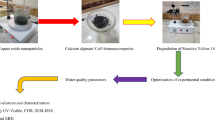
Abstract
Wastewater contaminated by dye produced by the rapid industrialization is a global problem. One type of the dyes used in the industry is malachite green (MG). In this study, we reported the results of designing and synthesizing the Ca-Alginate (Metal Organic Framework (MOF)) as well as its application as an adsorbent to reduce the amount of MG in wastewater. The alginate used for the synthesis of Ca-Alginate was first obtained by extracting it from brown algae using sodium carbonate as a solvent. In this study, Ca-Alginate was obtained by ion exchange process using calcium chloride solution and calcination at a temperature of 900 °C. Based on the characterization data, it was found that the adsorbent has a macropore structure with a specific surface area of 0.614 m2/g and a total pore volume of 1.744 × 10−3 cc/g. The result of the FTIR characterization showed the presence of hydroxyl groups, symmetrical and asymmetrical carboxyl groups, and mannuronic and guluronic groups. These groups indicated the presence of alginate. The adsorption ability test on the MG dye was performed at various changes in contact time and concentration of the adsorbent, while the MG concentration on the adsorption process was examined using the batch system. The results showed that the highest reduction in MG occurred at the contact time of 120 min, 0.06 g/L of adsorbent dosage and 0.0055 g/L of initial MG concentration. Under these conditions the Ca-Alginate could adsorb up to 84.47% of MG. The adsorption kinetic of MG onto Ca-Alginate obeyed the modified pseudo-first-order (MPFO) model.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dyes that contain the benzene ring carry the properties of benzene which are toxic and cannot be degraded naturally [34, 37]. These dyes are dangerous for humans because they can cause hormonal disorders, carcinogenesis, immune system diseases and kidney failure [2]. Therefore, due to their hazardous nature, a treatment to the waste containing such substances is necessary.
Some of the methods to treat color wastes are ion exchange, photocatalytic degradation, electrochemical engineering, membrane separation, biological processes and adsorption. From all the methods, adsorption is the most beneficial because of its simple process, low operating cost and only produces few by-products [19, 29, 36]. Several adsorbents that have been used, such as such as chitosan, activated carbon and silicic microspheres, have been commonly used in textile industrial waste treatment [14]. However, these materials usually have insufficient adsorption capacity. Thus, from now the use of MOF as an adsorbent is being developed.
The use of MOF as an adsorbent is deemed beneficial because of the ease in controlling the porosity, the morphological properties, the large surface are, and the larger properties of its pores and crystals [24]. Several MOFs which are stable in the water have been used as adsorbents to adsorb and separate the contaminants (including organic compounds and inorganic ions) in the solution [40]. Furthermore, MOF can be synthetized with various types of metal which can be adjusted for used with the type of dye to be removed.
The adsorption process is usually divided into three general steps: (1) convective diffusion of the adsorbate from the liquid phase to outer surface of the solid object; (2) diffusion from the outer surface of the adsorbant pores/channels; and (3) adsorption of the adsorbate in the active part of the adsorbent. The mass transfer process depends on the properties of the adsorption and the operating condition [40]. Among the studies that have been conducted regarding the use of MOF as an adsorbent in liquid waste treatment is a study conducted by Zhang et al. [39] which utilized ZIF-8@Fe/Ni to adsorb liquid waste containing malachite green (MG) with a concentration of 0.5 g/L. The adsorbed amount of MG was 92% with adsorption capacity of ZIF-8@Fe/Ni is 151.520 mg/g. Furthermore, Shi et al. [35] used Cu-MOFs/Fe3O4 to adsorb waste containing MG with a concentration of 4.64 mg/L. The adsorbed amount of MG was 94.35% with adsorption capacity of Cu-MOFs/Fe3O4 is 113.67 mg/g.
From the examples mentioned above, it has been proven that the biopolymer is proven to be able to adsorb MG dye using the adsorption process [35, 40]. In this study, the researchers conducted an experiment using an alginate molecular framework obtained from brown algae with the calcium metal to synthesize MOF with the aim to adsorb the MG dye. Alginate was chosen because the raw materials are easy to find and the process is relatively simple, namely using extraction and ion exchange [10, 11]. Calcium metal was chosen because it belongs to the non-heavy metal group and is widely available in the nature. In the earth's crust there is ±3.4% mass of elemental calcium. Calcium is the fifth most abundant element in the earth's crust. Calcium can be found in various compounds in the nature, such as anorthite (CaAl2Si2O8), thermolite/asbestos (Ca2Mg2(Si4O11)2(OH)2), wolastonite (CaSiO3), limestone (CaCO3), gypsum (CaSO4·2H2O), fluorite (CaSO4·CaF2), dolomite (CaCO3·MgCO3), marble (CaCO3·MgCO3), gypsum (CaSO4·2H2O), calcite (CaCO3) and phosphorite (Ca(PO4)2) [6]. As far as the author knows, there is not enough information about the adsorption of MG using Ca-Alginate-based adsorbent. Therefore, Ca-Alginate based adsorbent was synthesized in this study and several parameters that have an effect on its application for the adsorption of MG were also studied. In addition, it should be noted that the alginate used for the synthesis of Ca-Alginate-based adsorbent comes from brown algae which is a local natural resource in Ngeden Beach, Gunung Kidul, Special Region of Yogyakarta, Indonesia. In addition, this study also studied kinetic modeling for the adsorption of MG using Ca-Alginate-based adsorbent.
Materials and methods
Materials
The brown algae were obtained from Ngeden Beach, Gunung Kidul, Special Region of Yogyakarta, Indonesia. Sodium carbonate (Na2CO3) (CAS No. 497-19-8), calcium chloride (CaCl2) (CAS No. 10043-52-4) and MG dye (CAS No. 2437-29-8) have been obtained from Merck.
Extraction of alginate from brown algae
The process of making the Ca-Alginate (MOF) was done by extracting the alginate from brown algae using sodium carbonate (Na2CO3) as the solvent. Brown algae were washed thoroughly and were drained until there were no water dripping. Then, the aquades was added with a 1:6 (w/w) weight ratio of brown algae and aquades and was left for 2 h. After that, it was blended until it reached the texture of pulp. The brown algae pulp was added with 2% Na2CO3 solution with a 1:20 (b/v) weight ratio, which was then extracted for 2 h in an 80 °C temperature. After 2 h, the extraction was stopped and the mixture was cooled to room temperature. Once it was cooled, the mixture was separated using filter paper. The filtrate contained Sodium alginate and the residue was disposed as waste.
Synthesis of Ca-Alginate (MOF)
This step was performed with the method of ion exchange between the extracted sodium alginate and CaCl2. The sodium alginate solution was dripped with 0.1 M of CaCl2 solution until it formed sediments. The dripping was stopped when no more sedimentation was formed. The formed sedimentations were separated using filter paper, and the filtrated was disposed as waste. The sediments were then dried in an oven at a temperature of 104 °C until it reached the constant weight. The dried sediments were calcined at 900 °C to produce the Ca-Alginate (MOF).
Analysis of Ca-Alginate (MOF) as an adsorbent
Ca-Alginate (MOF) was characterized using FTIR to determine the availability of the adsorbent functional groups. The FTIR analysis was performed in range of 4000–400 cm−1 using Nicolet Avatar 360 IR tool. Meanwhile, characterization with BET method was conducted to determine the active surface area of the adsorbent. The BET analysis used the NOVA 1200e microanalyzer tool (Quantachrome Instrument) which then obtained the isotherm data of the adsorption and desorption as well as the distribution of the pores size of the sample. In this study, activities of the adsorbent were tested by the adsorption process using MG dye.
Determination of maximum wavelength for MG and making of the calibration curve
Before performing the study on the adsorption parameter of MG using Ca-Alginate (MOF), it was necessary to determine the former’s maximum wavelenghth. According to the literature, the maximum wavelength of MG is 618 nm [23]. Determination of MG maximum wavelength was conducted in the wavelength range of 587–647 nm using the UV–Vis spectrophotometer.
The making of standard calibration curve with a maximum wavelength of 617 nm was carried out in the absorbance range of 0.2–0.8 with various concentrations of MG dye solution. This was done to find out the linear regression equation which shows the correlation between the concentration of MG and its absorbance.
Adsorption of MG using Ca-Alginate (MOF)
The study or adsorption parameter was conducted by varying the contact time, dosage of adsorbent and the initial concentration of MG. The adsorption of MG using Ca-Alginate (MOF) as adsorbent was measured at a maximum wavelength of 617 nm. Determining the optimum contact time was done by contacting 0.002 g adsorbent with 100 ml of 0.004 g/L MG solution. The contacting was carried out in a 600 rpm of stirring speed for the contact time variations of 1; 3; 5; 10; 15; 30; 45; 60; 90; 120; 150; 180; 210; 240 min.
And the determination of the optimum adsorbent dosage has been carried out by contacting the adsorbent dosage of 0.0080; 0.0200; 0.0400; 0.0600; 0.0800; 0.1200; 0.4000 g/L to 100 ml of 0.004 g/L MG solution. The contacting was carried out with 600 rpm stirring speed for 120 min. While the determination of the optimum MG concentration has been carried out by contacting the optimum weight of the adsorbent, which was 0.006 g, to 100 ml of various concentration of MG (0.0020; 0.0025; 0.0030; 0.0035; 0.0040; 0.0045; 0.0005; 0.0055; 0.0060; 0.0065 g/L). The contacting was carried out with 600 rpm stirring speed for 120 min.
Once the adsorption process was performed, the sample was tested with UV–Vis spectrophotometer to determine the absorbance and also the concentration. Therefore, it could be used to determine the adsorbed percentage, which could be found by:
where C0 is the initial concentration, and Ct is the concentration of MG after the adsorption process.
Results and discussion
Characterization of Ca-Alginate adsorbent using FTIR
The Ca-Alginate adsorbent before and after the calcination was analyzed using the infrared spectrum or Fourier Transform Infra Red (FTIR) to determine the availability of the functional groups. Analysis of the functional groups was carried out at a wavelength of 4000–400 cm−1. It is shown in Fig. 1 that before the calcination, there was a hydroxyl function (–OH) at a wavelength of 3000–3600 cm−1 [25] with the absorption at a wavelength of 3425.54 cm−1. There was a carboxylate group in the 1789.00 cm−1 wavelength region for the asymmetric COO– group and 1419.22 cm−1 for the symmetrical COO– group. There were absorption peaks at 873.65 cm−1 and 711.37 cm−1 which indicate the presence of mannuronic and guluronic fingerprint groups [8, 9, 20]. These mannuronic and guluronate groups both showed an alginate structure [21]. It is also shown in Fig. 1 that after the calcination, there was a sharp absorption area at 3642.14 cm−1 which was a hydroxyl group (–OH). Also, there was a carboxylate group at the 1475.87 cm−1 wavelength.
Characterization of Ca-Alginate adsorbent using BET
Identification of the surface area was carried out through the physisorption of N2 gas using the NOVA 1200 microanalyzer (quantachrome) using Brunaur Emmet Teller (BET) method. Based on the analysis, the Ca-Alignate adsorbent before calcination had a specific surface area of 2.739 m2/g with a total pores volume of 1.242 × 10−2 cc/g. The total pores volume (VT) was defined as the maximum amount of nitrogen which is adsorted at relative pressure P/P0 = 0.99033. Meanwhile, the Ca-Alignate adsorbent after calcination had a specific surface area of 0.614 m2/g with a total pores volume of 1.744 × 10−3 cc/g. The total pores volume (VT) was defined as the maximum amount of nitrogen which is adsorted at relative pressure P/P0 = 0.99433. This showed that the calcination process affected the surface area of Ca-Alginate adsorbent by making it to become smaller. Calcination in the temperature of up to 900 °C will cause the pores to shrink and undergo a transformation, so that the pores will be completely closed and the grain boundaries will disappear [18]. This caused the total pores volume after calcination to become smaller than that before the calcination. The adsorptivity level of the calcined Ca-Alginate was better because it had the average pore radius of 3396.6 Å, while the Ca-Alginate before calcination was smaller, which was 2001.8 Å. A higher average pore radius has the potential to be filled and the adsorption on the adsorbent pores will be higher [26].
Determination of maximum wavelength for MG and calibration curve of MG
Before determining the absorbed percentage of MG using Ca-Alginate (MOF), determination of maximum wavelength for MG was conducted to provide maximum sensitivity of the samples containing MG. Determination of the maximum wavelength for MG was carried out in the wavelength range of 587–647 nm using UV–Vis spectrophotometer. The maximum wavelength of MG was 617 nm (Fig. 2). The obtained maximum wavelength slightly differed from the literature which was 618 nm [23].
The making of the calibration curved was used to find out the equation of the linear regression. As a result, it could be used in finding a certain amount whose absorbance had been measured. The linear regression equation indicates the correlation between the amount of MG and its absorbance. The calibration curve could be seen in Fig. 3 with slope value of 140,244 and intercept value of − 0.0872 with R2 value of 0.9777. The yielded absorbance data were classified as good since all the measured amounts of MG had the absorbance value of 0.2–0.8. In addition, the yielded R2 was also categorized as good since it was close to 1.
Adsorption study
Determination of the optimum contact time for adsorption of MG
The effect of the contact time between the Ca-Alginate adsorbent and MG was necessary to be examined in order to obtain the kinetics of the adsorption process. By conducting a research on the effect of the contact time with percentage of the adsorbed adsorbate, the time required for the adsorbat to reach the equilibrium could be discovered. When the adsorption rate has reached the equilibrium, it is at the same level with the rate of adsorped adsorbate release. Thus, in this certain condition, the adsorbed percentage is relatively constant.
In Fig. 4, it could be seen that at the 120 min contact time, the Ca-Alginate adsorbent could adsorp up to 77.05% of MG. At the 150 min contact time, the adsorption percentage of MG did not increase significantly. At the next times of contact, the adsorption of MG decreased and at the 240 min contact tome, the percentage was 75.8%. This was due to the reduced amount of active sides because the dye solution had formed a layer on the surface of the adsorbent which covered its layer. Thus, it can decrease the adsorption strength. Although the decreasing was not much, it indicates that the adsorbent which had adsorbed the dye solution had undergone desorption which is the re-releasing of ions that have been adsorbed due to experiencing saturation. That after the adsorption reaches equilibrium at the optimum time of contact, the addition of the contact time between the adsorbent and the adsorbate subsequently does not have a significant effect on the absorption of the dye [13]. Physical contact between the dye and the adsorbent for too long caused the dye to be released back into the solution. This resulted in a greater amount of dye exchange, which indicated that the absorption capacity was decreasing as well [12]. Figure 4 shows that the optimum contact time for the adsorption of MG with Ca-Alginate was at 120 min with 205.95 mg/g of qt value.
Determination of the optimum dosage of adsorbent
Figure 5 shows the optimum dosage of Ca-Alginate adsorbent was 0.06 g/L at the adsorption process with 120 min of contact time and 4 mg/L of initial concentration for MG. Under these conditions the Ca-Alginate could adsorb up to 82.68% of MG with 66.51 mg/g of qt value. At the next addition of adsorption, percentage of the adsorbed MG decreased up to 65.82%. There was also a decrease in the adsorption capacity because the active sides of the Ca-Alginate adsorbent had reached their saturation.
In a study conducted by Amiri et al. [4], adsorption of MG using 0.05 g of cobalt ferrite silica nanocomposite (SA20) adsorbent, the qe value was 71.6 mg/g. Meanwhile, [21] used 0.5 g of rice husk bio-char (EBC260) adsorbent to adsorp MG and the qe value was 56.8 mg/g. In addition, another study used 0.01 g of cellulose aerogel adsorbent to adsorp MG and the qe value was 212.7 mg/g [16]. Compared to the results of this study, which used 0.06 g of Ca-Alginate adsorbent with 66.51 mg/g of qe value, it indicates that this result is still far from optimal and further studies are necessary. The comparison between several adsorbents for adsorption of MG is shown in Table 1.
Determination of the optimum concentration for MG
Figure 6 shows the optimum concentration for MG was 5.5 mg/L at the adsorption process with 120 min of contact time and 0.06 g/L of adsorbent dosage. Under these conditions the Ca-Alginate could adsorb up to 84.47% of MG with 94.37 mg/g of qt value. The increase in the adsorption of MG dye could happen due to the high driving force for mass transfer in the added dye concentration. Meanwhile, the resistance to the adsorption of MG decreased with the increasing concentration of the dye. When the surface active site of the adsorbent is completely covered, the adsorption rate reaches the limit resulting in adsorption at the saturation level [32]. It could be seen in Fig. 6 that the trendline for the correlation of the MG concentration in the adsorption process could be related to the fact that there is a high driving force for mass transfer in high dye concentration. Furthermore, if the consistency of the dye in the solution is higher, the active sites of the adsorbent are surrounded by higher amount of dye molecules which leads to more efficient adsorption [31]. On the other hand, the adsorption capacity could reach the qt value of 94.32 mg/g. The increase in adsorption capacity that occurs along with the increase in the concentration of MG might occur because it occurred at a low concentration which allows the adsorbate in the solution to enter the active side of the Ca-Alginate adsorbent. This is in accordance with the statement of Irawati et al. [15] who said that when the active group of the adsorbent is saturated, the increase in the active group of the adsorbate will decrease. However, if at the time the active group of the adsorbent is not saturated by the adsorbate, the increase in the concentration of adsorbate will increase the amount of the adsorbed adsorbate [15].
Modeling of the adsorption kinetics
The study of adsorption kinetics provides the information related to the way, mechanism and rate of the adsorption process in a batch study [22]. The modeling of adsorption kinetics is carried out using seven kinetic models, namely pseudo-first-order (PFO), modified pseudo-first-order (MPFO), modified pseudo-first order-2 (MPFO-2), pseudo-second-order (PSO), modified pseudo-second-order (MPSO), modified pseudo-second-order-2 (MPSO-2) and Elovic, which is shown in Table 2.
The kinetics study was performed by varying the contact time for adsorption of MG. The results of adsorption kinetics modeling could be seen in Table 2 which show that the modeling in this study obeyed the MPFO model which is indicated by its highest coefficient of correlation among other models with R2 = 0.9944. Thus, it is safe to say that the MPFO model could estimate the equilibrium value of adsorption of experiments related to theoretical calculation. The MPFO mode has the tendency in which the initially rapid MG adsorption gradually slows down and eventually reaches the equilibrium value. This equation gives a straight line with k slope and intercept from ln qe [4].
Conclusion
MOF-based Ca-Alginate as an adsorbent to adsorp malachite green (MG) dye was successfully synthetized using the ion exchange method. The Ca-Alginate was characterized using BET and FTIR. In this study, the optimum condition for the adsorption of MG dye using Ca-Alginate adsorbent was obtained at initial MG concentration of 5.5 mg/L, adsorbent dosage of 0.06 g/L and contact time of 120 min. Under these conditions qt of Ca-Alginate for adsorption of MG is 94.37 mg/g. The kinetics study indicated that the adsorption of MG dye with Ca-Alginate adsorbent obeyed the modified pseudo-first-order (MPFO) model. Based on this study it could be said that MOF-based Ca-Alginate has the potential to be used as adsorbent for adsorption of MG dye.
References
Abbasi M, Sabzehmeidani MM, Ghaedi M, Jannesar R, Shokrollahi A (2021) Synthesis of grass-like structured Mn–Fe layered double hydroxides/PES composite adsorptive membrane for removal of malachite green. Appl Clay Sci 203:105946. https://doi.org/10.1016/j.clay.2020.105946
Abdelrahman EA (2018) Synthesis of zeolite nanostructures from waste aluminum cans for efficient removal of malachite green dye from aqueous media. J Mol Liquids 253:72–82. https://doi.org/10.1016/j.molliq.2018.01.038
Akram M, Bhatti HN, Iqbal M, Noreen S, Sadaf S (2017) Biocomposite efficiency for Cr(VI) adsorption: kinetic, equilibrium and thermodynamics studies. J Environ Chem Eng 5(1):400–411. https://doi.org/10.1016/j.jece.2016.12.002
Amiri M, Salavati-Niasari M, Akbari A, Gholami T (2017) Removal of malachite green (a toxic dye) from water by cobalt ferrite silica magnetic nanocomposite: herbal and green sol-gel autocombustion synthesis. Int J Hydrogen Energy 42(39):24846–24860. https://doi.org/10.1016/j.ijhydene.2017.08.077
Azizian S, Fallah RN (2010) A new empirical rate equation for adsorption kinetics at solid/solution interface. Appl Surf Sci 256(17):5153–5156. https://doi.org/10.1016/j.apsusc.2009.12.080
Bua P (2020) Makalah Kimia Dasar 2 (Kimia Unsur). Universitas Negeri Gorontalo, Gorontalo
Budiana IGMN, Jasman J, Neolaka YAB, Riwu AAP, Elmsellem H, Darmokoesoemo H, Kusuma HS (2021) Synthesis, characterization and application of cinnamoyl C-phenylcalix[4]resorcinarene (CCPCR) for removal of Cr(III) ion from the aquatic environment. J Mol Liquids 324:114776. https://doi.org/10.1016/j.molliq.2020.114776
Cardenas-Jiron G, Leal D, Matsuhiro B, Osorio-Roman IO (2011) Vibrational spectroscopy and density functional theory calculations of poly-D-mannuronate and heteropolymeric fractions from sodium alginate. J Raman Spectrosc 42(4):870–878. https://doi.org/10.1002/jrs.2760
Chandia NP, Matsuhiro B, Vasquez AE (2001) Alginic acids in Lessonia trabeculata: characterization by formic acid hydrolysis and FT-IR spectroscopy. Carbohyd Polym 46(1):81–87. https://doi.org/10.1016/S0144-8617(00)00286-1
Cui H, Li Q, Qian Y, Tang R, An H, Zhai J (2011) Defluoridation of water via electrically controlled anion exchange by polyaniline modified electrode reactor. Water Res 45(17):5736–5744. https://doi.org/10.1016/j.watres.2011.08.049
Daradmare S, Xia M, Le VN, Kim J, Park BJ (2021) Metal–organic frameworks/alginate composite beads as effective adsorbents for the removal of hexavalent chromium from aqueous solution. Chemosphere 270:129487. https://doi.org/10.1016/j.chemosphere.2020.129487
Dwijayanti U, Gunawan G, Widodo DS, Haris A, Suyati L, Lusiana RA (2020) Adsorpsi methylene blue (MB) menggunakan abu layang batubara teraktivasi larutan NaOH. Anal Environ Chem 5(1):1–14. https://doi.org/10.23960/aec.v5.i1.2020.p01-14
Fitriansyah A, Amir H, Elvinawati E (2021) Karakterisasi adsorben karbon aktif dari sabut pinang (Areca catechu) terhadap kapasitas adsorpsi zat warna indigosol blue 04-B. Alotrop 5(1):42–54
Habila MA, Al Othman ZA, El-Toni AM, Labis JP, Li X, Zhang F, Soylak M (2016) Mercaptobenzothiazole-functionalized magnetic carbon nanospheres of type Fe3O4@SiO2@C for the preconcentration of nickel, copper and lead prior to their determination by ICP-MS. Microchim Acta 183(8):2377–2384. https://doi.org/10.1007/s00604-016-1880-x
Irawati H, Aprilita NH, Sugiharto E (2018) Adsorpsi zat warna kristal violet menggunakan limbah kulit singkong (Manihot esculenta). Bimipa 25(1):17–31
Jiang F, Dinh DM, Hsieh Y. Lo. (2017) Adsorption and desorption of cationic malachite green dye on cellulose nanofibril aerogels. Carbohyd Polym 173:286–294. https://doi.org/10.1016/j.carbpol.2017.05.097
Khoshnamvand N, Jafari A, Kamarehie B, Mohammadi A, Faraji M (2019) Removal of malachite green dye from aqueous solutions using zeolitic imidazole framework-8. Environ Process 6(3):757–772. https://doi.org/10.1007/s40710-019-00384-9
Kurniawan A, Nizar M, Rijal M, Bagas R, Setyarsih W (2014) Studi pengaruh variasi suhu kalsinasi terhadap kekerasan bentuk morfologi, dan analisis porositas nanokomposit Cao/Sio2 untuk aplikasi bahan biomaterial. J Penelit Fis Dan Apl (JPFA) 4(2):22
Landin-Sandoval VJ, Mendoza-Castillo DI, Seliem MK, Mobarak M, Villanueva-Mejia F, Bonilla-Petriciolet A, Navarro-Santos P, Reynel-Ávila HE (2021) Physicochemical analysis of multilayer adsorption mechanism of anionic dyes on lignocellulosic biomasses via statistical physics and density functional theory. J Mol Liquids. https://doi.org/10.1016/j.molliq.2020.114511
Larosa C, Salerno M, de Lima JS, Merijs Meri R, da Silva MF, de Carvalho LB, Converti A (2018) Characterisation of bare and tannase-loaded calcium alginate beads by microscopic, thermogravimetric, FTIR and XRD analyses. Int J Biol Macromol 115:900–906. https://doi.org/10.1016/j.ijbiomac.2018.04.138
Leng L, Yuan X, Zeng G, Shao J, Chen X, Wu Z, Wang H, Peng X (2015) Surface characterization of rice husk bio-char produced by liquefaction and application for cationic dye (Malachite green) adsorption. Fuel 155:77–85. https://doi.org/10.1016/j.fuel.2015.04.019
Li N, Yue Q, Gao B, Xu X, Kan Y, Zhao P (2018) Magnetic graphene oxide functionalized by poly dimethyl diallyl ammonium chloride for efficient removal of Cr(VI). J Taiwan Inst Chem Eng 91:499–506. https://doi.org/10.1016/j.jtice.2018.05.028
Liang SX, Jia Z, Zhang WC, Wang WM, Zhang LC (2017) Rapid malachite green degradation using Fe73.5Si13.5B9Cu1Nb3 metallic glass for activation of persulfate under UV–Vis light. Mater Des 119:244–253
Mahreni M, Yuli R (2020) A review on Metal-Organic Framework (MOF): synthesis and solid catalyst applications. Proc Eng Sci Ser (ESS) 1(1):638–645
Manuja A, Kumar S, Dilbaghi N, Hanjana G, Chopra M, Kaur H, Kumar R, Manuja BK, Singh SK, Yadav SC (2014) Quinapyramine sulfate-loaded sodium alginat nanoparticles show enhanced trypanocidal activity. Nanomedicine 9:1625–1634
Marsen, Alimano dan Syafila Mindriany. (2014). Adsorbent size reduction to enlarge its pore diameter in effort to improve used cooking oil adsorption efficiency. Jurnal Teknik Lingkungan. Volume 20 Nomor 2, Oktober, pp. 173–182.
Neolaka YAB, Supriyanto G, Kusuma HS (2019) Synthesis and characterization of natural zeolite with ordered ion imprinted polymer structures (IIP@AFINZ) for selective Cr(VI) adsorption from aqueous solution. Moroccan J Chem 7(1):194–210
Neolaka YAB, Supriyanto G, Darmokoesoemo H, Kusuma HS (2018) Characterization, kinetic, and isotherm data for Cr(VI) removal from aqueous solution by Cr(VI)-imprinted poly(4-VP-co-MMA) supported on activated Indonesia (Ende-Flores) natural zeolite structure. Data Brief 17:969–979. https://doi.org/10.1016/j.dib.2018.01.076
Noreen S, Khalid U, Ibrahim SM, Javed T, Ghani A, Naz S, Iqbal M (2020) ZnO, MgO and FeO adsorption efficiencies for direct sky blue dye: equilibrium, kinetics and thermodynamics studies. J Mater Res Technol 9(3):5881–5893. https://doi.org/10.1016/j.jmrt.2020.03.115
Pan X, Zuo G, Su T, Cheng S, Gu Y, Qi X, Dong W (2019) Polycarboxylic magnetic polydopamine sub-microspheres for effective adsorption of malachite green. Colloids Surf A Physicochem Eng Aspects 560:106–113. https://doi.org/10.1016/j.colsurfa.2018.10.014
Patidar D, Goyal S (2021) Adsorptive removal of malachite green dye from aqueos solution using a non carbon adsorbent: equilibrium, kinetics and thermodynamics. J Adv Sci Res 1(2):19–23
Raval NP, Shah PU, Shah NK (2016) Nanoparticles loaded biopolymer as effective adsorbent for adsorptive removal of malachite green from aqueous solution. Water Conserv Sci Eng 1(1):69–81. https://doi.org/10.1007/s41101-016-0004-0
Rehman Shah HU, Ahmad K, Naseem HA, Parveen S, Ashfaq M, Rauf A, Aziz T (2021) Water stable graphene oxide metal-organic frameworks composite (ZIF-67@GO) for efficient removal of malachite green from water. Food Chem Toxicol 154(March):112312. https://doi.org/10.1016/j.fct.2021.112312
Sayğılı H, Güzel F (2018) Usability of activated carbon with well-developed mesoporous structure for the decontamination of malachite green from aquatic environments: kinetic, equilibrium and regeneration studies. J Porous Mater 25(2):477–488. https://doi.org/10.1007/s10934-017-0459-1
Shi Z, Xu C, Guan H, Li L, Fan L, Wang Y, Liu L, Meng Q, Zhang R (2018) Magnetic Metal Organic Frameworks (MOFs) composite for removal of lead and malachite green in wastewater. Colloids urf A: Physicochem Eng Aspects 539:382–390. https://doi.org/10.1016/j.colsurfa.2017.12.043
Tang J, Zhang YF, Liu Y, Li Y, Hu H (2020) Efficient ion-enhanced adsorption of congo red on polyacrolein from aqueous solution: experiments, characterization and mechanism studies. Sep Purif Technol 252(April):117445. https://doi.org/10.1016/j.seppur.2020.117445
Wu Y, Yang F, Liu X, Tan G, Xiao D (2018) Fabrication of N, P-codoped reduced graphene oxide and its application for organic dye removal. Appl Surf Sci 435:281–289. https://doi.org/10.1016/j.apsusc.2017.10.118
Zadvarzi SB, Khavarpour M, Vahdat SM, Baghbanian SM, Rad AS (2021) Synthesis of Fe3O4@chitosan@ZIF-8 towards removal of malachite green from aqueous solution: theoretical and experimental studies. Int J Biol Macromol 168:428–441. https://doi.org/10.1016/j.ijbiomac.2020.12.067
Zhang T, Jin X, Owens G, Chen Z (2021) Remediation of malachite green in wastewater by ZIF-8@Fe/Ni nanoparticles based on adsorption and reduction. J Colloid Interface Sci 594:398–408. https://doi.org/10.1016/j.jcis.2021.03.065
Zhao X, Zheng M, Gao X, Zhang J, Wang E, Gao Z (2021) The application of MOFs-based materials for antibacterials adsorption. Coord Chem Rev 440:213970. https://doi.org/10.1016/j.ccr.2021.213970
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahreni, M., Ramadhan, R.R., Pramadhana, M.F. et al. Synthesis of Metal Organic Framework (MOF) based Ca-Alginate for adsorption of malachite green dye. Polym. Bull. 79, 11301–11315 (2022). https://doi.org/10.1007/s00289-022-04086-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04086-5