Abstract
Despite their importance in ecosystems, population sizes and trends are unknown for many seabirds, including in the Antarctic. Here we report on the first comprehensive survey of south polar skuas Stercorarius maccormicki and Antarctic shags Leucocarbo bransfieldensis in Ryder Bay, and collate previous count data. In austral summer 2017/18, totals of 259 skuas at club sites and 978 occupied skua territories were counted in 2.3 km2 of suitable habitat at Rothera Point and adjacent islands. Based on the mean nearest neighbour distance (23.2 m), skua nest densities were comparable with colonies elsewhere. Long-term monitoring of skuas at Rothera Point indicated considerable annual variation and overall increases of 1.9 and 1.3% per annum, respectively, in breeding pairs from 1975/76 to 2017/18, and occupied territories from 1988/89 to 2017/18. In total, 405 pairs of Antarctic shags bred at two known and one newly discovered colony in 2017/18. Previous counts at the two known colonies indicated substantial annual variation and increases of 5.5 and 3.3% per annum, respectively, from 1985/86 to 2017/18 and 1989/90 to 2017/18. Factors leading to overall increases in both species, and the intermittent seasons of near-complete failure to breed, are unclear, but likely to reflect impacts of environmental change on their marine prey or sea ice. The breeding populations of south polar skuas and Antarctic shags in Ryder Bay represent 10.3 and 3.5%, respectively, of revised global estimates of 9500 and 11,684 breeding pairs. We recommend that the breeding colonies be included as important bird areas (IBAs) and within the Antarctic Specially Protected Area (ASPA) system, and provision made to conserve foraging areas at sea.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seabirds are often considered to be useful sentinels of the health of the wider marine ecosystem (Constable et al. 2000; Einoder 2009). They are major consumers of marine resources, harvesting as much prey in total as that harvested globally by fisheries (Brooke 2004). In addition, predatory seabirds can have dramatic impacts on populations of their avian prey (Phillips et al. 1999b; Votier et al. 2004). Worldwide, many seabird populations are in decline; this reflects diverse threats, particularly pollution, incidental mortality (bycatch) and competition with commercial fisheries at sea, invasive species, habitat degradation, human disturbance and direct exploitation at breeding colonies (Croxall et al. 2012; Phillips et al. 2016).
Despite their utility as biomonitors and their importance in ecosystems, basic information on population sizes and trends are missing for many seabirds, even for some of the most tractable species such as the surface-nesting albatrosses (Phillips et al. 2016). Data gaps are being filled by taking advantage of new technologies, such as drones (unmanned aerial vehicles or UAVs) and time-lapse camera networks (Ratcliffe et al. 2015; Black et al. 2017; Borowicz et al. 2018). In addition, satellite remote-sensing is used increasingly to detect previously unknown seabird colonies and, in some species, to estimate population sizes (Lynch and Schwaller 2014; Fretwell et al. 2015, 2017; Borowicz et al. 2018). However, most population data are still collected by ground counts (Lynch et al. 2008; Carneiro et al. 2016; Che-Castaldo et al. 2017; Schrimpf et al. 2018).
Although the Antarctic region is often considered to be amongst the most pristine on the planet, its seabird populations have shown direct and indirect responses to climatic change, particularly its effect on winter sea ice (Fraser and Hofmann 2003; Forcada et al. 2008; Che-Castaldo et al. 2017). At the Antarctic Peninsula, there are striking disparities among species and sites in recent decades. Adelié penguins Pygoscelis adeliae have decreased in the north and west, and chinstrap penguins P. antarctica also appear to have declined recently at most sites, whereas gentoo penguins P. papua are increasing and establishing new colonies (Hinke et al. 2007; Lynch et al. 2012; Casanovas et al. 2015). The more limited data for southern giant petrels Macronectes giganteus indicate generally stable or increasing populations (Lynch et al. 2008; Gonzalez-Zevallos et al. 2013). South polar skuas Stercorarius maccormicki have, similarly, been stable or increased at most sites (reviewed by Carneiro et al. 2016), except for a recent, steep decline at King George Island, reflecting high rates of breeding deferral (Graña Grilli 2014; Krietsch et al. 2016). Trends in Antarctic shags Leucocarbo bransfieldensis are highly variable, with evidence of stable or declining populations over much of the Peninsula, contrasting with increases in southern Marguerite Bay (Schrimpf et al. 2018). Indeed, there may be a transition for both shags and Adélie penguins at Marguerite Bay, with positive population trajectories to the south (Casanovas et al. 2015; Schrimpf et al. 2018). Many of the overall trends are likely to be in response to warming air temperatures until the late 1990s, and changes in sea-ice extent affecting growth and recruitment of krill, and hence food supply for krill-dependent predators (Smith et al. 1999; Forcada et al. 2008; Turner et al. 2016). Conditions in Marguerite Bay have probably been improving for marine predators, and it has been suggested that persistent phytoplankton blooms are maintained in this region even if sea-ice extent in the summer has decreased (Casanovas et al. 2015).
Despite the presence of a research station at Ryder Bay (Adelaide Island), at the northern limit of Marguerite Bay, little has been published on the local seabird populations (Milius 2000). Two species breed there in considerable numbers: the south polar skua, an opportunistic predator and scavenger, widely distributed as a breeding species in the Antarctic but absent from subantarctic islands (Higgins and Davies 1996), and the Antarctic shag, which feeds on demersal fish, particularly nototheniids, making it one of the few potential indicators of changes in their stocks, which are otherwise difficult to assess (Casaux and Barrera-Oro 2006). This study reports on the first comprehensive survey of south polar skuas and Antarctic shags at Ryder Bay. Previous published and unpublished counts are also collated, providing an indication of trends, and the results discussed in the context of regional climatic changes and the importance of the populations in global terms. Although the Antarctic shag is often treated as a subspecies of blue-eyed or imperial shag Leucocarbo or Phalacrocorax atriceps, here we follow the recent classification of cormorants based on extensive analysis of mitochondrial and nuclear DNA sequence (Kennedy and Spencer 2014), and consider it to be a separate species.
Materials and methods
The area of Ryder Bay surveyed for Antarctic shags and south polar skuas included Rothera Point (Adelaide Island), Killingbeck Island, the Léonie Islands group and, for shags only, the Mikkelsen Islands (Fig. 1). Most of the islands have patches of persistent snow, and Léonie Island (max. height 502 m a.s.l.) has a large permanent ice cap. The number of breeding pairs (with eggs or chicks) of south polar skuas at Rothera Point were first counted in the 1975/76 breeding season, and have been monitored annually since 1988/89, involving visits at least weekly to count occupied territories and record breeding success. Dedicated visits to Killingbeck Island and the Léonie Islands to count skuas were made between 16 January and 5 February 2018, corresponding to the late incubation to early chick-rearing periods. The count unit was ‘occupied territory’, which is standard in skua censuses (Phillips et al. 1999a; Carneiro et al. 2016; Krietsch et al. 2016). Occupied territories are indicated by the presence of vigilant adults or an incubating bird visible from a distance, and in the great majority of cases, birds actively defended their territory by mobbing when approached. At Rothera Point, Killingbeck, Kirsty, West Lagoon and East Lagoon islands, the position of nests or the centre of occupied territories was recorded using hand-held GPS loggers. At Anchorage, Léonie and Donnelly islands, the number of occupied territories was noted in subareas, using topography and the presence of snow patches to minimise the risk of missing or double-counting territories. In addition, the number of birds was counted at club sites (traditional roosting and bathing areas used by nonbreeding and to some extent also breeding skuas; Fig. 1).
a Breeding locations of Antarctic shags and south polar skuas in Ryder Bay (Antarctic Peninsula), with inset map showing general location, b Rothera Point, c Killingbeck Island, d Kirsty, Muckelscarf, East Lagoon and West Lagoon islands, e Léonie Island and f Trolval, Anchorage and Donnelly islands. Numbers of occupied skua territories in subareas shown in panels e and f
The number of eggs and chicks at each skua territory was noted opportunistically, but the data are not presented here because of possible losses prior to the survey. Territory occupancy rates were very high, based on the high densities of skuas on the islands and the presence of at least one member of failed or nonbreeding pairs during twice-weekly visits to the intensive-study colony at Rothera Point during the survey period. No corrections were therefore made for previous nest failure.
An image mosaic of the study area was available as the electronic version of a published map (British Antarctic Survey BAS 25 Series, Sheet 3, Ryder Bay, Scale 1:25,000). This was compiled from colour vertical aerial photographs acquired on 20 January 2005, and a 3D surface model derived from photogrammetry. Snow patches marked on this map appeared to cover almost exactly the same areas at the time of the skua surveys in 2018, and were discriminated from rock substrate in a GIS coverage according to spectral analysis of the aerial photographs used as a base layer. Skua nesting density (pairs per km2) in 2018 was calculated in three ways according to (i) the planar (2D) area of each island (excluding the ice cap on Léonie Island and adjacent, unoccupied islets), and of the rock outcrop and adjacent beach east of the station buildings at Rothera Point; (ii) the planar area of suitable habitat (excluding snow patches at all sites, the ice cap and scree slopes above 100 m a.s.l. on Léonie Island, and unoccupied islets); and (iii) the ground surface area of suitable habitat corrected for elevation (estimated from the 3D surface model using the 3D Analyst component of ArcGIS). In addition, minimum distances between neighbouring nests or territories were calculated for sites where locations were available from the hand-held GPS loggers.
Previous surveys had recorded Antarctic shags breeding at three locations in the Ryder Bay area: Mucklescarf Island (Lagoon Islands), Killingbeck Island and adjacent islet (these separated by only 100 m). The count unit was either ‘active nest’ or ‘occupied nest site’, which are the usual census methods for this species (Schrimpf et al. 2018). In addition, nest contents (eggs or number of chicks at each nest) were noted in some years. No data were available in previous years to correct for earlier breeding failure, and only counts made between early December and mid-February are included here, as later counts will underestimate breeding numbers if some chicks have already fledged. An additional shag breeding site on Skart Island (Mikkelsen Islands) was discovered and photographed from the air in February 2018, and the number of occupied nest sites counted from the images.
Results
South polar skuas
A total of 978 occupied skua territories were counted in surveys during the late incubation to early chick-rearing periods (16 January–5 February 2018) at Rothera Point, Killingbeck, Léonie, Kirsty, West Lagoon, East Lagoon, Trolval, Anchorage, and Donnelly islands in Ryder Bay (Table 1). Skua territories were widely distributed across snow-free ground on all islands, except on Trolval Island (the smallest of the islands), and scree above 100 m on Léonie Island (Fig. 1). Without correcting for habitat suitability, other than excluding the ice cap on Léonie Island, nesting densities ranged from 115 territories per km2 at Rothera Point to 544 territories per km2 on East Lagoon (overall 288 territories per km2). Restricting the analysis to areas of suitable habitat (excluding ice cap, snow patches, and scree above 100 m a.s.l.), nesting densities ranged from 174 to 634 (overall 447) territories per km2; correcting for elevation, densities reduced slightly to 169–610 (overall 420) occupied territories per km2 (Table 1). Densities on the islands where the skuas were counted in small subareas differentiated by local topography (Léonie, Anchorage and Donnelly) were within the range of those on islands where a GPS location was taken on each territory. Average nearest neighbour distances were greater at Rothera Point than on the other more densely populated islands (40.4 and 19.2–28.5 m, respectively). In addition, a total of 259 birds was recorded at club sites on the three islands with the largest breeding skua populations.
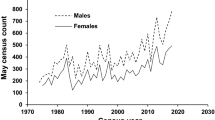
Long-term data indicated that the population size at Rothera Point varied considerably between years, increasing overall by 1.9% per annum from 11 breeding pairs in 1975/76 to 24 breeding pairs in 2017/18 (Fig. 2). Counts of the number of occupied territories (i.e. including nonbreeding birds holding territory) were available from 1988/89; these showed less annual variation than breeding pairs, and an increase of 1.3% per annum from 17 in 1988/89 to 25 in 2017/18.
Antarctic shags
In total, 405 pairs of Antarctic shags were breeding at the colonies in Ryder Bay—Killingbeck Island, Mucklescarf Island (Lagoon Islands) and Scart Island (Mikkelsen Islands)—in 26 January–3 February 2018 (Table 2). An additional nine and seven well-built, but empty nests were recorded at Muckelscarf Island and Killingbeck Island on the same dates as the 251 and 74 active nests, suggesting failure rates prior to the surveys of 3.5 and 8.6%, respectively (4.7% or 16/341 overall). Nevertheless, no correction was made for previous failures to ensure counts could be compared with those of active nests in earlier years. The latter were available for Killingbeck Island (including the adjacent islet) or Muckelscarf Island in nine seasons, and showed substantial annual variation, including near-complete failure to breed in the 1983/84, 1998/99 and 2016/17 seasons, and an apparent stepwise increase in the population sometime in the late 1990s to early 2000s (Fig. 3). Excluding the three seasons when very few or no birds bred, the long-term trend at Muckelscarf Island was an increase of 5.5% per annum from 45 to 251 breeding pairs in 1985/86 to 2017/18. Numbers at Killingbeck Island also increased, at 3.3% per annum from 30 to 74 breeding pairs in 1989/90 to 2017/18 (for comparison, the trend at Muckelscarf Island over the same period was an increase of 5.4% per annum).
Discussion
The survey methodologies followed standard approaches for both skuas (Phillips et al. 1999a, 2004; Carneiro et al. 2016; Krietsch et al. 2016) and shags (Schrimpf et al. 2018). These have an associated observation error. For the skuas, this includes potentially lower detectability of birds obscured by local topography and possible over-counting of birds in the air. However, densities on islands on which the skuas were counted in subareas were in the middle of the range of those where a GPS fix was obtained from each territory, suggesting that count method was not a confounding factor. No attempt was made to correct for previous nest failure in either shags or skuas. In the skuas, this was because in repeated visits at Rothera Point, at least one partner was present at territories where the breeding attempt had failed earlier in the season. In addition, based on the small proportion of empty but well-built nests (4.7%), apparently very few shags failed before the survey. Overall, therefore, the error in our counts was expected to be low.
The populations of south polar skuas (978 occupied territories) and Antarctic shags (405 pairs) in the Ryder Bay area counted in the 2017/18 breeding season are both of major regional and international importance, and are well above the threshold numbers −1% or more of the global population—used recently to identify Important Bird [and Biodiversity] Areas (IBAs) in Antarctica using criteria adopted by BirdLife International (Harris et al. 2015). A further 259 skuas were recorded at club sites during the census (Table 1), most of which would not have been territory-holders, indicating a substantial pool of nonbreeders. Densities were 289, 447 and 420 occupied skua territories per km2, respectively, either overall (excluding the icecap), in suitable habitat (planar area) or suitable habitat after correcting for elevation (Table 1). The average nearest neighbour distance of 23.2 m is comparable with those of south polar skuas elsewhere, including at Cape Royds, Cape Hallett, Cape Crozier, Pointe Géologie, Anvers Island and Edmonson Point (Higgins and Davies 1996; Pezzo et al. 2001). These densities are considerably higher than in brown skuas Stercorarius antarcticus, of which the most densely populated colony worldwide appears to be at Bird Island (South Georgia), with 105 pairs per km2 across the whole island, 132 pairs per km2 in suitable habitat, and an average nearest nest distance of 44 m (Phillips et al. 2004).
The trend in skua numbers breeding at Rothera Point is based on relatively few pairs, but if it applies to the other islands, the local population of skuas is increasing. Information on trends of south polar skuas is available for very few sites, particularly for the last 10–20 years; however, the limited data suggest that populations were generally stable or increasing until the 1990s or 2000s, although with a recent decline in breeding numbers attributed to a high rate of deferral at King George Island (Graña Grilli 2014; Carneiro et al. 2016; Krietsch et al. 2016). Population increases at skua colonies in the Antarctic and subantarctic, particularly of brown skuas, are often attributed to anthropogenic factors, including feeding on refuse at research stations, the presence of alien mammals providing supplementary prey, rapid increase in penguins (and therefore potential prey) following the near-extirpation of most Antarctic whales, or the increase in Antarctic fur seals Arctocephalus gazella following the cessation of sealing, providing abundant carrion (Hemmings 1990; Phillips et al. 2004; Ryan et al. 2009; Carneiro et al. 2016). There must be an alternative explanation for the increase of south polar skuas at Rothera Point as efficient waste management at the research station ensures there is minimal refuse available, and there are no alien mammals nor evidence for an increase in carrion from seals. In the absence of brown skuas, which are larger and competitively dominant, south polar skuas will exploit penguins and other seabirds, scavenging or depredating eggs, chicks or adults, depending on the species (Reinhardt et al. 2000). However, there are no penguin colonies in the Ryder Bay area, and no seabird or seal remains were seen in skua pellets or as dropped items, suggesting a diet of fish (Young 1990). Hence, this large population must feed predominantly or entirely at sea, possibly on Antarctic silverfish Pleuragramma antarcticum, as do the south polar skuas breeding around Palmer station (c. 370 km away) and some other colonies to the north (Pietz 1987; Reinhardt et al. 2000). Increased breeding numbers at Ryder Bay are therefore likely to reflect greater availability of fish or other prey in the marine environment.
The long-term increase in skuas at Rothera Point is despite intermittent years of low breeding numbers (Fig. 2); the latter presumably reflects occasionally high rates of breeding deferral by established adults and potentially low recruitment of new breeders, as in other skua populations (Ainley et al. 1990; Catry et al. 1998). Around Palmer Station, few skuas bred and no chicks hatched in a year of persistent heavy ice (Parmalee et al. 1978). The proportion of adults breeding also varies greatly in other species of Antarctic seabirds depending on environmental conditions, including sea-ice extent and duration (Olivier et al. 2005; Che-Castaldo et al. 2017). The reasons for breeding deferral in certain seasons by many skuas at Rothera Point are unclear, but could include birds returning from migration in poor body condition, late snow covering nest sites, low marine productivity, extreme weather or unfavourable sea-ice conditions. With regard to the sea ice, this can function as a physical barrier over the water surface, preventing feeding, or affect productivity and hence food availability for the fish on which the birds seem to depend. Although sea-ice cover around Antarctica has expanded slightly in recent decades, there are opposing regional trends, partly driven by changing wind regimes (Holland and Kwok 2012). Winter sea ice in the Peninsula region has reduced in duration and extent, and shown increased variability in recent decades (Fraser and Hofmann 2003; Stammerjohn et al. 2012). As this variability is predicted to increase (Bracegirdle et al. 2008), future conditions for the skuas are uncertain.
Although the first counts of south polar skuas at Rothera Point were in the 1975/76 season, and annual monitoring began in 1988/89, the presence of a major global population of this species in Ryder Bay was undocumented in the published literature until estimates of 100s present on Lagoon and Anchorage islands were noted as pers. comm. in the supplementary material of Fretwell et al. (2015). Previous global totals estimated for this species are 5000–8000 breeding pairs in the mid-1980s (Furness 1987), and, just recently, 8529 breeding pairs, including improved estimates for Victoria, Marie Byrd and Ellesworth Lands based on relationships between skua density and penguin numbers (Wilson et al. 2017). With the addition of the 978 pairs in Ryder Bay, an improved estimate of the global population of south polar skuas is therefore 9500 breeding pairs, of which the Ryder Bay population represents 10.3%. Our revised global total is nearly double that used by Harris et al. (2015) to establish a 1% threshold of 50 pairs of south polar skuas used to identify sites as IBAs; as such, several IBAs in the current list (one near Anvers Island and others in the Ross Sea region) established solely on the basis of the local skua population would no longer meet this criterion.
The Antarctic shag colony at the Mikkelsen Islands (Skart Island) was a new discovery in 2018. The other two colonies (Muckelscarf and Killingbeck islands) were taken into account, using the combined total of 88 pairs in 1989/90 (see Table 2), in the recent review by Schrimpf et al. (2018). Hence, our new data have only a small effect on the estimated global population, increasing it from 11,366 breeding pairs at 173 active colonies (Schrimpf et al. 2018) to 11,684 pairs at 174 active colonies. This excludes the shags breeding in the South Orkney Islands, which are usually assumed to be South Georgia shags Leucocarbo [atriceps] georgianus, although apparently without strong genetic or other evidence (Schrimpf et al. 2018). Based on this revised total, the current population of Antarctic shags on the islands in Ryder Bay represents 3.5% of global breeding numbers. In addition, at 251 pairs, the colony on Muckelscarf Island is unusually large for this species; only 11 other colonies (<10% of those recorded) hold ≥200 pairs (Schrimpf et al. 2018). Although our results indicate only a minor change in global breeding numbers of Antarctic shags, Schrimpf et al. (2018) point out that seven of the sites identified as IBAs by Harris et al. (2015) no longer qualify for this species because the number of pairs has since declined to below the original 1% threshold (133 pairs).
Both south polar skua and blue-eyed shag sensu stricto are currently listed as Least Concern by the World Conservation Union (IUCN). Regardless, BirdLife International, which is a global partnership of conservation organisations that strives to conserve birds and their habitats, seeks to identify sites of international conservation significance for all birds, not limited to those on the IUCN Red List. Given the presence of 10.3 and 3.5%, respectively, of global numbers, the populations of south polar skua and Antarctic shag in Ryder Bay should be included in the list of BirdLife IBAs for the Antarctic region. The other sites already designated as both IBAs and ASPAs for one or both of these species in Marguerite Bay are Avian, Lagotellerie, Dion and Stonington islands (Harris et al. 2015). In addition, part or all of the Ryder Bay islands should be considered for designation as an Antarctic Specially Protected Area (ASPA), to ensure their intrinsic environmental value (as ‘major colonies of breeding native birds’) is maintained while making provision for scientific research (Hughes et al. 2013).
There is increasing recognition of the need for improved marine protection not just within the Antarctic Treaty System, but to address the goal of the Convention on Biological Diversity (CBD) to identify a comprehensive global network of ecologically or biologically significant areas (EBSAs), including those outside national jurisdiction (Grant et al. 2012). To date, development of the network of IBAs and ASPAs in the region covered by the Antarctic Treaty System has focused much more on terrestrial than marine areas (Grant et al. 2012; Shaw et al. 2014). Scaling up from the estimated 3–5 tons of pelagic fish consumed annually by c. 50 breeding pairs of south polar skuas at King George Island (Hahn et al. 2008), the total for the much larger population of skuas in Ryder Bay (978 pairs) in 2017/18 would be in the order of 60–100 tons. Hence, the marine foraging areas on which they depend also need to be safeguarded, potentially by designation as Marine Protected Areas (MPAs). In addition, given the striking regional variation in population trajectories of south polar skuas, Antarctic shags and other seabirds, and the lack of reliable time-series for most colonies (Hinke et al. 2007; Lynch et al. 2012; Casanovas et al. 2015; Schrimpf et al. 2018; this study), monitoring of their breeding populations in Ryder Bay should be a priority.
References
Ainley DG, Ribic CA, Wood RC (1990) A demographic study of the South Polar Skua Catharacta maccormicki at Cape Crozier. J Anim Ecol 59:1–20
Black C, Rey AR, Hart T (2017) Peeking into the bleak midwinter: Investigating nonbreeding strategies of Gentoo Penguins using a camera network. Auk 134:520–529
Borowicz A, McDowall P, Youngflesh C, Sayre-McCord T, Clucas G, Herman R, Forrest S, Rider M, Schwaller M, Hart T (2018) Multi-modal survey of Adélie penguin mega-colonies reveals the Danger Islands as a seabird hotspot. Sci Rep 8:3926
Bracegirdle TJ, Connolley WM, Turner J (2008) Antarctic climate change over the twenty first century. J Geophys Res Atmos 113:D03103
Brooke ML (2004) The food consumption of the world's seabirds. Proc R Soc Lond B 271:S246–S248
Carneiro AP, Manica A, Phillips RA (2016) Long-term changes in population size, distribution and productivity of skuas (Stercorarius spp.) at Signy Island, South Orkney Islands. Polar Biol 39:617–625
Casanovas P, Naveen R, Forrest S, Poncet J, Lynch HJ (2015) A comprehensive coastal seabird survey maps out the front lines of ecological change on the western Antarctic Peninsula. Polar Biol 38:927–940
Casaux R, Barrera-Oro E (2006) Shags in Antarctica: their feeding behaviour and ecological role in the marine food web. Antarct Sci 18:3–14
Catry P, Phillips RA, Hamer KC, Ratcliffe N, Furness RW (1998) The incidence of nonbreeding by adult great skuas and parasitic jaegers from Foula, Shetland. Condor 100:448–455
Che-Castaldo C, Jenouvrier S, Youngflesh C, Shoemaker KT, Humphries G, McDowall P, Landrum L, Holland MM, Li Y, Ji R, Lynch HJ (2017) Pan-Antarctic analysis aggregating spatial estimates of Adélie penguin abundance reveals robust dynamics despite stochastic noise. Nat Commun 8:832
Constable AJ, de la Mare WK, Agnew DJ, Everson I, Miller D (2000) Managing fisheries to conserve the Antarctic marine ecosystem: practical implementation of the Convention on the Conservation of Antarctic Marine Living Resources (CCAMLR). ICES J Mar Sci 57:778–791
Croxall JP, Butchart SHM, Lascelles B, Stattersfield AJ, Sullivan B, Symes A, Taylor P (2012) Seabird conservation status, threats and priority actions: a global assessment. Bird Conserv Int 22:1–34
Einoder LD (2009) A review of the use of seabirds as indicators in fisheries and ecosystem management. Fish Res 95:6–13
Forcada J, Trathan PN, Murphy EC (2008) Life history buffering in Antarctic mammals and birds against changing patterns of climate and environmental variation. Glob Change Biol 14:2473–2488
Fraser WR, Hofmann EE (2003) A predator’s perspective on causal links between climate change, physical forcing and ecosystem response. Mar Ecol Prog Ser 265:1–15
Fretwell PT, Phillips RA, Brooke MDL, Fleming AH, McArthur A (2015) Using the unique spectral signature of guano to identify unknown seabird colonies. Remote Sens Environ 156:448–456
Fretwell PT, Scofield P, Phillips RA (2017) Using super-high resolution satellite imagery to census threatened albatrosses. Ibis 159:481–490
Furness RW (1987) The Skuas. T. & A.D, Poyser, Calton, Staffordshire
Gonzalez-Zevallos D, Santos MM, Rombolá EF, Juáres MA, Coria NR (2013) Abundance and breeding distribution of seabirds in the northern part of the Danco Coast, Antarctic Peninsula. Polar Res 32:11133
Graña Grilli M (2014) Decline in numbers of Antarctic skuas breeding at Potter Peninsula, King George Island, Antarctica. Mar Ornithol 42:161–162
Grant SM, Convey P, Hughes KA, Phillips RA, Trathan PN (2012) Conservation and management of Antarctic ecosystems. In: Rogers AD, Murphy EJ, Clarke A, Johnston NM (eds) Antarctic ecosystems: an extreme environment in a changing world. Wiley, London, pp 492–525
Hahn S, Ritz M, Reinhardt K (2008) Marine foraging and annual fish consumption of a south polar skua population in the maritime Antarctic. Polar Biol 31:959–969
Harris CM, Lorenz K, Fishpool LDC, Lascelles B, Cooper J, Coria NR, Croxall JP, Emmerson LM, Fijn RC, Fraser WL, Jouventin P, LaRue MA, Le Maho Y, Lynch HJ, Naveen R, Patterson-Fraser DL, Peter H-U, Poncet S, Phillips RA, Southwell CJ, van Franeker JA, Weimerskirch H, Wienecke B, Woehler EJ (2015) Important bird areas in Antarctica 2015. BirdLife International and Environmental Research & Assessment Ltd., Cambridge
Hemmings AD (1990) Human impacts and ecological constraints on skuas. In: Kerry KR, Hempel G (eds) Antarctic ecosytems: ecological change and conservation. Springer, Berlin, pp 224–230
Higgins PJ, Davies SJJF (1996) Handbook of Australian, New Zealand and Antarctic birds. Vol 3. Snipe to pigeons. Oxford University Press, Melbourne
Hinke JT, Salwicka K, Trivelpiece SG, Watters GM, Trivelpiece WZ (2007) Divergent responses of Pygoscelis penguins reveal a common environmental driver. Oecologia 153:845–855
Holland PR, Kwok R (2012) Wind-driven trends in Antarctic sea-ice drift. Nat Geosci 5:872–875
Hughes K, Pertierra L, Walton D (2013) Area protection in Antarctica: How can conservation and scientific research goals be managed compatibly? Environ Sci Policy 31:120–132
Kennedy M, Spencer HG (2014) Classification of the cormorants of the world. Mol Phylogenet Evol 79:249–257
Krietsch J, Esefeld J, Braun C, Lisovski S, Peter H-U (2016) Long-term dataset reveals declines in breeding success and high fluctuations in the number of breeding pairs in two skua species breeding on King George Island. Polar Biol 39:573–582
Lynch HJ, Schwaller MR (2014) Mapping the abundance and distribution of Adélie Penguins using Landsat-7: first steps towards an integrated multi-sensor pipeline for tracking populations at the continental scale. PLoS ONE 9:e113301
Lynch HJ, Naveen R, Fagan WF (2008) Censuses of penguin, blue-eyed shag Phalacrocorax atriceps and southern giant petrel Macronectes giganteus populations on the Antarctic Peninsula, 2001–2007. Mar Ornithol 36:83–97
Lynch HJ, Naveen R, Trathan PN, Fagan WF (2012) Spatially integrated assessment reveals widespread changes in penguin populations on the Antarctic Peninsula. Ecology 93:1367–1377
Milius N (2000) The birds of Rothera, Adelaide Island, Antarctic Peninsula. Mar Ornithol 28:63–67
Olivier F, van Franeker JA, Creuwels JCS, Woehler EJ (2005) Variations of snow petrel breeding success in relation to sea-ice extent: detecting local response to large-scale processes? Polar Biol 28:687–699
Parmalee DF, Bernstein NP, Neilson DR (1978) Impact of unfavorable ice conditions on bird productivity at Palmer Station during the 1977–78 field season. Antarct J US 13:146–147
Pezzo F, Olmastroni S, Corsolini S, Focardi S (2001) Factors affecting the breeding success of the south polar skua Catharacta maccormicki at Edmonson Point, Victoria Land, Antarctica. Polar Biol 24:389–393
Phillips RA, Bearhop S, Hamer KC, Thompson D (1999a) Rapid population growth of Great Skuas Catharacta skua at St Kilda: implications for management and conservation. Bird Study 46:174–183
Phillips RA, Thompson DR, Hamer KC (1999b) The impact of great skua predation on seabird populations at St Kilda: a bioenergetics model. J Appl Ecol 36:218–232
Phillips RA, Phalan B, Forster IP (2004) Diet and long-term changes in population size and productivity of brown skuas Catharacta antarctica lonnbergi at Bird Island, South Georgia. Polar Biol 27:555–561
Phillips RA, Gales R, Baker G, Double M, Favero M, Quintana F, Tasker ML, Weimerskirch H, Uhart M, Wolfaardt A (2016) The conservation status and priorities for albatrosses and large petrels. Biol Conserv 201:169–183
Pietz PJ (1987) Feeding and nesting ecology of sympatric south polar and brown skuas. Auk 104:617–627
Ratcliffe N, Guihen D, Robst J, Crofts S, Stanworth A, Enderlein P (2015) A protocol for the aerial survey of penguin colonies using UAVs. J Unmanned Veh Syst 3:95–101
Reinhardt K, Hahn S, Peter H-U, Wemhof H (2000) A review of the diets of Southern Hemisphere skuas. Mar Ornithol 28:7–19
Ryan P, Whittington P, Crawford RJ (2009) A tale of two islands: contrasting fortunes for subantarctic skuas at the Prince Edward Islands. Afr J Mar Sci 31:431–437
Schrimpf M, Naveen R, Lynch HJ (2018) Population status of the Antarctic shag Phalacrocorax (atriceps) bransfieldensis. Antarct Sci 30:151–159
Shaw JD, Terauds A, Riddle MJ, Possingham HP, Chown SL (2014) Antarctica’s protected areas are inadequate, unrepresentative, and at risk. PLoS Biol 12:e1001888
Smith RC, Ainley D, Baker K, Domack E, Emslie S, Fraser B, Kennett J, Leventer A, Mosley-Thompson E, Stammerjohn S (1999) Marine ecosystem sensitivity to climate change: historical observations and paleoecological records reveal ecological transitions in the Antarctic Peninsula region. BioSci 49:393–404
Stammerjohn S, Massom R, Rind D, Martinson D (2012) Regions of rapid sea ice change: an inter-hemispheric seasonal comparison. Geophys Res Lett 39:L06501
Turner J, Lu H, White I, King JC, Phillips T, Hosking JS, Bracegirdle TJ, Marshall GJ, Mulvaney R, Deb P (2016) Absence of 21st century warming on Antarctic Peninsula consistent with natural variability. Nature 535:411–415
Votier SC, Bearhop S, Ratcliffe N, Phillips RA, Furness RW (2004) Predation by great skuas at a large Shetland seabird colony. J Appl Ecol 41:1117–1128
Wilson DJ, Lyver POB, Greene TC, Whitehead AL, Dugger KM, Karl BJ, Barringer JR, McGarry R, Pollard AM, Ainley DG (2017) South Polar Skua breeding populations in the Ross Sea assessed from demonstrated relationship with Adélie Penguin numbers. Polar Biol 40:577–592
Young EC (1990) Diet of the south polar skua Catharacta maccormicki determined from regurgitated pellets: limitations of a technique. Polar Rec 26:124–125
Acknowledgements
We are very grateful to the many field assistants for help with monitoring of skuas at Rothera Point over many years, to Ali Rose for help with the skua census in 2018, to Peter Fretwell for photographing and counting the shag colony at the Mikkelsen Islands and to Sally Poncet for providing unpublished counts of shag nests. The boating officers and crew at Rothera diligently transported us to the Ryder Bay islands. We are grateful to Johannes Krietsch, Jan Esefeld and an anonymous referee for their comments on the manuscript. This study represents a contribution to the Ecosystems Component of the British Antarctic Survey Polar Science for Planet Earth Programme and the Environment Office Long-term Monitoring and Survey project (EO-LTMS), funded by the Natural Environment Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors are not aware of any conflicts of interest with regard to this study.
Rights and permissions
About this article
Cite this article
Phillips, R.A., Silk, J.R.D., Massey, A. et al. Surveys reveal increasing and globally important populations of south polar skuas and Antarctic shags in Ryder Bay (Antarctic Peninsula). Polar Biol 42, 423–432 (2019). https://doi.org/10.1007/s00300-018-2432-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-018-2432-0