Abstract
Key message
Identification of three genomic regions and underlying candidate genes controlling the high level of resistance to ToLCNDV derived from a wild melon. SNP markers appropriated for MAS management of resistance.
Abstract
Tomato leaf curl New Delhi virus (ToLCNDV) is a bipartite begomovirus that severely affects melon crop (Cucumis melo) in the main production areas of Spain since 2012. In this work, we evaluated the degree of resistance of four accessions (two belonging to the subsp. agrestis var. momordica and two to the wild agrestis group) and their corresponding hybrids with a susceptible commercial melon belonging to the subsp. melo (Piel de Sapo, PS). The analysis using quantitative PCR (qPCR) allowed us to select one wild agrestis genotype (WM-7) with a high level of resistance and use it to construct segregating populations (F 2 and backcrosses). These populations were phenotyped for symptom severity and virus content using qPCR, and genotyped with different sets of SNP markers. Phenotyping and genotyping results in the F 2 and BC1s populations derived from the WM-7 × PS cross were used for QTL analysis. Three genomic regions controlling resistance to ToLCNDV were found, one major locus in chromosome 11 and two additional regions in chromosomes 12 and 2. The highest level of resistance (no or mild symptoms and very low viral titer) was obtained with the homozygous WM-7WM-7 genotype at the major QTL in chromosome 11, even with PSPS genotypes at the other two loci. The resistance derived from WM-7 is useful to develop new melon cultivars and the linked SNPs selected in this paper will be highly useful in marker-assisted breeding for ToLCNDV resistance in melon.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tomato leaf curl New Delhi virus (ToLCNDV) causes a devastating viral disease that threatens the main cucurbit crops in Spain since 2012 (Juárez et al. 2014). It is a Begomovirus (family Geminiviridae) with two circular single-stranded DNA molecules of about 2.6–2.7 kb, referred to as DNA-A and DNA-B, both required for essential viral functions and encapsidated in geminate particles (Papidam et al. 1995; Jyothsna et al. 2013). The virus is transmitted by the whitefly Bemisia tabaci Genn. in a persistent manner (Chang et al. 2010; Rosen et al. 2015) and it can be artificially inoculated mechanically (López et al. 2015).
ToLCNDV was first detected on tomato (Solanum lycopersicum L.) in 1995 in north India (Srivastava et al. 1995) and soon after was found in other south and southeast Asian countries. The host range was extended to other plant species, mainly crops of the Cucurbitaceae and Solanaceae families (Chang et al. 2010; Pratap et al. 2011; Khan et al. 2012; Jyothsna et al. 2013; Bandaranayake et al. 2014). Before 2012, ToLCNDV was limited to Asian countries; thereafter, it was reported in different Mediterranean countries, affecting cucurbits [mainly zucchini (Cucurbita pepo L. subsp. pepo), melon (Cucumis melo L.), and cucumber (Cucumis sativus L.)]. It was initially detected in Spain and more recently in Tunisia and Italy (Juárez et al. 2014; Mnari-Hattab et al. 2015; Panno et al. 2016). In the background of its potential spread, European and Mediterranean Plant Protection Organization (EPPO) has added this virus to the EPPO Alert List (EPPO 2017).
The Spanish strain of ToLCNDV was first isolated from zucchini plants grown in Murcia and Almería in 2012–2013 (Juárez et al. 2014). Further analysis of Spanish isolates from different regions and hosts showed genetic uniformity, compatible with a recent introduction. This uniformity contrasts with the high heterogeneity found in isolates from other parts of the world (Fortes et al. 2016; Ruiz et al. 2016).
In southern Spain, severe epidemic outbreaks of the disease cause serious economic losses to greenhouses and open field zucchini squash and melon crops. In melon, the disease has spread to open fields in central Spain, the most traditional Spanish area of extensive melon cultivation. Yield losses around 20% have been reported in that area (Hernández 2016). Melon plants affected by ToLCNDV show curled leaves with yellow mosaic, stunted growth, and longitudinal cracking of ovaries and fruits (Juárez et al. 2014; Font et al. 2015; EPPO 2017). Some control strategies are being applied to prevent infections, relying on whitefly control, structural measures in greenhouses, and elimination of infected plants, but resistant commercial cultivars have not been released yet (EPPO 2017).
The knowledge of the genetic basis of the resistance to ToLCNDV is necessary to perform breeding programs. Only in sponge gourd (Luffa cylindrica M. Roem.) and in some Solanum species, the resistance to ToLCNDV has been characterized. This resistance has been found to be monogenic in Luffa cylindrica and polygenic in Solanum spp. (Islam et al. 2010, 2011; Rai et al. 2013). In Cucumis melo, López et al. (2015) screened a large germplasm collection representing the diversity of the species as a previous step. They identified resistance to the Spanish isolate of ToLCNDV in five melon genotypes, all from India and belonging to the subsp. agrestis (Naudin) Pangalo (three accessions of the momordica horticultural group and two wild agrestis). In this work, we aimed to identify QTLs controlling the resistance to ToLCNDV in C. melo using segregating populations derived from one of these resistant sources and a susceptible Piel de Sapo melon (belonging to the subsp. melo).
Materials and methods
Plant material
Five Indian accessions of Cucumis melo subsp. agrestis were identified as resistant by López et al. (2015) after mechanical inoculation with ToLCNDV: three cultivated momordica and two wild agrestis types. In this work, to validate the resistant response to the virus infection, we assayed two momordica (PI-414723 and Kharbuja) and two wild types (WM-7 and WM-9). The cultivated accession Piñonet Piel de Sapo (PS) (Cucumis melo subsp. melo var. inodorus) was used as susceptible control.
Seeds of PI-414723 and Kharbuja were supplied by USDA-NPGS and PS by COMAV genebanks, and seeds of WM-7 and WM-9 came from the Indian collection described in Roy et al. (2012). All accessions were multiplied by selfing at COMAV. Seeds were disinfected in a 5% solution of sodium hypochlorite for 3 min and washed for 5 min in distilled water. Germination was performed in Petri plates with moistened cotton at 37 °C for 48 h. Seedlings were transplanted to pots in a growth chamber under a photoperiod of 16 h day at 25 °C and 8 h night at 18 °C and 70% RH. Eight plants per genotype were mechanically inoculated and assessed for resistance, using symptom scores and quantitative PCR (qPCR). Six additional plants per genotype were grown in a greenhouse with plants of the PS cultivar to generate the four F 1 hybrid progenies. Eight plants of each hybrid were phenotyped for resistance to ToLCNDV. Six plants of the PS cultivar were used as susceptible controls in each inoculation assay. The screening results suggested that wild agrestis had better resistance levels than momordica sources, so we selected one of the wild accessions (WM-7) to generate the segregating populations for genetic studies. We selected WM-7 after genotyping both wild accessions, because WM-9 was highly heterozygous, whereas WM-7 was homozygous for most of the analyzed loci. Plants of WM-7, PS, and their F 1 generation were cultivated in a greenhouse to generate F 2, BC1PS, and BC1WM-7 (backcrosses to PS and to WM-7, respectively) segregating populations. Both F 2 and BC1 populations were screened against ToLCNDV (158 plants of the F 2 and 70 of each BC1PS and BC1WM-7 populations, respectively).
Inoculation method
A ToLCNDV-infectious clone, with a 99% nucleotide identity with the sequence of the Spanish isolate KF749224 and KF749225 (Juárez et al. 2014), was agroinfiltrated by injection into petioles of MU-CU-16 zucchini plants, which showed ToLCNDV symptoms 15 days later (Sáez et al. 2016). ToLCNDV transmission to melon plants was performed by mechanical inoculation as described by López et al. (2015). Leaf tissue from the MU-CU-16 plants was mashed in the presence of inoculation buffer in a 1:4 (w:v) proportion. All plants were mechanically inoculated at the stage of two true leaves, leaving one not inoculated as negative control. For each plant, one cotyledon and one fully expanded leaf, previously dusted with carborundum 600 mesh, were gently rubbed with a cotton-swab dipped in the homogenate inoculum. All plants were re-inoculated 10 days after the first mechanical inoculation to prevent them from escaping to the infection.
To evaluate the behavior of the F 1 WM-7 × PS under natural infection conditions, ten plants of the F 1 and PS were grown in a greenhouse located in El Ejido (Almería) along with 4000 commercial plans of Cucumis melo subsp. melo var. cantalupensis. ToLCNDV infection in this commercial greenhouse was due to the presence of viruliferous whiteflies. The F 1 plants and the PS controls were planted at the end of February (2016) and evaluated for symptomatology and virus titer at the beginning of May.
Disease assessment: symptoms and qPCR
All the mechanically inoculated plants and controls were grown in a climatic chamber. Symptoms of ToLCNDV were scored at 15 and 30 day post-inoculation (dpi), following the visual scale described in López et al. (2015), ranging from 0 (absence of symptoms) to 4 (highly severe symptoms). We used the Chi-squared (χ 2) test to assess whether the number of resistant/susceptible plants fitted the expected segregation.
Quantitative PCR was used to quantify the amount of virus. The five parents and the corresponding F 1s were analyzed at 15 and 30 dpi. Viral accumulation was also analyzed at 30 dpi in all plants of the segregating populations derived from the WM-7 × PS (F 2, BC1PS, and BC1WM-7). Total DNA from apical leaves was extracted using the Cetyltrimethyl ammonium bromide (CTAB) method (Doyle and Doyle 1990) and quantified using a NanoDrop 1000 spectrophotometer (Thermo Scientific, Waltham, MA, USA). DNA was diluted with sterile-deionized water to a final concentration of 5 ng μL−1. Equal amounts of genomic DNA (15 ng) were used as templates in qPCR reactions of 15 μL, containing 7.5 μl of 2× iTaq™ universal SYBR® Green Supermix (Bio-Rad), 1.5 μl of each primer, and 1.5 μl of H2O. Primers ToLCNDVF1 (5′-AATGCCGACTACACCAAGCAT-3′, positions 1145–1169) and ToLCNDVR1 (5′-GGATCGAGCAGAGAGTGGCG-3′, positions 1399–1418) were used for the amplification of a 273 bp fragment of viral DNA-A. The two single copy Cucumis melo genes CmPEROX and CmWIP1 (Mascarell-Creus et al. 2009) were amplified in all samples as internal DNA extraction controls using the primers CmPEROXF (5′-ACTCGATCAACTTCGAGCAAA-3′), CmPEROXR (5′-GCCTATCCAAAGACCTCGGCCTTCCC-3′), CmWIP1F (5′-TAGGGCTTCCAACTCCTTCCTCTT-3′), and CmWIP1R (5′CTTGCAATTGATGGGTGTGATCTTCTTG-3′). All samples were run in triplicate in a Roche Lightcycler 480. Cycling conditions consisted on an incubation step at 95 °C for 5 min, and 45 cycles at 95 ºC for 5 s and 60 ºC for 30 s to amplify CmPEROX and CmWIP1, and 40 cycles at the same conditions to amplify ToLCNDV DNA. The Ct (cycle threshold), which is the cycle number at which the fluorescence passes the threshold, was calculated for the viral target and the internal control genes in each sample. Relative accumulation of ToLCNDV was estimated by the comparative Ct method, using the formula 2−ΔΔCt, where ΔΔCt is the difference between the ΔCt of each sample (ΔCtsample: Ctreference genes – Ctviral target) and the ΔCt of the calibrator sample (ΔCtcalibrator). The resistant parental WM-7 at 15 dpi was used as the calibrator sample in all assays.
SNP genotyping and QTL analysis
Plants of the WM-7 and PS parental accessions, and the derived F 1, F 2, BC1PS, and BC1WM-7 populations, were genotyped with a set of 124 SNPs evenly distributed throughout the melon genome. This SNP panel was designed in previous studies and validated to perform QTL analysis in subsp. melo × subsp. agrestis melon crosses and to perform association studies (Leida et al. 2015; Perpiñá et al. 2016). The genomic position in the melon genome version 3.5.1 (García-Mas et al. 2012) of the polymorphic markers and their genetic position, estimated with the genotype of the F 2 population employing MAPMAKER 3.0 (Van Ooijen and Voorrips 2001) with the Kosambi map function, are indicated in Additional file 1, along with their flanking sequences. Total DNA was extracted from young leaves tissue before inoculation, using the protocol described in Esteras et al. (2012). DNA was quantified and adjusted to 15 ng μL−1. Genotyping was done using the iPLEX® Gold MassARRAY® Sequenom system at the Epigenetic and Genotyping unit of the University of Valencia (Unitat Central d´Investigació en Medicina (UCIM), Spain).
To identify markers linked to the resistance to ToLCNDV derived from WM-7, a QTL analysis was performed using symptom score at 15 and 30 dpi and ToLCNDV relative accumulation at 30 dpi of the F 2 population. We used the Kolmogorov–Smirnov test to check the normality assumption of traits distribution. Since the traits were not normally distributed, Kruskal–Wallis non-parametric test was used for quantitative trait loci (QTL) detection using the MapQTL version 4.1 software (Van Ooijen 2009), considering as significant associations those with p < 0.05.
In addition, a composite interval mapping approach (CIM, Zeng 1994) was applied. For the relative viral accumulation, the transformed distribution of ΔΔCt (ln (ΔΔCt) +2) instead of 2−ΔΔCt was used. It was implemented in Windows QTL Cartographer 2.5 (Wang et al. 2012), using a windows size of 15 cM and five cofactors. QTLs having LOD scores greater than a threshold value that had been determined by a permutation test were retained (1000 permutations were applied at the genome-wide level or each linkage group separately). Loci identified with both methods (Kruskal–Wallis and CIM) were considered true QTLs. For each QTL peak, we estimated the percentage of phenotypic variation explained (R 2), the additive and dominance effects, as well as the degree of dominance. A drop interval of 1.5 from the peak LOD was used to map the position of QTLs.
Additional mapping in chromosome 11
A new set of SNPs was designed to confirm QTL analysis and refine the position of the main locus associated with resistance to ToLCNDV located in chromosome 11. These new SNPs were obtained from a resequencing assay of the two parents, WM-7 and PS, which provided 12,927 polymorphisms between them. A set of 19 SNPs uniformly covering the chromosome 11 was implemented in a new Sequenom assay and used to genotype parents, F 1 and F 2 populations. These genotyping results were used to construct a new genetic map of chromosome 11. MAPMAKER 3.0 (Van Ooijen and Voorrips 2001) software and the Kosambi map function was employed to generate the new map. The genetic distances of the new map were used in a second QTL analysis following the same procedure described above. The genetic and genomic positions of these markers and their flanking sequences are shown in Additional file 2. Increasing the marker density in the candidate region is useful to narrow the size of the QTL confidence interval. However, CIM results can be affected by an uneven distribution of markers. We adjusted the CIM model to these new set of markers. The best results were obtained using a windows size of 10 cM and three cofactors. Those markers closely linked (<1 cM) that cause collinearity, and those having more than a 10% of missing data were removed from the analysis. In addition, 50 plants of the BC1PS and 50 of BC1WM-7 populations were genotyped to validate QTL analysis with both SNPs sets (the first background SNPs and the additional set of chromosome 11 SNPs).
Study of QTL interactions
Epistatic interactions between the main QTLs involved in ToLCNDV resistance were investigated by two-way ANOVA with the STATGRAPHIC Centurion XVI.I statistic software and using the genotypic data from the linked markers in the F 2 population. These markers were D16 for the QTL in chromosome 11, CMPSNP658 for the QTL in chromosome 2, and AI_35-A08 for the QTL in chromosome 12. Interactions were considered to be statistically significant when p ≤ 0.05.
Results
Resistance of the different melon sources and their F 1 progeny
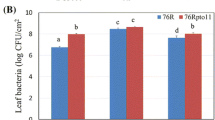
All the assayed accessions showed resistance to ToLCNDV, with no symptoms or only mild-to-moderate symptoms (most scores from 0 to 2), compared to the severe symptoms found in the susceptible PS cultivar (scores from 3 to 4), and with viral titers that were on average 15 times lower than those of the PS control (Fig. 1). However, differences in the response to ToLCNDV infection were observed between the wild agrestis and the momordica types. Plants of the Kharbuja accession developed mild symptoms at 30 dpi, and showed the highest ToLCNDV virus titer among the assayed resistant sources (Fig. 1). Most plants of PI-414723 remained symptomless, and although a few showed moderate symptoms at 15 dpi, all recovered and were symptomless at the end of the assay. The F 1 hybrids of these two momordica accessions with PS showed similar behavior. In general, symptoms in F 1 plants were slightly more severe than those of the corresponding resistant parents, but remained mild to moderate at the end of the assay. These momordica derived F 1 had viral titers significantly higher than those of the resistant parents, increasing from 15 to 30 dpi, but still around 4–6 times lower than PS.
Relative ToLCNDV accumulation (calculated as 2−ΔΔCt) at 15 and 30 days after mechanical inoculation (dpi) with ToLCNDV (light and dark bars, respectively), in four Cucumis melo subsp. agrestis resistant accessions (two momordica, Kharbuja and PI414723, and two wild types WM-7 and WM-9), the susceptible control C. melo subsp. melo Piñonet Piel de sapo (PS) and their respective hybrids. Mean and range of symptom score at 15/30 dpi are indicate in the x axis legend
The two wild agrestis WM-7 and WM-9 exhibited a higher level of resistance. Symptoms were milder (scores 0–1) than those of the momordica accessions, and viral titers were very low, both in the parents and F 1 progenies (Fig. 1). The fact that resistance to ToLCNDV remained in F 1 progenies suggests a dominant genetic control of this trait. The genetic control of virus resistance was studied in the F 2 and BC1 populations obtained from WM-7 × PS.
In the greenhouse assay, F 1 plants derived from WM-7 remained symptomless and with a low viral titer in the greenhouse (symptom score of 0 and 2−ΔΔCt value of 210 ± 35), whereas the PS controls and the commercial cantaloupes showed severe symptoms (including fruit cracking) and high viral accumulation (scores 1–3, and 3–4, and 2−ΔΔCt values 11,250 ± 532 and 14,325 ± 450, respectively) after the natural infection with viruliferous whiteflies.
Response of segregating populations
The F 2 and BC1PS populations derived from WM-7 × PS segregated for both symptoms severity and ToLCNDV accumulation. All plants of the BC1WM-7 generation remained symptomless at the end of the assay.
In the F 2 population, 108 plants remained symptomless or with mild symptoms at the end of the assay (scores 0 and 1), whereas 50 plants showed moderate to very severe symptoms at 30 dpi (scores 2–4), with leaf curling and yellow spotting typical of ToLCNDV infection. Plants with symptom scores of 0 and 1 (similar to those found in the resistant parent and in the corresponding F 1) were classified as resistant, and those with symptom scores between 2 and 4 as susceptible. The χ 2 test indicated that this segregation fitted to a 3:1 (resistant:susceptible) ratio expected for a single dominant gene for resistance (p = 0.07) (Table 1).
BC1PS also segregated for symptoms severity with 38 resistant (scores 0–1) and 32 susceptible (scores 2–4) plants at 30 dpi. This segregation also fitted to a 1:1 ratio expected for a single dominant gene (p = 0.4) (Table 1).
To further characterize the response to ToLCNDV, virus accumulation was estimated in segregating populations by qPCR (Fig. 2). Viral titer positively correlated with symptoms severity (r 2 = 0.9301 and 0.978, p = 0.0002 and 0.0215, in F 2 and BC1PS, respectively). In both populations F 2 and BC1PS, the resistant plants had viral amounts significantly lower than susceptible plants (mean 2−ΔΔCt values 263.7 versus 1037.8, and 1687.8 versus 5778.4 for F 2 and BC1PS, respectively), and a high variability in the viral titer was found in susceptible plants.
Mean viral titer of the BC1PS population was significantly higher than that of the F 2. This effect was observed in the resistant and susceptible categories (Fig. 2). Variability was also found in virus titer among plants of the BC1WM-7 population, with a mean 2−ΔΔCt value of 140.75, ranging from 0.002 to 4083.8. Taking together all these results, an effect of the genetic background (additional genes from the resistant parent) on the viral accumulation is suggested.
QTL analysis of F 2 population
To study the contribution of different genomic regions to ToLCNDV resistance, a QTL analysis was performed, using genotypic and phenotypic data of the F 2 population. Genotyping results of the F 2 population with the background set of SNPs and phenotypes for symptom score at 15 and 30 dpi and virus titer at 30 dpi were used for the analysis. Since the studied traits did not fit to a normal distribution, the non-parametric Kruskal–Wallis test (KW) was used to identify QTLs using untransformed data, and Composite Interval Mapping Method (CIM) was used with transformed data. Three genomic regions involved in the variation of these traits were detected with both methods (Table 2).
Three major overlapping QTLs were found in chromosome 11, one for each trait (ToLCNDVSy15_11, ToLCNDVSy30_11, and ToLCNDVVT30_11), located between CMPSNP30 and CMPSNP475 markers, with LOD peaks of 19.3, 50.7, and 12.9 (located at 60.5, 57.4, and 59.5 cM). These QTLs explained 67, 67, and 54% of the observed variation of symptoms at 15 dpi, 30 dpi, and viral accumulation, respectively (Table 2). We found additional overlapping QTLs for all traits in chromosome 12 (ToLCNDVSy15_12, ToLCNDVSy30_12, and ToLCNDVVT30_12) and for viral accumulation in chromosome 2 (ToLCNDVVT30_2). The QTLs in chromosome 12, with 5.3, 3.9, and 4.6 LOD scores were located close to the AI_35-A08 marker (LOD peaks at 14.9, 15.7, and 12.9 cM), and explained percentages of variation of 18, 10, and 10%, respectively, for each trait. The QTL in chromosome 2 (LOD peak 4.8 at 82.1 cM, closest marker CMPSNP658) was only significant for virus titer, and explained 13% of the observed variance for this trait.
Further analysis of the major QTL in chromosome 11
The resequencing data of the parental lines provided additional SNPs evenly distributed in the candidate region of chromosome 11 that were used to genotype the F 2 population. Genotyping results were employed to generate a new genetic map of this region covering 112.2 cM, with an average distance between consecutive markers of 4.7 cM (Fig. 3). The new QTL analysis confirmed the occurrence of major QTLs in chromosome 11 (ToLCNDVSy15_11, ToLCNDVSy30_11, and ToLCNDVVT30_11), with LOD scores of 3.2, 5.2, and 11.6 (LOD peaks located at 68.0, 69.0, and 68.4 cM) and 11, 21, and 32 of explained variance, respectively (Table 3; Fig. 3). Despite this second mapping experiment decreased the proportion of explained variance, it allowed the delimitation of the position interval of the ToLCNDVSy30_11 and ToLCNDVVT30_11 QTLs between the new D14 and D23 markers (located in chromosome 11 physical positions 29,550,418 and 30,192,781, respectively), being the D16 (29,690,406) the closest marker to the LOD peaks.
Genetic map of the chromosome 11 constructed with the genotyping data of the F 2 population (derived from the cross WM-7 × PS). The position of the major QTLs for the three traits (symptoms at 15 and 30 dpi and virus titer at 30 dpi) associated with ToLCNDV resistance are indicated. Map distances (cM) were obtained using Kosambi’s function and QTL location was obtained by Composite Interval Mapping (CIM) method
Epistatic interactions between QTLs involved in ToLCNDV resistance
To detect the putative effects of the regions of chromosomes 2 and 12 on the major QTL in chromosome 11, the digenic interactions between them were studied. Those that were statistically significant (p ≤ 0.05) are shown in Table 4.
QTL in chromosome 11 versus chromosome 12
Significant interactions were identified between ToLCNDVSy15_11 and ToLCNDVSy15_12 (p = 0.005) and between ToLCNDVSy30_11 and ToLCNDVSy30_12 (p = 0.0425) (Table 4; Fig. 4), which explained 10.8 and 7.12% of the variance of each trait, respectively. The effect of interaction was similar for both traits (Fig. 4). The occurrence of the WM-7 homozygous genotype at D16 resulted in a high level of resistance (no or mild symptoms), independently of the genotype at the chromosome 12 locus. However, the symptom severity of plants both heterozygous and PSPS at D16 was dependent on the genotype at locus AI_35-A08. The D16 heterozygous plants displayed no or mild symptoms when combined with WM-7WM-7 AI_35-A08 genotype, but mild to moderate when combined with PSPS or heterozygous genotype at chromosome 12. Only the D16 PSPS/AI_35-A08PSPS plants were highly susceptible (severe symptoms) as the presence of the WM-7 alleles at the AI_35-A08 locus (both in heterozygosity or homozygosity) in D16 PSPS plants resulted in intermediate symptoms.
Epistatic interactions identified in F 2 population between D16 marker (chromosome 11) and CMPSNP658 and AI_35-A08, of chromosome 2 and 12, respectively, for symptoms at 15 and 30 dpi and viral titer at 30 dpi (genotypic classes of D16 marker: PS plants homozygous for the PS allele, h heterozygous plants, WM-7 plants homozygous for the WM-7 allele)
QTL in chromosome 11 versus chromosome 2
A significant interaction was also identified between ToLCNDVVT30_11 and ToLCNDVVT30_2 (p = 0.0168) (Table 4), which explained 2.8% of the genetic variance of this trait. The homozygous plants for the WM-7 allele at marker D16 were all resistant with a very low viral titer, independently of the genotype at marker CMPSNP658 (Fig. 4). However, the response of the heterozygous genotype at D16 was dependent on the genotype at CMPSNP658. Viral titer was low, although higher than that of the resistant WM-7WM-7 D16 genotype when the genotype at the CMPSNP658 locus was homozygous for the WM-7 allele or heterozygous, but high viral titers were found when the CMPSNP658 genotype was homozygous for the PS allele.
Validation of the resistance in the BC1PS and BC1WM-7 populations
To confirm the effect of the chromosome 11 major QTL in BC1PS and BC1WM-7 populations, the means of symptom score at 15 and 30 dpi and viral accumulation (2−ΔΔCt) were calculated for each genotypic class of the D16 marker (Fig. 5), and compared with those of the F 2 generation. In addition, interactions with QTLs of chromosomes 2 and 12 were studied in backcrosses (Fig. 6).
Mean of symptom score at 15 and 30 dpi and viral titers in F 2, BC1PS, and BC1WM-7 populations, according to each genotypic class of D16 marker (chromosome 11). Genotypic classes of D16 marker are as in Fig. 4
Mean of symptom score and viral titers at 30 dpi in plants, of the BC1PS, F 2, and BC1WM-7 populations, heterozygous for the D16 marker (chromosome 11) and classified according their genotype in markers CMPSNP658 (chromosome 2, dotted bars) and AI_35-A08 (chromosome 12, solid bars). For these markers, dark bars represent PSPS genotype, grey bars heterozygous genotype, and white bars WM-7WM-7 genotype
The high level of resistance of the WM-7 homozygous genotype at D16 was confirmed at the BC1WM-7 population. All the WM-7WM-7 BC1WM-7 plants had no symptoms and viral titers as low as the WM-7WM-7 F 2 plants (Fig. 5). Intermediate levels of resistance were also found, as expected, in D16 heterozygous BC1WM-7 plants. The heterozygous BC1WM-7 plants showed less symptoms than F 2 plants but similar viral titers. A reduction of viral titer, similar to that found in the F 2, was observed in the BC1WM-7 population due to the presence of the WM-7WM-7 genotype in the two additional loci, more pronounced with locus CMPSNP658 (Fig. 6a, b).
The D16 heterozygous plants of the BC1PS generation showed symptoms and viral titers significantly higher than that of heterozygous F 2 plants (Fig. 5). The effect of the other two loci was also confirmed in BC1PS, where a significant drop of virus titer caused by heterozygous genotypes in chromosomes 2 and 12 was observed (Fig. 6a, b).
Discussion
In the present work, we studied the genetic control of resistance to ToLCNDV from the wild agrestis Indian melon accession WM-7 using mechanical inoculation. WM-7 was first selected as resistant after screening a wide collection of C. melo (López et al. 2015). In agreement with this previous work, the two agrestis accessions used here were found to be more resistant that the two momordica accessions. In the current paper, the resistant response of the F 1 derived from WM-7 to natural whitefly inoculation is also demonstrated. Indian germplasm has been previously used as source of resistances to viral and fungal pathogens for breeding melons (Dhillon et al. 2012; McCreight et al. 2017). However, this germplasm has not yet been exploited for breeding melons against ToLCNDV.
Mendelian analysis of symptom segregation in F 2 and BC1s populations derived from WM-7, as well as QTL results, suggested the presence of a major dominant gene in chromosome 11 controlling symptoms development and virus titer and an effect of additional genomic regions (in chromosomes 12 and 2) on disease progress. Those results are consistent with the existence of a major dominant gene reported to control the resistance to whitefly transmission of ToLCNDV in sponge gourd (Luffa cylindrica), another cucurbit crop popular in India (Islam et al. 2010), for which two linked SRAP markers have been reported (Islam et al. 2011). In our study, the use of mechanical inoculation could have increased the genetic complexity of the trait. Any of the minor genes could, for example, be implicated in resistance to mechanical transmission (i.e., in the restriction of the virus movement from cell to cell in the leaf parenchyma), which would be not important for the disease when inoculated in the phloem directly by the whitefly. A similar oligogenic control, three dominant genes, has been reported in Solanum habrochaites S. Knapp & D. M. Spooner, a wild species related to tomato, after ToLCNDV agroinoculation (Rai et al. 2013).
Our study also indicate the existence of epistatic interactions between these three regions, both for symptoms development and for viral accumulation. Similarly, Palomares-Rius et al. (2016) described two genomic regions involved in the resistance to Cucurbit yellow stunting disorder virus (CSYDV), one responsible for virus multiplication and the other controlling symptoms development, also derived from a subsp. agrestis melon type. The knowledge of these epistatic interactions between the three loci involved in the resistance to ToLCNDV will be necessary to develop new melon cultivars derived from WM-7. In this context, the linked SNPs selected in this paper will be highly useful in managing these loci in breeding programs.
Available background SNPs derived from previous resequencing experiments (Blanca et al. 2011, 2012; Esteras et al. 2013; Leida et al. 2015), and the additional SNPs generated in new resequencing assays, allowed an accurate analysis of the region of chromosome 11. Among the genes annotated in the candidate region of chromosome 11 (Additional file 3), we have not found nucleotide binding site-leucine-rich repeat gene sequences, present in the majority of plant R genes. These sequences were strongly associated with resistance to ToLCNDV in Luffa cylindrica Roem (Saha et al. 2013), and were also found to be induced in Capsicum annuum following ToLCNDV infection (Kushwaha et al. 2015). In addition, the leucine-rich repeat receptor-like kinase NIK confers high resistance to begomoviruses by trigging translational suppression on begomovirus infection (Brustolini et al. 2015; Zorzatto et al. 2015). In tomato, defense-related host gene expression in response to ToLCNDV includes one gene differentially expressed in a resistant cultivar that encodes a 26S proteasomal subunit RPT4a (SlRPT4) that interferes with the ToLCNDV genome transcription and activates hypersensitive response (Sahu et al. 2016). We did not find either sequences related to factors involved in recessive resistance to viruses, such as eukaryotic translation initiation factors (Yeam 2016), or messenger RNA surveillance factors, like Pelota (located in locus Ty-5), which confers resistance to geminiviruses in tomato (Lapidot et al. 2015). Further analysis of this region is necessary to check if it contains genes similar to those mentioned above that have not been annotated yet.
However, interestingly, the sequence analysis of this region revealed a predicted transcription factor similar to the TIFY4B from Arabidopsis thaliana (MELO3C022348, located at chromosome 11 position 30,052,212–30,054,984) (Chung and Sunter 2014) that has shown a potential role in host defense against geminiviruses. TIFY4B is a plant-specific DNA binding protein that interacts with begomoviruses for their transcription activity (Lacatus and Sunter 2009). The overexpression of this factor may delay symptom development by inhibiting viral replication, determining as a consequence the degree of symptom severity to geminiviruses depending on the host. For example, the expression of TIFY4B in Nicotiana benthamiana and tomato increased in response to geminivirus infection, reducing viral replication (Chung and Sunter 2014). In addition, a protein similar to the serine/threonine-protein kinase PBS1 of A. thaliana (MELO3C022340, located at chromosome 11 position 29,959,231–29,964,198) is annotated in this region. PBS1 is required for plant defense mechanism mediated by R proteins, and appeared to be highly upregulated in tomato cultivars that were resistant to ToLCNDV (Sahu et al. 2010). A similar protein OsPBL1 (PBS1-like) is involved in antiviral defense signaling pathways in rice (Lee and Kim 2015).
In the candidate region of chromosome 2, there is an RNA-dependent RNA polymerase 2 gene (MELO3C017106, located in chromosome 2 in position 25,048,095–25,052,883). This function has been described to contribute to basal virus resistance in many species (Hunter et al. 2016). For example, the widely used Solanum chilense-derived Ty-1 and Ty-3 alleles, which confer resistance to the geminivirus tomato yellow leaf curl virus (TYLCV) and Tomato mottle virus (ToMoV) (Zamir et al. 1994; Ji et al. 2007), also encode for an RNA-dependent RNA polymerase involved in the RNAi response to virus infections (Verlaan et al. 2013). RNAi has a key role in the defense of plants against begomoviruses (Zaidi et al. 2016). In tomato, both virus-specific siRNAs and miRNAs seem to be involved in the resistance against ToLCNDV (Naqvi et al. 2010; Sahu et al. 2010; Pradhan et al. 2015), and transgenic plants expressing RNAs for silencing were highly resistant (Mubin et al. 2007; Vu et al. 2013; Sharma et al. 2015). Most of these resistance sources are known to support virus replication. However, the level of virus accumulation is lower than the levels in susceptible cultivars. The candidate region of chromosome 12 contains a CCR4-associated factor 1 (CAF1) (MELO3C025580, located at chromosome 12 in position 13,943,018–13,944,220). This factor has been reported to be associated with plant defense responses (Sarowar et al. 2007; Chou et al. 2014).
Preliminary approximation to candidate genes involved in virus resistance and disease defense provides interesting information to identify the gene/s involved in resistance to ToLCNDV. Future work will focus on fine mapping the three QTLs responsible for the resistance and in expression studies and co-segregation assays of the candidate genes to facilitate the transfer of these QTLs into elite breeding lines.
Author contribution statement
CS, MF, CL, and BP conceived and designed the experiments. CS and CM performed the experiments. CS, CE, CM, and BP analyzed data. ND provided resistant accessions. CS and BP wrote the manuscript. MF helped in discussion. ND, MF, CL, and BP edited the manuscript. All authors read and approved the manuscript.
References
Bandaranayake WMEK, Wickramarachchi WART, Wickramasinghe HAM, Rajapakshe RGAS, Dissanayake DMKK (2014) Molecular detection and characterization of begomoviruses associated with cucurbitaceae vegetables in Sri Lanka. J Natl Sci Found Sri Lanka 42:265–271. doi:10.4038/jnsfsr.v42i3.7400
Blanca J, Cañizares J, Ziarsolo P, Esteras C, Mir G, Nuez F, García-Mas J, Picó B (2011) Melon transcriptome characterization: simple sequence repeats and single nucleotide polymorphisms discovery for high throughput genotyping across the species. Plant Genome 4:118–131. doi:10.1186/1471-2164-12-104
Blanca J, Esteras C, Ziarsolo P, Pérez D, Fernández V, Collado C, Rodríguez R, Ballester A, Roig C, Cañizares J, Picó B (2012) Transcriptome sequencing for SNP discovery across Cucumis melo. BMC Genom 13:280. doi:10.1186/1471-2164-13-280
Brustolini OJB, Machado JPB, Condori-Apfata JA, Coco D, Deguchi M, Loriato VAP, Pereira WA, Alfenas-Zerbini P, Zerbini FM, Inoue-Nagata AK, Santos AA, Chory J, Silva FF, Fontes EPB (2015) Sustained NIK-mediated antiviral signalling confers broad-spectrum tolerance to begomoviruses in cultivated plants. Plant Biotechnol J 13:1300–1311. doi:10.1111/pbi12349
Chang HH, Ku HM, Tsai WS, Chien RC, Jan FJ (2010) Identification and characterization of a mechanical transmissible begomovirus causing leaf curl on oriental melon. Eur J Plant Pathol 127:219–228. doi:10.1007/s10658-010-9586-0
Chou WL, Huang LF, Fang JC, Yeh CH, Hong CY, Wu SJ, Lu CA (2014) Divergence of the expression and subcellular localization of CCR4-associated factor 1 (CAF1) deadenylase proteins in Oryza sativa. Plant Mol Biol 85:443–458. doi:10.1007/s11103-014-0196-7
Chung HY, Sunter G (2014) Interaction between the transcription factor AtTIFY4B and begomovirus AL2 protein impacts pathogenicity. Plant Mol Biol 86:185–200. doi:10.1007/s11103-014-0222-9
Dhillon NPS, Monforte AJ, Pitrat M, Pandey S, Singh PK, Reitsma KR, Garcia-Mas J, Sharma A, McCreight JD (2012) Melon landraces of India: contributions and importance. In: Janick J (ed) Plant breeding reviews, vol 35. Wiley, New Jersey, pp 85–150. doi:10.1002/9781118100509
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
EPPO (2017) European and Mediterranean Plant Protection Organization. https://www.eppo.int/QUARANTINE/Alert_List/viruses/ToLCNDV.htm. Accessed 28 Feb 2017
Esteras C, Gómez P, Monforte AJ, Blanca J, Vicente-Dólera N, Roig C, Nuez F, Picó B (2012) High-throughput SNP genotyping in Cucurbita pepo for map construction and quantitative trait loci mapping. BMC Genom 22(13):80. doi:10.1186/1471-2164-13-80
Esteras C, Formisano G, Roig C, Díaz A, Blanca J, García-Mas J, Gómez-Guillamón ML, López-Sesé AI, Lázaro A, Monforte AJ, Picó B (2013) SNP genotyping in melons: genetic variation, population structure, and linkage disequilibrium. Theor Appl Genet 126:285–303. doi:10.1007/s00122-013-2053-5
Font San Ambrosio MI, Alfaro Fernández AO (2015) El virus de Nueva Delhi (Tomato leaf curl New Delhi virus, ToLCNDV) amplía su gama de hospedantes en los cultivos españoles. Phytoma 272:25–30
Fortes IM, Sánchez-Campos S, Fiallo-Olivé E, Díaz-Pendón JA, Navas-Castillo J, Moriones E (2016) A novel strain of Tomato leaf curl New Delhi virus has spread to the Mediterranean basin. Viruses 8:307. doi:10.3390/v8110307
García-Mas J, Benjak A, Sanseverino W, Bourgeois M, Mir G, González VM et al (2012) The genome of melon (Cucumis melo L). Proc Natl Acad Sci 109:11872–11877. doi:10.1073/pnas.1205415109
Hernández V (2016) El melón deja espacio a la sandía en Castilla La Mancha. MERCADOS. http://www.revistamercadoscom/noticia/melon-deja-espacio-la-sandia-castilla-la-mancha/. Accessed 24 Feb 2017
Hunter LJR, Brockington SF, Murphy AM, Pate AE, MacFarlane SA, Palukaitis P, Carr JP (2016) RNA-dependent RNA polymerase 1 in potato (Solanum tuberosum) and its relationship to other plant RNA-dependent RNA polymerases. Sci Rep 6:23082. doi:10.1038/srep23082
Islam S, Munshi AD, Mandal B, Behera TK, Kumar R (2010) Genetics of resistance in Luffa cylindrical Roemagainst tomato leaf curl New Delhi virus. Euphytica 174:83–89. doi:10.1007/s10681-010-0138-7
Islam S, Munshi AD, Verma M, Arya L, Mandal B, Behera TK, Kumar R, Lal SK (2011) Screening of Luffa cylindrical Roem for resistance against Tomato leaf curl New Delhi virus, inheritance of resistance, and identification of SRAP markers linked to the single dominant resistance gene. J Hortic Sci Biotechnol 86:661–667
Ji Y, Schuster DJ, Scott JW (2007) Ty-3, a begomovirus resistance locus near the Tomato yellow leaf curl virus resistance locus Ty-1 on chromosome 6 of tomato. Mol Breed 20:271–284. doi:10.1007/s11032-007-9089-7
Juárez M, Tovar R, Fiallo-Olivé E, Aranda MA, Gosálvez B, Castillo P, Moriones E, Navas-Castillo J (2014) First detection of tomato leaf curl New Delhi virus infecting Zucchini in Spain. Plant Dis 98:857–858. doi:10.1094/pdis-10-13-1050-PDN
Jyothsna P, Haq QMI, Singh P, Sumiya KV, Praveen S, Rawat R, Briddon RW, Malathi VG (2013) Infection of tomato leaf curl New Delhi virus (ToLCNDV), a bipartite begomovirus with betasatellites, results in enhanced level of helper virus components and antagonistic interaction between DNA B and betasatellites. Appl Microbiol Biotechnol 97:5457–5471. doi:10.1007/s00253-012-4685-9
Khan MS, Ji SH, Chun SC (2012) Begomoviruses and their emerging threats in South Korea: a review. Plant Pathol 28:123–136. doi:10.5423/PPJ.2012.28.2.123
Kushwaha N, Singh AK, Basu S, Chakraborty S (2015) Differential response of diverse solanaceous hosts to tomato leaf curl New Delhi virus infection indicates coordinated action of NBS-LRR and RNAi-mediated host defense. Arch Virol 160:1499–1509. doi:10.1007/s00705-015-2399-x
Lacatus G, Sunter G (2009) The Arabidopsis PEAPOD2 transcription factor interacts with geminivirus AL2 protein and the coat protein promoter. Virology 392:196–202. doi:10.1016/j.virol.2009.07.004
Lapidot M, Karniel U, Gelbart D, Fogel D, Evenor D, Kutsher Y, Makhbash Z, Nahon S, Shlomo H, Chen L, Reuveni M, Levin I (2015) A novel route controlling begomovirus resistance by the messenger RNA surveillance factor pelota. PLoS Genet 11:e1005538. doi:10.1371/journal
Lee KJ, Kim K (2015) The rice serine/threonine protein kinase OsPBL1 (ORYZA SATIVA ARABIDOPSIS PBS1-LIKE 1) is potentially involved in resistance to rice stripe disease. Plant Growth Regul 77(1):67–75. doi:10.1007/s10725-015-0036-z
Leida C, Moser C, Esteras C, Sulpice R, Lunn JE, De Langen F et al (2015) Variability of candidate genes, genetic structure and association with sugar accumulation and climacteric behavior in a broad germplasm collection of melon (Cucumis melo L). BMC Genet 16:28. doi:10.1186/s12863-015-0183-2
López C, Ferriol M, Picó MB (2015) Mechanical transmission of Tomato leaf curl New Delhi virus to cucurbit germplasm: selection of tolerance sources in Cucumis melo. Euphytica 204:279–691. doi:10.1007/s10681-015-1371-x
Mascarell-Creus A, Cañizares J, Vilarrasa-Blasi J, Mora-Garcia S, Blanca J, Gonzalez-Ibeas D, Saladie M, Roig C, Picó B, Lopez-Vigas N, Aranda MA, Garcia-Mas J, Nuez F, Puigdomenech P, Caño-Delgado AI (2009) An oligo-based microarray offers novel transcriptomic approaches for the analysis of pathogen resistance and fruit quality traits in melon (Cucumis melo L.). BMC Genom 10:467. doi:10.1186/1471-2164-10-467
McCreight JD, Wiutemantel WM, Natwick ET, Sinclair JW, Crosby KM, Gómez-Guillamón ML (2017) Recessive resistance to CYSDV in melon TGR 1551. Acta Hortic 1151:101–108. doi:10.17660/ActaHortic.2017.1151.17
Mnari-Hattab M, Zammouri S, Belkadhi MS, Bellon Doña D, ben Nahia MR, Hajlaoui E (2015) First report of Tomato leaf curl New Delhi virus infecting cucurbits in Tunisia. New Dis Rep 31:21. doi:10.5197/j2044-05882015031021
Mubin M, Mansoor S, Hussain M, Zafar Y (2007) Silencing of AV2 gene by antisense RNA protects transgenic plants against a bipartite begomovirus. Virol J 4:10. doi:10.1186/1743-422X-4-10
Naqvi AR, Haq QMR, Mukherjee SK (2010) MicroRNA profiling of Tomato leaf curl New Delhi virus (ToLCNDV) infected tomato leaves indicates that deregulation of mir159/319 and mir172 might be linked with leaf curl disease. Virol J 7:281. doi:10.1186/1743-422X-7-281
Palomares-Rius FJ, Garcés-Claver A, Gómez-Guillamón ML (2016) Detection of two QTLs associated with resistance to cucurbit yellow stunting disorder virus in Melon Line TGR 1551. In: Kozik EU, Paris HS (eds) XIth EUCARPIA Meeting on Genetics and Breeding of Cucurbitaceae, pp 334–337
Panno S, Iacono G, Davino M, Marchione S, Zappardo V, Bella P, Tomassoli L, Accotto GP, Davino S (2016) First report of Tomato leaf curl New Delhi virus affecting zucchini squash in an important horticultural area of southern Italy. New Dis Rep 33:6. doi:10.5197/j2044-05882016033006
Papidam M, Beachy RN, Fauquet CM (1995) Tomato leaf curl geminivirus from India has a bipartite genome and coat protein is not essential for infectivity. J Gen Virol 76:25–35. doi:10.1099/0022-1317-76-1-25
Perpiñá G, Esteras C, Gibon Y, Monforte AJ, Picó B (2016) A new genomic library of melon introgression lines in a cantaloupe genetic background for dissecting desirable agronomical traits. BMC Plant Biol 16:154. doi:10.1186/s12870-016-0842-0
Pradhan B, Naqvi AR, Saraf S, Mukherjee SK, Dey N (2015) Prediction and characterization of Tomato leaf curl New Delhi virus (ToLCNDV) responsive novel microRNAs in Solanum lycopersicum. Virus Res 195:183–195. doi:10.1016/j.virusres.2014.09.001
Pratap D, Kashikar AR, Mukherjee SK (2011) Molecular characterization and infectivity of a Tomato leaf curl New Delhi virus variant associated with newly emerging yellow mosaic disease of eggplant in India. Virol J 8:305. doi:10.1186/1743-422X-8-305
Rai NK, Sahu PP, Gupta S, Reddy MK, Ravishankar KV, Singh M, Sadashiva AT, Prasad M (2013) Identification and validation of an ISSR marker linked to Tomato leaf curl New Delhi virus resistant gene in a core set of tomato accessions. Veg Sci 40:1–6
Rosen R, Kanakala S, Kliot A, Pakkianathan BC, Farich BA, Santana-Magal N, Elimelech M, Kontsedalov S, Lebedev G, Cilia M, Ghanim M (2015) Persistent, circulative transmission of begomoviruses by whitefly vectors. Curr Opin Virol 15:1–8. doi:10.1016/j.coviro.2015.06.008
Roy A, Bal SS, Fergany M, Kaur S, Singh H, Malik AA, Singh J, Monforte AJ, Dhillon NPS (2012) Wild melon diversity in India (Punjab State). Genet Resour Crop Evol 59:755–767. doi:10.1007/s10722-011-9716-3
Ruiz L, Simon A, Velasco L, Janssen D (2016) Biological characterization of Tomato leaf curl New Delhi virus from Spain. Plant Pathol. doi:10.1111/ppa12587
Sáez C, Martínez C, Ferriol M, Manzano S, Velasco L, Jamilena M, López C, Picó B (2016) Resistance to Tomato leaf curl New Delhi virus in Cucurbita spp. Ann Appl Biol 169:91–105. doi:10.1111/aab12283
Saha D, Rana RS, Sureja AK, Verma M, Arya L, Munshi AD (2013) Cloning and characterization of NBS-LRR encoding resistance gene candidates from Tomato Leaf Curl New Delhi Virus resistant genotype of Luffa cylindrica Roem. Physiol Mol Plant Pathol 81:107–117. doi:10.1016/j.pmpp.2012.11.007
Sahu PP, Rai NK, Chakraborty S, Singh M, Chandrappa PH, Ramesh B, Chattopadhyay D, Prasad M (2010) Tomato cultivar tolerant to Tomato leaf curl New Delhi virus infection induces virus-specific short interfering RNA accumulation and defence-associated host gene expression. Mol Plant Pathol 11:531–544. doi:10.1111/j.1364-3703.2010.00630.x
Sahu PP, Sharma N, Puranik S, Chakraborty S, Prasad M (2016) Tomato 26S Proteasome subunit RPT4a regulates ToLCNDV transcription and activates hypersensitive response in tomato. Sci Rep 6:27078. doi:10.1038/srep27078
Sarowar S, Oh HW, Cho HS, Baek KH, Seong ES et al (2007) Capsicum annuum CCR4-associated factor CaCAF1 is necessary for plant development and defence response. Plant J 51:792–802. doi:10.1111/j.1365-313X.2007.03174.x
Sharma V, Basu S, Chakraborty S (2015) RNAi mediated broad-spectrumtransgenic resistance in Nicotiana benthamiana to chilli-infecting begomoviruses. Plant Cell Rep 34:1389–1399. doi:10.1007/s00299-015-1795-8
Srivastava KM, Hallan V, Raizada RK, Chandra G, Singh BP, Sane PV (1995) Molecular cloning of Indian tomato leaf curl virus genome following a simple method of concentrating the supercoiled replicative form of viral DNA. J Virol Methods 51:297–304
Van Ooijen JW (2009) MapQTL® 6 Software for the mapping of quantitative trait loci in experimental population of diploid species Kayzma BV. Wageningen, The Netherlands
Van Ooijen JW, Voorrips RE (2001) JoinMap Version 30, Software for the calculation of genetic linkage maps Release 30 Plant Research International Wageningen, The Netherlands
Verlaan MG, Hutton SF, Ibrahem RM, Kormelink R, Visser RGF, Scott JW, Edwards JD, Bai Y (2013) The tomato yellow leaf curl virus resistance genes Ty-1 and Ty-3 are allelic and code for DFDGD-class RNA–dependent RNA polymerases. PLOS Genetics 9:e1003399. doi:10.1371/journal.pgen.1003399
Vu TV, Roy Choudhury N, Mukherjee SK (2013) Transgenic tomato plantsexpressing artificial microRNAs for silencing the pre-coat and coat proteins of abegomovirus, Tomato leaf curl New Delhi virus, show tolerance to virus infection. Virus Res 172:35–45. doi:10.1016/j.virusres.2012.12.008
Wang S, Basten CJ, Zeng Z-B (2012) Windows QTL cartographer 25 department of statistics, North Carolina State University, Raleigh, NC. http://statgen.ncsu.edu/qtlcart/WQTLCart.htm. Accessed 20 Feb 2017
Yeam I (2016) Current advances and prospectus of viral resistance in horticultural crops. Hortic Environ Biotechnol 57:113–122. doi:10.1007/s13580-016-0105-x
Zaidi SSEA, Martin DP, Amin I, Farooq M, Mansoor S (2016) Tomato leaf curl New Delhi virus: a widespread bipartite begomovirus in the territory of monopartite begomoviruses. Mol Plant Pathol. doi:10.1111/mpp12481
Zamir D, Ekstein-Michelson I, Zakay Y et al (1994) Mapping and introgression of a tomato yellow leaf curl virus tolerance gene, TY-1. Theor Appl Genet 88:141–146. doi:10.1007/BF00225889
Zeng ZB (1994) Precision mapping of quantitative trait loci. Genetics 136:1457–1468
Zorzatto C, Machado JP, Lopes KV, Nascimento KJ, Pereira WA, Brustolini OJ, Reis PA, Calil IP, Deguchi M, Sachetto-Martins G, Gouveia BC, Loriato VA, Silva MA, Silva FF, Santos AA, Chory J, Fontes EP (2015) NIK1-mediated translation suppression functions as a plant antiviral immunity mechanism. Nature 520:679–682. doi:10.1038/nature14171
Acknowledgements
CS was the recipient of a predoctoral fellowship (ACIF/2016/188) from Generalitat Valenciana, and CM was the recipient of a Juan de la Cierva contract from the Spanish Ministerio de Economía y Competitividad (FJCI-2014-19817). This work was supported by Project E_RTA2013-00020-C04-03 from the Spanish Instituto Nacional de Investigaciones Agrarias (INIA) cofunded with FEDER funds. We also thank Maureen Mecozzi for helpful edits and the USDA genebank for providing seeds of some of the accessions used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Kathryn K. Kamo.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sáez, C., Esteras, C., Martínez, C. et al. Resistance to tomato leaf curl New Delhi virus in melon is controlled by a major QTL located in chromosome 11. Plant Cell Rep 36, 1571–1584 (2017). https://doi.org/10.1007/s00299-017-2175-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-017-2175-3