Abstract
Background
Surgical resection is the mainstay of treatment for patients with gastric cancer (GC). Development of a simple, high-performance, integrated scoring system is needed to provide appropriate management. This study aimed to evaluate predictive values of the systemic inflammation score (SIS) for short- and long-term outcomes of patients who underwent surgery for GC.
Methods
A total of 187 patients who underwent gastrectomy for pT2–4 GC without preoperative treatment were analyzed. SIS was formulated based on serum albumin level and lymphocyte–monocyte ratio, and graded into SIS 0, 1, and 2.
Results
Preoperative SIS was significantly associated with incidence of postoperative complications, showing a stepwise increased incidence in proportion to SIS in the entire cohort and all subgroups according to operative procedure and disease stage. Overall and disease-free survival times of patients in SIS 0, 1, and 2 shortened in a stepwise fashion. SIS was linked to prevalence of hematogenous metastasis as initial recurrence site. Survival differences between patients with SIS 2 and the others were particularly large in patients who underwent adjuvant chemotherapy. The continuation rate of adjuvant S-1 was lower in the SIS 2 group.
Conclusion
SIS represents a simple predictor for incidence of postoperative complications and survival in patients with pT2–4 GC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) is one of most common cancers worldwide, even though the incidence rate is decreasing in certain regions [1, 2]. Although it is well known that surgery is the most important component of the treatment for this disease [3, 4], curative intent resection alone, which in itself is associated with relatively high morbidity [1, 5], is not sufficient to cure locally advanced GC. Development of predictive markers with sufficient accuracy that can be calculated using preoperative blood tests would help improve the quality of the clinical management of GC patients with regard to both short- and long-term outcomes.
There is growing consensus that inflammation is involved in the development of malignancy and that signs of ongoing systemic inflammatory responses are associated with worse prognosis [6, 7]. Accordingly, the predictive values of several inflammation-nutritional biomarkers and hematological indices, including C-reactive protein, prognostic nutrition index, neutrophil–lymphocyte ratio, platelet–lymphocyte ratio, and lymphocyte–monocyte ratio (LMR), have been reported for various malignancies [7,8,9]. Since there is room for improvement in optimized cutoff values and predictive performance of these markers, development of simple, high-performance, integrated scoring systems based on an ordinal variable is desirable. Recently, a scoring system integrating serum albumin level and LMR, termed the systemic inflammation score (SIS), has been proposed and reported to be a potent prognostic factor in clear-cell renal cell carcinoma and colorectal cancer [8, 10]. However, to the best of our knowledge, there have been no reports to date on SIS in patients with GC.
The aim of this study was to evaluate the clinical significance of preoperative SIS for both short- and long-term outcomes of patients with pT2–4 GC after gastrectomy with intent to cure and evidence-based postoperative treatments.
Materials and methods
Patients
Between June 2001 and April 2017, a total of 1093 patients underwent gastrectomy for GC at the Department of Gastroenterological Surgery, Nagoya University. The eligibility criteria for the current study were (1) pT2–4 GC according to the TNM Classification of Malignant Tumors, 7th Edition [11], (2) underwent radical gastrectomy with systematic lymphadenectomy, (3) no preoperative treatment, and (4) sufficient data for analysis. We enrolled 187 patients who fulfilled the criteria. Written informed consent was obtained from all patients as approved by the Institutional Review Board of Nagoya University. All clinical data were retrospectively collected from our medical records. Preoperative blood tests were performed within 3 days before surgery.
Patient management
R0 gastrectomy with systematic lymphadenectomy as defined by the Japanese Gastric Cancer Treatment Guidelines was performed [12]. Postoperative complications were defined as adverse events categorized as Clavien–Dindo grade II–V [13]. Since 2006, postoperative adjuvant S-1 monotherapy was recommended for all patients with stage II/III GC unless contraindicated by a patient’s condition or refusal [14]. The dose of S-1 was administered according to body surface area, as follows: <1.25 m2 (80 mg daily), 1.25–1.50 m2 (100 mg daily), and >1.50 m2 (120 mg daily) [15]. The S-1 adjuvant chemotherapy was administered for 4 weeks followed by abstention for 2 weeks. This 6-week cycle was continued for 12 months unless necessity to terminate arose due to recurrence, adverse events or patient refusal [16]. Postoperative follow-up included physical examination, laboratory tests, and imaging diagnostics at least once every 6 months. Patients underwent enhanced computed tomography of the chest and abdomen at 6-month intervals and endoscopy at 12-month intervals until 5 years after surgery [12]. The chemotherapy protocol implemented after recurrence was determined by the attending physician according to available evidence, taking into account the patient’s general condition and with the patient’s consent.
Definition of SIS
SIS was formulated based on serum albumin level (ALB) and LMR as follows: a score of 0 indicated patients with ALB ≥ 4.0 g/dL and LMR ≥ 4.44; a score of 1 indicated those with either ALB < 4.0 g/dL or LMR < 4.44; and a score of 2 indicated those with both ALB < 4.0 g/dL and LMR < 4.44 [10].
Statistical analysis
Disease-specific survival, disease-free survival, and S-1 continuation rates were estimated using Kaplan–Meier curves, and differences were analyzed using the log-rank test. When calculating disease-specific survival, only gastric cancer-related deaths were counted, and subjects who died of some other causes were censored. Recurrence-free survival was defined as the period between the day of curative gastrectomy and the detection of disease recurrence. The χ2 test and Mann–Whitney test were used to compare the patient groups. The Cox proportional hazards model was employed for multivariable analysis, and variables with p < 0.05 were entered into the final model [17]. Statistical analysis was performed using JMP 10 software (SAS Institute, Cary, NC, USA). A value of p < 0.05 was considered statistically significant.
Results
Patients’ characteristics
Demographics and clinical characteristics of the 187 patients are summarized in Supplemental Table 1. Total gastrectomy was performed to treat 70 patients (37%); 35, 30, 38, 24, 32, and 28 patients were classified as TNM stages IB, IIA, IIB, IIIA, IIIB, and IIIC, respectively. Postoperative adjuvant chemotherapy was administered in 88 (47%) patients. The patients were followed for a median of 45.2 months or until death. Median ALB and LMR were 4.3 g/dL and 4.42, respectively. The numbers of patients assigned to SIS 0, 1, and 2 were 78, 73, and 36, respectively.
Comparison of clinicopathological parameters for each SIS
The association between SIS and clinicopathological parameters was evaluated. No significant differences were found with respect to sex, preoperative body mass index, tumor location, T factor, lymph node metastasis, and disease stage (Table 1). By contrast, SIS was significantly associated with age, macroscopic tumor size, postoperative complications, and administration of postoperative adjuvant chemotherapy (Table 1).
Association between SIS and postoperative complications
Thirty-seven patients (20%) experienced postoperative complications, including anastomotic leakage in 9 patients (5%), intra-abdominal abscess in 8 (4%), leakage of pancreatic fluids in 7 (4%), pneumonia in 4 (2%), and bowel obstruction in 3 (2%). The incidence of postoperative complications tended to be higher in those with hypoalbuminemia than in those without hypoalbuminemia (27 vs 17%, respectively) and was significantly higher in those with LMR < 4.44 than in those with LMR ≥ 4.44 (27 vs 13%, respectively; p = 0.018, Fig. 1a). There was a stepwise increase in the prevalence of postoperative complications in proportion to SIS (p = 0.043, Fig. 1a). Subgroup analyses according to operative procedure (total or partial gastrectomy), and disease stage was conducted to evaluate the clinical impact of preoperative SIS on short-term outcomes. The incidence of postoperative complications was gradually increased in parallel with SIS in all subgroups (Fig. 1b).
Incidence of postoperative complications. a Patients were categorized by LMR, serum albumin level, and SIS. Morbidity rate increased along with SIS. b Impact of SIS on postoperative complications according to operative procedure and disease stage. LMR lymphocyte-to-monocyte ratio, SIS systemic inflammation score. *p < 0.05
Relevance of preoperative SIS to long-term outcomes
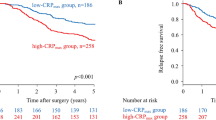
The median postoperative follow-up periods for each patients’ subgroup with SIS 0, 1, and 2 were 54.0, 30.4, and 27.0 months, respectively. The disease-specific survival times of patients in SIS 0, 1, and 2 shortened in a stepwise fashion (5-year survival rates of 87, 79, and 57%, respectively; p = 0.009, Fig. 2a). On multivariable analysis, SIS was not identified as an independent prognostic factor for disease-specific survival (Supplemental Table 2). With respect to disease-free survival, prognostic impact of constituents of SIS, ALB, and LMR was evaluated in addition to that of SIS. Patients with hypoalbuminemia had significantly shorter disease-free survival than those without, whereas LMR was not significantly associated with survival differences (Fig. 2b). The disease-free survival had a graded decrease according to SIS (3-year survival rates of 83, 79, and 61%, respectively; p = 0.024, Fig. 2b). However, in multivariable analysis, SIS was not identified as an independent prognostic factor for disease-free survival (Supplemental Table 3). The overall recurrence rate of patients with SIS 2 was 36%, significantly higher than in those with SIS 0/1 (20%) (p = 0.045, Fig. 3). On analyzing the sites of initial recurrence, SIS was found to be linked with prevalence of hematogenous recurrences, which in patients with SIS 0, 1, and 2 was 6, 10, and 19%, respectively (Fig. 3).
Association between SSI and postoperative adjuvant chemotherapy
We conducted a subgroup analysis of disease-free survival according to whether postoperative adjuvant chemotherapy was administered, focusing on patients with stage II/III GC. In the surgery-alone group, the difference in survival between patients with SIS 2 and the others was reduced. On the contrary, the prognosis of patients with SIS 2 was significantly worse than that of those with SIS 0 and 1 in the adjuvant chemotherapy group (Fig. 4a). As an attempt to explain for this finding, we evaluated the continuation rate of S-1 adjuvant therapy. S-1 continuation rate was lower in patients with SIS 2 than in those with SIS 0 and 1 (6-month S-1 continuation rates of patients with SIS 0, 1, and 2 were 82, 72, and 33%, respectively; Fig. 4b).
Discussion
SIS relies on three variables of blood tests, namely ALB, lymphocyte count, and monocyte count, which can easily and inexpensively be measured in routine clinical practice [8, 10]. In 187 patients with pT2–4 GC, preoperative SIS and its constituent LMR were significantly associated with incidence of postoperative complications. Of note, a stepwise increase in the prevalence of postoperative complications was found in proportion to SIS in the entire cohort and all subgroups according to operative procedure and disease stage. With respect to long-term outcomes, overall and disease-free survival decreased incrementally in relation to SIS. In particular, the prognostic impact of SIS 2 was evident in patients who received adjuvant chemotherapy, which we attributed to the low compliance to the chemotherapy.
Chang et al. [10] first developed SIS incorporating ALB and LMR, and reported that a high preoperative SIS was significantly associated with aggressive tumor behaviors and served as an independent prognostic factor in 441 clear-cell renal cell carcinomas. In addition, incorporation of the SIS into a nomogram successfully predicted postoperative survival. Suzuki et al. [8] compared predictive performance between two inflammation-based scores, SIS and modified Glasgow Prognostic Score, in 727 patients with colorectal cancer, and found that the time-dependent receiver-operating characteristics curve of the SIS was superior to that of the modified Glasgow Prognostic Score throughout the observation period. In these studies, the association between SIS and postoperative complications was not evaluated.
SIS is characterized by a simple and objective marker presented in an ordinal qualitative variable that may reflect the balance between host inflammatory and nutritional status. In fact, SIS was reported to have superior predictive value over other single parameters such as C-reactive protein, ALB, and LMR [7, 9, 18]. Accordingly, SIS might be more effective in patients having the complex of malnutrition and pro-inflammatory status. This background led us to employ an integrated marker, SIS, in the hope of developing a cost-effective marker to identify patients at high risk of postoperative complications and adverse prognosis. The association between high SIS (integration of low ALB, monocytosis, and lymphocytopenia) and the development of postoperative complications is likely complex and remains unclear. One plausible explanation is that a high SIS may reflect combined disadvantages for the postoperative course, including an inflammatory status and malnutrition. Pro-inflammatory status leads to compromised cell-mediated immunity and an impaired T-lymphocytic response via cytokines [19, 20]. A decreased lymphocyte-mediated antibacterial immune reaction may weaken the lymphocyte-mediated antibacterial cellular immune response and contribute to increased bacterial invasion and growth [21, 22]. Malnutrition is a major cause of tissue vulnerability, impaired wound healing, and susceptibility to infection [23, 24]. Eventually the crosstalk of these complex factors increases the incidence of postoperative complications.
With respect to long-term outcomes in patients with pT2–4 GC, SIS had superiority in detailed stratification and clear cutoff compared with its constituents, ALB and LMR. Our findings indicated that SIS may serve as a simple, preoperatively accessible, and promising prognosticator in patients with pT2–4 GC, potentially helpful in the decision-making process regarding appropriate postoperative management. The underlying biological mechanisms to explain the link between high SIS (integration of low ALB, monocytosis, and lymphocytopenia) and adverse prognosis should be further explored. Impaired nutritional status is closely related to immune incompetence and leads to accelerated tumor progression through the suppression of tumor immunity [25,26,27,28]. Recent evidence indicates that circulating monocytes can be recruited in tumor tissues and differentiate into tumor-associated macrophages, which have been reported to be a key player in the tumor microenvironment, encouraging metastasis and tumor progression [29, 30]. Lymphocytes can also secrete several cytokines, such as interferon-γ and tumor necrosis factor-α, to control tumor growth and improve the prognosis of cancer patients, while the decreased lymphocyte count and function will impair cancer immune surveillance and defense [6, 18, 31]. SIS might reflect this complex situation in a well-balanced state. Another important finding of the present study is the influence of SIS on tolerability of postoperative adjuvant chemotherapy. Decreased tolerance to adjuvant chemotherapy is at least one of the reasons for the inferior long-term outcome in the SIS 2 group. Moreover, it has been reported that postoperative complications adversely affect long-term outcomes of patients with gastric cancer [32]. Association between SIS and incidence of postoperative complications is also a possible reason for poor prognosis in the SIS 2 group.
To translate our findings into clinical practice, we need to clarify whether preoperative modification of SIS by anti-inflammatory treatment and nutritional support reduces the adverse effect on the postoperative course. Several clinical trials demonstrated that preoperative enteral alimentation for malnourished surgical patients with gastrointestinal cancers improves postoperative outcomes through significant elevations of ALB and lymphocyte counts [33,34,35]. Moreover, nutritional supportive care and intensive symptomatic treatment for adverse effects may enhance the tolerability of adjuvant chemotherapy in patients with high SIS. Prospective clinical trials evaluating the efficacy of anti-inflammatory treatment and nutritional support based on SIS are warranted.
The present study included several limitations. We were unable to eliminate potential selection bias, as this was a small, single-center retrospective study without external validation. Statistical power was limited by the relatively small sample size, particularly in subgroup analyses. Moreover, lack of postoperative SIS data might limit our deliberation on the influence of SIS on the tolerability and efficacy of adjuvant chemotherapy. Nevertheless, it is worth noting that our study is the first to evaluate the significance of SIS in both short- and long-term outcomes of GC patients.
Conclusion
Our results indicated that SIS can predict the incidence of postoperative complications and survival in patients with pT2–4 GC after radical gastrectomy. SIS is inexpensive and can be routinely performed in the clinical setting and hence can potentially provide readily available and objective information to help clinicians estimate postoperative short- and long-term outcomes.
References
Van Cutsem E, Sagaert X, Topal B et al (2016) Gastric cancer. Lancet 388:2654–2664
Kanda M, Mizuno A, Fujii T et al (2016) Tumor infiltrative pattern predicts sites of recurrence after curative gastrectomy for stages 2 and 3 gastric cancer. Ann Surg Oncol 23:1934–1940
Shen L, Shan YS, Hu HM et al (2013) Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol 14:e535–e547
Kurita N, Miyata H, Gotoh M et al (2015) Risk model for distal gastrectomy when treating gastric cancer on the basis of data from 33,917 Japanese patients collected using a nationwide web-based data entry system. Ann Surg 262:295–303
Kanda M, Tanaka C, Kobayashi D et al (2017) Proposal of the coagulation score as a predictor for short-term and long-term outcomes of patients with resectable gastric cancer. Ann Surg Oncol 24:502–509
Mantovani A, Allavena P, Sica A et al (2008) Cancer-related inflammation. Nature 454:436–444
Huang L, Liu S, Lei Y et al (2016) Systemic immune-inflammation index, thymidine phosphorylase and survival of localized gastric cancer patients after curative resection. Oncotarget 7:44185–44193
Suzuki Y, Okabayashi K, Hasegawa H et al (2018) Comparison of preoperative inflammation-based prognostic scores in patients with colorectal cancer. Ann Surg 267:527–531
Chan JC, Chan DL, Diakos CI et al (2017) The lymphocyte-to-monocyte ratio is a superior predictor of overall survival in comparison to established biomarkers of resectable colorectal cancer. Ann Surg 265:539–546
Chang Y, An H, Xu L et al (2015) Systemic inflammation score predicts postoperative prognosis of patients with clear-cell renal cell carcinoma. Br J Cancer 113:626–633
Sobin LH, Gospodarowicz MK, Wittekind C (2009) International Union Against Cancer, TNM classification of malignant tumors, 7th edn. Wiley-Blackwell, New York
Japanese Gastric Cancer Association (2017) Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer 20:1–19
Clavien PA, Barkun J, de Oliveira ML et al (2009) The Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg 250:187–196
Kanda M, Murotani K, Kobayashi D et al (2015) Postoperative adjuvant chemotherapy with S-1 alters recurrence patterns and prognostic factors among patients with stage II/III gastric cancer: a propensity score matching analysis. Surgery 158:1573–1580
Sasako M, Sakuramoto S, Katai H et al (2011) Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol 29:4387–4393
Kanda M, Kobayashi D, Tanaka C et al (2016) Adverse prognostic impact of perioperative allogeneic transfusion on patients with stage II/III gastric cancer. Gastric Cancer 19:255–263
Kanda M, Shimizu D, Tanaka H et al (2018) Significance of SYT8 for the detection, prediction, and treatment of peritoneal metastasis from gastric cancer. Ann Surg 267:495–503
Geng Y, Shao Y, Zhu D et al (2016) Systemic immune-inflammation index predicts prognosis of patients with esophageal squamous cell carcinoma: a propensity score-matched analysis. Sci Rep 6:39482
Moyes LH, Leitch EF, McKee RF et al (2009) Preoperative systemic inflammation predicts postoperative infectious complications in patients undergoing curative resection for colorectal cancer. Br J Cancer 100:1236–1239
Kim EY, Lee JW, Yoo HM et al (2015) The platelet-to-lymphocyte ratio versus neutrophil-to-lymphocyte ratio: which is better as a prognostic factor in gastric cancer? Ann Surg Oncol 22:4363–4370
Neal CP, Mann CD, Garcea G et al (2011) Preoperative systemic inflammation and infectious complications after resection of colorectal liver metastases. Arch Surg 146:471–478
Pang W, Lou N, Jin C et al (2016) Combination of preoperative platelet/lymphocyte and neutrophil/lymphocyte rates and tumor-related factors to predict lymph node metastasis in patients with gastric cancer. Eur J Gastroenterol Hepatol 28:493–502
Kanda M, Mizuno A, Tanaka C et al (2016) Nutritional predictors for postoperative short-term and long-term outcomes of patients with gastric cancer. Medicine (Baltimore) 95:e3781
Inaoka K, Kanda M, Uda H et al (2017) Clinical utility of the platelet-lymphocyte ratio as a predictor of postoperative complications after radical gastrectomy for clinical T2–4 gastric cancer. World J Gastroenterol 23:2519–2526
Felekis D, Eleftheriadou A, Papadakos G et al (2010) Effect of perioperative immuno-enhanced enteral nutrition on inflammatory response, nutritional status, and outcomes in head and neck cancer patients undergoing major surgery. Nutr Cancer 62:1105–1112
Gonzalez-Torres C, Gonzalez-Martinez H, Miliar A et al (2013) Effect of malnutrition on the expression of cytokines involved in Th1 cell differentiation. Nutrients 5:579–593
Brenner D, Blaser H, Mak TW (2015) Regulation of tumour necrosis factor signalling: live or let die. Nat Rev Immunol 15:362–374
Kanda M, Fujii T, Kodera Y et al (2011) Nutritional predictors of postoperative outcome in pancreatic cancer. Br J Surg 98:268–274
Forssell J, Oberg A, Henriksson ML et al (2007) High macrophage infiltration along the tumor front correlates with improved survival in colon cancer. Clin Cancer Res 13:1472–1479
Pollard JW (2009) Trophic macrophages in development and disease. Nat Rev Immunol 9:259–270
Wang D, Dubois RN (2010) Eicosanoids and cancer. Nat Rev Cancer 10:181–193
Tokunaga M, Tanizawa Y, Bando E et al (2013) Poor survival rate in patients with postoperative intra-abdominal infectious complications following curative gastrectomy for gastric cancer. Ann Surg Oncol 20:1575–1583
Sultan J, Griffin SM, Di Franco F et al (2012) Randomized clinical trial of omega-3 fatty acid-supplemented enteral nutrition versus standard enteral nutrition in patients undergoing oesophagogastric cancer surgery. Br J Surg 99:346–355
Zhao H, Zhao H, Wang Y et al (2013) Randomized clinical trial of arginine-demented enteral nutrition versus standard enteral nutrition in patients undergoing gastric cancer surgery. J Cancer Res Clin Oncol 139:1465–1470
Wang F, Hou MX, Wu XL et al (2015) Impact of enteral nutrition on postoperative immune function and nutritional status. Genet Mol Res 14:6065–6072
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any commercial interest or sources of financial or material support to report.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sato, B., Kanda, M., Tanaka, C. et al. Significance of Preoperative Systemic Inflammation Score in Short-Term and Long-Term Outcomes of Patients with Pathological T2–4 Gastric Cancer After Radical Gastrectomy. World J Surg 42, 3277–3285 (2018). https://doi.org/10.1007/s00268-018-4597-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-018-4597-7