows with a tricuspid rachidian tooth. Additionally, the jaws:body length ratio is higher in T. vorax (Table 2).
Tritonia dantarti was described by possessing a conspicuous orange colouration in the dorsum of living specimens (see Fig. 6a,c,e in Ballesteros and Avila 2006; Fig. 6b in this study). This was, in fact, the main difference from T. challengeriana, but here molecular evidence of both white and orange colour-morphs is given for T. dantarti . An additional diagnostic character is the presence of a warty reticulation in the notal surface of living specimens of T. dantarti, which has not been obviously observed in our preserved specimens and is completely lacking in T. challengeriana. Moreover, T. challengeriana generally presents fewer velar processes and fewer clusters of gills, but some overlap exists for both species, and a broad range of morphological differences is especially misleading in preserved specimens of both species. Our results agree with previous descriptions for both species (Wägele 1995; Schrödl 2003; Ballesteros and Avila 2006) (Table 2).
Discussion
Taxonomy and morphology of Antarctic Tritonia species
The specimens reviewed in this study from the high Antarctic belonged to the only current valid species Tritonia challengeriana, while the specimens from Bouvet Island belonged to T. dantarti. Phylogenetic analyses and species delimitation tests recovered these two species with strong support (Fig. 2), including the specimens of T. challengeriana from the Weddell Sea and the only sequenced specimen from the Ross Sea (PP = 1, BS = 100), and the specimens of T. dantarti from Bouvet Island (PP = 1, BS = 98). Morpho-anatomical analyses showed that on the dorsal body surface in living specimens of T. dantarti warts and reticulation are visible. Nonetheless, the bright orange colouration (Ballesteros and Avila 2006) may no longer be a valid diagnostic character, since both milky white and orange colour morphotypes from Bouvet Island were found here, as it has been described for T. challengeriana from both South America and high Antarctic regions (Fig. 7). These results were supported by our molecular analyses. Besides this, no other clear diagnostic characters were found in the morpho-anatomical analysis to allow discrimination among these two species. For instance, shape and body measurements, the number of velar processes, the shape and number of gills, the radular formula, and the shape of the jaws are not quite discernible between T. dantarti and T. challengeriana. Both species overlap in the range of the aforementioned characters (Table 2), as also reported by Wägele (1995), Schrödl (2003), and Ballesteros and Avila (2006). Nonetheless, T. challengeriana seems to present lesser oral tentacles and gill clusters than T. dantarti, but still, this might be subjected to ontogenetic development.
Underwater photographs of alleged Tritonia challengeriana from its current range of distribution. a Puerto Raúl Marín Balmaceda, Chile (photograph by T. Heran). b Comau Fjord, Chile (photograph by D. Thompson). c Punta Porra, Chile (photograph by T. Heran). d Ross Sea, Antarctica (photograph by S. Harper). e Ross Sea, Antarctica (photograph by P. Brueggeman). f Antarctic Peninsula, Antarctica (photograph by G. Giribet)
The validity of T. antarctica has been questioned in a few studies (see Remarks section of T. challengeriana). Wägele (1995) sustained the existence of T. antarctica for the presence, in Antarctic specimens, of subepithelial glands (externally visualized as knobs), which were lacking in Magellanic specimens. Later on, Schrödl (2003) suggested the contrary, showing that the glands were present on the dorsal surface of the specimens from the Magellanic area, even if sporadically and in a lower number. Our specimens seem to be similar to the T. antarctica specimens described by Wägele (1995). Pictures of living specimens from the Magellanic region (Fig. 7a–c) do not show visible knobs, which are easily detectable on specimens from Antarctica (Fig. 7d–f). However, the morphological analysis suggests that T. challengeriana and T. antarctica could be considered to be different species, given the evidence of the visible knobs on the dorsal surface present on Antarctic specimens. Unfortunately, we cannot confirm the validity of T. antarctica, since there are no molecular data available for T. challengeriana from the Magellanic region to date. Southern American material and additional samples from around Antarctica could be very useful to shed light into the Southern Hemisphere Tritonia species systematics.
The colouration issue
Members of the family Tritoniidae feed almost exclusively on octocorals, including sea pens, alcyonarian soft corals, and gorgonians, sometimes being cryptic in shape and colouration upon them (García-Matucheski and Muniain 2011). In the SO, Tritonia species feed mostly on alcyonarian soft corals (Wägele 1995; Schrödl 2003García-Matucheski and Muniain op. cit.). Here, we found soft coral sclerites in the gut contents of both T. challengeriana and T. dantarti. The Alcyonium species living in the SO are A. antarcticum Wright & Studer, 1889, A. grandis Casas, Ramil & van Ofwegen, 1997, A. haddoni Wright & Studer, 1889, A. paucilobatum, A. sollasi Wright & Studer, 1889, and A. southgeorgensis Casas, Ramil & van Ofwegen, 1997, and they can all present a yellow, cream or orange colouration, while they tend to be brighter in the Magellanic region (Casas et al. 1997). Through the evolution, and related to the loss of the shell, nudibranchs have developed a plethora of defensive strategies against predators (Avila et al. 2018). These defences include chemicals (natural products), which can be either de novo synthesized by the own slug or gathered from their prey (i.e. kleptochemistry). An example of kleptochemistry in Antarctica is found in Tritoniella belli Eliot, 1907 which obtains its defensive natural products from its prey, the anthozoan Clavularia frankliniana Roule, 1902 (McClintock et al. 1994). Some dietary metabolites can be brightly coloured pigments, as for some Alcyonium spp. natural products (Abdel-Lateff et al. 2019), and their sequestration and display in the external tissue of the slug may provide an additional mimetic defensive strategy. Although the development of a bright colouration may sometimes represent a warning mechanism (i.e. aposematism; Aguado and Marin 2007; Haber et al. 2010; Avila et al. 2018), the bright orange colouration found in both T. challengeriana and T. dantarti may not represent an aposematism mechanism, since the majority of visually guided predators, such as fishes or decapods, are not especially diversified in Antarctica (De Broyer and Danis 2011; Moles 2016). Nevertheless, antipredation assays suggest T. challengeriana is chemically protected against sympatric sea star predators, although the compounds have not been identified yet (Avila et al. 2018). This strategy has been proved in other Antarctic species, such as Bathydoris hodgsoni Eliot, 1907 and Tritoniella belli Eliot, 1907 (McClintock et al. 1994; Avila et al. 2000). We propose here that the colouration in T. challengeriana and T. dantarti varies locally in direct relation to diet and therefore cannot be used as a diagnostic character for these species.
Distribution and cryptic speciation
From our molecular phylogenetic analyses, we observe that while T. dantarti seems to have a restricted, endemic distribution in Bouvet Island, T. challengeriana seems to present a disjunct (i.e. found in both the Weddell and Ross Seas) and probably circumpolar distribution. This distribution could be partially explained by the action of the ACC (Thatje 2012). When the Drake Passage was narrower, the ACC flow was particularly intense in the Antarctic region, carrying adults or egg masses attached to floating debris (i.e. rafting phenomenon) to new habitats all around Antarctica (i.e. circumpolar distribution). Through genetic drift and selection these populations could diverge sufficiently to either yield a new species by allopatric speciation (Allcock and Strugnell 2012)—which might have been the case for the restricted T. dantarti—or widening its geographical range, as for T. challengeriana. Even if the rafting phenomenon allowed a long-distance dispersal in organisms that not produce free-swimming larvae (Thatje op. cit.) during glacial cycles, the existence of shelters where the ice shelf did not cover homogeneously the shelf, acted as refugia (Thatje et al. 2005; Fraser et al. 2014; Chown et al. 2015). There, species isolation could have led to cryptic speciation (Wilson et al. 2009). In fact, because of the existence of cryptic species, the current species richness of gastropods in the Antarctic and Sub-Antarctic regions is higher than previously thought (Linse et al. 2007). Likewise, new cephalaspidean molluscs with low character displacement have been recently described based on molecular data in the same region (Moles et al. 2017, 2019). The nudibranch Doris kerguelenensis (Bergh, 1884) seems to also present this trend; molecular data evidenced a complex genetic structure that suggests much diversity than a single recognized species (Wilson et al. 2009). This hypothesis is corroborated by the wide variety of natural products used against predators. Yet, due to the lack of morphological analyses, the taxonomy of D. kerguelenensis is still not solved (Wilson et al. 2013). Both T. challengeriana and T. dantarti are two different species, but the relationship between T. challengeriana specimens from the Antarctic and the Magellanic regions remains still unclear, thus the validity of T. antarctica requires further systematic work. Additional samples from other locations in Antarctica, Sub-Antarctic Islands, and South America are urgently needed to shed light on the systematics of the T. challengeriana species group. Sampling in poorly known areas of the SO, such as the Amundsen Sea or the western Weddell Sea, and the continental shelves underneath floating ice shelves (Griffiths 2010), with the increasing application of molecular techniques and complementary molecular markers with higher resolution (e.g. EPIC markers, microsatellites, and/or genome- or transcriptome-derived SNPs; Riesgo et al. 2015; Moles et al. 2019; Moles and Giribet 2021) are required to further evaluate cryptic speciation and increasing our knowledge on the biodiversity of most invertebrate taxa in the SO.
References
Aguado F, Marin A (2007) Warning coloration associated with nematocyst-based defences in aeolidiodean nudibranchs. J Moll Stud 73:23–28
Allcock AL, Strugnell JM (2012) Southern Ocean diversity: new paradigms from molecular ecology. Trends Ecol Evol 27:520–528
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Arntz W, Gutt J (1999) The expedition ANTARKTIS XV/3 (EASIZ II) of RV “Polarstern” in 1998. Rep Pol Res 301. Alfred Wegener Institute for Polar and Marine Research, Bremerhaven
Avila C, Iken K, Fontana A, Cimino G (2000) Chemical ecology of the Antarctic nudibranch Bathydoris hodgsoni Eliot, 1907: defensive role and origin of its natural products. J Exp Mar Biol Ecol 252:27–44
Avila C, Núñez-Pons L, Moles J (2018) From the Tropics to the Poles: chemical defense strategies in sea slugs (Mollusca: Heterobranchia). In: Puglisi MP, Becerro MA (eds) Chemical ecology: the ecological impacts of marine natural products. CRC Press, Boca Raton, pp 71–163
Ballesteros M, Avila C (2006) A new tritoniid species (Mollusca: Opisthobranchia) from Bouvet Island. Pol Biol 29:128–136
Beu AG, Griffin M, Maxwell PA (1997) Opening of Drake Passage gateway and Late Miocene to Pleistocene cooling reflected in Southern Ocean molluscan dispersal: evidence from New Zealand and Argentina. Tectonophysics 281:83–97
Casas C, Ramil F, van Ofwegen LP (1997) Octocorallia (Cnidaria: Anthozoa) from the Scotia Arc, South Atlantic Ocean: the genus Alcyonium Linnaeus, 1758. Zool Meded 71:299–311
Chown SL, Clarke A, Fraser CI, Cary SC, Moon KL, McGeoch MA (2015) The changing form of Antarctic biodiversity. Nature 522:431
Dall WH (1909) Report on a collection of shells from Peru, with a summary of the littoral marine Mollusca of the Peruvian Zoological Province. Proc U S Natl Mus 37:147–294
Dayton PK, Mordida BJ, Bacon F (1994) Polar marine communities. Am Zool 34:90–99
De Broyer C, Danis B (2011) How many species in the Southern Ocean? Towards a dynamic inventory of the Antarctic marine species. Deep-Sea Res 2. Top Stud Oceanogr 58:5–17
De Broyer C, Clarke A, Koubbi P, Pakhomov E, Scott F, Vanden Berghe W, Danis B (2019) The SCAR-MarBIN Register of Antarctic Marine Species (RAMS). http://www.scarmarbin.be/scarramsabout.php. Accessed 6 Apr 2016
Eliot C (1905) The Nudibranchiata of the Scottish National Antarctic Expedition. Trans R Soc Edinb 41:519–532
Eliot C (1907) Nudibranchs from New Zealand and the Falkland Islands. Proc Malac Soc Lond 7:350–361
Fassio G, Modica MV, Alvaro MC, Buge B, Salvi D, Oliverio M, Schiaparelli S (2019) An Antarctic flock under the Thorson’s rule: diversity and larval development of Antarctic Velutinidae (Mollusca: Gastropoda). Mol Phylogenet Evol 132:1–13
Fraser CI, Terauds A, Smellie J, Convey P, Chown SL (2014) Geothermal activity helps life survive glacial cycles. Proc Natl Acad Sci USA 111:5634–5639
Fujisawa T, Barraclough TG (2013) Delimiting species using single-locus data and the Generalized Mixed Yule Coalescent approach: a revised method and evaluation on simulated data sets. Syst Biol 62:707–724
García-Matucheski S, Muniain C (2011) Predation by the nudibranch Tritonia odhneri (Opisthobranchia: Tritoniidae) on octocorals from the South Atlantic Ocean. Mar Biodivers 41:287–297
Goodhear JA, Bazinet AL, Collins AG, Cummings MP (2015) Relationships within Cladobranchia (Gastropoda: Nudibranchia) based on RNA-Seq data: an initial investigation. R Soc Open Sci 2:150196
Griffiths HJ (2010) Antarctic marine biodiversity—what do we know about the distribution of life in the Southern Ocean? PLoS ONE 5:e11683
Haber M, Cerfeda S, Carbone M, Calado G, Gaspar H, Neves R, Maharajan V, Cimino G, Gavagnin M, Ghiselin MT, Mollo E (2010) Coloration and defense in the nudibranch gastropod Hypselodoris fontandraui. Biol Bull 218:181–218
Jörger KM, Schrödl M, Schwabe E, Würzberg L (2014) A glimpse into the deep of the Antarctic Polar Front-Diversity and abundance of abyssal molluscs. Deep-Sea Res 2. Top Stud Oceanogr 108:93–100
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Mentjies P, Drummond A (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2828:1647–1649
Layton KK, Rouse GW, Wilson NG (2019) A newly discovered radiation of endoparasitic gastropods and their coevolution with asteroid hosts in Antarctica. BMC Evol Biol 19:180
Linse K, Cope T, Lörz AN, Sands C (2007) Is the Scotia Sea a centre of Antarctic marine diversification? Some evidence of cryptic speciation in the circum-Antarctic bivalve Lissarca notorcadensis (Arcoidea: Philobryidae). Pol Biol 30:1059–1068
Marcus EDBR, Marcus E, Kirsteuer E (1969) Opisthobranchian and lamellarian gastropods collected by the “Vema”. Am Mus Novit (2368):1–33
McClintock JB, Baker BJ, Slattery M, Heine JN, Bryan PJ, Yoshida W, Davies-Coleman M, Faulkner DJ (1994) Chemical defense of common Antarctic shallow-water nudibranch Tritoniella belli, Eliot (Mollusca: Tritoniidae) and its prey, Clavularia frankliniana, Rouel (Cnidaria: Octocorallia). J Chem Ecol 20:3361
Moles J (2016) Antarctic heterobranch molluscs: diving into their challenging ecology, taxonomy, and systematics. Dissertation, Universitat de Barcelona
Moles J, Wägele H, Ballesteros M, Pujals Á, Uhl G, Avila C (2016) The end of the cold loneliness: 3D comparison between Doto antarctica and a new sympatric species of Doto (Heterobranchia: Nudibranchia). PLoS ONE 11:e0157941
Moles J, Wägele H, Cutignano A, Fontana A, Ballesteros M, Avila C (2017) Giant embryos and hatchlings of Antarctic nudibranchs (Mollusca: Gastropoda: Heterobranchia). Mar Biol 164:114
Moles J, Avila C, Malaquias MAE (2018) Systematic revision of the Antarctic gastropod family Newnesiidae (Heterobranchia: Cephalaspidea) with the description of a new genus and a new abyssal species. Zool J Linnean Soc 183:763–775
Moles J, Avila C, Malaquias MAE (2019) Unmasking Antarctic mollusc lineages: novel evidence from philinoid snails (Gastropoda: Cephalaspidea). Cladistics 35:487–513
Odhner NHJ (1926) The Opisthobranchien, vol 2. PA Norstedt & Söner, Stockholm
Odhner NHJ (1963) On the taxonomy of the family Tritoniidae (Mollusca: Opisthobranchia). Veliger 6:48–52
Moles J, Giribet G (2021) A polyvalent and universal tool for genomic studies in gastropod molluscs (Heterobranchia). Mol Phylogenet Evol 155:106996
Peck LS, Clarke A, Chapman AL (2006) Metabolism and development of pelagic larvae of Antarctic gastropods with mixed reproductive strategies. Mar Ecol Prog Ser 318:213–220
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Posada D, Buckley TR (2004) Model selection and model averaging in phylogenetics: advantages of Akaike information criterion and Bayesian approaches over likelihood ratio tests. Syst Biol 53:793–808
Pulliandre N, Lambert A, Brouillet S, Achaz G (2011) ABGD, Automated Barcode Gap Discovery for primary species delineation. Mol Ecol 21:1864–1877
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Tracer v1.7. http://tree.bio.ed.ac.uk/software/tracer/
Riesgo A, Taboada S, Avila C (2015) Evolutionary patterns in Antarctic marine invertebrates: an update on molecular studies. Mar Genomics 23:1–13
Rochebrune AT, de Mabille J (1889) Mollusques. Mission Scientifique du Cap Horn 1882–1883. Tome 6 (Zoologie 2, part 8). Gauthiers-Villars, Paris, pp 11–12
Ronquist F, Huelsenbeck J, Teslenko M (2011) Draft MrBayes version 3.2 manual: tutorials and model summaries. http://brahms.biology.rochester.edu/software.html
Schiaparelli S, Lörz AN, Cattaneo-Vietti R (2006) Diversity and distribution of mollusc assemblages on the Victoria Land coast and the Balleny Islands, Ross Sea, Antarctica. Antarct Sci 18:615–631
Schrödl M (1996) Nudibranchia and Sacoglossa of Chile: external morphology and distribution. Gayana Zool 60:17–62
Schrödl M (2003) Sea slugs of Southern South America. ConchBooks, Hackenheim, p 165
Schrödl M (2009) Opisthobranchia—sea slugs. In: Häussermann V, Försterra G (eds) Marine benthic fauna of Chilean Patagonia, pp 505–542
Shields CC (2009) Nudibranchs of the Ross Sea, Antarctica: phylogeny, diversity, and divergence. Master thesis, Clemson University
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web servers. Syst Biol 57:758–771
Suchard MA, Lemey P, Baele G, Ayres DL, Drummond AJ, Rambaut A (2018) Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Virus Evol. https://doi.org/10.1093/ve/vey016
Talavera G, Castresana J (2007) Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst Biol 56:564–577
Thatje S (2012) Effects of capability for dispersal on the evolution of diversity in Antarctic benthos. Integr Comp Biol 52:470–482
Thatje S, Hillenbrand CD, Larter R (2005) On the origin of Antarctic marine benthic community structure. Trends Ecol Evol 20:534–540
Vicente N, Arnaud PM (1974) Invertébrés marins des XIIeme et XVeme expéditions Antarctiques Françaises en Terre Adélie. 12. Gastéropodes Opisthobranches Tethys 5:531–548
Wägele H (1995) The morphology and taxonomy of the Antarctic species of Tritonia Cuvier, 1797 (Nudibranchia: Dendronotoidea). Zool J Linnean Soc 113:21–46
Wilson NG, Schrödl M, Halanych KM (2009) Ocean barriers and glaciation: evidence for explosive radiation of mitochondrial lineages in the Antarctic sea slug Doris kerguelenensis (Mollusca, Nudibranchia). Mol Ecol 18:965–984
Wilson NG, Maschek JA, Baker BJ (2013) A species flock driven by predation? Secondary metabolites support diversification of slugs in Antarctica. PLoS ONE 8:e80277
WoRMS Editorial Board (2018). World Register of Marine Species. http://www.marinespecies.org. Accessed 27 Feb 2018
Yang Z (1996) Among-site rate variation and its impact on phylogenetic analyses. Trends Ecol Evol 11:367–372
Acknowledgements
We are indebted to Prof W. Arntz, T. Brey, M. Rauschert, and the crew of R/V Polarstern, for allowing the participation of CA in the Antarctic cruises ANT XV/3 and ANT XXI/2 (AWI, Bremerhaven, Germany) and providing support during the sampling. E. Prats (CCiT-UB, Barcelona, Spain) is acknowledged for her help during the SEM sessions. We also thank M. Ballesteros, P. Brueggeman, G. Giribet, S. Harper, T. Heran, and D. Thompson for kindly providing the pictures of the live animals, and S. Taboada for helpful discussion and support. Two anonymous referees and Stefano Schiaparelli provided valuable comments that improved the ms. Funding was provided by the Spanish government through the ECOQUIM (REN2003-00545, REN2002-12006-E ANT) and DISTANTCOM (CTM2013-42667/ANT) grants to CA. JM postdoctoral fellowships were supported by the Ramón Areces (Spain) and the Alexander von Humboldt Foundation (Germany). MER was supported by an Erasmus + Grant from La Sapienza, University of Rome, at the University of Barcelona. This paper is part of the AntEco (State of the Antarctic Ecosystem) Scientific Research Programme.
Author information
Authors and Affiliations
Contributions
CA conducted the field work taking part in the expeditions and collecting the samples. JM conceived and designed the study. MER and JM conducted the experiments and analysed the data. MER and JM wrote the manuscript. CA contributed with financial support. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rossi, M.E., Avila, C. & Moles, J. Orange is the new white: taxonomic revision of Tritonia species (Gastropoda: Nudibranchia) from the Weddell Sea and Bouvet Island. Polar Biol 44, 559–573 (2021). https://doi.org/10.1007/s00300-021-02813-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02813-8