Abstract
Many studies have estimated the correlation between HLA-B27 polymorphisms and ankylosing spondylitis (AS). However, the results were controversial. Therefore, we performed this meta-analysis to determine the association of HLA-B*27 polymorphisms with AS and investigate the impacts of HLA-B27 on the clinical symptoms of AS patients. A comprehensive search was performed in PubMed, Web of Science and Embase databases to retrieve the eligible studies, which addressed the association between HLA-B27 polymorphisms and AS susceptibility. The correlation in fixed-effect model was estimated using the relative risk (RR) and 95% confidence intervals (CI). Finally, 41 studies were included in this meta-analysis, among which 35 studies were used to analyze the correlation between HLA-B27 and AS. And 11 studies were applied to estimate the effects of HLA-B27 on the clinical characteristics of AS patients. Besides, our meta-analysis was composed of 8993 AS patients and 19,254 healthy controls. The results suggested that HLA-B27, HLA-B27*02 and HLA-B27*04 were positively in relation to AS (RRHLA-B27 (95% CI) 16.02 (13.85, 18.54), P < 0.001; RRHLA-B*2702 (95% CI) 1.28 (1.08, 1.53), P = 0.005; RRHLA-B27*04 (95% CI) 1.14 (1.01, 1.29), P = 0.041). Moreover, positive association was observed between HLA-B27 and sex (male) [RR (95% CI) 1.10 (1.05, 1.15), P < 0.001], family history [RR (95% CI) 1.10 (1.06, 1.140), P < 0.001], uveitis [RR (95% CI) 1.07 (1.03, 1.11), P < 0001], peripheral joint involvement [RR (95% CI) 1.04 (1.01,1.07), P = 0.013] and hip joints involvement [RR (95% CI) 1.06 (1.02, 1.10), P = 0.003]. In addition, we also found that HLA-B27*04 showed association with peripheral joint involvement [RR (95% CI) 1.13 (1.05–1.23), P = 0.002]. In conclusion, the current meta-analysis indicates that HLA-B27, especially, its subtypes (HLA-B27*02 and HLA-B27*04) may be potential risk factors for AS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ankylosing spondylitis (AS), a chronic and immune-mediated disorder, is characterized by new bone formation and inflammation in the axial skeleton, which can induce irretrievable structural damage, deterioration of function, disability, spinal and pelvic joint dysfunction [1,2,3,4]. Whereas effective treatments are mainly used to control symptom, it has not verified that any treatment could decrease the occurrence of AS. Meanwhile, the therapy of AS mainly depends on the use of drugs including nonsteroidal antiinflammatory drugs (NSAIDs), conventional disease-modifying antirheumatic drugs (DMARDs), which are often insufficient to suppress inflammation and improve symptoms [5]. In addition, although anti-tumor necrosis factor (TNF) therapy is widely recognized as the effective method for patients with disease activity, its application often causes unacceptable side effects and fails to control disease [6,7,8,9,10].
Accumulating evidence has demonstrated that human leukocyte antigen (HLA) class I molecule B27 (HLA-B27) is strongly related to the progression of AS via restricting immune responses and inducing autophagy responses [11]. And over 100 different allelic variants of B27 have been recognized (HLA-B2701 to HLA-B27106) in AS patients, among which B27*05 and B27*02 are widely prevalent in European populations, and B27*04 is predominate in Asian populations [11]. The articles reported by Kim et al. [12] and Al-Qadi et al. [13] found that the incidence of HLA-B27 was higher in AS cases versus controls. Akassou et al. also [14] concluded the RR and 95% CI to show that high frequency of HLA-B27 was related to high risk of AS (RR: 16.8, 95% CI 5.83–51.03; P < 0.001). In contrast, the study reported by Diyarbakir et al. showed no difference of the frequency of HLA-B27 and its subtypes (HLA-B2702, HLA-B2704 and HLA-B2705) between AS patients and the controls [15]. In addition, Qi et al. [16] discovered the different expression of HLA-B27 alleles in AS patients and healthy control patients, and found that HLA-B2704 expression was equal in both groups and the frequency of HLA-B2705 was higher in AS patients than control patients. However, another study reported that HLA-B2704 and HLA-B2705 were both more likely to appear in AS patients compared with healthy controls [17]. Additionally, Al-Qadi et al. also discovered that the presence of HLA-B27 are prone to affect the function of combined axial and peripheral joint involvement of the AS patients and cause inflammatory back pain and buttock pain [13]. Qi et al. [16] also found that compared to HLA-2705-positive patients, HLA-2704-positive patients were more likely to get the risk of AS at a younger age, and HLA-B2705-positive patients had a higher risk of dactylitis and uveitis compared with HLA-B2704-positive patients. To the opposite, another study reported that AS patients with uveitis were observed more in HLA-B2704+ patients than HLA-B2705+ patients [18]. Thus, the association between HLA-B27 and its subtypes with AS and clinical features of AS patients are controversial.
Meta-analysis has the potential roles to detect an association with overcoming the problems of small sample studies and inadequate statistics in complex traits. In the current study, a meta-analysis was performed to investigate the risk of AS with HLA-B27/HLA-B27 subtypes and the association between HLA-B27/HAL-B27 subtypes and clinical features in AS patients.
Materials and methods
Identification of eligible studies
PubMed, Web of science and Embase databases were applied to retrieve cross-sectional and cohorts studies linking HLA-B27 polymorphisms to AS with updating to November, 2016. The query for searching was as follows: “HLA-B27” or “HLA-B*27” or “human leukocyte antigen B27” (Mesh) and “ankylosing spondylitis” or “AS” (Mesh). Review articles and references from included articles were manually searched to check for other relevant researches. Two independent reviewers (HL and YZG) first read the title and abstract of the candidate papers. Full text would be retrieved when articles could not be decided from title and abstract. Any disparity in quality assessment and data collection was reached a final agreement via discussion.
Studies were included for the meta-analysis with meeting the following inclusion criteria: (1) the diagnosis of AS was based on the modified New York criteria; (2) studies evaluated the frequency of HLA-B27 in cases of AS patients and control groups; (3) relative risk (RR) and 95% confidence interval (95% CI) were available; (4) articles written in English; (5) cross-sectional or cohorts studies.
Data extraction
The available data were extracted by two independent reviewers (Hai Lin and Yi-Zhen Gong), and the following information was included: first author, country, publication time, sample size, the frequency of HLA-B27(+)in both cases and controls, the clinical features of AS patients. Inconsistencies in the data extraction were resolved through debates and consultations.
Statistical analysis
RR and 95% CI were performed to estimate the impact of HLA-B27 on risk of AS in the current meta-analysis. By convention, it implies a high risk for the group with more frequency of HLA-B27 when RR > 1. The influence of HLA-B27 expression on the risk of AS was regarded as statistically significant when the 95% CI for the HR did not overlap 1 and P < 0.05. Stata version 11.0 was applied to perform the [15] data analysis, while Q tests and I squared test were conducted to evaluate the heterogeneity. A fixed effects model was used when no heterogeneity was present and the results were similar with the random effects model. It was considered that there existed no statistically significant heterogeneity when P ≥ 0.05 or I 2 ≤ 50%. Subgroup analysis was performed on the basis of region. In addition, the sensitivity analysis was conducted to find the sources of heterogeneity. The Begg’s funnel plot and Egger’s test were used to evaluate the publication bias when more than ten articles were included in the meta-analysis.
Results
Characteristics of studies
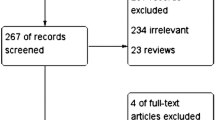
The flow chart for the search strategy was summarized in Fig. 1. We first reviewed 1760 articles and excluded 1691 articles after reviewing the title and abstract. Finally, we included 41 articles after excluding 1 article without full text, 2 meta-analyses, 1 review and 24 articles without available data. The main features of the eligible studies were presented in Tables 1 and 2. Finally, 41 articles were included in our current study, including 35 studies (described the relationship of HLA-B27 and AS) and 11 studies (identified the association between HLA-B27 frequency and clinical features of AS patients). The total numbers of AS patients and healthy controls were, respectively, 8993 and 19,254. Among the 35 studies mentioned above, 17 studies reported the expression of HLA-B27 in case–control groups (cases: 3983; controls: 17,856) and the influence of most common HLA-B27 subtypes (HLA-B27*02, HLA-B27*04, HLA-B27*05) were, respectively, demonstrated in 19 articles (cases:2958; controls:14,258), 19 articles (cases:4656; controls:7569) and 23 articles (cases:5162; controls:8358). In the 11 studies related to the clinical characteristics of AS, HLA-B27-positive patients accounted for 80% (3711 out of 4384). The clinical features included in the meta-analysis mainly as follows: sex (male), family history, uveitis, peripheral joint involvement, hip joints involvement, low back pain (LBP) or buttock pain, dactylitis, enthesitis.
Analysis for the association between HLA-B27 expression and AS
The results of this meta-analysis showed there existed positive association between HLA-B27, HLA-B27*02, HLA-B27*04, HLA-B27*05 and susceptibility to AS [RRHLA-B27 (95% CI) 8.24 (7.75–8.76), P < 0.001; RRHLA-B*2702 (95% CI) 1.49 (1.31, 1.70), P < 0.001; RRHLA-B27*04 (95% CI) 58 (4.25, 4.94), P < 0.001; RRHLA-B*2705 (95% CI) 1.53 (1.46, 1.61), P < 0.001]. However, there was heterogeneity in our current study (PHLA-B27 < 0.001, I 2 = 99.4%; PHLA-B27*02 < 0.001, I 2 = 80%; PHLA-B27*04 < 0.001, I 2 = 99.5%; PHLA-27*05 < 0.001, I 2 = 96.1%). The subgroup analysis was performed according to the distribute region of the patients. As a result, the heterogeneity was mainly induced by the samples from East Asia (PHLA-B27 = 0.004, I 2 = 70.7%; PHLA-B27*02 < 0.001, I 2 = 80%; PHLA-B27*04 < 0.001, I 2 = 99.8%; PHLA-27*05 < 0.001, I 2 = 94.9%), Southwest Asia (PHLA-B27 = 0.030, I 2 = 62.7%; PHLA-B27*02 < 0.001, I 2 = 91%; PHLA-27*05 < 0.001, I 2 = 97.9%), South Europe (PHLA-B27*02 = 0.003, I 2 = 82.8%; PHLA-27*05 < 0.001, I 2 = 97.5%) and Africa (PHLA-B27*02 < 0.001, I 2 = 94.5%; PHLA-27*05 < 0.001, I 2 = 92.3%). After removal of the studies contributed to the heterogeneity analyzed by the sensitivity analysis, the prevalence of HLA-B27, HLA-B27*02 and HLA-B27*04 were still positively related to the susceptibility of patients to AS (RRHLA-B27 (95% CI) 16.02 (13.85, 18.54), P < 0.001; RRHLA-B*2702 (95% CI) 1.28 (1.08, 1.53), P = 0.005; RRHLA-B27*04 (95% CI) 1.14 (1.01, 1.29), P = 0.041) (Fig. 2). While no association was observed between HLA-B27*05 and susceptibility to AS (RRHLA-B*2705 (95% CI) 1.04 (0.97, 1.12), P = 0.256).
Analysis for the association between HLA-B27 expression and the clinical parameters of AS patients
The results showed that there was a positive relation between HLA-B27 and family history [RR (95% CI) 1.10 (1.07, 1.14), P < 0.001), uveitis [RR (95% CI) 1.05 (1.02, 1.09), P = 0.006), peripheral joint involvement [RR (95% CI) 1.04 (1.01, 1.07), P = 0.013] and hip joints involvement [RR (95% CI) 1.04 (1.01, 1.08), P = 0.011]. Moreover, HLA-B27 could increase the susceptibility of male populations to AS [RR (95% CI) 1.06 (1.01, 1.11) P = 0.013]. However, we observed heterogeneity in the present meta-analysis: sex (I 2 = 87.70%, P < 0.001), family history (I 2 = 73.5%, P = 0.005), uveitis (I 2 = 59.9%, P = 0.008), and hip joints involvement (I 2 = 88.45%, P < 0.001). There was no statistically significant heterogeneity after removal of the studies induced the heterogeneity based on the sensitivity analysis, and the results were in favor of the findings mentioned above: sex (male) [RR (95% CI) 1.10 (1.05, 1.15), P < 0.001]; family history [RR (95% CI) 1.10 (1.06, 1.140), P < 0.001], uveitis [RR (95% CI) 1.07 (1.03, 1.11), P < 0.001], and hip joints involvement [RR (95% CI) 1.06 (1.02, 1.10), P = 0.003]. In addition, we also found HLA-B27*04 was remarkably in relation to peripheral joint involvement in AS patients without heterogeneity [RR (95% CI) 1.13 (1.05–1.23), P = 0.002; I 2 = 0.0%, Pheterogeneity = 0.972]. And association was observed between HLA-B27*05 and uveitis [RR (95% CI) 1.31 (1.07–1.61), P = 0.009; I 2 = 93.1%, P < 0.001]. It also showed that HLA-B27*05 might decrease the risk of male to AS [RR (95% CI) 0.48 (0.344–0.655), P < 0.001; I 2 = 95.9%, P < 0.001]. As was shown, there existed heterogeneity in the analysis of HLA-B27*05 and sex/uveitis in AS patients. Interestingly, no association was found between HLA-B27*05 and sex/uveitis after eliminating the heterogeneity [sex: RR (95% CI) 1.37 (0.81–2.32), P = 0.241; uveitis: RR (95% CI) 0.90 (0.61–1.31), P = 0.578]. The statistically significant results were shown in Fig. 3.
Publication bias
Publication bias was evaluated by Begg’s funnel plot and Egger’s test. The combined results of Begg’s funnel plots and P values of the Egger’s test suggested no obvious publication bias (Tables 3 and 4 and Fig. 4).
Begg’s funnel plots for publication bias test on studies assessing the association between HLA-B27/HAL-B27*02/HLA-27*04 and AS/clinical features. The relationship of HLA-B27/B27*02/B27*04 with AS: a HLA-B27 (I 2 = 26.7, P = 0.198); b HLA-B27*02; c HLA-B27*04. Positive association between HLA-B27 and clinical features of AS patients: d sex (male); e uveitis; f peripheral joint involvement
Discussion
Summary of the results
Previously, two meta-analyses reported by Yang et al. [49] and Zhang et al. [50] have demonstrated the association between HLA-B27 genetic polymorphisms and susceptibility to AS. In comparison with the previous meta-analysis, the present meta-analysis was the first one to identify the effects of HLA-B27 and its subtypes (HLA-B27*04 and HLA-B27*05) on the clinical features of AS patients including 11 articles (4384 AS patients). Furthermore, 35 studies have been included in our meta-analysis with 3983 AS patients and 17,856 controls, which was more than those of the article reported by Yang et al. [49] (31 studies involved with 3140 AS patients and 1735 controls) and the report of Zhang et al. [50] (14 studies consisted of 1900 AS patients and 831 healthy controls). In consideration of these, our current study gains advantages over the studies published by Yang et al. and Zhang et al. Therefore, our meta-analysis could provide more powerful evidence to identify the association between HLA-B27 and AS susceptibility. Notably, it could also offer some indications on the influence of HLA-B27 on the clinical characteristics in AS patients.
In our current study, we found HLA-B27 was positively associated with AS susceptibility [RR (95% CI) 16.02 (13.85, 18.54), P < 0.001] without identifying the subtypes of HLA-B27, which has not been reported in other articles. Moreover, no matter there was heterogeneity or not, the prevalence of HLA-B27*04 could increase the risk of AS, which was inconsistent with the results concluded by Yang et al. [49] and Zhang et al. [50]. The same to HLA-B27*04, we also found positive association between HLA-B27*02 and susceptibility to AS. In contrast, Yang et al. [49] and Zhang et al. [50] have suggested that HLA-B27*02 showed no relation to AS. In addition, it was observed that HLA-B27*05 was positively in relation to AS with the existence of heterogeneity, and no association was found when the heterogeneity was not statistically significant, which was in line with the study reported by Yang et al. [49]. To the opposite, Zhang et al. have found there was negative association between HLA-B27*05 and AS susceptibility [50].
Importantly, we are the first to explore the relationship of HLA-B27 and the clinical features in AS patients. The results suggested that male populations and patients with AS family history were more susceptible to AS, which were in consistent with previous studies [13, 14, 27]. While another study reported by Wu et al. showed no association between family history and the susceptibility to AS [47]. In addition, we also found that HLA-B27 positive AS patients were more likely accompanied with uveitis, peripheral joint involvement and hip joints involvement. Three previous studies showed the same results that the incidence of uveitis was positively related to HLA-B27 expression in AS patients [16, 45, 51]. In contrast, another three studies demonstrated that HLA-B27 had no relationship with the risk of AS [13, 30, 46]. The researches studied by Al-Qadi et al. and Kim et al. identified that HLA-B27+ patients are more likely to suffer the functional disability of peripheral join and hip joints, respectively [13, 51]. However, Chavan et al. testified that no significant difference was observed between the distribution of HLA-B27 and peripheral join involvement and hip joints involvement [30]. In addition, we also investigated that AS patients with HLA-B27*04 tended to peripheral joint involvement, which was in contrast with previous studies [16, 47]. Although we found HLA-B27*05 was associated with sex and uveitis in AS patients, there existed heterogeneity. Therefore, further investigation was needed to be conducted.
The limitation
We attempted to make a comprehensive analysis via extensive strategy. However, as we did not involve the articles not published in English, some relevant articles might be missed. Besides, the samples were collected from different region in the worldwide and the sample size differed in different studies, which contribute to the heterogeneity. As we excluded the literatures in related to other subtypes of HLA-27 studied in less than three articles, which might make it unavailable to understand the effects of other HLA-B27 subtypes on AS. In addition, the studies demonstrated the clinical features of AS patients were limited, so it was needed to make further investigation for exploring the association between HLA-B27 and clinical parameters in AS patients.
Conclusion
In conclusion, our current meta-analysis not only demonstrated that HLA-B27, HLA-B27*02 and HLA-B27*04 showed association with AS, but also indicated the prevalence of HLA-B27 might influence the clinical symptoms of AS patients. While considering the existence of limitation in our meta-analysis, further studies should be performed to explore the relationship of HLA-B27 with AS and the symptoms in AS patients.
References
Braun J, Sieper J (2007) Ankylosing spondylitis. Lancet 369:1379–1390
Dougados M, Baeten D (2011) Spondyloarthritis. Lancet 377:2127–2137
Gao X, Wendling D, Botteman MF, Carter JA, Rao S et al (2012) Clinical and economic burden of extra-articular manifestations in ankylosing spondylitis patients treated with anti-tumor necrosis factor agents. J Med Econ 15:1054–1063
Jang JH, Ward MM, Rucker AN, Reveille JD, Davis JC Jr et al (2011) Ankylosing spondylitis: patterns of radiographic involvement—a re-examination of accepted principles in a cohort of 769 patients. Radiology 258:192–198
Braun J, van den Berg R, Baraliakos X, Boehm H, Burgos-Vargas R et al (2011) 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann Rheum Dis 70:896–904
Jeong H, Eun YH, Kim IY, Kim H, Ahn JK et al (2016) Drug survival of tumor necrosis factor alpha inhibitors in patients with ankylosing spondylitis in Korea. Korean J Intern Med. doi:10.3904/kjim.2016.042
Liu J, Dong Z, Zhu Q, He D, Ma Y et al (2016) TNF-alpha promoter polymorphisms predict the response to etanercept more powerfully than that to infliximab/adalimumab in spondyloarthritis. Sci Rep 6:32202
Cavazzana I, Taraborelli M, Fredi M, Tincani A, Franceschini F (2014) Aseptic meningitis occurring during anti-TNF-alpha therapy in rheumatoid arthritis and ankylosing spondylitis. Clin Exp Rheumatol 32:732–734
Zavada J, Uher M, Sisol K, Forejtova S, Jarosova K et al (2016) A tailored approach to reduce dose of anti-TNF drugs may be equally effective, but substantially less costly than standard dosing in patients with ankylosing spondylitis over 1 year: a propensity score-matched cohort study. Ann Rheum Dis 75:96–102
Nossent JC, Sagen-Johnsen S, Bakland G (2014) Tumor necrosis factor-alpha promoter-308/238 polymorphism association with less severe disease in ankylosing spondylitis is unrelated to serum TNF-alpha and does not predict TNF inhibitor response. J Rheumatol 41:1675–1682
Bowness P (2015) Hla-B27. Annu Rev Immunol 33:29–48
Kim K, Bang SY, Lee S, Lee HS, Shim SC et al (2015) An HLA-C amino acid variant in addition to HLA-B*27 confers risk for ankylosing spondylitis in the Korean population. Arthritis Res Ther 17:342
Al-Qadi R, Salih SF, AlDoski HJ, Nabeel M, Ali JH et al (2015) Association of HLA-B*27 with ankylosing spondylitis in Kurdish patients. Int J Rheum Dis. doi:10.1111/1756-185X.12605
Akassou A, Yacoubi H, Jamil A, Dakka N, Amzazi S et al (2015) Prevalence of HLA-B27 in Moroccan healthy subjects and patients with ankylosing spondylitis and mapping construction of several factors influencing AS diagnosis by using multiple correspondence analysis. Rheumatol Int 35:1889–1894
Diyarbakir E, Eyerci N, Melikoglu M, Topcu A, Pirim I (2012) HLA B27 subtype distribution among patients with ankylosing spondylitis in eastern Turkey. Genet Test Mol Biomark 16:456–458
Qi J, Li Q, Lin Z, Liao Z, Wei Q et al (2013) Higher risk of uveitis and dactylitis and older age of onset among ankylosing spondylitis patients with HLA-B*2705 than patients with HLA-B*2704 in the Chinese population. Tissue Antigens 82:380–386
Yi L, Wang J, Guo X, Espitia MG, Chen E et al (2013) Profiling of hla-B alleles for association studies with ankylosing spondylitis in the chinese population. Open Rheumatol J 7:51–54
Li H, Li Q, Ji C, Gu J (2016) Ankylosing spondylitis patients with HLA-B*2704 have More uveitis than patients with HLA-B*2705 in a North Chinese population. Ocul Immunol Inflamm 1–5: doi:10.1080/09273948.2016.1188967
Diaz-Pena R, Vidal-Castineira JR, Lopez-Vazquez A, Lopez-Larrea C (2016) HLA-B*40:01 is associated with ankylosing spondylitis in HLA-B27-positive populations. J Rheumatol 43:1255–1256
Wei JC, Sung-Ching HW, Hsu YW, Wen YF, Wang WC et al (2015) Interaction between HLA-B60 and HLA-B27 as a better predictor of ankylosing spondylitis in a Taiwanese population. PLoS ONE 10:e0137189
Videm V, Cortes A, Thomas R, Brown MA (2014) Current smoking is associated with incident ankylosing spondylitis—the HUNT population-based Norwegian health study. J Rheumatol 41:2041–2048
van Gaalen FA, Verduijn W, Roelen DL, Bohringer S, Huizinga TW et al (2013) Epistasis between two HLA antigens defines a subset of individuals at a very high risk for ankylosing spondylitis. Ann Rheum Dis 72:974–978
Acar M, Cora T, Tunc R, Acar H (2012) HLA-B27 subtypes in Turkish patients with ankylosing spondylitis and healthy controls. Rheumatol Int 32:3103–3105
al Attia HM, Sherif AM, Hossain MM, Ahmed YH (1998) The demographic and clinical spectrum of Arab versus Asian patients with ankylosing spondylitis in the UAE. Rheumatol Int 17:193–196
Liu Y, Jiang L, Cai Q, Danoy P, Barnardo MC et al (2010) Predominant association of HLA-B*2704 with ankylosing spondylitis in Chinese Han patients. Tissue Antigens 75:61–64
Mou Y, Wu Z, Gu J, Liao Z, Lin Z et al (2010) HLA-B27 polymorphism in patients with juvenile and adult-onset ankylosing spondylitis in Southern China. Tissue Antigens 75:56–60
Harfouch EI, Al-Cheikh SA (2011) HLA-B27 and its subtypes in Syrian patients with ankylosing spondylitis. Saudi Med J 32:364–368
Cipriani A, Rivera S, Hassanhi M, Marquez G, Hernandez R et al (2003) HLA-B27 subtypes determination in patients with ankylosing spondylitis from Zulia, Venezuela. Hum Immunol 64:745–749
Kchir MM, Hamdi W, Laadhar L, Kochbati S, Kaffel D et al (2010) HLA-B, DR and DQ antigens polymorphism in Tunisian patients with ankylosing spondylitis (a case-control study). Rheumatol Int 30:933–939
Chavan H, Samant R, Deshpande A, Mankeshwar R (2011) Correlation of HLA B27 subtypes with clinical features of ankylosing spondylitis. Int J Rheum Dis 14:369–374
Pazar B, Safrany E, Gergely P, Szanto S, Szekanecz Z et al (2010) Association of ARTS1 gene polymorphisms with ankylosing spondylitis in the Hungarian population: the rs27044 variant is associated with HLA-B*2705 subtype in Hungarian patients with ankylosing spondylitis. J Rheumatol 37:379–384
Ben Radhia K, Ayed-Jendoubi S, Sfar I, Ben Romdhane T, Makhlouf M et al (2008) Distribution of HLA-B*27 subtypes in Tunisians and their association with ankylosing spondylitis. Joint Bone Spine 75:172–175
Liu X, Hu LH, Li YR, Chen FH, Ning Y et al (2010) The association of HLA-B*27 subtypes with ankylosing spondylitis in Wuhan population of China. Rheumatol Int 30:587–590
Alaez C, Gazit E, Ibarrola B, Yaron M, Livneh A et al (2007) Distribution of HLA-B27 subtypes in ankylosing spondylitis in an Israeli population. Arch Med Res 38:452–455
Birinci A, Bilgici A, Kuru O, Durupinar B (2006) HLA-B27 polymorphism in Turkish patients with ankylosing spondylitis. Rheumatol Int 26:285–287
Gonzalez S, Garcia-Fernandez S, Martinez-Borra J, Blanco-Gelaz MA, Rodrigo L et al (2002) High variability of HLA-B27 alleles in ankylosing spondylitis and related spondyloarthropathies in the population of northern Spain. Hum Immunol 63:673–676
Lopez-Larrea C, Blanco-Gelaz MA, Torre-Alonso JC, Bruges Armas J, Suarez-Alvarez B et al (2006) Contribution of KIR3DL1/3DS1 to ankylosing spondylitis in human leukocyte antigen-B27 Caucasian populations. Arthritis Res Ther 8:R101
Varnavidou-Nicolaidou A, Karpasitou K, Georgiou D, Stylianou G, Kokkofitou A et al (2004) HLA-B27 in the Greek Cypriot population: distribution of subtypes in patients with ankylosing spondylitis and other HLA-B27-related diseases. The possible protective role of B*2707. Hum Immunol 65:1451–1454
Lopez-Larrea C, Gonzalez-Roces S, Pena M, Dominguez O, Coto E et al (1995) Characterization of B27 haplotypes by oligotyping and genomic sequencing in the Mexican Mestizo population with ankylosing spondylitis: juvenile and adult onset. Hum Immunol 43:174–180
Gonzalez-Roces S, Alvarez MV, Gonzalez S, Dieye A, Makni H et al (1997) HLA-B27 polymorphism and worldwide susceptibility to ankylosing spondylitis. Tissue Antigens 49:116–123
Ren EC, Koh WH, Sim D, Boey ML, Wee GB et al (1997) Possible protective role of HLA-B*2706 for ankylosing spondylitis. Tissue Antigens 49:67–69
Hou TY, Chen HC, Chen CH, Chang DM, Liu FC et al (2007) Usefulness of human leucocyte antigen-B27 subtypes in predicting ankylosing spondylitis: Taiwan experience. Intern Med J 37:749–752
Ma HJ, Hu FP (2006) Diversity of human leukocyte antigen-B27 alleles in Han population of Hunan province, southern China. Tissue Antigens 68:163–166
Chou CT, Chen JM, Hsu CM, Chen SJ (2003) HLA-B27 and its subtypes in 4 Taiwanese aborigine tribes: a comparison to Han Chinese patients with ankylosing spondylitis. J Rheumatol 30:321–325
Park SH, Kim J, Kim SG, Kim SK, Chung WT et al (2009) Human leucocyte antigen-B27 subtypes in Korean patients with ankylosing spondylitis: higher B*2705 in the patient group. Int J Rheum Dis 12:34–38
Yang M, Xu M, Pan X, Hu Z, Li Q et al (2013) Epidemiological comparison of clinical manifestations according to HLA-B*27 carrier status of Chinese ankylosing spondylitis patients. Tissue Antigens 82:338–343
Wu Z, Lin Z, Wei Q, Gu J (2009) Clinical features of ankylosing spondylitis may correlate with HLA-B27 polymorphism. Rheumatol Int 29:389–392
Sun L, Wu R, Xue Q, Wang F, Lu P (2016) Risk factors of uveitis in ankylosing spondylitis: an observational study. Medicine (Baltimore) 95:e4233
Yang T, Duan Z, Wu S, Liu S, Zeng Z et al (2014) Association of HLA-B27 genetic polymorphisms with ankylosing spondylitis susceptibility worldwide: a meta-analysis. Modern Rheumatol 24:150–161
Zhang L, Liu JL, Zhang YJ, Wang H (2011) Association between HLA-B*27 polymorphisms and ankylosing spondylitis in Han populations: a meta-analysis. Clin Exp Rheumatol 29:285–292
Kim TJ, Na KS, Lee HJ, Lee B, Kim TH (2009) HLA-B27 homozygosity has no influence on clinical manifestations and functional disability in ankylosing spondylitis. Clin Exp Rheumatol 27:574–579
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
No procedure in this study has involved human participants.
Rights and permissions
About this article
Cite this article
Lin, H., Gong, YZ. Association of HLA-B27 with ankylosing spondylitis and clinical features of the HLA-B27-associated ankylosing spondylitis: a meta-analysis. Rheumatol Int 37, 1267–1280 (2017). https://doi.org/10.1007/s00296-017-3741-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3741-2