Abstract
New graft copolymer hydrogels based on sodium acrylate, acrylamide, and styryl-terminated poly(2-cyclopropyl-2-oxazoline) macromonomer (MM) were synthetized by free radical polymerization using N,N′-methylenebisacrylamide as cross-linker. The polymerization was carried out in water at 5 °C and was initiated by sodium peroxodisulfate/N,N,N′,N′-tetramethylethylenediamine. The MM and the hydrogels were characterized by NMR and FTIR spectroscopy. In the hydrogels, the sodium acrylate provided the sensitivity to changes in pH value while the MM provided sensitivity to temperature. In dependence of their composition, the bi-sensitive hydrogels showed conformational transitions with variation of temperature or pH value. This property was shown macroscopically as a hydrogel volume contraction or expansion as it was determined by swelling experiments in water at different pH values and temperatures. Due to phase separation within the hydrogels facilitated by the graft copolymer network structure, both sensitivities could be addressed individually by both triggers and defined swelling states could be addressed over a wide range by adjusting both temperature and pH.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Responsive polymeric materials are polymers that go through a phase transition in response to an external stimulus such as temperature, pH, magnetic field, electrical impulses, and light [1,2,3,4,5]. This type of material is being investigated for its potential applications in biomedicine, water management, in sensors and as actuators, among others [6, 7]. Currently, many polymers that respond with changes in their solubility in aqueous solution depending on the changes of temperature are been studied in the literature [2, 5, 8]. Among them, one of the most studied is poly(N-isopropylacrylamide) (PNiPAAm), since its minimum critical solution temperature (LCST) in water, which is the temperature at which the polymer changes from hydrophilic to hydrophobic, is close to the temperature of the human body [2, 5, 8, 9].
In recent years, the thermal sensitivity of some polyoxazolines has also been investigated, such as, for example, poly(isopropyloxazoline) (poly(iPrOxa)), poly(cyclopropyloxazoline) (poly(CPOxa)), and poly(ethyloxazoline) [4, 10,11,12]. Poly(CPOxa) presents a LCST value of approximately 30 °C [10] that is close to human body temperature, and therefore, this polymer could be used eventually in biomedical application. The cyclopropyloxazoline monomer (CPOxa) polymerizes extremely fast and does not present the crystallization phenomenon that poly(iPrOxa) has when it is heated at relatively high temperatures [13], which limits its practical application.
The synthesis of poly(2-oxazoline)s by ring-opening cationic polymerization proceeds in a ‘living’ form without termination or chain transfer reactions; thus, the molecular weight as well as the functionality of these polymers can be predetermined from the start of polymerization [14,15,16,17].
Hydrogels that exhibit pH sensitivity contain in their structure ionizable groups that can donate or accept protons (for example, carboxylic acids or amines) in response to changes in the pH value of the medium. When the pH of the medium changes, a change in the degree of ionization occurs. This results in a conformational change of the polymer and a change of hydrogel volume, and such pH responsive hydrogels find manifold application [2, 5, 8]. A prominent application of responsive hydrogels with intensive volume change and multiple responsivity is as actuator in microfluidic where the hydrogel can act as a valve opening and closing microfluidic channels upon a stimulus in the fluid, e.g., changes in pH or temperature [6, 18,19,20].
In this work, bi-sensitive hydrogels are realized by the concept of graft copolymer gels, using a temperature-sensitive poly(cyclopropyloxazoline) macromonomer with a styrene polymerizable group and a sodium acrylate/acrylamide pH-sensitive gel frame. The composition was adjusted in a way that both sensitivities had been realized and could be addressed individually with sufficient volume phase transition temperature for a technical application, e.g., as actuator valves in microfluidics.
Experimental
Methods
The FTIR-ATR measurements were taken using a Bruker Vertex 80v with MCT detector in the range of 600 to 4000 cm−1 with a resolution of 4 cm−1. Hundred scans were accumulated for each measurement.
UV–Vis turbidity measurements have been taken on 1 wt% MM concentration in distilled water using Helios Gamma UV–Vis spectrometer from Thermo-Electron Corporation. Transmittance was measured at 550 nm, varying temperature between 15 and 65 °C.
NMR spectra were recorded on a Bruker Avance III 500 NMR spectrometer operating at 500.13 MHz for 1H and at 125.75 MHz for 13C. CD3OD (δ(1H) = 3.31 ppm; δ(13C) = 49.0 ppm) was used as a solvent and internal reference. For high-resolution magic angle spinning (HR-MAS) measurements, the PTFE insert of the ZrO2 rotor (4 mm outer diameter, 50 μl insert volume) was filled with small pieces of dry hydrogel (~ 2 mg). After addition of D2O and about 30 min swelling time, the sample was measured with a HR-MAS probe (νR = 4000 Hz). The 1H NMR spectra were referenced on the HDO signal (4.72 ppm).
Changes in pH were measured with a pH meter and conductimeter (Seven Multi, Mettler Toledo). Temperature control was achieved with a thermostat (Fischer 604) with an accuracy of 0.1 °C. The pH values were adjusted with respective buffer solutions.
Materials
Chloromethylstyrene (CMS) (Aldrich, mixture of isomers 70 mol% meta and 30 mol% para) was bi-distilled before use. 2-Cyclopropyl-2-oxazoline was synthesized by a method described in the literature [11, 21] and was distilled over calcium hydride before use. All other substances and solvents were of high purity and were used as received.
Cyclopropyloxazoline synthesis (CPOxa) [11, 21]
In a round-bottomed flask, 67.35 g of cyclopropanecarbonitrile was reacted with 61 g of ethanolamine, in the presence of 6.5 g of cadmium acetate dehydrate. The ethanolamine was added dropwise. The reaction was maintained at 125 °C under a nitrogen atmosphere and under reflux for 20 h. The synthetized CPOxa was distilled (yield 85%), and structure and purity were confirmed by 1H NMR spectroscopy and fit literature data.
Macromonomer synthesis (MM)
The MM was synthesized via ring-opening cationic polymerization of CPOxa initiated by CMS in the presence of sodium iodide [17]. The polymerization was carried out in acetonitrile at 78 °C. In a reactor, 13 g (117 mmol) CPOxa and 1.54 g (9.20 mmol) NaI were dissolved under nitrogen in 35 mL acetonitrile; then, 0.7 g (4.59 mmol) of chloromethylstyrene was added. The reaction mixture was heated for 7 h. After this time, the polymerization was terminated with a solution of KOH in methanol. The obtained MM was purified by reprecipitation (yield 97%). The degree of polymerization (DP) of the styryl-terminated MM was 25 (Mn = 2910 g/mol) as determined by 1H NMR spectroscopy.
1H NMR (CD3OD): δ 7.5–7.1 (HAr), 6.74 (=CH–), 5.80 and 5.25 (=CH2), 4.0–3.4 (–CH2–N(R)–CH2–), 2.1–1.6 (CHCycl), 0.85 ppm (CH2,Cycl).
13C NMR (CD3OD): 176.7–176.1 (C=O), 48–45 (–CH2–N(R)–CH2–), 11.9 and 11.7 (CHCycl), 9.0 and 8.8 ppm (CH2,Cycl).
Hydrogel synthesis (HG)
In a typical procedure, 0.167 g of MM was dissolved in water (pH 6.7) and 0.133 g of sodium acrylate (NaAc), 0.2 g of acrylamide (AAm), and 0.041 g of N,N′-methylenebisacrylamide (BIS) were added at 5 °C under nitrogen atmosphere. This mixture was stirred for 40 min under nitrogen, and then, a mixture of 0.013 g of sodium peroxodisulfate and 0.02 g of N,N,N′,N′-tetramethylethylenediamine (TEMED), previously dissolved with 1 mL of water, was added. The reaction mixture was stirred and transferred by means of a syringe to a glass tube mold (NMR tube, inner diameter: about 4 mm) that was keep at 5 °C. The hydrogel was obtained in 15 min and was maintained in the mold for 24 h. After removing the hydrogel from the mold, it was washed with distilled water. It was first put in 500 mL of ethanol for 24 h and then in 1 L of distilled water, changing the water every 4 h. Finally, the gels were dried at 40 °C under vacuum until weight becomes constant. For the swelling experiments, the hydrogel tube after removal from the glass tube was cut in disk-like samples of 3–6 mm height and those were washed and tried to constant weight as described above. The experimental details and results are summarized in Table 1.
1H NMR (D2O): δ 4.1–3.2 (–CH2–N(R)–CH2–), 2.6–2.0 (backbone-CH of AAm, NaAc and MM units), 2.1–1.2 (backbone-CH2 of AAm, NaAc and MM units and CHCycl), 0.84 ppm (CH2,Cycl).
Hydrogel sensitivity to pH value and temperature
The sensitivity of the hydrogel was determined on the above-described disk-like samples by measuring its contraction or expansion in aqueous medium in dependence on temperature or pH value. For measurements of contraction or expansion of the hydrogel, it was assumed that the weight of the hydrogel is proportional to its volume and the weight was measured.
First, the weight of the swollen hydrogel disk at 25 °C in equilibrium with water (24 h) was determined at pH = 6, and then after a gradually heating process, the new weight of the hydrogel was measured in other temperature and it was related to the initial weight. The measurements were taken after 4 h equilibration time.
Similarly, the pH sensitivity was determined by changing the pH of the medium (emerging it in the respective buffer solution) and measuring the new weight of the hydrogel at each different pH value and relating it to the initial weight at pH 2. The measurements were taken after 2 h equilibration time at 20 °C.
Results and discussion
Synthesis and characterization of macromonomer MM
The polyoxazoline macromonomer (MM) was synthesized introducing the styryl function through the initiator. 2-Cyclopropyl-2-oxazoline (CPOxa) was polymerized by ring-opening cationic polymerization initiated by chloromethylstyrene, using sodium iodide as coinitiator for halogen exchange for the reaction (Scheme 1). It is known from the literature that this polymerization of 2-oxazolines proceeds by a living mechanism without termination or chain transfer reactions [10, 12, 15], and thus, polymers can be obtained with predetermined degree of polymerization and functionality. The yield of the reaction was 97%.
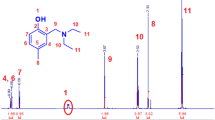
The structure of MM was confirmed by 1H and 13C NMR spectroscopy (Figs. 1a and 2, respectively, and Fig. SI-1). A degree of polymerization DP = 25 was calculated from 1H NMR signal intensities of the styryl group (Ha, Ha′, and Hb) and of the repeating unit (Hf) which fits the predetermined DP from the used monomer to chloromethylstyrene ratio.
The characterization of the temperature sensitivity of the macromonomer (MM) was performed by turbidimetric analysis by UV–Vis spectroscopy (Fig. SI-2). It was found that the macromonomer had a cloud point, which is related to the lower critical solution temperature (LCST), at relatively low 18 °C compared to the 30 °C from the literature [10]. This is in agreement with its low molecular weight and with the fact that the hydrophobic styrene group at the chain end further decreases the cloud point of MM.
Synthesis and structural characterization of hydrogels
The hydrogels were obtained by radical copolymerization of the macromonomer MM, sodium acrylate, and acrylamide in the presence of N,N′-methylenebisacrylamide as the cross-linker. Acrylamide was added in the composition to reduce the content of ionic groups in the pH-sensitive part of the hydrogel which allows to control the pH depended degree of swelling in a technically interesting range [19]. The polymerization was initiated by sodium peroxodisulfate and TEMED, a well-known radical redox initiating system [23], at a temperature of 5 °C in water (Scheme 2). At these reaction conditions, it was avoided to exceed the transition temperature of the macromonomer and the complete dissolution of the reactants was allowed. The reaction occurred in a time of 15 min, with reaction yields between 83 and 94% (Table 1). Completely transparent hydrogels were obtained. The use of a poly(2-cyclopropyl-2-oxazoline) macromonomer (MM) leads to a so-called graft copolymer hydrogel structure, with tangling chains of poly(CPOxa) with DP of 25 (Scheme 2). This specific hydrogel structure should ensure that the hydrogels retain the temperature sensitivity due to the possibility to build a certain phase-separated structure with a poly(CPOxa) phase. The positive effect of keeping multisensitivity in graft copolymers hydrogels has been shown recently for even tetra-sensitive hydrogels [22].
The hydrogels were characterized by FTIR spectroscopy (Fig. SI-3). The primary amide bands at 3450–3400 cm−1 (νas NH2) and 3250–3180 cm−1 (νs NH2) and the band at 1685–1680 cm−1 (ν C=O) result from the acrylamide unit. Bands at 1560 and 1410–1400 cm−1 are the stretching vibrations of carboxylate group of the acrylate unit. A band at 1710 cm−1 (ν C=O) results from H-bond-associated carboxylic acid groups and indicates not fully neutralized acrylate. The bands of MM are superposed by bands of the acrylamide and sodium acrylate units in the polymer.
The NMR characterization of the hydrogels was carried out on samples swollen in D2O using the high-resolution magic angle spinning (HR-MAS) technique. Figure 1b depicts the 1H NMR spectrum of HG2 synthesized from a feed composition of 1.8 mol% MM, 42.7 mol% NaAc, and 48.5 mol% AAm and a cross-linked content of 7.0 mol%. The macromonomer (DP = 25) results in the typical signal pattern of the NCH2 protons (Hd) in the 4.1–3.2 ppm region (comp. Fig. 1a). The methylene protons (Hg) of the cyclopropyl unit can be well identified by their intense signal at 0.84 ppm, whereas the signals of the methine proton Hf at ~ 1.9 ppm are overlapped by the CH2 signal groups of the NaAc and AAm monomers in the copolymer. Also the methine proton signals of both acryl comonomers overlap in the 2.6–2.0 ppm region. This makes it impossible to determine quantitatively the content of the comonomers in the final hydrogel. The short cross-linker (Hm–Ho) results in very broad signals and could not be detected. Generally, the chain mobility decreases with decreasing distance between cross-linking points, i.e., increasing BIS content, and the line width of the signals increases as observed for HG3 and HG4 (Fig. SI-4). It should be mentioned that the hydrogels still contain TEMED due to salt formation with acrylic acid units in the copolymer as can be concluded from two singlets at 3.56 (NCH2) and 2.94 ppm (NCH3).
Regarding the reactivity ratios for the MM and the monomers NaAc and AAm, there are no data in the literature, but they can be approximated when only the styryl chain end of the macromonomer is considered. The reactivity ratios of styrene and sodium acrylate are similar (r1 = 0.82 and r2 = 0.77, [24]), whereas acrylamide is favorably incorporated in comparison with styrene. Still, the reactivity ratios depend also on the reaction medium (solvent, other monomers, [25]). It is possible that the macromonomer MM, due to its large size and therefore low diffusibility, polymerizes more slowly than the monomers NaAc and AAm which would produce a polymeric structure where the MM is not be homogeneously distributed in the hydrogel, leading to a more gradient structure (see discussion below).
pH sensitivity of the hydrogels
The sensitivity of the hydrogels to pH changes was determined from the experimentally determined degree of swelling Q at different pH values. Figure 3 depicts the dependency of Q from pH value for the five hydrogels under investigation. The swelling in acid media is low, but they swell with increasing pH value, having a maximum degree of swelling at pH ~ 8 followed by slightly decreasing swelling up to pH = 10.
At low pH values (pH = 2 or 3), all hydrogels show a strong contraction, possibly due to the formation of hydrogen bonds between the acid and amide groups [26]. At pH = 8, all the acid groups of the hydrogels reach a complete dissociation forming highly polar sodium carboxylate groups, which allows them to absorb the greatest amount of water. However, at pH = 10, possibly due to a higher ionic content, the hydrogel undergoes a slight contraction. This phenomenon was also observed in previous investigations of hydrogels made with 2-oxazoline macromonomers [17].
The hydrogels HG1 to HG4 have similar molar percentages of acrylamide and of sodium acrylate (Table 1), so that from pH > 6 they had similar degrees of swelling (Q) because the predominant factor in the expansion of the hydrogel becomes the degree of cross-linking.
Hydrogel HG5, having higher content of sodium acrylate (Table 1), has at pH > 4 greater expansion than the other hydrogels due to the greater mutual electrostatic repulsion between the carboxylate groups.
Temperature sensitivity of the hydrogels
The temperature sensitivity of the hydrogels was determined in the temperature range of 5 to 50 °C. Figure 4 shows the graphs of the degree of swelling (Q) against the temperature of hydrogels at pH value of 6, a pH which allows a high degree of swelling of the hydrogels. At low MM content, at a specific temperature the degree of swelling decreases stepwise to a lower level since at higher temperatures the temperature-sensitive poly(CPOxa) side chains tend to contract due to the conformational transition phenomenon (LCST) of the macromonomer contained therein. Hydrogels at low temperatures, such as 10 °C, expand completely because this temperature is lower than the cloud point of the MM macromonomer (18 °C). Interestingly, as the concentration of MM in the hydrogels increases, the conformational transition occurs with greater intensity and the degree of swelling decreases rather constantly with increasing temperature. In addition, the main stepwise transition is shifted to higher temperatures, close to the physiologically interesting range of 30 to 35 °C. For example, HG4 has a main volume phase transition temperature (VPTT) of approximately 35 °C at pH 6, while cloud point of MM is only 18 °C. The VPTT of the hydrogels is higher than the cloud point of the macromonomer because the former contains hydrophilic comonomers such as sodium acrylate and acrylamide close to the poly(CPOxa) chains which raise this value [5, 8].
Following in more detail the degree of swelling of the hydrogel HG4 with the highest MM content in dependence of temperature at different pH (Fig. 5), in practically all the samples a double thermal transition (double “drop” of the degree of swelling) is observed, being most prominent at pH 6. This could be due to a non-homogeneous, gradient structure of this hydrogel with MM-enriched areas built at the end of the network formation. In such a case, the conformational transition within the hydrogel occurs in two stages: first, the poly(CPOxa) MM-rich areas collapse close to the cloud point temperature of the original MM, and later, the collapse of the full hydrogel with also the more diluted MM chains is observed at a higher temperature due to the polar surrounding. Similar results are found for HG3.
Furthermore, it can be concluded from the results in Fig. 5 that in our graft copolymer hydrogels the pH and temperature sensitivities are strongly intercorrelated and thus, both triggers can be used to set a specific state of swelling. Still, the pH sensitivity dominates. At pH ≥ 6, the sodium carboxylate groups of the main chain are in a high degree of dissociation and cause the hydrogel to expand and swell due to the internal repulsions of the polar groups. At this state, also the temperature sensitivity can be very pronounced. At pH = 3, a fully collapsed state of the hydrogel is reached and then the temperature sensitivity is strongly suppressed.
Conclusion
New graft copolymer hydrogels with sensitivity to both temperature and pH have been synthesized by radical copolymerization of NaAc, AAm, and poly(2-cyclopropyl-2-oxazoline) macromonomers (MM) with a styryl end group in the presence of a bisacrylamide.
The macromonomer MM synthesis proceeded via cationic polymerization of the 2-cyclopropyl-2-oxazoline using chloromethylstyrene as initiating moiety under mild conditions at relative low reaction temperature, and the MM had a degree of polymerization of 25 and a cloud point of 18 °C.
Upon incorporation of the MM into the hydrophilic graft copolymer hydrogel structure and optimization of the composition, the temperature sensitivity could be maintained and the volume phase transition temperature (VPTT) of the gels could be shifted into a more technically interesting area between 30 and 37 °C when the MM content was sufficiently high. The pH-dependent contraction and swelling of HG are caused by the different dissociation state of the sodium acrylate groups and potential hydrogen bonding of the acrylamide groups with the highest degree of swelling Q observed at pH 6. The degree of swelling toward both triggers is significantly high and intercorrelated and can be fine-tuned by both temperature and pH while keeping a high gel integrity. This makes these bi-sensitive responsive hydrogels interesting for applications in biomedicine, in sensorics, and especially as actuators in microfluidics.
Supplementary material
Additional NMR and FTIR spectra of the hydrogels as well as transmittance change with temperature of the MM can be found in supplemental material.
References
Stuart M, Huck WT, Genzer J, Müller M, Ober C, Stamm M, Sukhorukov GB, Szleifer I, Tsukruk V, Urban S, Zauscher S, Luzinov I, Minko S, Winnik F (2010) Emerging applications of stimuli-responsive polymer materials. Nat Mater 9:101–113. https://doi.org/10.1038/nmat2614
Koetting MC, Peters JT, Steichen SD, Peppas NA (2015) Stimulus-responsive hydrogels: theory, modern advances, and applications. Mater Sci Eng R Rep 93:1–49. https://doi.org/10.1016/j.mser.2015.04.001
Gil E, Hudson SM (2004) Stimuli-reponsive polymers and their bioconjugates. Prog Polym Sci 29:1173–1222. https://doi.org/10.1016/j.progpolymsci.2004.08.003
Weber C, Hoogenboom R, Schubert U (2012) Temperature responsive bio-compatible polymers based on poly (ethylene oxide) and poly(2-oxazoline)s. Prog Polym Sci 37:686–714. https://doi.org/10.1016/j.progpolymsci.2011.10.002
Schild HG (1992) Poly(N-isopropylacrylamide): experiment, theory and application. Prog Polym Sci 17:63–249. https://doi.org/10.1016/0079-6700(92)90023-R
Richter A (2009) Hydrogels for actuators. In: Gerlach G, Arndt KF (eds) Hydrogel sensors and actuators. Chapter 7. Springer, Berlin
Langer R, Tirrell DA (2004) Designing materials for biology and medicine. Nature 428:487–492. https://doi.org/10.1038/nature02388
Dimitrov I, Trzebicka B, Müller A, Dworak A, Tsvetanov C (2007) Thermosensitive water-soluble copolymers with doubly responsive reversibly interacting entities. Prog Polym Sci 32:1275–1343. https://doi.org/10.1016/j.progpolymsci.2007.07.001
Rueda JC, Zschoche S, Komber H, Schmaljohann D, Voit B (2005) Synthesis and characterization of thermoresponsive graft copolymers of NIPAAm and 2-alkyl-2-oxazolines by the “grafting from” method. Macromolecules 38:7330–7336. https://doi.org/10.1021/ma050570p
Park JS, Kataoka K (2007) Comprehensive and accurate control of thermosensitivity of poly(2-alkyl-2-oxazoline)s via well-defined gradient or random copolymerization. Macromolecules 40:3599–3609. https://doi.org/10.1021/ma0701181
Bloksma MM, Weber C, Perevyazko IY, Kuse A, Baumgärtel A, Vollrath A, Hoogenboom R, Schubert US (2011) Poly(2-cyclopropyl-2-oxazoline): from rate acceleration by cyclopropyl to thermoresponsive properties. Macromolecules 44:4057–4064. https://doi.org/10.1021/ma200514n
Hoogenboom R, Schlaad H (2017) Thermoresponsive poly(2-oxazoline)s, polypeptoids, and polypeptides. Polym Chem 8:24–40. https://doi.org/10.1039/C6PY01320A
Diehl C, Cernoch P, Zenke I, Runge H, Pitschke R, Hartmann J, Tierschc B, Schlaad H (2010) Mechanistic study of the phase separation/crystallization process of poly(2-isopropyl-2-oxazoline) in hot water. Soft Matter 6:3784–3788. https://doi.org/10.1039/c0sm00114g
Aoi K, Okada M (1996) Polymerization of oxazolines. Prog Polym Sci 21:151–208. https://doi.org/10.1016/0079-6700(95)00020-8
Hoogenboom R, Fijten MWM, Schubert US (2004) Parallel kinetic investigation of 2-oxazoline polymerizations with different initiators as basis for designed copolymer synthesis. J Polym Sci Part A Polym Chem 42:1830–1840. https://doi.org/10.1002/pola.20024
Verbraeken B, Monnery B, Lava K, Hoogenboom R (2017) The chemistry of poly(2-oxazoline)s. Eur Polym J 88:451–469. https://doi.org/10.1016/j.eurpolymj.2016.11.016
Rueda JC, Campos E, Komber H, Zschoche S, Häussler L, Voit B (2014) Synthesis and characterization of new pH- and thermo-responsive hydrogels based on N-isopropylacrylamide and 2-oxazolines. Des Monomers Polym 17:208–216. https://doi.org/10.1080/15685551.2013.840471
Kuckling D (2009) Responsive hydrogel layers—from synthesis to applications. Colloid Polym Sci 287:881–891. https://doi.org/10.1007/s00396-009-2060-x
Krause AT, Zschoche Z, Rohn M, Hempel C, Richter A, Appelhans D, Voit B (2016) Swelling behavior of bisensitive IPNs for microfluidic applications. Soft Matter 12:5529–5536. https://doi.org/10.1039/C6SM00720A
Richter A, Kuckling D, Howitz S, Gehring T, Arndt KF (2003) Electronically controllable microvalves based on smart hydrogels: magnitudes and potential applications. J Microelectromech Syst 12:748–753. https://doi.org/10.1109/JMEMS.2003.817898
Gräfe D, Erdmann T, Richter A, Appelhans D, Voit B (2017) Tetra-sensitive graft copolymer gels as chemomechanical valve. ACS Appl Mater Interfaces 9:7565–7576. https://doi.org/10.1021/acsami.6b14931
Witte H, Seeliger W (1974) Cyclische Imidsäureester aus Nitrilen und Aminoalkoholen. Liebigs Ann Chem. https://doi.org/10.1002/jlac.197419740615
Feng XD, Guo XQ, Qiu KY (1988) Study of the initiation mechanism of the vinyl polymerization with the system persulfate/N, N, N′, N′-tetramethylethylendiamine. Makromol Chemie 189:77–83. https://doi.org/10.1002/macp.1988.021890108
Nuño-DonLucas S, Rhoton AI, Crona-Galvan S, Puig JE, Kaler EW (1993) Emulsion copolymerization of styrene and sodium acrylate. Polym Bull 30:207–214. https://doi.org/10.1007/BF00296851
Minsk LM, Kotlarchik C, Meyer GN (1973) Copolymerization of acrylamide and styrene II. Reactivity ratios with unperturbed acrylamide. J Polym Sci Polym Chem Ed 11:3037–3130. https://doi.org/10.1002/pol.1973.170111201
Weber C, Becer CR, Guenther W, Hoogenboom R, Schubert US (2019) Dual responsive methacrylic acid and oligo(2-ethyl-2-oxazoline) containing graft copolymers. Macromolecules 43:160–167. https://doi.org/10.1021/ma902014q
Acknowledgements
The authors thank Dr. M. Malanin (IPF Dresden) for the ATR-FTIR measurements on the hydrogels. J. C. R. and C. S. gratefully acknowledge the Deutschen Akademischen Austauschdienst (DAAD), the Pontifical Catholic University of Peru (PUCP), and the National Council of Science, Technology and Technological Innovation of Peru (CONCYTEC) for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rueda, J.C., Suárez, C., Komber, H. et al. Synthesis and characterization of pH- and thermo-responsive hydrogels based on poly(2-cyclopropyl-2-oxazoline) macromonomer, sodium acrylate, and acrylamide. Polym. Bull. 77, 5553–5565 (2020). https://doi.org/10.1007/s00289-019-03034-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-019-03034-0