Abstract
Sensitivity toward humidity, response and recovery time, and reproducibility behaviors of a novel copolymer sensor, obtained by oxidative chemical polymerization of o-phenylenediamine and aniline using D-camphor-10-sulfonic acid dopant, were reported. It is a novel copolymer, and there are very few studies available concerning its synthesis and humidity sensing applications. Humidity-sensitive properties of the copolymer were tested by measuring the resistance of the fabricated films, and good humidity sensitivity was obtained. Moreover, a decrease in resistance with the increase in humidity was observed. Compared to polyaniline (PANI), the humidity sensing property of the copolymer was found to show quicker response and recovery time, higher reproducibility, and higher sensitivity. However, PANI alone exhibited relatively poor sensing property. The response time for the copolymer films at the relative humidity ranging from 11 to 97% was about 38 s, and the corresponding recovery time was about 11 s. The value of sensitivity was about 10. These results indicate excellent sensing character of the new type of copolymer humidity sensors. The obtained sensor is expected to exhibit good humidity sensing behavior in moisture-containing environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Recently, humidity sensors have attracted progressively more attention on account of their extensive usage in industrial, technology, and laboratory fields and also in everyday life [1,2,3,4,5]. Among different types of humidity sensors, polymer humidity sensors make full use of their strength and show widespread applications especially when they are compared with the ceramic humidity sensors. This is basically attributed to their good flexible properties, smaller-size design, and room temperature operation and flexibility [6, 7]. Therefore, design and fabrication of polymer sensors which possess good sensitivity, low hysteresis, quick response time, and excellent stability are significantly important. In order to promote the sensing characteristics, a general route involves exploration of the fabrication of nanostructured thin films because of high surface area which promotes the process of adsorbing water molecules [8,9,10].
Polyaniline (PANI) serves as a significantly important conducting polymer enjoying various potential usages in anticorrosion and antistatic coatings, batteries, and sensors [11,12,13,14] owing to its desirable characteristics. However, currently, copolymer of aniline (An) and its corresponding derivatives have displayed their charm and turned out to be in spotlight [15, 16]. o-Phenylenediamine (oPD) and An copolymer have been successfully fabricated by electropolymerization and oxidative chemical polymerization [17, 18]. Nevertheless, owing to limited electrode surface area and non-uniform current density, it is difficult to obtain large-area and uniform polymer films by electropolymerization [19]. Therefore, oxidative chemical polymerization method has become significantly more important than before.
Jain et al. [20] reported the behavior of humidity sensors with PANI-based conducting polymers doped with weak acidic dopants. It was observed that over the major range of relative humidity (RH, between 20 and 95%), behavior of these sensors was almost linear and high sensitivity was obtained for PANI doped with D-camphor-10-sulfonic acid (CSA). Poly(o-anisidine) (PoA) and poly(o-phenylenediamine) (PoPD), which are derivatives of PANI, were also reported to be humidity sensitive by Kulkarni et al. and Tonosaki et al. [21, 22]. However, a lot more systematic explorations are still demanded to investigate substituted anilines and to explore the possibility of utilizing them as an alternative to PANI for humidity sensors [23].
In the present study, a novel copolymer sensor was prepared from oPD and An monomers, and CSA dopant. The structure of this new copolymer was characterized in detail by various techniques. The humidity sensing behavior of this novel copolymer was examined, and the results revealed rapid and highly sensitive detection of humidity. To the best of our knowledge, the humidity sensing property of poly(o-phenylenediamine-co-aniline) copolymer has rarely been reported. Therefore, this study utilized a novel perspective to design highly sensitive humidity sensor, which has potential in further research for humidity sensing.
Experimental section
Materials
The oPD (98%), An (98%) and CSA (99%) were purchased from J&K Scientific Ltd. An was distilled before use and stored at 4 °C. Ammonium persulfate, LiCl, K2SO4 and N,N-dimethylformamide were obtained from Beijing Chemical Plant. All these reagents were used as obtained in analytical grade. The de-ionized water was used throughout the present study.
Fabrication of humidity sensor of PANI
0.186 g of distilled An monomer was put together with 10 mL CSA aqueous solution (0.1 M) and stirred for 0.5 h to obtain a mixed solution. Ten milliliter ammonium persulfate aqueous solution (0.2 M) was put into the above solution quickly. A substrate (10 mm × 6 mm × 0.5 mm) with five pairs of Au interdigital electrodes was immersed into the solution. The reaction was kept unstirred for additional 24 h. Finally, a uniform film was formed on the interdigital electrode substrate. The obtained substrate was washed by using de-ionized water and dried in the air.
Fabrication of humidity sensor of oPD/An copolymer
Typical synthetic processes of oPD/An copolymer nanoparticles were as follows: 0.093 g of distilled An monomer and 0.108 g oPD were mixed with 10 mL CSA aqueous solution (0.1 M) under stirring which lasted 0.5 h. A clear solution was then obtained. Ten milliliter ammonium persulfate aqueous solution (0.2 M) was added to the solution at room temperature. The interdigital electrode substrate was immersed into the mixture. The reaction was kept unstirred for additional 24 h. Finally, a uniform film was formed on the interdigital electrode substrate. The substrate was washed by using de-ionized water and was dried at room temperature.
Instruments and characterization
Ultraviolet–visible (UV–Vis) absorption spectra were obtained on a Shimadzu UV-3100 spectrometer. The slit width was 2 nm. Fourier transform infrared (FTIR) spectra were recorded on a Nicolet 5PC spectrometer by using KBr powder-pressed pellet samples. Raman spectra of the polymer films were gained with a Renishaw 1000 model armed with a holographic notch filter and a CCD detector. Air-cooled argon ion laser (Spectra-Physics Model 163-C4260) with the radiation of 514.5 nm was used as the excitation source. Scanning electron microscopy (SEM) was obtained through the microscope of SHIMADZU SSX-550. Thermogravimetric analysis (TGA) test was performed on Pyris 6 Thermogravimetric Analyzer (Perkin-Elmer). The heating rate was 10 °C min−1 up to 900 °C by using alumina crucibles under constant N2 flow. The RH was gained with saturated aqueous solutions of LiCl (11%) and K2SO4 (97%), separately, by using a homemade gas-sensing setup [24].
Results and discussion
Figure 1 shows the FTIR spectra of CSA-doped PANI and CSA-doped oPD/An copolymers. Figure 1a exhibits the characteristic peaks of PANI being consistent with the literature report [25]. The bands at 1577 and 1510 cm−1 correspond to the stretching vibration modes of C=N and C=C in the phenazine ring of PANI, respectively. The band at 1305 cm−1 is attributed to the C–N stretching vibration. The band at 1147 cm−1 is ascribed to electronic-like absorption peak of N=Q=N (where Q represents the quinoid ring). Figure 1b shows two high wave number bands at 3315 and 3147 cm−1, assigned to the typical N–H stretching of the –NH– and –NH2 groups in oPD units, respectively. The band at 1237 cm−1 corresponds to the C–N stretching vibration in the benzenoid units. Two bands at 1124 and 619 cm−1 represent the in plane and out of plane C–H bending motions of benzenoid rings, respectively [26]. Appearance of the IR bands proved the successful fabrication of the oPD/An copolymers.
The UV–Vis spectra of CSA-doped PANI and CSA-doped oPD/An copolymers were obtained by dispersing each of them in N,N-dimethylformamide, separately. The obtained spectra are shown in Fig. 2. The major absorption bands at about 330 and 597 nm in Fig. 2a are assigned to the π–π* electron transition of benzenoid units and the exciton-like transition (the conducting form) in quinoid diimino units of PANI [27]. Figure 2b exhibits the spectra of CSA-doped oPD/An copolymers, showing three typical absorption bands. The absorption band centered at 273 nm is assigned to the π–π* transitions of the benzenoid and quinoid structures. The absorption band centered at 366 nm is assigned to the π–π* transition associated with the phenazine ring conjugated to the two lone pairs of nitrogen in the –NH2 groups [28, 29]. These two bands are characteristic of PoPD. The band at 560 nm is assigned to the red-shift of the band of 597 nm corresponding to exciton-like transition in quinoid diimino units of PANI. These results confirmed that the oPD/An copolymers were prepared successfully.
The TG curves of CSA-doped PANI and CSA-doped oPD/An copolymers are shown in Fig. 3. Overall, the curves reveal that the decomposition of the two samples occurs in two steps. The first step occurs at about 270 °C, and it corresponds to the decomposition of the acid dopant. The next phase happens at 464 °C because of the decomposition of the polymer chains [30]. Noteworthily, the oPD/An copolymers are more thermally stable than PANI. Decomposition of the polymer components in oPD/An above 464 °C is much slower than those in PANI. Therefore, a good thermally stable material was prepared by chemical oxidation polymerization in the present study.
Figure 4 shows Raman spectra of the CSA-doped PANI and CSA-doped oPD/An copolymers by using the 514.5 nm laser line excitation. The spectrum of PANI (Fig. 4a) shows typical Raman shifts at 1344 and 1575 cm−1, which are assigned to the C–N•+ and C=C stretching modes in benzenoid ring, respectively [31]. Figure 4b demonstrates that the Raman shift at 1486 cm−1 is assigned to the C=N stretching mode, indicating the presence of imine sites in the polymer structure [32]. A strong Raman shift at 1365 cm−1 corresponds to the C–N•+ stretching vibrations-delocalized polaronic charge carriers. Existence of this band proves high concentration of the C–N•+ carrier in the oPD/An copolymers. The Raman shift at 1514 cm−1 is assigned to the N–H bending deformation of protonated amines. The Raman shift at 1248 cm−1 is ascribed to the C–N stretching mode of the polaronic units. The Raman shift concerning the C–C deformation of benzenoid rings is observed at 1615 cm−1 [33].
Figure 5a shows a representative SEM image of PANI with added CSA, exhibiting the existence of nanotubes with mean diameter of 200 nm and mean length of more than 1 μm. However, the morphology of the CSA-doped oPD/An copolymers was found to be spherical, as shown in Fig. 5b. The particles are uniform in size and are about 320 nm in diameter.
Figure 6 shows typical response curves of the CSA-doped PANI film and the CSA-doped oPD/An copolymer film. Figure 6a exhibits the response curve for CSA-doped PANI, revealing low sensitivity and long response-recovery time. The sensitivity is defined as S = (R − R0)/R0, where R is the resistance of the sample for RH at 11%, and R0 is the resistance of the sample for RH at 97%. The sensitivity of the CSA-doped PANI film is 0.7, and the response time is 74 and 126 s for the humidity absorption and desorption processes, respectively. Figure 6b shows that the sensitivity of the CSA-doped oPD/An copolymer films is about 10 for the RH between 11 and 97%. The time required for humidity adsorption and desorption was 38 and 11 s, respectively. Compared to PANI nanotubes, oPD/An copolymers nanoparticles exhibited significantly improved sensitivity and response–recovery behavior.
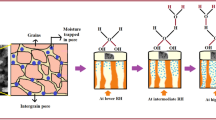
Figure 7 exhibits that the sensitivity of the CSA-doped oPD/An copolymer films remains constant during seven cycles. This indicates that the copolymer may act as a potential candidate for a reusable sensor material in humidity sensing. The cycling curve proves that the copolymer has good reproducibility and reliable sensitivity. Such good humidity sensing property has relation with the water–adsorption and proton transfer mechanism. Initially, water molecules are physically adsorbed on the surface of the copolymer, and then, the hydronium (H3O+) ion is formed at the surface with increasing number of water molecules. Under this condition, proton transfer occurs finally. The mechanism can be explained in terms of acid–base reaction as shown in Fig. 8. From this mechanism, we deduced that the interaction between water molecules and the surface of the CSA-doped oPD/An copolymer was dominated by chemisorption, while physisorption played a minor role. Numerous charged sites and hydrogen bond accepting/donating sites (–NH–, –N=, =NH+–, –NH2+–, etc.) are present within the copolymer. These charged or polar sites aid in shifting the equilibrium toward the formation of protons, which can either increase the doping level of the copolymer or conduct charge themselves through the network of absorbed water molecules. Hydrogen bond between copolymer chains through water molecules have also been suggested to contribute to the increase in conductivity [34,35,36,37].
Noteworthily, the humidity adsorption period takes much more time than the humidity desorption period on the thin film of the CSA-doped oPD/An copolymer. Although numerous charged sites and hydrogen bond accepting/donating sites are present in the copolymer, it becomes difficult when the H3O+ enters into the main chain of the copolymer at high humidity, because of larger steric hindrance of CSA. However, the water molecules are easy to take off from the copolymer in dry atmosphere. Thus, quick recovery occurs at low humidity.
Conclusion
An o-phenylenediamine/aniline (oPD/An) copolymer doped with D-camphor-10-sulfonic acid (CSA) was synthesized by oxidative chemical polymerization. FTIR spectroscopy and UV–Vis spectroscopy showed the successful fabrication of the doped copolymer. Thermal studies indicated that the copolymer exhibited better thermal stability than PANI. Humidity-sensitive behavior of the copolymer was tested by measuring the resistance of the fabricated films at 11 and 97% relative humidity. The response and recovery time was 38 and 11 s, respectively. High sensitivity and quick response to water vapor make the films to be potential candidate for humidity sensors.
References
Hsu C-L, Su IL, Hsueh T-J (2017) Tunable Schottky contact humidity sensor based on S-doped ZnO nanowires on flexible PET substrate with piezotronic effect. J Alloy Compd 705:722–733
Zhang D, Jiang C, Sun Y (2017) Room-temperature high-performance ammonia gas sensor based on layer-by-layer self-assembled molybdenum disulfide/zinc oxide nanocomposite film. J Alloy Compd 698:476–483
Xu JM, Cheng JP (2016) The advances of Co3O4 as gas sensing materials: a review. J Alloy Compd 686:753–768
Seki T, Tanaka K, Ichimura K (1997) Photomechanical response in monolayered polymer films on mica at high humidity. Macromolecules 30:6401–6403
Park MS, Lim TH, Jeon YM, Kim JG, Joo SW, Gong MS (2008) A facile and simple method for the preparation of copoly(TEAMPS/VP)/silver nanocomposites for the humidity-sensing membranes. J Colloid Interface Sci 321:60–66
Sun Y-L, Wu R-J, Huang Y-C, Su P-G, Chavali M, Chen Y-Z, Lin C-C (2007) In situ prepared polypyrrole for low humidity QCM sensor and related theoretical calculation. Talanta 73:857–861
Shoji E, Hirayama D (2007) Effects of humidity on the performance of ionic polymer-metal composite actuators: experimental study of the back-relaxation of actuators. J Phys Chem B 111:11915–11920
Biju KP, Jain MK (2007) Effect of polyethylene glycol additive in sol on the humidity sensing properties of a TiO2 thin film. Meas Sci Technol 18:2991–2996
Kuang Q, Lao C, Wang ZL, Xie Z, Zheng L (2007) High-sensitivity humidity sensor based on a single SnO2 nanowire. J Am Chem Soc 129:6070–6071
Li Z, Zhang H, Zheng W, Wang W, Huang H, Wang C, MacDiarmid AG, Wei Y (2008) Highly sensitive and stable humidity nanosensors based on LiCl added TiO2 electrospun nanofibers. J Am Chem Soc 130:5036–5037
Wang JG (2002) Polyaniline coatings: anionic membrane nature and bipolar structures for anticorrosion. Synth Met 132:53–56
Soto-Oviedo MA, Araujo OA, Faez R, Rezende MC, De Paoli M-A (2006) Antistatic coating and electromagnetic shielding properties of a hybrid material based on polyaniline/organoclay nanocomposite and EPDM rubber. Synth Met 156:1249–1255
Grgur BN, Ristic V, Gvozdenovic MM, Maksimovic MD, Jugovic BZ (2008) Polyaniline as possible anode materials for the lead acid batteries. J Power Sour 180:635–640
Li Z-F, Blum FD, Bertino MF, Kim C-S, Pillalamarri SK (2008) One-step fabrication of a polyaniline nanofiber vapor sensor. Sens Actuators B 134:31–35
Ayad MM, Salahuddin NA, Abou-Seif AK, Alghaysh MO (2008) pH sensor based on polyaniline and aniline-anthranilic acid copolymer films using quartz crystal microbalance and electronic absorption spectroscopy. Polym Adv Technol 19:1142–1148
Borole DD, Kapadi UR, Mahulikar PP, Hundiwale DG (2004) Electrochemical behavior of polyaniline, poly(o-anisidine) and their copolymer thin films in inorganic and organic supporting electrolytes. Polym Plast Technol Eng 43:1443–1458
Zhang G-R, Xu C-T, Zhang A-J, Chen L, Lu J-X (2008) Study on. electrochemical copolymerization of aniline with p-phenylenediamine on ITO electrode by in situ UV–Vis spectroscopy and characterization of the copolymer. Acta Chim Sinica 66:376–384
Lu J-F, Wang L, Lai Q-Y, Chu H-Y, Zhao Y (2009) Study of capacitive properties in supercapacitor for copolymer of aniline with m-phenylenediamine. J Solid State Electrochem 13:1803–1810
Li X-G, Wang H-Y, Huang M-R (2007) Synthesis, film-forming, and electronic properties of o-phenylenediamine copolymers displaying an uncommon tricolor. Macromolecules 40:1489–1496
Jain S, Chakane S, Samui AB, Krishnamurthy VN, Bhoraskar SV (2003) Humidity sensing with weak acid-doped polyaniline and its composites. Sens Actuators B 96:124–129
Kulkarni MV, Viswanath AK (2005) Spectroscopic, thermal and electrical properties of sulphonic acids doped poly(o-anisidine) and their application as humidity sensor. Sens Actuators B 107:791–797
Tonosaki T, Oho T, Isomura K, Ogura K (2002) Effect of the protonation level of poly(o-phenylenediamine) (PoPD) on the ac impedance of humidity-sensitive PoPD/poly(vinyl alcohol) composite film. J Electroanal Chem 520:89–93
Patil D, Seo YK, Hwang YK, Chang JS, Patil P (2008) Humidity sensitive poly(2,5-dimethoxyaniline)/WO3 composites. Sens Actuators B 132:116–124
Zhou TL, Xie XH, Cai JY, Yin LY, Ruan WD (2016) Preparation of poly(o-toluidine)/TiO2 nanocomposite films and application for humidity sensing. Polym Bull 73:621–630
Zhang LJ, Wan MX (2005) Chiral polyaniline nanotubes synthesized via a self-assembly process. Thin Solid Films 477:24–31
Kulkarni MV, Viswanath AK, Marimuthu R, Seth T (2004) Synthesis and characterization of polyaniline added with organic acids. J Polym Sci Pol Chem 42:2043–2049
Kulkarni MV, Viswanath AK, Marimuthu R, Seth T (2004) Spectroscopic, transport, and morphological studies of polyaniline added with inorganic acids. Polym Eng Sci 44:1676–1681
Lu X, Mao H, Chao D, Zhao X, Zhang W, Wei Y (2007) Preparation and characterization of poly(o-phenylenediamine) microrods using ferric chloride as an oxidant. Mater Lett 61:1400–1403
Wang L, Guo S, Dong S (2008) Facile synthesis of poly(o-phenylenediamine) microfibrils using cupric sulfate as the oxidant. Mater Lett 62:3240–3242
Ginic-Markovic M, Matisons JG, Cervini R, Simon GP, Fredericks PM (2006) Synthesis of new polyaniline/nanotube composites using ultrasonically initiated emulsion polymerization. Chem Mater 18:6258–6265
Zhang H, Li HX, Cheng HM (2006) Water-soluble multiwalled carbon nanotubes functionalized with sulfonated polyaniline. J Phys Chem B 110:9095–9099
Mazeikiene R, Malinauskas A (2002) The stability of poly (o-phenylenediamine) as an electrode material. Synth Met 128:121–125
Mallick K, Witcomb MJ, Erasmus R, Scurrell MS (2007) Hydrophilic behaviour of gold-poly (o-phenylenediamine) hybrid nanocomposite. Mater Sci Eng, B 140:166–171
Travers JP, Nechtschein M (1987) Water effects in polyaniline: a new conduction process. Synth Met 21:135–141
Pinto NJ, Shah PD, Kahol P, McCormick BJ (1996) Conducting state of polyaniline films: dependence on moisture. Phys Rev B 53:10690–10694
Matveeva ES (1996) Residual water as a factor influencing the electrical properties of polyaniline. The role of hydrogen bonding of the polymer with solvent molecules in the formation of a conductive polymeric network. Synth Met 79:127–139
Zeng FW, Liu XX, Diamond D, Lau KT (2010) Humidity sensors based on polyaniline nanofibres. Sens Actuators B 143:530–534
Acknowledgements
The study was supported by the Natural Science Foundation of China (Grant No. 21103062) and the Korean Federation of Science and Technology Societies (KOFST) Grant funded by the Korean government (MSIP: Ministry of Science, ICT and Future Planning). W. R. is grateful to the Postdoctoral Science Foundation of China (Grant No. 2014M561286).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, T., Wu, S., Cai, J. et al. Rapid humidity sensors based on poly(o-phenylenediamine-co-aniline) spherical nanoparticles. Polym. Bull. 77, 1095–1105 (2020). https://doi.org/10.1007/s00289-019-02794-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-019-02794-z