Abstract
Background
Two-thirds of patients undergoing liver resection for intrahepatic cholangiocarcinoma experience recurrence after surgery. Our aim was to identify factors associated with early recurrence after resection for intrahepatic cholangiocarcinoma.
Methods
Patients with intrahepatic cholangiocarcinoma undergoing curative intent resection (complete resection and lymphadenectomy) were included in two centers between 2005 and 2021 and were divided into three groups: early recurrence (< 12 months after resection), delayed recurrence (> 12 months), and no recurrence. Patients experiencing early (< 90 days) postoperative mortality were excluded.
Results
Among 120 included patients, 44 (36.7%) experienced early recurrence, 24 (20.0%) experienced delayed recurrence, and 52 (43.3%) did not experience recurrence after a median follow-up of 59 months (IQR: 26-113). The median recurrence-free survival was 16 months (95% CI: 9.6–22.4). Median overall survival was 55 months (95% CI: 45.7–64.3), while it was 25 months for patients with early recurrence (p < 0.001). Patients with early recurrence had significantly larger tumors (59.1% of tumors > 70 mm in early vs. 58.3% in delayed vs. 26.9% in no recurrence group, p = 0.002), multiple lesions (65.9% vs. 29.2% vs. 11.5%, p < 0.001), and positive lymph nodes (N +) (38.6% vs. 37.5% vs. 11.5%, p = 0.005). In multivariable analysis, presence of multiple lesions (OR: 9.324; 95% CI: 3.051–28.489; p < 0.001) and positive lymph nodes (OR: 3.307. 95% CI: 1.001–11.011. p = 0.05) were associated with early recurrence.
Conclusion
Early recurrence after curative resection of intrahepatic cholangiocarcinoma is frequent and is associated with the presence of multiple lesions and positive lymph nodes, raising the question of surgery’s futility in this context.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intrahepatic cholangiocarcinoma (ICC) represents 20% of all biliary malignancies which are the second most frequent primary liver tumors [1]. Incidence of ICC has increased during the past decades [2]. ICC prognosis is poor as it is most usually diagnosed at an advanced stage due to absence of symptoms and rare development on pre-existing chronic liver disease. As a consequence, complete surgical resection (with negative surgical margins) and regional lymphadenectomy [3] are feasible for only 30% of patients [4]. Moreover, postoperative morbidity and mortality after ICC resection are high usually due the extension of the resected liver volume, given the large diameter of the tumors, and the frequent underestimation of biliary and vascular extension necessitating reconstruction [5].
So far, no neoadjuvant chemotherapy protocol has demonstrated any benefit on recurrence and overall-free survival for initially resectable ICC [6]. Moreover, despite complete resection and administration of adjuvant capecitabine [7], two-thirds of ICC patients will experience disease recurrence [8], with more than 60% representing local liver recurrences and more than 50% occurring within the first 12 months [9]. Since modern chemotherapy protocols in advanced ICC provide a median overall survival of 12 months [10], the question of surgery’s futility should be raised when disease recurrence occurs within 12 months after a curative intent surgery.
The aim of the present study was to identify factors associated with early recurrence after complete surgical resection of ICC.
Methods
Study population
All consecutive patients who underwent curative intent surgical resection of ICC from January 2005 to May 2021 at Beaujon Hospital, Clichy, France and Cochin Hospital, Paris, France were retrieved from prospectively maintained databases.
Patients with ICC considered as initially resectable after a complete preoperative evaluation including both multiphasic CT-scan and magnetic resonance imaging (MRI) were selected. Resectability was evaluated and validated in a multidisciplinary board meeting and was defined as the possibility to perform a negative margin resection while preserving an adequate future liver remnant. All patients undergoing a curative intent surgery with associated lymphadenectomy were finally included. Patients with macroscopic positive resection margins, without associated lymphadenectomy or experiencing early postoperative mortality (< 90 days) were excluded from the analysis. This study was approved by the institutional review board and conducted in accordance with the Declaration of Helsinki.
Surgical technique, postoperative outcomes and histological analysis
All operative procedures were performed by at least one senior liver surgeon. Liver resection was either performed by laparotomy or laparoscopy. Parenchymal transection was performed using the ultrasonic dissector (Thunderbeat®, Olympus Co, Tokyo, Japan) when performed under laparoscopy and the Cavitron Ultrasonic Surgical Aspirator (CUSA®) when performed under laparotomy. Pringle maneuver and hepatic pedicle clamping were not routinely performed but were used in cases of liver parenchymotomy bleeding. Extent of liver resection was defined according to the Brisbane classification of liver resection [11] and major hepatectomy was defined as a resection of 3 or more contiguous segments. All patients underwent a lymphadenectomy of the hepatoduodenal ligament and coeliac region. Patients who did not undergo lymphadenectomy were excluded from the analysis.
Operative time, intraoperative blood loss and transfusion were recorded. Postoperative morbidity and mortality were evaluated within 90 days after surgery using the Dindo-Clavien classification [12] with severe complications defined as a grade 3 or more. Postoperative liver failure rate and postoperative bile leak were defined according to the International Study Group of Liver Surgery [13, 14]. Tumor classification was described according to the TNM-AJCC-UICC classification [15] and ICC with more than one lesion or satellite nodules was considered as multiple lesions (T2 stage tumor). On histological analysis, R0 resection margin was defined as a margin of at least 1 mm distance from the tumor, while R1 as a distance of less than 1 mm or in case of direct microscopic invasion. R2 patients, defined by macroscopical invasion of the resection margins, were excluded from the analysis.
Follow-up and definition of recurrence
Patients were followed up according to national French surveillance protocols [15], including physical examination, liver function tests and CA 19–9 testing and finally multiphasic thorax/abdominal/pelvic computed tomography (CT), performed every 3 months for 2 years and every 6 months for the following 3 years.
Adjuvant chemotherapy was considered for all patients and was decided after a multidisciplinary board meeting. Until February 2014, gemcitabine + oxaliplatin were administered for 6 months to all patients as part of the PRODIGE 12-accord 18-unicancer GI trial [16]. Since the results of the BILCAP study, launched in 2019 [3], patients received capecitabine for 6 months.
Disease recurrence was defined as an intrahepatic or extrahepatic lesion being suspicious on cross-sectional imaging. Time to recurrence was defined as the time separating the operation and first imaging revealing a new lesion. Early recurrence was defined as any recurrence occurring within 12 months after liver resection [17]. Disease recurrence was either treated by chemotherapy alone or combined with surgery or radiofrequency ablation when accessible to complete resection, or by best supportive care in case of advanced metastatic disease. Finally, overall survival (OS) defined from the time of surgery to the time of death or most recent follow-up was calculated.
Study Design and statistical analysis
Patients were divided into three groups according to presence or absence of recurrence and timing of recurrence: patients who had no recurrence (NoR Group), patients experiencing early recurrence (ER Group) and patients with delayed recurrence (DR Group).
All data analyses were performed using SPSS 23.0 statistical software (SPSS, Inc., Chicago, IL, USA). Categorical variables were compared using χ2 test or Fischer’s exact test when appropriate. Continuous data are expressed as mean ± standard deviation (SD) or medians with interquartile range (IQR) and were compared using the Mann–Whitney test or Kruskal–Wallis test as appropriate. A p value of less than 0.05 was considered as statistically significant. Multivariable analysis by multinomial logistic regression was performed to determine independent clinical risk factors for early recurrence and death within 12 months after surgery.
Results
Study population
From January 2005 to May 2021, 190 consecutive patients operated for ICC were considered for inclusion. Among all 190 selected patients, 43 (22.6%) without lymphadenectomy and 6 (3.2%) with incomplete resection macroscopically (R2) were excluded. Twenty-one patients (11.1%) experiencing early (< 90 days) postoperative death were also excluded from the study. Therefore, the selected population included 120 ICC patients with complete surgical resection and lymphadenectomy (Fig. 1). Among these patients, 52 (43.3%) did not experience any recurrence (NoR), while 44 (36.7%) experienced early recurrence (ER) and 24 (20.0%) experienced delayed recurrence (DR).
Baseline characteristics, intraoperative and postoperative data
The distribution of patient baseline characteristics, intraoperative and postoperative data are presented in Tables 1 and 2. Among the 120 patients selected, 66 (55.0%) patients were female and mean age was 63.0 ± 11.5 years. Overall, 93 (77.5%) patients underwent a major liver resection. Patients with ER underwent more frequently a major liver resection than those without (61.5% vs. 90.9% vs. 87.5% in the NoR, ER and DR groups, respectively, p = 0.001). However, no difference was found between groups regarding postoperative complications (Table 2). After resection, 41 patients (34.2%) received adjuvant chemotherapy with ER and DR patients receiving more frequently adjuvant chemotherapy compared with the NoR group (21.1% vs. 40.9% vs. 50.0% in NoR, ER and DR groups, respectively, p = 0.028).
Histological findings
Mean tumor size was 65.5 ± 38.2 mm, 32 (26.7%) patients had positive lymph nodes, and 95 (79.2%) had R0 margin on final pathological examination. Patients with recurrence had significantly larger tumors (with 26.9% of tumors > 70 mm in the NoR group, 59.1% in the ER group and 58.3% in the DR group, p = 0.002), more frequently multiple lesions (11.5% vs. 65.9% vs. 29.2% in the NoR, ER and DR groups, respectively, p < 0.001) and more frequently associated with positive lymph node status (11.5% vs. 38.6% vs. 37.5% in the NoR, ER and DR groups, respectively, p = 0.005). Histological findings are described in Table 3.
Disease recurrence analysis, prognostic factors and long-term outcomes
After a median follow-up of 59 months (IC 95% 61.5–96.4), 68 patients experienced disease recurrence, while 80.9% of these patients developed disease recurrence within 24 months after resection. Median time to recurrence was 7.3 months (IQR 4-9) in the ER group and 29.2 months (IQR 18-43) in the DR group. Recurrence patterns and treatment according to the delay of recurrence are described in Table 4. Both ER and DR groups had similar disease recurrence patterns, principally characterized by exclusive intrahepatic recurrence (34.1% and 37.5%, respectively, p = 0.412) and followed by exclusive extrahepatic recurrence (25.0% and 20.8%, respectively, p = 0.973) or both intra- and extrahepatic recurrence (29.5% and 16.7%, respectively, p = 0.394). Disease recurrence was most frequently treated by chemotherapy alone (52.3% in the ER vs. 45.8% in the DR group, p = 0.569), combination of chemotherapy and surgery (15.9% vs. 4.2% in the ER and DR group, respectively, p = 0.569) or best supportive care (15.9% vs. 12.5% in the ER and DR group, respectively, p = 0.569) due to extensive metastatic disease.
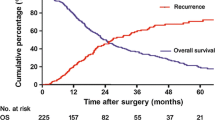
Median DFS was 16 months (95% CI 9.6–22.4) and median OS was 55 months (95% CI 45.7–64.3) in the whole population. Median OS was significantly shorter in the ER group (25 months (95% CI 13.0–36.9)) compared with the other groups (73 months (95% CI 51.7–94.3)) (p < 0.001). ER patients who died within 12 months after surgery (18.4%) had T4 stage tumors in 22.2% and positive lymph nodes in 55.5% of the cases. Disease recurrence for ER patients was short (median DFS 3.44 ± 1.3 months) and mostly extrahepatic (66.7%).
On multivariable analysis, among the different clinical and pathological variables analyzed as potential prognostic factors of disease recurrence, factors associated with ER were satellite lesions (OR: 9.324; 95% CI: 3.051–28.489; p < 0.001) and lymph node involvement (OR: 3.307. 95% CI: 1.001–11.011. p = 0.05) (Table 5). Moreover, lymph node involvement was the only predictor of death within 12 months following surgery (OR = 12.700; 95% CI: 1.801–89.552; p = 0.011) (Table 6).
Discussion
Despite optimal treatment including both complete surgical excision and adjuvant chemotherapy, prognosis of patients with ICC remains poor, due to high rate of disease recurrence [18]. The present series shows that 36.7% of patients undergoing curative intent surgery experienced ER and not surprisingly this latter was associated with dismal prognosis. Previous studies have established a 24-month cutoff after resection to differentiate early and delayed recurrence [19]. However, this cutoff described empirically is based on the definition employed in the setting of HCC [20] and aims to discriminate disease recurrence related to tumor characteristics or de novo tumor development related to underlying liver disease, i.e., cirrhosis. In the case of ICC, disease recurrence after 24 months is rare, and most patients will experience recurrence within the first 2 years after surgery. Indeed, a recent study of Wang et al. [17] determined that the optimal cutoff time point to define early and delayed recurrence in ICC after curative resection was 12 months. In our study, we used the same cutoff in order to investigate the futility of surgery in the setting of ICC. We thus observed that 82.6% of patients experienced disease recurrence within 24 months after surgical resection and ER patients within a median interval time of 6 months.
In this study, patients who experienced ER had more aggressive tumors, as evidenced by the rate of poor differentiation and presence of multinodularity, as well as the higher rate of adjuvant chemotherapy administered to these patients. Moreover, patients with ER had larger tumors compared with NoR patients although not statistically larger than DR patients. Indeed, large ICCs are more frequently associated with satellite lesions [21, 22]. Multifocal tumors were described in the present series as the presence of multiple confluent lesions or satellite lesions. Multifocal ICC lesions are associated with poorer prognosis [17, 19] as is the case of multiple hepatocellular carcinomas, associated with more frequent tumor recurrence, whatever the type of multifocality (satellite nodules or multiple lesions) [23]. In our study, presence of multiple lesions was independently associated with ER and is thus related with a greater liver tumor burden [24].
Our results also confirmed that more than 30% of patients with ER presented exclusive intrahepatic disease recurrence [8]. On the other hand, patients with multiple lesions presented more frequently extrahepatic recurrence, which raises the possibility that satellite nodules could be equivalent to early stage intrahepatic synchronous metastases. In TNM-UICC-AJCC classification [15], multifocal disease only classified the primitive lesion as T2. Multiple lesions however seem associated with tumor aggressiveness, and as a consequence with poorer prognosis. It could be more clinically accurate to define multiple ICC as metastatic, such as for hilar cholangiocarcinomas [15], but this should be evaluated with a comparative study evaluating oncological outcomes between those patients and extrahepatic metastatic patients treated with chemotherapy only.
Just as the presence of multiple tumors is a strong determinant of survival, presence of positive lymph nodes, reported in 45–65% of patients with ICC, has a significant prognostic impact [25] and is a well-known factor associated with the development of extrahepatic metastases and low survival [26]. Indeed, in our study, we observed that positive lymph node status was independently associated with early disease recurrence (OR: 3.883. 95% CI: 1.115–13.523. p = 0.033) and death (OR: 9.189; 95% CI: 1.516–55.689; p = 0.016) within 12 months after surgery. Thus, presence of both multiple tumors and positive lymph node status may question the futility of surgery for these patients and indicate a neoadjuvant chemotherapy. In our study, patients who died within 12 months had more extrahepatic recurrence (66.7%). However, one of the main limitations in the preoperative diagnosis of lymph node invasion is the low diagnostic accuracy of MRI, CT-scan and PET-scan imaging [27, 28], usually necessitating hepatic pedicle lymphadenectomy in order to ensure precise lymph node staging.
Our results suggest that, given the importance of lymph node status and multiple lesion tumors on disease prognosis [25, 29] as well as their potential impact on OS [30, 31], it may be preferable to consider performing lymph node dissection and frozen section analysis before any liver resection is performed. If presence of positive lymph nodes is confirmed, interruption of surgery should be considered and a neoadjuvant (or palliation) treatment should be initiated in order to select the best candidates for a future curative liver resection according to treatment response. Indeed, however, no randomized study has been conducted yet, the rationale of neoadjuvant chemotherapy with a downstaging intent exists in ICC, including systemic and transarterial therapies [32].
The limitations of our study are its retrospective nature covering a long period, during which surgical techniques and imaging modalities have evolved. Moreover, it can imply time lead bias regarding follow-up and management protocols, especially in regards to chemotherapy regimens, which have evolved throughout the study period. Nevertheless, it was performed by two high-volume tertiary French centers following common practices. Finally, the present study failed to assess some preoperative patient-related factors, such as CA 19–9 level, which could have impacted ICC prognosis.
In conclusion, one-third of patients with ICC undergoing curative intent surgery experienced disease recurrence within 12 months (ER). ER was associated with the presence of multiple lesion tumors and positive lymph nodes. Given the poor prognosis of this group of patients, futility of extended liver resections should be put in question. Neoadjuvant or palliation treatment may be of interest and should be considered in these situations.
References
Lepage C, Cottet V, Chauvenet M et al (2011) Trends in the incidence and management of biliary tract cancer: a French population-based study. J Hepatol 54(2):306–310
Wu L, Tsilimigras DI, Paredes AZ et al (2019) Trends in the incidence, treatment and outcomes of patients with intrahepatic cholangiocarcinoma in the USA: facility type is associated with margin status, use of lymphadenectomy and overall survival. World J Surg 43(7):1777–1787. https://doi.org/10.1007/s00268-019-04966-4
Farges O, Fuks D, Boleslawski E et al (2011) Influence of surgical margins on outcome in patients with intrahepatic cholangiocarcinoma: a multicenter study by the AFC-IHCC-2009 study group. Ann Surg 254(5):824–829
Doussot A, Jarnagin WR, Azoulay D et al (2019) Improving actual survival after hepatectomy for intrahepatic cholangiocarcinoma-still a long way to go. Hepatobiliary Surg Nutr 8(2):161–163
Reames BN, Ejaz A, Koerkamp BG et al (2017) Impact of major vascular resection on outcomes and survival in patients with intrahepatic cholangiocarcinoma: a multi-institutional analysis. J Surg Oncol 116(2):133–139
Sutton TL, Billingsley KG, Walker BS et al (2021) Neoadjuvant chemotherapy is associated with improved survival in patients undergoing hepatic resection for intrahepatic cholangiocarcinoma. Am J Surg 221(6):1182–1187
Primrose JN, Fox RP, Palmer DH et al (2019) Capecitabine compared with observation in resected biliary tract cancer (BILCAP): a randomised, controlled, multicentre, phase 3 study. Lancet Oncol 20(5):663–673
Doussot A, Gonen M, Wiggers JK et al (2016) Recurrence patterns and disease-free survival after resection of intrahepatic cholangiocarcinoma: preoperative and postoperative prognostic models. J Am Coll Surg 223(3):493-505.e2
Hu L-S, Zhang X-F, Weiss M et al (2019) Recurrence patterns and timing courses following curative-intent resection for intrahepatic cholangiocarcinoma. Ann Surg Oncol 26(8):2549–2557
Valle J, Wasan H, Palmer DH et al (2010) Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 362(14):1273–1281
Pang YY (2000) The Brisbane 2000 terminology of liver anatomy and resections. HPB 2:333–339
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Rahbari NN, Garden OJ, Padbury R et al (2011) Posthepatectomy liver failure: a definition and grading by the international study group of liver surgery (ISGLS). Surgery 149(5):713–724
Koch M, Garden OJ, Padbury R et al (2011) Bile leakage after hepatobiliary and pancreatic surgery: a definition and grading of severity by the international study group of liver surgery. Surgery 149(5):680–688
Malka D, Bernardini D, Boudjema K, et al (2019) Cancer des voies biliaires. TNCD
Edeline J, Benabdelghani M, Bertaut A et al (2019) Gemcitabine and oxaliplatin chemotherapy or surveillance in resected biliary tract cancer (PRODIGE 12-ACCORD 18-UNICANCER GI): a randomized phase III study. J Clin Oncol 37(8):658–667
Wang C, Pang S, Si-Ma H et al (2019) Specific risk factors contributing to early and late recurrences of intrahepatic cholangiocarcinoma after curative resection. World J Surg 17(1):2. https://doi.org/10.1186/s12957-018-1540-1
Bridgewater J, Galle PR, Khan SA et al (2014) Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J Hepatol 60(6):1268–1289
Zhang X-F, Beal EW, Bagante F et al (2018) Early versus late recurrence of intrahepatic cholangiocarcinoma after resection with curative intent. Br J Surg 105(7):848–856
Portolani N, Coniglio A, Ghidoni S et al (2006) Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg 243(2):229–235
Valverde A, Bonhomme N, Farges O et al (1999) Resection of intrahepatic cholangiocarcinoma: a Western experience. J Hepatobiliary Pancreat Surg 6(2):122–127
Addeo P, Jedidi I, Locicero A et al (2019) Prognostic impact of tumor multinodularity in intrahepatic cholangiocarcinoma. J Gastrointest Surg 23(9):1801–1809
Xue R, Li R, Guo H et al (2016) Variable intra-tumor genomic heterogeneity of multiple lesions in patients with hepatocellular carcinoma. Gastroenterology 150(4):998–1008
Bagante F, Spolverato G, Merath K et al (2019) Intrahepatic cholangiocarcinoma tumor burden: a classification and regression tree model to define prognostic groups after resection. Surgery 166(6):983–990
Sposito C, Droz Dit Busset M, Virdis M et al (2021) The role of lymphadenectomy in the surgical treatment of intrahepatic cholangiocarcinoma: a review. Eur J Surg Oncol. https://doi.org/10.1016/j.ejso.2021.08.009
Shimada M, Yamashita Y, Aishima S et al (2001) Value of lymph node dissection during resection of intrahepatic cholangiocarcinoma. Br J Surg 88(11):1463–1466
Noji T, Kondo S, Hirano S et al (2008) Computed tomography evaluation of regional lymph node metastases in patients with biliary cancer. Br J Surg 95(1):92–96
Songthamwat M, Chamadol N, Khuntikeo N et al (2017) Evaluating a preoperative protocol that includes magnetic resonance imaging for lymph node metastasis in the cholangiocarcinoma screening and care program (CASCAP) in Thailand. World J Surg Oncol 15(1):176
Hobeika C, Cauchy F, Fuks D et al (2021) Laparoscopic versus open resection of intrahepatic cholangiocarcinoma: nationwide analysis. Br J Surg 108(4):419–426
Bagante F, Spolverato G, Weiss M et al (2017) Defining Long-Term survivors following resection of intrahepatic cholangiocarcinoma. J Gastrointest Surg 21(11):1888–1897
Jutric Z, Johnston WC, Hoen HM et al (2016) Impact of lymph node status in patients with intrahepatic cholangiocarcinoma treated by major hepatectomy: a review of the national cancer database. HPB 18(1):79–87
Akateh C, Ejaz AM, Pawlik TM et al (2020) Neoadjuvant treatment strategies for intrahepatic cholangiocarcinoma. World J Hepatol 12(10):693–708
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Contributions
AN, ST, RS, DF contributed to conception and design. ST, RS, UM, DF provided administrative support. UM, JV, FC, DF contributed to provision of study materials or patients. AN, CH, TC, AB, AD, MS contributed to collection and assembly of data. AN, ST, RS, FC, DF contributed to data analysis and interpretation. All authors contributed to manuscript writing. All authors approved the final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Consent to publish
This study has not been previously published elsewhere. All authors actively contributed to this work and gave their final approval before submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nassar, A., Tzedakis, S., Sindayigaya, R. et al. Factors of Early Recurrence After Resection for Intrahepatic Cholangiocarcinoma. World J Surg 46, 2459–2467 (2022). https://doi.org/10.1007/s00268-022-06655-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-022-06655-1