Abstract
The aim of the present study was to determine the prevalence of human leukocyte antigen HLA-B27 in Moroccan healthy controls and in patients with ankylosing spondylitis (AS), and to analyze the correlation between HLA-B27 and AS in Moroccan patients. The prevalence of HLA-B27 was determined by evaluating the number of HLA-B27-positive samples in 128 healthy subjects and in 53 patients diagnosed with AS according to the ESSG and AMOR criteria. HLA-B27 was determined by the polymerase chain reaction using sequence-specific primers. Multivariate analysis of our data (HLA-B27, age, sex, and family history) for AS and healthy controls was performed by multiple correspondence analysis (MCA). The frequency of HLA-B27 was significantly greater in AS patients (45.3 %) than in healthy controls (4.7 %) [p < 0.0001, OR 16.8, and CI 95 % (5.83–51.03)]. In addition, HLA-B27 was more common in male patients than in female ones (p < 0.05). 100 % of the AS patients reported a family history of AS, whereas only 20 % of the healthy controls reported a family history of AS. The graphical interpretation of MCA showed a significant relation between the presence of HLA-B27 and AS. This study strengthens the link between HLA-B27 and AS and represents a very valuable informative diagnostic tool, especially in regard to male patients who have a family history of AS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ankylosing spondylitis (AS) is a common chronic inflammatory rheumatic disease affecting sacroiliac joints, spine, and peripheral arthritis and is a representative member of the group of spondyloarthropathies (SpA) [1]. Geoepidemiological studies of AS reveal a higher incidence of AS in northern European populations compared to sub-Saharan African ones [2, 3]. It affects young adults and men, with a sex ratio of approximately 3:1 (M/F) [4]. Genetic and environmental factors have been reported to play an important role in terms of predisposition to the disease. In fact, a strong genetic association with HLA-B27 has been shown previously many times [5, 6], and it is one of the best examples of a disease association with a genetic marker [7]. To the best of our knowledge, the exact mechanism of the role of HLA-B27 in AS pathogenicity is still poorly understood. Various hypotheses have been suggested to explain the association of HLA-B27 with AS. The arthritogenic peptide hypothesis states that certain T cell receptors can recognize a complex of foreign and unknown MHC self-peptides when together [7]. The molecular mimicry hypothesis suggests a cross-reactivity between antigens of associated bacteria and the HLA-B27 molecule [8, 9], while a third hypothesis is based on aberrant folding of HLA-B27 inducing disorder of intracellular traffic of this molecule [10]. However, none of these hypotheses fully elucidates the pathogenicity mechanism of HLA-B27 in AS.
The prevalence of HLA-B27 and its association with AS varies markedly between racial and ethnic groups with a clear decline in frequency from north to south across the world [11]. For example, in the Caucasian population, HLA-B27 is found in approximately 90 % of patients with AS [5, 6], as compared to 8 % in the general population, but in Sub-Saharan Africa the prevalence of HLA-B27 is rare in the general population, and it is present in only 6 % of patients with AS [5, 6, 12]. Thus, the relative risk of subjects carrying HLA-B27 has been shown to be >70, with an odds ratio of about 100 [13]. Identification of HLA-B27 in patients in which AS is suspected could be helpful for diagnosis and disease management [14, 15].
The Moroccan population has an interesting gene pool inherited from different ethnic groups (Arabs, Sahrawi, Berbers, and Caucasians), a situation which can be useful to investigate the role of HLA-B27 in AS. The HLA-B27 antigen prevalence in the Moroccan population was reported to be 3.7 % [16], which is not significantly different from values reported in other Mediterranean, North African, and Arabic countries: 4.7 % in Greek Cypriots [17], 3–6 % in Tunisia [18, 19], and 3 % in the United Arab Emirates (UAE) [20].
The purpose of this study was to investigate the prevalence of HLA-B27 in healthy controls and in patients with AS in the Moroccan population. Additionally, we have analyzed the association between several variables (HLA-B27, sex, age, and family history) and AS by using a categorical data analysis technique, i.e., multiple correspondence analysis (MCA).
Materials and methods
Study population
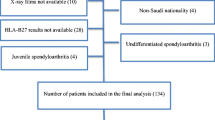
Fifty-three (53) informed, volunteering, and unrelated AS patients of both genders (21 females and 32 males with ages ranging between 16 and 59 years) were recruited from the Department of Rheumatology, among the outpatient clinics and Ibn Tofail University, Hospital of Marrakech, Morocco. Diagnosis of AS was done in accordance with AMOR and ESSG criteria [21, 22]. One hundred twenty-eight (128) informed, volunteering, and healthy unrelated blood donors (44 females and 84 males with ages ranging between 17 and 56 years), without AS, and living in the same geographic area of Morocco were included as controls.
All subjects were asked to identify members of their immediate family, their parents, their siblings, and their children with any history of AS or AS symptoms.
The demographic characteristics of the AS patients and of the healthy controls are given in Table 1.
The survey conducted in this study was approved by an ethical committee, and consent was obtained from all participants.
HLA-B27 typing
Genomic DNA was extracted from whole blood samples using a commercial kit (GenElute™ Mammalian Genomic DNA Purification Kit, Sigma-Aldrich). HLA-B27 was typed using polymerase chain reaction-sequence-specific primers designed by O. Olerup, 1994 [23]. This genotyping was undertaken using the Bio-Rad S1000 Thermal Cycler (California, USA). The assay was validated using samples of a known genotype based on DNA sequencing.
Statistical analysis
The frequencies of HLA-B27 and statistical comparisons were determined by direct counting. Frequencies in patients and controls were compared using the Chi-squared test, and the level of significance was set at 0.05.
Multiple correspondence analysis was applied to explore and describe the relationship between four variables: HLA-B27 positivity (Ne = negative and Po = positive), age (<45, 45–55, and >55), sex (female or male), and family history of AS (parents, siblings, other, and without). MCA, a technique for data analysis and an extension of correspondence analysis (CA), allows for the identification of the most important directions of variability in a multivariate data matrix (Burt table), and the results are presented in a low-dimensional map fashion. The distances between the points in the dimensional space summarize all of the information about the similarities between the variables; thus, variables that are projected close to each other on the graphical map are positively correlated. This multivariate analysis transforms the variables into new axes, which are orthogonal, and the number of axes to be retained for analysis is determined by calculating modified eigenvalues. The associations between variables are revealed by calculating the Chi-square distance between different categories of the variables and between individuals [24].
In addition, this exploratory method was applied to analyze the data set (healthy controls and AS patients) as a first step, and to explore separately AS patients as a second step. The statistical analyses in this study were performed using the commercial STATISTICA ® software package for Windows, version 8.0.
Results
A total of 181 subjects participated in this study, 128 healthy controls and 53 AS patients. The control group was 66 % males and 34 % females (M/F = 1.9) with a mean age of 30.7 ± 9.4 years. The patient group was 60 % males and 40 % females (M/F = 1.5) with a mean age of 37.81 ± 11.5 years. Table 2 summarizes the distribution of HLA-B27 in these AS patients compared to that in the healthy controls. The frequency of HLA-B27 was significantly higher in AS patients than in healthy controls: 45.3 and 4.7 %, respectively [p < 0.0001, OR 16.8 and CI 95 % (5.83–51.03)]. In the AS patient group, as expected, the frequency of HLA-B27 showed a significant increase in the male group (56.3 %) compared to the female group (28.6 %), (p < 0.05).
In addition, all AS patients reported at least one first-degree relative (father or mother) with confirmed AS, whereas only 20 % of the healthy controls reported a family history of AS (parents, siblings, and others).
MCA
The first step of this analysis was the application of MCA to the complete data set (AS patients and healthy controls). The results of MCA revealed that the total inertia in Burt’s table was equal to 1.800 with a significant difference between the first axis (25.70 %) and the second one (13.36 %). We decided to retain only the first and the second axes from the nine axes (Table 3).
The values of Chi-square presented in Table 3 showed the existence of associations between variables. Indeed, if the axes had been totally independent, as there are nine dimensions in total, the first two axes would have shown an inertia value of 22 % while 39.7 % was calculated.
These results are presented in Fig. 1. In fact, the profiles of AS patients and healthy controls are quite different. AS patients are characterized by the presence of HLA-B27, with a family history of AS in first-degree relatives (father or mother) and an age range between 45 and 55 years, and the distances between these variables are quite shorter and clustered on the positive side in the factorial design. On the other hand, healthy controls clustered on the negative side and are characterized by the absence of HLA-B27, an age range <45 years, and these healthy controls were without a family history of AS.
Plot of first and second axes (Dim 1*2) for the classification of AS patients and of healthy controls according to their variables. Projection of the variables in principal plane 1*2: the first and the second dimensions present, respectively, 25.70 and 13.37 % of the total inertia. The sharps of the clouds suggest a repulsion between two groups of variables. Group 1: Absence of HLA-B27 (HLA-B27: Ne), absence of family history (family history: W), and age <45 seem to characterize healthy controls in the factorial design (blue circle). Group 2: On the opposite side, the AS patients were characterized by the presence of HLA-B27 (HLA-B27: Po), one of the parents diagnosed with AS (family history: P), and age between 45 and 55 (red circle). AS: ankylosing spondylitis/AS: healthy (healthy controls)/AS: AS-confirmed (AS patients)/HLA-B27: Ne (HLA-B27 negative)/HLA-B27: Po (HLA-B27 positive)/Family history: (parents family history of AS)/S (sibling family history of AS)/O (other family history of AS)/W (without family history of AS)/Sex: F (female)/Sex: M (male)
To improve the results and the characterization of the interactions between chosen variables and AS, we performed MCA on the AS patients. In this analysis, total inertia was 1.200, with greater differences for the second and the third axes compared to the first axis. The first three axes explained 28.21, 22.58, and 21.77 % of the total inertia, respectively. It was therefore decided to retain only the first three axes to reduce the dimensions of the problem while retaining a percentage value for inertia quite important (72.58 %) for this analysis (data not shown).
Figures 2 and 3 show the location of variables in the multivariate space of the first three axes (dimensions 1 and 2, dimensions 1 and 3). Both Figs. 2 and 3 are similar as they revealed the presence of a conjunction between HLA-B27-positive and the male sex, mother family history of AS, and age <45 years. On the other hand, HLA-B27 negative was related to the female sex, father family history of AS, and age ranging between 45 and 55 years.
Plot of first and second axes (Dim 1*2) for the classification of AS patients according to their variables. Projection of variables in principal plane 1*2: The first and second dimensions present, respectively, 28.22 and 22.58 % of the total inertia. In this factorial design, the AS patients with HLA-B27 were predominantly of the male sex with a mother family history of AS and with an age <45 years (blue circle). On the opposite side, the HLA-B27-negative AS patients were predominantly of the female sex, with a father family history of AS and with an age ranging between 45 and 55 years (red circle). Family history: f (father family history of AS)/Family history: m (mother family history of AS)
Plot of first and third axes (dim 1*3) for the classification of AS patients according to their variables. Projection of variables in principal plane 1*3: the first and the third dimensions present, respectively, 28.22 and 21.78 % of the total inertia. The interpretation of this factorial design is like that of Fig. 2, but the distance between variables in this graph plot seems to be smaller than that represented in Fig. 2
Discussion
In this study, the patients were significantly more likely to have a young age at onset of AS (74 % were 16–45 years); this result was in agreement with the literature [25]. We also found that AS was more common in men than in women with a ratio of 1.5, which is lower to those determined in other studies (such as in German and French populations) [25] but comparable to those of Chinese and Turkish populations [26, 27]. The male-to-female ratio was 3 in HLA-B27-positive AS patients and 0.9 in HLA-B27-negative AS patients; this finding was compatible with previous studies [26].
Regarding family history of AS, we observed that all patients reported a history of AS in their family, while only 20 % of the healthy controls reported a family history of AS. This result supports a higher incidence of HLA-B27 in families with a history of AS and argues in favor of a strong involvement of genetic factors in the development of AS [28, 29].
According to the results of the present study, the frequency of HLA-B27 was 45.3 %, which is significantly higher than that in healthy controls, which was 4.7 % [p < 0.0001, OR 16.83, 95 % CI (5.83–51.03)]. This frequency was lower than that reported in other studies: over 90 % in Caucasian patients [5, 6], 70 % in Turkish patients [27], and 83.3 % in Korean patients [30]. Nevertheless, the frequency observed in our study was similar to that reported in Tunisian and African patients [31]. Recently, a Moroccan study undertaken by a rheumatologic team from Rabat and involving 46 AS patients has shown a frequency of 58.6 % [32]. This difference could be explained by the ethnicity of the studied population. In fact, the population of our cohort was higher in Sahrawi and Berbers, while the population of the Rabat investigation was higher in Arabs and Caucasians. This lower frequency in African countries could suggest the involvement of genes not belonging to the HLA system and of environmental factors specific to these countries in the predisposition to AS. Furthermore, the frequency of HLA-B27 in our healthy controls, 4.6 %, was less than that reported in a Tunisian population 6 % [19] and in other European populations (8 %) [13].
Despite the fact that prevalence of HLA-B27 in the AS patients surveyed in our study was lower than those previously reported for other populations, it was however significantly higher compared to that found in the healthy controls, demonstrating that HLA-B27 is strongly associated with AS in our country. Thus, typing of HLA-B27 could be of significant value in the diagnosis of AS in cases where the clinical picture is incomplete.
The MCA approach confirmed the findings of others studies [25] and was an efficient tool for the characterization of the AS patients. The patients with confirmed diagnosis of AS showed a high association with HLA-B27 and were also more likely to have first-degree relatives with this disease.
Additionally, patients with AS studied separately provided a correlation between HLA-B27, male sex, age <45 years, and positive family history. In this regard, the HLA-B27-positive patients were more likely to be male, to have shown a younger disease onset, and report a maternal AS diagnosis.
Finally, AS still remains a complex phenomenon resulting from the interaction of several genes and environmental factors, including HLA-B27. Further studies are expected to discover other genes that are responsible for AS, which will facilitate diagnosis and screening of subjects at risk, and they will also increase our understanding of the pathophysiological mechanism of AS and, at the same time, lead to a better understanding of the potential pathogenic role of HLA-B27. All of these achievements will undoubtedly favor the development of targeted therapies for this condition [33].
Conclusion
The MCA findings provided a significant association between HLA-B27 and AS in our study, thus suggesting that HLA-B27 could be a biomarker for this pathogenesis. In this study, the AS and healthy control groups were relatively well distinguished based on HLA-B27 and family history of AS. The application of MCA to the AS patient cohort indicated that the HLA-B27-positive cases were predominantly males, with an earlier age of AS onset. It should be noted that a family history of AS plays a major role in susceptibility to the AS disease.
The next step should be the application of MCA to a greater number of AS patients in order to explore other risk factors which could predispose to and influence AS onset.
References
Akgul O, Ozgocmen S (2011) Classification criteria for spondyloarthropathies. World J Orthop 2:107–115
Braun J, Bollow M, Remlinger G, Eggens U, Rudwaleit M, Distler A et al (1998) Prevalence of spondyloarthropathies in HLA-B27 positive and negative blood donors. Arthritis Rheum 41:58–67
Helmick CG, Felson DT, Lawrence RC, Gabriel S, Hirsch R, Kwoh CK et al (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum 58:15–25
Dean LE, Jones GT, Macdonald AG, Downham C, Sturrock RD, Macfarlane GJ (2014) Global prevalence of ankylosing spondylitis. Rheumatology (Oxford) 53:650–657
Schlosstein L, Terasaki PI, Bluestone R, Pearson CM (1973) High association of an HL-A antigen, W27, with ankylosing spondylitis. N Engl J Med 288:704–706
Brewerton DA, Hart FD, Nicholls A, Caffrey M, James DC, Sturrock R (1973) Ankylosing spondylitis and HL-A27. Lancet 1:904–907
Brown MA (2008) Breakthroughs in genetic studies of ankylosing spondylitis. Rheumatology (Oxford) 47:132–137
Khan MA, Mathieu A, Sorrentino R, Akkoc N (2007) The pathogenetic role of HLA-B27 and its subtypes. Autoimmun Rev 6:183–189
Ebringer A (1983) The cross-tolerance hypothesis, HLA B27 and ankylosing spondylitis. Br J Rheumatol 22(suppl 2):53–66
Mear JP, Schreiber KL, Münz C, Zhu X, Stevanović S, Rammensee HG et al (1999) Misfolding of HLA-B27 as a result of its B pocket suggests a novel mechanism for its role in susceptibility to spondyloarthropathies. J Immunol 163:6665–6670
Blanco-Gelaz MA, López-Vázquez A, García-Fernández S, Martínez-Borra J, González S, López-Larrea C (2001) Genetic variability, molecular evolution, and geographic diversity of HLA-B27. Hum Immunol 62:1042–1050
Mijiyawa M, Oniankitan O, Khan MA (2000) Spondyloarthropathies in sub-Saharan Africa. Curr Opin Rheumatol 12:281–286
Khan MA, Ball EJ (2002) Genetic aspects of ankylosing spondylitis. Best Pract Res Clin Rheumatol 16:675–690
Reveille JD, Arnett FC (2005) Spondyloarthritis: update on pathogenesis and management. Am J Med 118:592–603
Reveille JD (2006) Major histocompatibility genes and ankylosing spondylitis. Best Pract Res Clin Rheumatol 20:601–609
Gómez-Casado E, del Moral P, Martínez-Laso J, García-Gómez A, Allende L, Silvera-Redondo C et al (2000) HLA genes in Arabic-speaking Moroccans: close relatedness to Berbers and Iberians. Tissue Antigens 55:239–249
Varnavidou-Nicolaidou A, Karpasitou K, Georgiou D, Stylianou G, Kokkofitou A, Michalis C et al (2004) HLA-B27 in the Greek Cypriot population: distribution of subtypes in patients with ankylosing spondylitis and other HLA-B27-related diseases. The possible protective role of B2707. Hum Immunol 65:1451–1454
Kchir MM, Hamdi W, Laadhar L, Kochbati S, Kaffel D, Saadellaoui K et al (2010) HLA-B, DR and DQ antigens polymorphism in Tunisian patients with ankylosing spondylitis (a case-control study). Rheumatol Int 30:933–939
Ayed K, Ayed-Jendoubi S, Sfar I, Labonne MP, Gebuhrer L (2004) HLA class-I and HLA class-II phenotypic, gene and haplotypic frequencies in Tunisians by using molecular typing data. Tissue Antigens 64:520–532
Al-Attia HM, Al-Amiri N (1995) HLA-B27 in healthy adults in UAE. An extremely low prevalence in Emirian Arabs. Scand J Rheumatol 24:225–227
Dougados M, van der Linden S, Juhlin R, Huitfeldt B, Amor B, Calin A et al (1991) The European spondyloarthropathy study group preliminary criteria for the classification of spondyloarthropathy. Arthritis Rheum 34:1218–1227
Amor B, Dougados M, Mujiyawa M (1990) Critères diagnostiques des spondylarthropathies. Rev Rhum 57:85–89
Olerup O (1994) HLA-B27 typing by a group-specific PCR amplification. Tissue Antigens 43:253
Benzecri JP (1973) Analyses des Données. Dunod, Paris, pp 1–2
Sieper J, Braun J, Rudwaleit M, Boonen A, Zink A (2002) Ankylosing spondylitis: an overview. Ann Rheum Dis 61(Suppl 3):iii8–iii18
Liu X, Li YR, Hu LH, Zhou ZM, Chen FH, Ning Y et al (2010) High frequencies of HLA-B27 in Chinese patients with suspected of ankylosing spondylitis. Rheumatol Int 30:1305–1309
Gunal EK, Sarvan FO, Kamali S, Gul A, Inanc M, Carin M et al (2008) Low frequency of HLA-B27 in ankylosing spondylitis patients from Turkey. Joint Bone Spine 75:299–302
Woodrow JC, Nichol FE, Whitehouse GH (1983) Genetic studies in ankylosing spondylitis. Br J Rheumatol 22(suppl 2):12–17
Tsui FWL, Haroon N, Reveille JD, Rahman P, Chiu B, Tsui HW et al (2010) Association of an ERAP1 ERAP2 haplotype with familial ankylosing spondylitis. Ann Rheum Dis 69:733–736
Baek HJ, Shin KC, Lee YJ, Kang SW, Lee EB, Yoo CD et al (2004) Clinical features of adult-onset ankylosing spondylitis in Korean patients: patients with peripheral joint disease (PJD) have less severe spinal disease course than those without PJD. Rheumatology (Oxford) 43:1526
Frikha F, Marzouk S, Jallouli M, Frigui M, Kaddour N, Jribi S et al (2006) La spondylarthrite ankylosante: étude d’une série de 83 cas. Revue du rhumatisme 73:1251
Atouf O, Benbouazza K, Brick C, Saoud B, Benseffaj N, Amine B et al (2012) Distribution of HLA class I and II genes in ankylosing spondylitis patients from Morocco. Pathol Biol 60:e80–e83
López de Castro JA (2007) HLA-B27 and the pathogenesis of spondyloarthropathies. Immunol Lett 108:27–33
Acknowledgments
The current study was supported by Mohammed V-Agdal University “plan d’urgence” research grant. We would like to thank all patients and healthy controls for their participation. We are sincerely grateful to the clinicians and nurses for their contribution to the clinical examinations and blood collection. The authors wish to express their gratitude to Denis Groleau, Professor at Chemical and Biotechnological engineering department, Faculté de Génie, University of Sherbrooke, for his help in this study and Mounia Akassou for computing assistance.
Authors’ contributions
All authors contributed to the conduct of the research and writing of this manuscript. Authors reviewed and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no conflict of interest exists.
Rights and permissions
About this article
Cite this article
Akassou, A., Yacoubi, H., Jamil, A. et al. Prevalence of HLA-B27 in Moroccan healthy subjects and patients with ankylosing spondylitis and mapping construction of several factors influencing AS diagnosis by using multiple correspondence analysis. Rheumatol Int 35, 1889–1894 (2015). https://doi.org/10.1007/s00296-015-3342-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-015-3342-x