Abstract
Purpose
The transition muscle between the palatopharyngeus (PP) and the superior constrictor of the pharynx (SCP) encircles the pharyngeal isthmus from behind and is designated as the palatopharyngeal sphincter (PPS). The PPS is inferred to play important roles for velopharyngeal closure, but its existence remains controversial and its roles have been regarded as being played by the SCP. The present study aimed to clarify the anatomical status and functional implications of the PPS.
Materials and methods
Macroscopic and microscopic examinations were performed on 39 and 4 cadavers, respectively. In the former, the bilateral PPSs and their adjacent structures were exposed from outside and/or inside. In the latter, the velums embedded in paraffin were cut into frontal or sagittal sections and alternately processed with HE and Azan stains.
Results
The PPS originated from the nasal aspect of the lateral half of the palatine aponeurosis and the inferior margin of the medial pterygoid plate and was distinguishable from the PP descending in and along the palatopharyngeal arch and the cranialmost portion of the SCP in its origin. It passed dorsally on the lateral side of the levator veli palatini and traversed around the salpingopharyngeal fold running longitudinally. It then entered below the SCP and ran toward the pharyngeal raphe with SCP muscle fibers intermingled.
Conclusions
The PPS is a muscle distinct from the SCP. Its contraction produces Passavant’s ridge and conceivably enhances the efficiency of velopharyngeal closure by pressing the salpingopharyngeal fold and the musculus uvulae ridge against the velum.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The palatopharyngeus (PP) and the superior constrictor of the pharynx (SCP), respectively, distributed on the inner and outer surfaces of the pharynx, overlap each other to take an appearance of a large single sheet of muscle folded back at the top of the pharynx [14, 18]. The transition zone between the two muscles occupies a range from the lateral to posterior wall of the pharyngeal isthmus, and the muscle bundles located there are designated as the palatopharyngeal sphincter (PPS) by Whillis [20]. Although that name is adopted in Gray’s anatomy [16], the term “Passavant’s muscle” is also used there, in the sense of a muscle whose contraction produces Passavant’s ridge. Passavant’s ridge is a mucosal ridge running transversely along the posterior wall of the pharyngeal isthmus during velopharyngeal closure and is well recognized in patients with an unrepaired cleft palate [23].
The pharyngeal isthmus is where velopharyngeal closure occurs during speaking and swallowing. Accordingly, the PPS is inferred to play important roles for velopharyngeal closure, but its existence remains controversial and its roles have been regarded as being played by the SCP. This confusion must be attributed to the difficulty in distinguishing the PPS from the SCP owing to their wide overlapping area in the posterior pharyngeal wall accompanied with dense interdigitations between the muscle fibers of them. It may be also one of the reasons of the confusion that the role of Passavant’s ridge during velopharyngeal closure is regarded as being compensatory [8].
Actually, even Whillis [20] regarded the PPS as a portion of the SCP. In addition, in the diagram of Fritzel [6] often used to explain the arrangement and function of the muscles of the velum, the PPS is replaced with the SCP. Although the distribution of the PPS would implicate it as a major muscle responsible for the inward movement of the lateral wall of the pharyngeal isthmus, the PPS is replaced with the SCP or the levator veli palatini (LVP) in this respect as well [7].
We previously studied the macroscopic architecture of the human PP and reported that it had two divisions, the transverse and the longitudinal [18]. The transverse division located in the transition zone from the SCP corresponded to the PPS, while the longitudinal division was the portion generally accepted as the PP in standard anatomy textbooks (PP in the narrow sense, hereafter abbreviated as PPn). The sufficient results were obtained in this study as regards the longitudinal division, while as regards the transverse division, the detailed status of its origin and insertion, the relationships to the anatomical structures adjacent to it and its functional implications remained unresolved although its course connecting the palatine aponeurosis and the pharyngeal raphe could be understood well.
The present study aimed to further clarify the anatomical status and functional implications of the PPS. The distinctions of the PPS from the PPn and the SCP and its topographical relationships to other anatomical structures in and around the pharyngeal isthmus were studied through the macroscopic and microscopic examinations of and around the transition zone between the PP and the SCP. We could not find any comprehensive study of the PPS among available past literatures. Detailed anatomical knowledge of the PPS and its surroundings undoubtedly makes a great contribution to understandings of the functional implications of the PPS and consequently the mechanism of velopharyngeal closure.
Materials and methods
A total of 43 Japanese cadavers devoted to the university for education and research of anatomy were used in the present study, the details of which are 29 for macroscopic examination of the PPS, four for microscopic examination, and ten for macroscopic examination of positional relationships.
Macroscopic examinations of the PPS throughout its entire distribution including origin and insertion
Fifty-one body sides of 29 cadavers (15 male and 14 female, male 26 sides and female 25 sides, right 26 sides and left 25 sides) were used. On 33 sides of 18 cadavers, the examination was not preceded by sectioning the head along the paramedian plane, while on the remaining 18 sides of 11 cadavers, it was performed after the sectioning of the head.
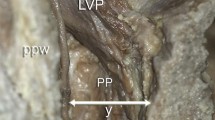
In the cases of non-sectioning of the head, the muscles were initially exposed from outside in the posterior and lateral walls of the upper pharynx, and then, the pharyngobasilar fascia was carefully removed together with the roof of the nasopharynx so as not to injury the relevant muscles and finally to visualize the nasal aspect of the velum (Fig. 1a). The entire nasal aspect including the rear of hard palate was dissected to expose all underlying velar muscles and adjacent structures including the palatine aponeurosis, the inferior margin of the medial pterygoid plate, and the pterygoid hamulus. The tensor veli palatini (TVP) and the LVP were then bilaterally cut at their origins on the cranial base and turned laterally and medially, respectively. After full removal of fat tissues and vessels filling the space between the two muscles, the muscular layer forming the bottom of that space, corresponding to the transition zone between the PP and the SCP, was traced from its origin to the posterior wall of the pharynx and visualized to meet the same muscular layer of the contralateral side at the pharyngeal raphe (Fig. 1b).
a Schematic drawing of muscles of the posterior pharyngeal wall. The pharyngobasilar fascia has been removed together with the roof of the nasopharynx to open the cavity of pharynx. b Viewed from this site of opening. b Muscle of the transition zone (PPS) between the palatopharyngeus (PP) and the superior constrictor of the pharynx (SCP) is viewed from above in the case of non-sectioning of the head. c Schematic drawing of the medial view of velar muscles, seen in the right half of the head sectioned along the paramedian plane. The pharyngobasilar fascia and the roof of the nasopharynx have been removed. The right upper arrowhead shows the direction of view in b. d PP, the palatopharyngeal sphincter (PPS), and the SCP are viewed in the angle similar to that in c. The levator veli palatini (LVP) is turned upward to visualize the arrangement of the muscles on the oral side of the velum. e PPS viewed from lateral in the head sectioned along the paramedian plane. The arrowhead in a shows the direction of view in this picture. According to the previous notion, the PPS was colored as a portion of the SCP in a, c. CB cranial base, HB hyoid bone, HP hard palate, ICP inferior constrictor of pharynx, LVP levator veli palatini, colored green in a, c; MCP middle constrictor of pharynx, ML median line of velum, MP medial pterygoid plate, MU musculus uvulae, NC nasal cavity, nPPn nasal fasciculus of origin of PPn, oPPn oral fasciculus of origin of PPn, PA palatine aponeurosis, colored blue in a, c; PF pharyngobasilar fascia, PG palatoglossus, PH pterygoid hamulus PMR pterygomandibular raphe, PPA palatophasryngeal arch, PPn PP in narrow sense, colored dark brown in c; pPPn posterior fasciculus of origin of PPn, PPS muscle of transition zone between PP and SCP or palatopharyngeal sphincter, colored brown in a, c; PPW posterior pharyngeal wall, PR pharyngeal raphe, SCP superior constrictor of pharynx, colored brown in a, c; SP salpingopharyngeus, StP stylopharyngeus, T tongue, TG thyroid gland, TVP tensor veli palatini, colored blue in a, c
In the cases of sectioning of the head, the exposure of muscles was started from the inner surface of the head, such that the muscles throughout the nasal aspect of the velum to the nasopharynx (Fig. 1c) and then those throughout the oral aspect of the velum to the oropharynx (Fig. 1d) were entirely exposed. In the outer surface of the head, the muscles of the posterior and lateral walls of the upper pharynx were first exposed (Fig. 1e). Then, after cutting the TVP at its origin on the cranial base to turn it laterally, the muscular layer forming the bottom of the space between the TVP and the LVP was traced as far as the palatine aponeurosis along the outer margin of the LVP.
Microscopic examination of the origin of the PPS
This examination was performed to visualize some of the macroscopic findings difficult to display in gross anatomical figures. Four of the cadavers (two males and two females) were used for this examination. The entire portion of the velum including the rear of the hard palate was bilaterally removed as one piece, and embedded in paraffin according to the usual method. One and three of the embedded specimens were, respectively, cut into serial sagittal and frontal sections of 7-µm thickness. The sections were then alternately processed with HE and Azan stains and examined at low magnifications using stereomicroscope (Vixen SL-60TL·SL-60ZTL).
Macroscopic examinations of the positional relationships between the PPS and its adjacent anatomical structures
The examinations were performed to obtain observations necessary for determining the functional implications of the PPS. The anatomical structures examined were the PPS, salpingopharyngeal fold, salpingopharyngeus, pharyngeal recess, and palatopharyngeal ridge.
In eight of the ten cadavers (five males and five females), the head was sectioned along the paramedian plane, while in the remaining two, the sectioning of the head was not performed. The positional relationships were then examined before removing the mucosa or after entirely exposing the PPS and its relevant muscles in the velum and the upper pharynx.
Results
Muscle bundles forming the transition zone between the PP and the SCP
The bilateral transition zones encircled together the pharyngeal isthmus from behind and lateral. The transition zone consisted of three muscle bundles. They, respectively, originated from the following three sites: the palatine aponeurosis, the inferior margin of the medial pterygoid plate, and the pterygoid hamulus (see “type A” picture of Fig. 2). In the palatine aponeurosis, the muscle bundle (the aponeurosis bundle) originated from the nasal aspect of the lateral half of the aponeurosis (Fig. 3a). This bundle was the transverse division of the PP in our previous study [18] and corresponded to the PPS. In the inferior margin of the medial pterygoid plate, the muscle bundle (the inferior pterygoid bundle) originated from the periosteum of the inferior margin of the plate (Fig. 3b) and was easily removable from the plate. In the pterygoid hamulus, the muscle bundle (the hamulus bundle) originated from dense connective tissue tightly attached to the pterygoid hamulus and the adjoining posterior margin of the medial pterygoid plate (Fig. 3b), and was removable only with much difficulty from the attached portions. This bundle was the pterygopharyngeal part of the SCP, which caudally continued to the buccopharyngeal part of it. The three muscle bundles ran dorso-medially or medially toward the cranialmost portion of the pharyngeal raphe, with adjacent ones partially overlapping each other and/or with partial intermixture of muscle fibers lying between adjacent ones (see “type A” picture of Fig. 2).
Muscle bundles forming the transition zone between the PP and the SCP are viewed from above. Three bundles exist (see “type A” picture): the aponeurosis bundle (Ab) originating from the palatine aponeurosis (PA), the inferior pterygoid bundle (IPb) originating from the inferior margin of the medial pterygoid plate (MP), and the hamulus bundle (Hb) originating from the pterygoid hamulus (PH) and the adjacent posterior margin of the medial pterygoid plate. The stratigraphic relationships among the three bundles are subdivided into five types, types A–E. Ab aponeurosis bundle, Hb hamulus bundle, HP hard palate, IPb inferior pterygoid bundle, LVP levator veli palatini, ML median line of velum, MP medial pterygoid plate, oPPn oral fasciculus of origin of PPn, PA palatine aponeurosis, PF pharyngobasilar fascia, PH pterygoid hamulus, PPW posterior pharyngeal wall, PR pharyngeal raphe, SCP superior constrictor of pharynx, TVP tensor veli palatini
Microscopic observations of origin sites of the three muscle bundles forming the transition zone between the PP and the SCP are shown in frontal sections of the velum. a At a level proximal to the pterygoid hamulus, it was found that the aponeurosis bundle (Ab) originated from the nasal aspect of the lateral half of the palatine aponeurosis (PA), while the oral fasciculus of origin of the PPn (oPPn) was adjacent to the oral side of the medial half of the PA. b At the level of the pterygoid hamulus (PH), it was found that the inferior pterygoid bundle (IPb) originated from the periosteum (P) of the inferior margin of the medial pterygoid plate (MP), while the hamulus bundle (Hb) originated from dense connective tissue tightly attached to the PH. Ab aponeurosis bundle, Hb hamulus bundle, IPb inferior pterygoid bundle, LVP levator veli palatini, ML median line of velum, MP medial pterygoid plate, NC nasal cavity, OC oral cavity, oPPn oral fasciculus of origin of PPn, P periosteum, PA palatine aponeurosis, PG palatine glands, PH pterygoid hamulus, TVP tensor veli palatini
The stratigraphic relationships among the three muscle bundles forming the transition zone between the PP and the SCP
The stratigraphic relationships were investigated to consider which muscle the inferior pterygoid bundle belonged to, the PPS or the SCP. The relationships were subdivided into five types, A, B, C, D, and E (Fig. 2; Table 1). Type A: the aponeurosis bundle was located in the medial layer, and the inferior pterygoid one in the intermediate layer and the hamulus one in the lateral layer. Type B: the hamulus bundle was located laterally, while the other two were located medially and not distinguishable from each other in order of layers. Type C: the aponeurosis bundle was located medially, while the other two were located laterally and not distinguishable from each other in order of layers. Type D: the inferior pterygoid bundle was located medially, while the other two were located laterally and not distinguishable from each other in order of layers. Type E: the three bundles could not be distinguished from each other in order of layers. Each type was found in 12, 33, four, one, and one of 51 body sides, respectively (Table 1).
It was clarified from the above results that the aponeurosis bundle was located more medially than the hamulus bundle in almost cases (49 of 51 sides). The inferior pterygoid bundle was also located more medially than the hamulus bundle in many cases (46 of 51 sides) and lay in the same layer as the aponeurosis bundle in about two-thirds of the cases (33 of 51 cases), showing that the inferior pterygoid bundle lay in the layer closer to the aponeurosis bundle than the hamulus one. This finding suggests that the inferior pterygoid bundle might be more properly said to be a portion of the PPS rather than the SCP.
Origins of the PPS (Fig. 4)
The PPS, composed of the aponeurosis and the inferior pterygoid bundles, originated from a region continuing from the palatine aponeurosis to the inferior margin of the medial pterygoid plate. In the palatine aponeurosis, the PPS originated from the nasal aspect of the lateral half of the aponeurosis and passed dorsally below the LVP, and therefore, it was definitely distinguishable from the PPn originating from the medial half of the palatine aponeurosis (Fig. 3). The PPn started as the nasal and oral fasciculi of origin, which arose from the nasal and oral aspects of the medial half of the aponeurosis, respectively, and then passed over and below the LVP, respectively. Also between the inferior pterygoid bundle of the PPS and the cranialmost part of the SCP (the hamulus bundle), the two muscles were easily distinguishable, because the former originated from the periosteum of the inferior margin of the medial pterygoid plate with its attachment easily removable, while the latter originated from the pterygoid hamulus and the adjoining posterior margin of the medial pterygoid plate with its attachment firmly adhered and located more laterally than the origin of the former.
Positional relationships among the PP in the narrow sense (PPn), the PPS and the SCP are shown in schema from the nasal side. Figure 11 of our previous paper [18] is altered to add new findings obtained in the present study. The addition of the musculus uvulae (MU) and the revision in the portion of the palatine aponeurosis (PA) are based on our study of the musculus uvulae [17]. In the oral fasciculus of origin of the PPn (oPPn), a portion and the remaining portion of it originated from the median plane of the velum and the oral aspect of the medial half of the PA, respectively, and they joined to appear from below the PA. The PPS is composed of the aponeurosis (Ab) and inferior pterygoid bundles (IPb). The hamulus bundle (Hb) is the cranialmost part of the SCP. Ab aponeurosis bundle, CAT cartilage of auditory tube, Hb hamulus bundle, HP hard palate, IPb inferior pterygoid bundle, LVP levator veli palatini, colored green, ML median line of velum, MP medial pterygoid plate, MU: musculus uvulae, nPPn nasal fasciculus of origin of PPn, oPPn oral fasciculus of origin of PPn, PA palatine aponeurosis, colored blue, PH pterygoid hamulus, PPn PP in narrow sense, colored dark brown, pPPn posterior fasciculus of origin of PPn, PPS palatopharyngeal sphincter, colored yellow, PPW posterior pharyngeal wall, PR pharyngeal raphe, SCP superior constrictor of pharynx, colored brown, SP salpingopharyngeus, TVP tensor veli palatini, colored blue
The course and insertion of the PPS
After leaving the sites of origin, the PPS ran dorsally along the oral (lateral) side of the LVP similar to the oral fasciculus of the PPn, but it did not pass toward the palatopharyngeal arch and instead took a more cranial course to enter the lateral wall of the pharyngeal isthmus and reach the posterior pharyngeal wall (Fig. 1d). When following that course, it ran dorsally between the LVP and the TVP with its surface horizontally directed (Fig. 1e). After that, however, it changed the direction of its surface to vertical with its more medial fibers tending to reach more cranial levels of the pharyngeal raphe (Fig. 1e). The above findings were also obtained in our previous paper [18].
Reaching the posterior pharyngeal wall, the PPS passed under the hamulus bundle to enter below the SCP (Fig. 1e) and it ran toward the cranialmost portion of the pharyngeal raphe with their distribution spreading a little rostrocaudally (Fig. 5). Accordingly, the wall of the cranialmost portion of the posterior pharyngeal wall was composed of two transverse layers of muscle, the inner PPS layer and the outer SCP layer (Fig. 4). However, distinguishing the two layers was difficult owing to the dense interdigitations between the muscle fibers of them, so that the PPS could be traced only with difficulty as far as the pharyngeal raphe.
Insertion of the PPS is visualized on the outer surface of the pharynx by removing the overlying SCP muscle fibers. The tensor veli palatini (TVP) is cut off near the pterygoid hamulus (PH) and the stylopharyngeus (StP) is turned ventrally. LVP levator veli palatini, NC nasal cavity, PA palatine aponeurosis, PF pharyngobasilar fascia, PH pterygoid hamulus, PPn PP in narrow sense, PPS palatopharyngeal sphincter, PR pharyngeal raphe, StP stylopharyngeus, TVP tensor veli palatini
The positional relationships between the PPS and its adjacent anatomical structures
The palatopharyngeal ridge, listed just below the pharyngeal recess in the section of the nasopharynx of Terminologia Anatomica Japonica [9], was not discernible in eight of ten cadavers investigated. In the two cadavers bilaterally having the well-developed pharyngeal recess, however, the palatopharyngeal ridge was bilaterally discernible as a clear ridge running transversely along the lowest lateral wall of the nasopharynx as if connecting the velum and the pharyngeal wall (Fig. 6a). The ridge appeared from below the torus levatorius and then passed lateral to the salpingopharyngeal fold to reach the median portion of the posterior pharyngeal wall after forming the bottom of the pharyngeal recess. The PPS was found in all cases beneath the mucosa of the palatopharyngeal ridge or the place corresponding to the ridge and took the course similar to that of the ridge described above (Fig. 6b), suggesting the possibility that the ridge is formed by the PPS.
a Palatopharyngeal ridge (PPR) is shown on the lowest inner wall of the nasopharynx to run transversely as if connecting the velum and the posterior pharyngeal wall (PPW). The pharyngeal recess (PRe) is well developed in this case, and the PPR forms the bottom of it. b Muscles are exposed on the inner surfaces of the nasopharynx and the pharyngeal isthmus, although the salpingopharyngeal fold (SPF) remains intact. The PPS is seen to take the course similar to that of the PPR shown in a. HP hard palate, LVP levator veli palatini, MP medial pterygoid plate, NC nasal cavity, OC oral cavity, PF pharyngobasilar fascia, PPA palatophasryngeal arch, PPR palatopharyngeal ridge, PPS palatopharyngeal sphincter, PPW posterior pharyngeal wall, PRe pharyngeal recess, SPF salpingopharyngeal fold, TL torus levatorius, TT torus tubarius, TVP tensor veli palatini
The salpingopharyngeal fold crossed over the inner surface of the PPS to descend onto the lateral wall of the pharyngeal isthmus (Fig. 6b). The above course of the fold allows the contracted PPS to press the fold against the nasal aspect of the velum (Fig. 7). The salpingopharyngeus, which passed downward within the salpingopharyngeal fold, was totally or partially replaced with connective tissue and/or adipose tissue in many cases as reported in our previous paper [18].
It is shown from behind after removals of the pharyngobasilar fascia and the roof of the nasopharynx that the salpingopharyngeal fold (SPF) running longitudinally is sandwiched between the PPS running transversely peripheral to the SPF and the nasal aspect of the velum. LVP levator veli palatini, NC nasal cavity, nPPn nasal fasciculus of origin of PPn, PH pterygoid hamulus, PPS palatopharyngeal sphincter, PPW posterior pharyngeal wall, PR pharyngeal raphe, SCP superior constrictor of pharynx, SP salpingopharyngeus, SPF salpingopharyngeal fold, TT torus tubarius, TVP tensor veli palatini
Discussion
Anatomical status of the PPS
The PPS is located in the transition zone between the PP and the SCP [20]. As regards the PP, there are three recent studies anatomically made by Sumida et al. [18], Choi et al. [4], and Sakamoto [14]. According to these studies, they are unanimously agreed that the PP excluding the portion of the PPS, namely, the PPn, originates from the palatine aponeurosis and sandwiches the LVP before descending toward the pharynx and the larynx within or along the palatopharyngeal arch. As regards the SCP, there is a disagreement of opinion whether the origin of the cranialmost part of it, namely, the pterygopharyngeal part, is restricted within the pterygoid hamulus or extends as far as the adjoining posterior margin of the medial pterygoid plate [19]. However, this disagreement is conceivably due to the range of the hamulus different among literatures. On the other hand, it is known that muscle fibers originating from the velum join to the pterygomandibular part [5, 15] (Fig. 1a, c). Those muscle fibers, which are thought of as corresponding to the PPS, have been regarded for long as a portion of the SCP [5]. Nobody excluding Sakamoto [14] described muscle fibers originating from the inferior margin of the medial pterygoid plate to join the SCP, but there is a possibility that those fibers were indistinguishably included in the fibers from the velum.
The present study clarified that the transition zone between the PP and the SCP was composed of the three muscle bundles, the aponeurosis bundle, the inferior pterygoid bundle, and the hamulus bundle. The aponeurosis and hamulus bundles correspond to the PPS and the pterygomandibular portion of the SPC, respectively. As regards the PPS located between the PPn and the SCP, even Whillis [20], who is the namer of the PPS, regarded it as a portion of SCP (Fig. 1a, c). However, Kriens [10], Huang et al. [7], and recently Sumida et al. [18] and Choi et al. [4] regarded it as a portion of the PP. Sakamoto [14] regarded it as a place where the muscle fibers of both the PP and the SCP intermingled, even though he regarded the inferior pterygoid bundle to be a portion of the SCP. The present study investigated the stratigraphic relationships among the three muscle bundles and clarified that the inferior pterygoid bundle lay in the layer closer to the aponeurosis bundle rather than the hamulus bundle. This finding suggests that the inferior pterygoid bundle participates in formation of the PPS and may be a portion of the PP instead of the SCP.
The present study further clarified that the aponeurosis bundle, constituting a portion of the PPS, originated from the nasal aspect of the lateral half of the palatine aponeurosis and, therefore, was clearly distinguishable from the PPn originating from the medial half of it. The similar finding was also obtained in the study of Sakamoto [14]. We stated in our previous paper [18] that the difference of course between the two muscles was useful as a criterion to distinguish them. The above finding shows that their different sites of origin are also useful as criteria for distinction between them. Also between the PPS and the SCP, we mentioned only their different sites of origin as a criterion for distinction between them in our previous paper [18]. The present study additionally clarified that the PPS was distributed in the layer more medially than the SCP. This is also useful as a criterion for distinction between them. These findings show that the PPn, PPS, and SCP are distinguishable from each other on the basis of their statuses of origin (Fig. 4).
Functional implications of the PPS
There are three movements of the walls of the pharyngeal isthmus concerned with the velopharyngeal closure: backward movement of the anterior wall, medial movement of the lateral wall, and forward movement of the posterior wall [21]. Considering the arrangement of muscles around the pharyngeal isthmus (Fig. 1b), it is easily understood that the elevation of the velum by the LVP is responsible for the movement of the anterior wall, while the PPS and the SCP are responsible for the movement of the lateral and posterior walls. The SCP must be effective for the movement of the posterior wall. However, it may be ineffective for the movement of the lateral wall owing to its bilateral attachments being fixed in an immovable position by the pterygoid hamulus (Fig. 4). On the contrary, the bilateral PPSs encircle the pharyngeal isthmus from behind and lateral (Fig. 1b), and, therefore, must be effective not only for the movement of the posterior wall but also for the movement of the lateral wall, conceivably making great contribution to velopharyngeal closure [7].
Casey [1] directly viewed velopharyngeal closure during speaking or swallowing from a defective area of the face after orbital exenteration and maxillectomy, and reported that when swallowing, Passavant’s ridge extended from the posterior wall of the pharyngeal isthmus toward the lateral wall. Since no muscles other than the PPS display such passage and movement (Fig. 6b), Cho et al. [3] used the term “Passavant’s ridge” as a synonym for “PPS” in their paper. The term “palatopharyngeal ridge”, listed in Terminologia Anatomica Japonica [9], may be also a synonym of Passavant’s ridge, because the palatopharyngeal ridge is also suggested to be formed by the PPS (Fig. 6a, b).
In the median portion of the pharyngeal isthmus, the portion of mucosa under which the PPS runs transversely along the posterior pharyngeal wall is opposed to the “musculus uvulae” ridge [13] running longitudinally on the nasal aspect of the velum (Fig. 8). When closing the pharyngeal isthmus, the contracted LVP elevates the velum to move the anterior wall of the isthmus backward, while the contracted SCP move the posterior wall forward and the contracted PPS further make the wall protrude forward. The dense interdigitations of muscle fibers between the both muscles in the posterior pharyngeal wall (Figs. 1b, 4) are conceivably concerned with the working together of them in this time. The contracted musculus uvulae then enlarges the “musculus uvulae” ridge formed with this muscle and the glandular connective tissue overlying the muscle, and presses it against the protruding posterior wall to enhance the efficiency of velopharyngeal closure in the median portion of the pharyngeal isthmus [17]. The above action of the musculus uvulae is called “space-occupying function” [11].
Pharyngeal isthmus viewed from the nasal side. The portion of mucosa (PPPS) under which the PPS runs transversely is opposed to the “musculus uvulae” ridge (MUR) running longitudinally on the median nasal aspect of the velum and the salpingopharyngeal fold (SPF) running longitudinally in the lateral portion of the isthmus. HP hard palate, MUR musculus uvulae ridge, PPPS portion of mucosa containing PPS, PPW posterior pharyngeal wall, SPF salpingopharyngeal fold, TL torus levatorius, TT torus tubarius
The space-occupying function may also occur in the lateral portion of the pharyngeal isthmus, in which the salpingopharyngeal fold running longitudinally is sandwiched between the PPS transversely running peripheral to the fold and the nasal aspect of the velum (Fig. 7). The contracted PPS makes the wall of the isthmus protrude not only forward but also medially and presses the cushiony salpingopharyngeal fold against the velum to enhance the efficiency of velopharyngeal closure. The space-occupying function in the lateral portion may be a passive function by the sandwiched cushiony tissue, not requiring contraction of muscle, because the salpingopharyngeal fold is a prominent mucosal fold rich in adipose loose connective tissue, but the salpingopharyngeus contained in it is often undeveloped and is due to be partially or totally replaced with adipose loose connective tissue [18].
Thus, the PPS conceivably acts to enhance the efficiency of velopharyngeal closure in collaboration with the “musculus uvulae” ridge and the salpingopharyngeal fold (Fig. 8). During velopharyngeal closure, the pharyngeal isthmus is primarily closed with the posterior pharyngeal wall moved forward by the SCP and the velum elevated by the LVP. On the other hand, the above ridge and fold, both rich in loose connective tissue including fat cells or glands, are considered to act as good cushiony pads occupying spaces left between the posterior pharyngeal wall and the velum even after primary closure. The contracted PPS, which allows the posterior and lateral walls of the pharyngeal isthmus to protrude forward and medially, respectively, acts to press the pads against the velum, as a result of which the closure of the pharyngeal isthmus is considered to be additionally tightened. Thus, it is conceivable that the protrusion made by the contracted PPS during velopharyngeal closure has been clinically called Passavant’s ridge from the past.
Which muscle does the PPS belong to, the PP or the SCP?
Most mammals have an intranarial larynx [12], in which the epiglottis passes through the pharyngeal isthmus to ride on the velum. The intranarial larynx provides a direct air channel from the nasal cavity to the larynx and allows fluid to pass through both sides of the larynx. This requires animals to have a sphincter-like muscle holding the epiglottis in that position. Cave [2] mentioned the PP as that muscle, which is actually known in many mammals to assume a sphincter-like appearance to encircle the pharyngeal isthmus [18]. This appearance is the same as that of the human PPS described in the present study. Laitman and Reidenberg [12] additionally described that the connection between the epiglottis and the velum may be broken even in animals having the intranarial larynx during some vocalizations or swallowing of a bolus of food. In such occasions, the PP conceivably acts as a nasopharyngeal sphincter to shut off any leak of food or air from the pharynx to the nasal cavity, this action being also similar to that of the human PPS.
In mammals having the intranarial larynx, Wood-Jones [22] mentioned three muscles radiating from the velum to the tongue, the larynx, and the posterior pharyngeal wall: the palatoglossus, the palatothyroideus and the palatopharyngeus. The latter two collectively correspond to the PP in the present study and are conceivably responsible for maintaining the intranarial position of the larynx as follows: the palatothyroideus draws the larynx near as a retractor, and the palatopharyngeus holds the larynx in that position as a sphincter. In the human with the larynx taking a low position largely separate from the velum, however, the anatomical states of the two muscles change in their own ways [22]. In the palatothyroideus, the site of insertion descends together with the larynx and the muscle takes on a vertical direction. Changes are not so marked in the palatopharyngeus, however. Although its lower muscle fibers are pulled down on the posterior pharyngeal wall in the wake of the descending larynx, its upper muscle fibers retain their primitive position and continue to play their original sphincteric action.
We previously reported that the human PP was composed of the transverse and longitudinal divisions, the latter of which, namely, the PPn, was further subdivided into two portions inserting the pharyngeal wall and the thyroid cartilage, respectively [18]. The transverse division corresponds to the aponeurosis bundle of the present study and forms the PPS together with the inferior pterygoid bundle. Applying the above subdivisions of the PP to the descriptions of Wood-Jones [22], the transverse division, the portion of the longitudinal division inserting the pharyngeal wall, and the portion of it inserting the thyroid cartilage correspond to the upper and lower muscle fibers of the palatopharyngeus of Wood-Jones, and the palatothyroideus, respectively. Thus, it is conceivable that the PPS, located in the transition zone between the PP and the SCP, is a portion retaining the primitive appearance and function of the original PP, while the PPn is a portion characteristic to the human that is modified, accompanied with the descent of the larynx.
The primary office of the mammalian pharynx is alimentary, namely, the conduction of food from the oral cavity to the esophagus [2]. From the descriptions of Wood-Jones [22] and Cave [2], it is supposed that the muscular mechanism for the subsidiary respiratory function of the pharynx (the nasopharyngeal sphincter) is entrusted to the PPS derived from the PP, and the SCP, which is the intrinsic musculature of the pharynx, continues to keep its primary configuration and function as an alimentary canal muscle. Accordingly, the PPS should be considered as a muscle separate from the SCP. The PPS is derived from the PP together with the PPn, the two muscles, however, are anatomically and functionally distinguishable from each other.
References
Casey DM (1983) Palatopharyngeal anatomy and physiology. J Prosthet Dent 49:371–378
Cave AJE (1967) The nature and function of the mammalian epipharynx. J Zool Lond 153:277–289
Cho JH, Kim JK, Lee H-Y, Yoon J-H (2013) Surgical anatomy of human soft palate. Laryngoscope 123:2900–2904
Choi D-Y, Bae J-H, Youn K-H, Kim H-J, Hu K-S (2014) Anatomical considerations of the longitudinal pharyngeal muscles in relation to their function on the internal surface of pharynx. Dysphagia 29:722–730
Dickson DR (1975) Anatomy of the normal velopharyngeal mechanism. Clin Plast Surg 2:235–248
Fritzell B (1969) The velopharyngeal muscles in speech. An electromyographic and cineradiographic study. Acta Oto-Laryngol Suppl 250:5–81
Huang MHS, Lee ST, Rajendran K (1998) Anatomic basis of cleft palate and velopharyngeal surgery: implications from a fresh cadaveric study. Plast Reconstr Surg 101:613–627
Isberg AM, Henningsson GE (1990) Intraindividual change in the occurrence of Passavant’s ridge due to change in velopharyngeal sphincter function: a videofluoroscopic study. Cleft Palate J 27:253–257
Japanese Association of Anatomist (2007) Terminologia Anatomica Japonica, 13th edn. Igaku-shoin, Tokyo
Kriens OB (1969) An anatomical approach to veloplasty. Plast Reconstr Surg 43:29–41
Kuehn DP, Folkins JW, Linville RN (1988) An electromyographic study of the musculus uvulae. Cleft Palate J 25:348–355
Laitman JT, Reidenberg JS (1993) Specializations of the human upper respiratory and upper digestive systems as seen through comparative and developmental anatomy. Dysphagia 8:318–325
Pigott RW (1969) The nasendoscopic appearance of the normal palate-pharyngeal valve. Plast Reconstr Surg 43:19–24
Sakamoto Y (2015) Spatial relationship between the palatopharyngeus and the superior constrictor of the pharynx. Surg Radiol Anat 37:649–655
Sappey PH C (1879) Traité d’anatomie descriptive avec figures intercalées dans le texte, vol 4, 3rd edn. Adrien Delahaye, Paris, pp 55, 142–143
Standring S (2008) Head and neck. In: Standring S, Borley NR, Cllins P et al (eds) Gray’s anatomy: the anatomical basis of clinical practice, 40th edn. Churchill Livingstone Elsevier, Philadelphia, pp 568–575
Sumida K, Kashiwara G, Seki S, Masui T, Ando Y, Yamashita K, Fujimura A, Kitamura S (2014) Anatomical status of the human musculus uvulae and its functional implications. Clin Anat 27:1009–1015
Sumida K, Yamashita K, Kitamura S (2012) Gross anatomical study of the human palatopharyngeus muscle throughout its entire course from origin to insertion. Clin Anat 25:314–323
Tsumori N, Abe S, Agematsu H, Hashimoto M, Ide Y (2007) Morphologic characteristics of superior pharyngeal constrictor muscle in relation to the function during swallowing. Dysphagia 22:122–129
Whillis J (1930) A note on the muscles of the palate and the superior constrictor. J Anat 65:92–95
Witzel MA, Posnick JC (1989) Patterns and location of velopharyngeal valving problem: atypical findings on video nasopharyngoscopy. Cleft Palate J 26:63–67
Wood-Jones F (1939) The nature of the soft palate. J Anat 74:147–170
Yanagisawa E, Weaver EM (1996) Passavant’s ridge: is it a functional structure? Ear Nose Throat J 75:766–767
Acknowledgements
The authors wish to express their special thanks to the donors of the cadavers used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study received no financial support from other than the university.
Conflict of interest
The authors declare that they do not have any conflict of interest.
Ethic approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Sumida, K., Ando, Y., Seki, S. et al. Anatomical status of the human palatopharyngeal sphincter and its functional implications. Surg Radiol Anat 39, 1191–1201 (2017). https://doi.org/10.1007/s00276-017-1855-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-017-1855-6