Abstract
The main objectives of the present study were to assess the water demand for heavy fruit load of ‘Hass’ avocado throughout the growth periods and to investigate the effects of deficit irrigation during sensitive phenological phases on yield. The experimental set-up allowed the comparison between trees responses to three irrigation strategies during the entire growth period (no water stress; excessive irrigation; constant water stress) as well as the comparison between regulated deficit irrigation (RDI) managements applied during the early or the late growth period. The yield of no water stress treatments during three experimental years was very high (25–31 t ha−1) while the yields of water-stressed trees were significantly lower (16–21 t ha−1). More importantly, the yield of no water stress trees was not susceptible to alternate bearing while the yield of water-stressed trees was considerably reduced during off-crop years. Irrigation rates and the actual evapotranspiration coefficient KL = ET/ET0 for the no water stress treatment may serve as a reasonable guide for irrigation management. Fruit load should be taken into account while planning irrigation and fertilization management and plant-based methods should be used for controlling the irrigation management (scheduling and quantities). Analyses of trunk diameter variation data that lead to evaluation of trunk growth rate and maximum daily shrinkage reflect phenological stages and periodicity of shoot, fruit and root growth, and also may provide an integrative, “holistic viewpoint” of overall tree status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Avocado (Persea Americana Mill.) is indigenous to the humid subtropical and tropical regions of central and northern South America (Lahav et al. 2013), but nowadays, is grown all over the world due to its very high fruit nutritional value (Schaffer et al. 2013). The development of irrigation technologies was the key factor that facilitated avocado plantation spreading to Mediterranean, semi-arid and arid areas where irrigation is crucial (Carr 2013; Lahav et al. 2013). The vegetative growth of avocado trees is generally vigorous, bearing a potential photosynthetic capability for producing more than 30 t ha−1 of fruit containing 17% oil (Wolstenholme 1986). Unfortunately, the worldwide average avocado yield is generally low (Campisi-Pinto et al. 2017) as a result of high rates of flower and fruit abscission (Garner and Lovatt 2008) and alternate bearing (annual cycle of high and low yield, Lovatt 2011). Recently, Holzapfell et al. (2017) reported that water stress significantly amplified the effect of alternate bearing.
The total number of flower, bud and fruitlet abscised is huge (approximately 1.3 million per a single tree) and only 1% of the fruitlet reach maturity (Lahav and Zamet 1999). Weekly abscission rate of 140 thousand flowers and 5 thousand fruits per single avocado tree which annually accumulated to almost 500 thousand flowers and 15 thousand fruits has been reported (Slabbert 1981). Optimal water status during flowering is required for proper fruit-set as there is an increased demand for water to meet the high water transpiration from flowers (Whiley et al. 1988). Flower or fruit abscission could not be related to alternate bearing (Garner and Lovatt 2008) or others factors such as soil moisture (Slabbert 1981) or leaf nutrient status (Garner and Lovatt 2008). Wolstenholme et al. (1990) suggested that carbohydrates stress during the period of rapid fruit growth and oil accumulation coinciding with peaks of high temperatures and high evaporative demand may be the most important cause for fruit abscission. Adato and Gazit (1977) hypothesized that the fruitlet abscission is caused by a malfunction of the embryo or the seed. Seeds have the highest priority for carbohydrates (Wolstenholme 1987) and it is likely that the very fast growth during the early stage of fruit development has created a huge demand for carbohydrates. Water stress in a lysimeter experiment increased significantly the extent of fruitlet abscission, probably because of the combination of water stress and insufficient carbohydrates supply to the developing fruits (Silber et al. 2012). Low carbohydrate reserves induced by high fruit load may affect the flowering process too and consequently, flower intensity in the next year (Ziv et al. 2014). Thus, it is plausibly to relate carbohydrate deficiency to the main two limiting causes for low avocado yield, i.e., fruit abscission and alternate bearing.
Considering that the vector of nutrients to the roots is the soil water, variations in soil water content (θ) may have considerable effects on plant-nutrient availability: (1) θ have a quadratic influence on the diffusion coefficient of nutrients in soil (Claassen and Steingrobe 1999); and (2) slight θ variations may induces several orders of magnitude in the unsaturated hydraulic conductivity (Mualem 1986; Assouline 2001). Therefore, irrigation and fertilization managements are mutually dependent and accordingly, optimal fertilization management requires an optimal irrigation one.
The main objectives of the present research study were to assess the water demand for heavy fruit load of ‘Hass’ avocado throughout the growth periods and to investigate the effects of deficit irrigation during sensitive phenological phases on yield.
Materials and methods
General information and site characteristics
The study was conducted between 2014 and 2017 at the Acre Experimental Station, located in the Western Galilee, in Israel (32°57′N; 35°05′E; 10 m ASL). The soil is grumusol containing approximately 60% clay, mostly smectite. The climate is Mediterranean, with mild, wet winters followed by dry hot summers. The rainfall season in the region is between October and May, and annual precipitation at Acre totaled 317, 687, 280 and 439 mm in 2013–2014, 2014–2015, 2015–2016 and 2016–2017, respectively.
Experimental design
The response of ‘Hass’ avocado trees grafted on ‘Degania 117’, a West-Indian avocado rootstock, to different irrigation treatments was studied. The trees were planted on the top of 60-cm high raised beds in 2010. The distance between rows was 6 m and that between plants within rows 4 m (density of 417 trees per hectare). Each experimental plot comprised 18 trees (3 rows with 6 trees per row). The external rows were considered as buffer rows and only the four internal trees were included in yield statistics.
Reference water demand of the trees was determined according to the actual water uptake measured from comparable ‘Hass’; avocado trees grown in 1000-L plastic containers (lysimeters) located nearby the experiment field. The containers were covered by a plastic sheet to prevent evaporation. Tree size, leaf area index (LAI) and fruit yield of the lysimeter-grown trees were practically similar to those of the soil-grown trees. The ratio of evaporation (E) to total evapotranspiration (ET) from soil surfaces of drip irrigated orchards is only 0.10–0.15 (Kool et al. 2014) and therefore, water uptake of the lysimeter-grown trees was used as reference for the soil-grown trees. Accordingly, the actual evapotranspiration coefficient of the lysimeter experiment (KL = ET/ET0) was used in the present experiment. The drainage from six lysimeters was collected in 100-L containers that were placed on a weight-recording scale (BH200, Shekel Electronic scales, Israel). Irrigation frequency was high enough to assume minimal temporal changes of water content in the lysimeters. Leachate weight was measured at a 10 min interval. The tree water uptake was calculated as the difference between the irrigation amount and the leachate weight. Since water uptake was the only unknown in the water balance of the lysimeter, it could be estimated quite accurately, and provided a reliable value for the field experiment. The potential evapotranspiration (ET0) in the experimental plots was calculated using the Penman–Monteith method. Additional information is detailed elsewhere (Silber et al. 2018).
The experimental design comprised five treatments, allocated to five complete randomized blocks:
-
1.
T1—no water stress treatment irrigated according to the actual water uptake estimated in the lysimeters experiment (as described above). The daily irrigation quantities applied to T1 treatment during the four experiment years are presented in Fig. 1a and the weekly average of the calculated irrigation coefficient (KL = ET/ET0) during 2016 is presented in Fig. 1b
-
2.
T2—excessive irrigation, irrigated 150% from T1.
-
3.
T3—Regulated deficit irrigation (RDI) during the summer, the main fruit growth period. Irrigation until 1 August was as T1 trees and after that until the harvest was 75% from T1.
-
4.
T4—No irrigation and fertilization was applied during the early growth period (from January to 1 May) and after that the trees were irrigated as in T1. Note that this period include the early stages of fruit development: emergence of flower buds (mid-March), bloom (beginning of April), and fruit set (middle of April).
-
5.
T5—constant water stress treatment, irrigated 75% of T1 through the entire season.
a Daily irrigation quantities applied to T1 treatment during 2014 (red), 2015 (green). 2016 (magenta) and 2017 (blue) experimental years. Insight: accumulative quantities of irrigation and rain; b Weakly averages of the calculated irrigation coefficient (KL) calculated for T1 (green), T2 (blue), and T5 (red) treatments from March to December 2016. The recommended Kc for avocado by the Israeli Extension Service and the ET0 are presented for comparison. DOY is day of the year. (Color figure online)
The experimental set-up allowed the comparison between trees responses to: (1) three irrigation strategies during the entire growth periods: T1, T2 and T5; and (2) comparison between RDI management during the early and the late growth period (T4 and T3, respectively). During the rainy season (November–February) the irrigation rate was equivalent for all the treatments except for T4 (dashed green line in Fig. 1b). Irrigation was stopped during rainy events and was renewed as the soil water matric potential (ψ) monitored by digital tensiometers (SOILSPEC, Healesville, Australia) located at 40 cm depth below the drippers reached a value of 20 kPa. In case of rain period longer than 2 weeks, “technical” irrigation dose of 2 mm with doubled nutrient concentration was applied.
The irrigation system consisted of two lines of drippers located 20 cm on each side of the trunk, 1.6 L h−1 pressure-compensated drippers (Netafim Inc., Israel), the distance between drippers was 40 cm (irrigation rate of 1.33 mm h−1). All the horticultural treatments (pruning, pest and weeds control) were performed uniformly in all treatments according to the recommendations of the Israeli Extension Service (Noy 2006). The harvest was between 5 and 15 November each year, as fruit dry weight reached 20.5%. All the fruits from all the trees were collected and the fruits were sorted by weight, but only the fruits from the four internal trees were included in yield statistics. The trunk diameters of six trees per each irrigation treatment were recorded every 1 min by LVDT dendrometers (PhyTech Ltd., Rosh Haayin, Israel).
Midday stem water potential (SWP) was measured from February to May every week and later on every 2 weeks. Two shaded shoot ends (5–7 leaves) from the inner part of the canopy inserted while intact to zip-loc plastic bags covered by aluminium foil 90 min before measurements were taken. The leaves then were cut and inserted into a plastic bag before pressure chamber (PMS 1000, PMS, Albany OR) measurement was taken.
Fertigation solutions and plant analyzes
The nutrient solutions were prepared from commercial fertilizers ((NH4)2SO4, NH4NO3, KNO3, KCl and H3PO4), and tap water containing (mg L−1): 100–120 Ca, 30–40 Mg, 20–30 Na, and 60–70 Cl−. Micronutrient concentrations (mg L−1) applied was 0.3 Zn, 0.6 Mn, 1.0 Fe, 0.04 Cu, 0.4 B, and 0.03 Mo, all EDTA-based. Nutrients quantities for all the treatments were similar, i.e., higher concentrations for the water-stress treatments (T3 and T5) while lower concentration for T2 treatment. The absent fertilizer quantity of T4 tree during January–April was added during May, immediately after irrigation started.
The pH and EC of the fertigation solution were 7.0 ± 0.3 and 1.0 ± 0.1 dS m−1, respectively. The N, P and K concentrations in the irrigation solution were 40, 10 and 50 mg L−1, respectively.
Statistics analysis
Statistical analyses were carried out with JMP(®) 12 software. All data were analyzed for the effects of treatments by means of the general linear model procedure of SAS (SAS Institute, Cary, NC, USA). Differences among means were tested with the standard least squares mode of ANOVA, followed by Tukey HSD pair wise comparison of means. Differences with a probability larger than 95% were considered as significant.
Results
Tree development and yield
Absence of irrigation during the early growth period (January–May, T4 treatment) induced leaf-chlorosis, while healthy, dense and green leaves characterized all the other trees. The first sign of chlorosis appeared at the beginning of February and its severity significantly increased at the emergence of flower buds (mid-March) and bloom (beginning of April). Leaf chlorosis was followed by massive defoliation and later on by fruitlet abscission (May–June).
The effect of the different irrigation treatments on the first yield (2014) was not statistically significant (Table 1). The effect of the blocks and the interaction Treatment × year were not statistically significant. The effect of year was significant for fruit number and average fruit weight. The average weight of a single fruit during 2015, 2016 and 2017 harvests was not statistically affected by the irrigation treatments and was: 202, 192 and 185 g, respectively. A significant linear regression was obtained between single fruit weight and fruit number during 2015–2017 experimental years (not presented). The equation (standard error of the parameters given in parentheses) was: y = 239(6.9) − 0.16(0.014) × x (r2 = 0.97), where y is the single fruit weight and x is the fruit number.
The yield of the two no-water stress treatments (T1 and T2) during the four experimental years was very high and above the common yield in Israel (200–250 fruits per tree, 10–15 t ha−1), while the yield of the RDI and water-stress treatments (T3, T4 and T5) was significantly lower (Table 1). More important, the yield of the two no-water stress trees was relatively stable and did not present an alternate bearing pattern while the yield of water-stress trees was considerably reduced during low-crop years (2015 and 2017).
Effects of irrigation treatments on soil- and plant-water status during the early season (January–April)
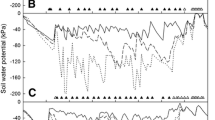
Irrigation for all the treatments except T4 started on 15 February (DOY = 46), stopped during rain events, and renewed several days after that. Note that the irrigation doses recommended by the Israeli extension service during the early fruit development: emergence of flower buds (mid-March), bloom (beginning of April), and fruit set (middle of April) were lower than the actual T1 doses as reflected by the recommended Kc in Fig. 1b (black line). Soil water tension measured by tensiometers located under the dripper at the vicinity of trees exposed to no-water stress (T1) and water stress treatments (T4) during April–May 2016 is presented in Fig. 2. The values of soil water tensions during 2015 and 2017 were similar to the values of 2016, and therefore, were not depicted. Water tension decreased sharply during rains or irrigation events (T1), as soil water content increased, and increased significantly shortly afterwards (Fig. 2, top). Water tensions in the upper soil layer (20 cm) were higher than in the lower part (40 and 60 cm), but the trend was almost the same. In the absence of irrigation, the soil water tension of T4 trees exceeded 60 (± 8.6), 20 (± 2.5) and 10 (± 0.9) kPa in soil depth of 20, 40 and 60 cm beneath the dripper, respectively, but decreased sharply in response to rain events (Fig. 2, bottom). From mid-March (DOY 75) the soil water tension in the upper layer of T4 trees was above 80 (± 11.5) kPa while in the middle and the bottom layers (40 and 60 cm, respectively) the water tension were lower but clearly pointed towards drying conditions, i.e., increasing water tensions with time (Fig. 2 bottom). Several days after starting the irrigation for T4 trees (1 May), the water tension decreased sharply and was almost similar to T1 trees (not presented). Soil water tensions of T2, T3 and T5 trees were not significantly different from T1 trees during April–May 2016, and therefore, were not shown.
Soil water tension measured by tensiometers located at different depth (red—20, green—40 and blue—60-cm) under dripper in the vicinity of the trunk and rain events during February–May 2016. Top: non-water stress treatment (T1); bottom: water-stress treatment (T4). DOY is day of the year. (Color figure online)
Midday SWP values of no water stress (T1) and the RDI treatment during the early growth period (T4) trees measured 3 days after starting the irrigation for T1 (DOY = 49,) were relatively high (− 0.71 and − 0.82 MPa for T1 and T4, respectively) but increased in the coming days to, even for the non-irrigated trees (Fig. 3). However, in very dry days where ET0 was 9 mm/day (20 April, DOY = 111) the SWP value of T1 and T4 trees decreased to − 0.85 and − 0.95 MPa, respectively.
Midday stem water potential (SWP) of non-(T1) and water-stress trees (T4) during the first growth period of 2016. Vertical lines designate standard error values. ** and *** designates Prob > F of: 0.001 > and 0.0001 >, respectively. The values of ET0 are presented for comparison. DOY is day of the year
Maximum daily trunk shrinkage (MDS) of T1 and T4 trees during April–May 2016 are presented in Fig. 4. MDS of 2015 and 2017 years were similar to 2016, and therefore, these data were not presented. MDS values of both treatments were low (below 150 µm) and almost similar as long as ET0 were below 5 mm/day (Fig. 4). However, in hot and dry days characterized by high evaporative demand (ET0 above 7 mm day−1) the MDS increased sharply and closely followed the ET0 trend. MDS values of T4 trees from 20 April until 1 May (DOY = 111–122) were statistically higher than the MDS of T1 trees (Prob > F less than 0.05). MDS of T4 trees after commencing the irrigation (1 May) were almost similar to T1 trees (Fig. 4). The MDS values of the excessive irrigation (T2), RDI during the summer (T3) and constant water stress (T5) trees were not significantly different from T1 trees, and therefore, not presented.
Effects of irrigation treatments on soil- and plant-water status during the summer (June–November)
The averaged values of soil water tension at a depth of 40 cm beneath the dripper at the vicinity of the trunk of no-water stress trees (T) during July–September 2015–2017 were low (below 10 kPa, not presented). Excessive irrigation (T2 trees) induced lower tension (below 8 kPa), while water-stressed trees (T5 trees) induced values higher than 10 kPa during June to above 20 kPa at the end of September. Water tension in the upper (20 cm depth) and in the deeper layer (60 cm depth) of each irrigation treatment were higher or lower, respectively. Reducing irrigation rate on 1 August (T3 treatment) almost did not affect soil water tension in the upper 20-cm layer but induced a continuous increase in the middle 40- and the deep 60-cm layers (only data for 2016 are presented in Fig. 5). The reference evapotranspiration (ET0) during June–October 2016 was generally stable without extreme events characterized in April–May season (Fig. 6). The effect of irrigation treatments on midday SWP during April–October 2016 was statistically significant and followed the trend T5 > T1 > T2 (Fig. 6). Since June 1st (DOY = 153), SWP values became more negative, in opposition to the ET0 values that decreased since then, and reached a peak on 8 August (DOY = 221) (Fig. 6). Soil water potential of the summer RDI treatment (T3) exhibit clear dehydration phenomena (Fig. 5), nevertheless, the SWP values from August 1st (DOY = 221) to middle October (DOY 288) were not statically different from the no water stress treatment (T1, Fig. 6).
Maximum daily shrinkage (MDS) of no water stress (T1), RDI during the summer (T3) and constant water stress (T5) trees during June–December 2016 are presented in Fig. 7. MDS of 2015 and 2017 years were similar to 2016, and therefore, not presented. MDS values at the beginning of June were relatively low (below 200 µm) but consistently increased during June–August, reached a peak of 200, 249 and 289 µm for T1, T3 and T5, respectively. Note that this trend was opposite to the decreased trend of ET0 values but in accord with the SWP trend presented in Fig. 6. The MDS values of the constant water stress trees (T5) significantly deviated from the no water stress trees (T1) from 7 June until the harvest (10 November, DOY = 315). MDS of the summer RDI trees (T3) was almost similar to T1 until 1 August, but increased as irrigation doses decreased and significantly deviated from the no-stress trees around end of August (DOY = 243, Fig. 7). The MDS increases of T3 trees was in accord with the soil-dehydration phenomena presented in Fig. 5 but in conflict with the SWP data presented in Fig. 6. Exceptional meteorological conditions during 19–29 November 2016 (very low humidity and high ET0) induced a sharp MDS increase that reduced as ET0 decreased back to normal values for the season (Fig. 7). MDS of T4 trees was similar to T1 trees while that of T2 was consistently lower (20–40 µm, not presented).
Discussion
The yield of the no water stress treatments during the four experimental years was very high above the common yield in Israel, while the yield of the RDI (T3 and T4) and water-stress (T5) treatments was significantly lower. Furthermore, the fruit number of the no water stress trees (T1 and T2) was not susceptible to alternate bearing while the fruit number of water-stressed trees was reduced during off-crop years (Fig. 8).
The primary and substantial role of irrigation is to supply water for plant development yet, in view of the significant role of irrigation management on nutrient availability, the mutual association between water and nutrient should be anticipated. The energy cost for producing avocado fruit is very high (8072 KJ kg−1), almost triple from that of sugar fruits such as apple and orange (Wolstenholme 1986). Therefore, improvement of water and nutrient availability, especially in periods where the activity of the root system was weak as a result of low carbohydrates supply presumably played a dominant role in seeds or fruit function (Silber et al. 2012).
Stress condition during the early growth season
During the rainy season (January–March) irrigation events according the common practice in Israel are rare because the soil–water content is generally high and both air and soil temperature are low (10–15 °C). Nevertheless, nutrient demand, especially nitrogen, phosphorus and zinc by the developing organs (flower buds and later on flowers and fruitlets) is very high. Earlier research clearly demonstrated that water stress during the early period of fruit growth (March–April) induced later on fruitlet abscission and accordingly, reduced avocado yield (Silber et al. 2012). Absence of fertilization during the winter period induced leaf-chlorosis, followed by defoliation and later on fruitlet abscission (Silber et al. 2018). Therefore, special effort was made in the present study to supply water and nutrients according to the actual plant demand, especially before the inflorescence period (February–March). The visual resemblance between T4 trees in the present study and the non-fertilized trees in Silber et al. (2018) study may indicate that nutrient deficiency can play an important role on yield decreases.
The earliest indication for stress signal on non-irrigated and fertilized trees (T4) arrived from trunk growth rate (TGR) measurements (Fig. 9). TGR of T4 trees significantly deviated from no water stress (T1) trees as early as 15 March (DOY = 75). Except typical fluctuations, the TGR values increased from January to 10 June, and later on decreased (Fig. 9). It is commonly accepted that TGR fluctuations during the growth season follow seasonal changes in partitioning of carbohydrates between reproductive and vegetative organs, as previously suggested for avocado by Ploetz et al. (1991), Whiley and Wolstenholme (1990) Wolstenholme and Whiley (1989, 1997) and Silber et al. (2013a). Based on the TGR measurements, it is suggested that the window opportunity for avocado root growth ended at the beginning of June, as fruit demand for carbohydrates amplify and accordingly, the carbohydrate quantities allocated to the roots are drastically reduced (Whiley and Wolstenholme 1990). Variation in soil water content (θ) may have considerable effects on plant-nutrient availability as detailed in the “Introduction”. Subsequently, it is reasonable to assume that nutrient deficiency caused by absence of fertilization in addition with a decrease in soil-nutrient availability induced by the decrease in soil–water content generated sharp decreases on fruit number (Table 1).
Stress condition during the main growth season
Intermittent or constant water stress during the main growth season (T3 and T5, respectively) decreased yield (Table 1), mainly because the decreases of fruit number. The number of fruits of constant water stress treatment (T5) was lower from the no-water stress treatments (T1) in 2015, 2016 and 2017 experimental years by 44, 49 and 153 fruits per tree, respectively, while that of T3 by 102, 56 and 133, respectively. Furthermore, the effect of constant water stress throughout the all growth period (T5) on ‘Hass’ avocado yield was less severe than shorter water stress periods applied during summer (T3). This conclusion might result from tree adaptation to conditions of constant water deficiency by decreasing the vegetative part and reducing plant evapotranspiration.
SWP measurements of T3 trees were almost similar to that of T1 trees (Fig. 6) while MDS measurements exhibited clear differences after the decrease of irrigation doses (Fig. 7), in accord with the tensiometer readings (Fig. 5). SWP measurements of T5 trees were significantly more negative than that of T1 trees (Fig. 6), but except for 10 August (DOY = 223) the measurement were below the water stress threshold of − 0.8 MPa (Fig. 6). MDS measurements of T5 trees from 5 July (DOY = 187) were above the threshold of 200 µM (Fig. 7) which may designate the higher sensitivity of MDS measurements to water stress.
Leaf-carbohydrate play an important role in the complex framework of stomata aperture in avocado trees (Silber et al. 2013b). The effect of fruit sink on water uptake may explain the apparent discrepancy between the decreased trends of the ET0 values and the consistent increase of the MDS depicted in Fig. 7. Measurements of stomatal conductance, photosynthesis and water uptake in an earlier research included fruiting and de-fruited avocado trees clearly demonstrated the significant role of fruit load on gas-exchange properties and water management (Silber et al. 2013b). Despite higher vegetative growth and leaf area index of the de-fruited trees, their daily water uptake was 40% lower than that of fruiting trees, mainly because of accumulation of leaf-carbohydrates in the absence of strong sink. Therefore, crop load should play an important role on irrigation scheduling. Thus, the traditional ‘KCET0’ approach (Allen et al. 1998) might be insufficient as a basis for proper irrigation management during the productive period and irrigation management should be adjusted, therefore, for the actual crop load.
Agronomic implications
The most important difference between the recommended Kc for avocado by the Israeli Extension Service and the calculated KL for the no water stress treatment (T1) appeared from the end of February until beginning of June (DOY 160 represent the 8 June, Fig. 1b). This period include the flowering and fruit-set stages and the “window opportunity” for root development (15 March–10 June, Fig. 9). Thus, special effort should be made to fulfil the periodic demand for water and nutrients during the inflorescence and fruit-set period and in case of long rainy period, “technical irrigation” i.e., irrigation with high concentration of nutrient to prevent nutrient deficiency (Silber et al. 2018) should be applied. Irrigation rates and the calculated KL for the no water stress treatment (T1) depicted in Fig. 1 may serve as a reasonable guide for irrigation management. A linear regression was obtained between the calculated KL for the no water stress treatment and time (days) from March to November 2016. The equation (standard error of the parameters given in parentheses) was: y = 0.49(0.026) + 0.0013(0.00012) × x (r2 = 0.87). The sensitivity of avocado trees to water stress during mid-June to the end of the summer (October–November) is less severe than that of March–June and applying no water stress treatment such as T1 and KL value of 0.8–0.9 illustrated in Fig. 1b may be favorable.
Plant-based methods such as dendrometers and\or SWP should be used in avocado orchards for controlling irrigation scheduling. SWP is a widely accepted method but slow and labor intensive (Jones 2004) and therefore, problematic for use. Automatic and on-line and measurements are the main advantage of the dendrometer method, and therefore, easy for horticultural uses (Fernández et al. 2011). Analyses of trunk diameter variation data that lead to evaluation of TGR and the MDS reflect phenological stages and periodicity of shoot, fruit and root growth, and also may provide an integrative, “holistic viewpoint” of overall tree status (Silber et al. 2013a). Moreover, the sensitivity of both, TGR and MDS measurements are very high. TGR data early indicated early effects of water stress on tree development (Fig. 9) and MDS measurements exhibited clear differences between RDI during the summer (T3) and no water stress (T1) trees after the decrease of irrigation doses (Fig. 7), while SWP data were less susceptible. Soil-based method such as tensiometer may be adequate too but requires numerous sensors for covering the whole wetted soil volume under the dripper. Furthermore, water retention curve for each soil type is necessary to transform from pressure units (kPa) to volumetric water units (ml or liter).
Conclusions
-
1.
Optimal irrigation managements is crucial for increasing ‘Hass’ avocado yield. Special effort should be made to provide high demand for water and nutrient during the inflorescence and fruit-set periods. In case of rain period longer than 2 weeks, “technical” irrigation, i.e., irrigation with high concentration of nutrient to prevent nutrient deficiency should be applied. For the main irrigation season (March–November) the irrigation rates and the calculated KL for the no water stress treatment (T1) may serve as a reasonable guide for irrigation management. The validity of the linear equation KL = 0.49 + 0.0013 × days (from 1 January) should be examined as a guide for irrigation management in different climatic conditions.
-
2.
The yield of no water stress trees was not susceptible to alternate bearing while the yield of water-stressed trees was considerably reduced during off-crop years.
-
3.
Analyses of trunk diameter variation (TDV) data that lead to the evaluation of trunk growth rate (TGR) and maximum daily shrinkage (MDS) reflect phenological stages and periodicity of shoot, fruit and root growth, and also may provide an integrative, “holistic viewpoint” of overall tree status.
-
4.
The window opportunity for root growth ended at the beginning of June, as fruit demand for carbohydrates amplify and the rate of root growth decline.
-
5.
Fruit load should be taken into account while planning irrigation and fertilization management.
-
6.
Constant water stress along the growth period on ‘Hass’ avocado yield was less severe than short water stress applied in summer, during the growth period, probably because of tree adaptation by decreasing the vegetative part and reducing plant evapotranspiration.
References
Adato I, Gazit S (1977) Role of ethylene in avocado fruit development and ripening. J Exp Bot 28:636–643
Allen RG, Pereira LS, Raes D, Smith M (1998) Crop evapotranspiration—guidelines for computing crop water requirements—FAO Irrigation and drainage paper 56. UN-FAO, Rome
Assouline S (2001) A model of soil relative hydraulic conductivity based on water retention curve characteristics. Water Resour Res 37:265–271
Campisi-Pinto S, Zheng Y, Rolshausen PE, Crowley DE, Faber B, Bender G, Bianchi M, Khuong T, Lovatt CJ (2017) Optimal nutrient concentration ranges of ‘Hass’ avocado cauliflower stage inflorescences—potential diagnostic tool to optimize tree nutrient status and increase yield. HortScience 52:1707–1715
Carr MKV (2013) The water relations and irrigation requirements of avocado (Persa americana Mill.): a review. Expl Agric 49:256–278. https://doi.org/10.1017/S0014479712001317
Claassen N, Steingrobe B (1999) Mechanistic simulation models for a better understanding of nutrient uptake from soil. In: Rengel Z (ed) Mineral nutrition of crops, fundamental mechanisms and implications. Haworth Press, New York, pp 327–369
Fernández JE, Torres-Ruiz JM, Diaz-Espejo A, Montero A, Álvarez R, Jiménez MD (2011) Use of maximum trunk diameter measurements to detect water stress in mature ‘Arbequina’ olive trees under deficit irrigation. Agric Water Manag 98:181–1821
Garner LC, Lovatt CJ (2008) The relationships between flower and fruit abscission and alternate bearing of ‘Hass’ avocado. J Am Soc Hortic Sci 133:3–10
Holzapfel E, de Souza JA, Jara J, Carvallo GuerraH (2017) Responses of avocado production to variation in irrigation levels. Irrig Sci. https://doi.org/10.1007/s00271-017-0533-0
Jones HG (2004) Irrigation scheduling: advantages and pitfalls of plant-based methods. J Exp Bot 55:2427–2436
Kool D, Agam N, Lazarovitcha N, Heitmanc JL, Sauerd TJ, Ben-Gal A (2014) A review of approaches for evapotranspiration partitioning. Agric For Meteorol 184:56–70
Lahav E, Zamet D (1999) Flowers, fruitlets and fruit drop in avocado trees. In: Proceedings of IV world avocado congress, Mexico, pp 95–100
Lahav E, Whiley AW, Turner DW (2013) Irrigation and mineral nutrition. In: Schaffer B, Wolstenholme BN, Whiley AW (eds) The avocado: botany, production, and uses, 2nd edn. CBI Pub., New York, pp 301–341
Lovatt CJ (2011) Alternate bearing of ‘Hass’ avocado. Calif Avocado Soc 2010 Yearb 93:125–140
Mualem Y (1986) Hydraulic conductivity of unsaturated soils, predictions and formulas. In: Klute A (ed) Methods of soil analysis, Agronomy monograph, vol 9, chapter 31. American Society of Agronomy and Soil Science Society of America, Madison, USA, pp 799–823
Noy M (2006) Recommendations for growing of avocado. Extension Service, Ministry of Agriculture, Israel
Ploetz RC, Ramos JL, Parrado JL, Shepard ES (1991) Shoot and root growth cycles of avocado in south Florida. In: Proceedings of Florida state horticultural sciences, vol 104, pp 21–24
Schaffer B, Wolstenholme BN, Whiley AW (2013) Introduction. In: Schaffer B, Wolstenholme BN, Whiley AW (eds) The avocado: botany, production, and uses, 2nd edn. CBI Pub., New York
Silber A, Israeli Y, Levi M, Keinan A, Shapira O, Chudi G, Golan A, Noy M, Levkovitch I, Assouline S (2012) Response of ‘Hass’ avocado trees to irrigation management and root constraint. Agric Water Manag 104:95–103
Silber A, Naor A, Israeli Y, Assouline S (2013a) Combined effect of irrigation regime and fruit load on the patterns of trunk-diameter variation of ‘Hass’ avocado at different phenological periods. Agric Water Manag 129:87–94
Silber A, Israeli Y, Levi M, Keinan A, Chudi G, Golan A, Noy M, Levkovitch I, Narkis K, Naor A, Assouline S (2013b) The roles of fruit sink in the regulation of gas exchange and water uptake: a case study for avocado. Agric Water Manag 116:21–28
Silber A, Naor A, Cohen H, Bar-Noy Y, Yechieli N, Levi M, Noy M, Peres M, Duari D, Narkis K, Assouline S (2018) Avocado fertilization: matching the periodic demand for nutrients. Sci Hortic 241:231–240
Slabbert MJ (1981) Flower and fruit drop. S Afr Avocado Growers Assoc. Yearb 4:89–91
Whiley AW, Wolstenholme BN (1990) Carbohydrate management in avocado trees for increased production? S Afr Avocado Growers Assoc. Yearb 13:25–27
Whiley AW, Chapman KR, Saranah JB (1988) Water loss by floral structures of avocado (Persea americana cv. Fuerte) during flowering. Aust J Agric Res 39:457–467
Wolstenholme BN (1986) Energy costs of fruiting as a yield-limiting factor with special reference to avocado. Acta Hortic 175:121–126
Wolstenholme BN (1987) Theoretical and applied aspects of avocado yield as affected by energy budgets and carbon partitioning. In: Proceedings of I world avocado congress, Pretoria, S. Afr. Avocado Grower’s Assoc. Yrbk., vol 10, pp 58–61
Wolstenholme BN, Whiley AW (1989) Carbohydrate and phenological cycling as management tools for avocado orchards. S Afr Avocado Growers Assoc. Yearb 12:33–37
Wolstenholme BN, Whiley AW (1997) What do carbohydrate reserves tell us about avocado orchard management? S Afr Avocado Growers Assoc. Yearb 20:63–67
Wolstenholme BN, Whiley AW, Saranah JB (1990) Manipulating vegetative: reproductive growth in avocado (Persea americana Mill.) with paclobutrazol foliar sprays. Sci Hortic 41:315–327
Ziv D, Zviran T, Zezak O, Samach A, Irihimovitch V (2014) Expression profiling of FLOWERING LOCUS T-like gene in alternate bearing ‘Hass’ avocado trees suggests a role for PaFT in avocado flower induction. PLoS One 9(10):e110613. https://doi.org/10.1371/journal/pone/0110613
Acknowledgements
The authors are grateful to the anonymous reviewer for critical readings of the article and the constructive comments. This research was supported by the Israeli Board of Avocado.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. López-Urrea.
Publisher's Note
Publisher's Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Silber, A., Naor, A., Cohen, H. et al. Irrigation of ‘Hass’ avocado: effects of constant vs. temporary water stress. Irrig Sci 37, 451–460 (2019). https://doi.org/10.1007/s00271-019-00622-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00271-019-00622-w