Abstract
Seedling banks are very important in forest regeneration following forest disturbances such as crop cultivation. In 2011 and 2013 the Uganda National Forestry Authority (NFA) evicted encroachers from parts of Mabira Central Forest Reserve that had been under crop cultivation for over 40 years. This gave an opportunity for the vegetation to recover. In this study, we assessed the recovery process based on seedling bank diversity, richness and density in three blocks differing in recovery time. Two disturbed blocks, the Western Block (WB) (abandoned by encroachers 1–3 years), and the Eastern Block (EB) (abandoned 4–5 years) before this study and a nearby undisturbed area (intact) were included in the study. We recorded 48 seedlings species; 37 in WB, 30 in EB and 27 in intact. Differences in species richness were not statistically significant among blocks (F2, 88 = 1.2420, p = 0.294). All seedling species found in the intact were found in the EB and WB. There were statistically significant differences in species diversity (Shannon–Wiener: F2, 88 = 5.354, p = 0.006), density (P < 0.001) and composition (ANOSIM; R = 0.55, p = 0.001) among blocks. Apart from Broussonetia papyrifera, other species contributing to the dissimilarity (Acalypha neptunica, Antiaris toxicaria, Blighia unijugata, Funtumia elastica were late succession species. Animal dispersed species dominated intact. Seed-regenerating species were found in both WB and EB, but re-sprouts were more common in EB. These results show that proximity to intact forest aids forest recovery, even for areas with long-term cultivation history.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A seedling bank, or a pool of suppressed individuals waiting for a chance to grow when conditions improve (Antos et al. 2005), is critical for plant community regeneration after disturbances or unfavorable conditions such as inadequate light, which may inhibit seedling growth. As a result, seedling banks contribute to forest community resilience by spreading ecological niches out over time by storing species as suppressed individuals, allowing species to fill gaps formed by disturbances that open the forest canopy and improve growth factors such as light availability (Li et al. 2010; Hu et al. 2019). Persistence of individuals of plant species either as seeds in the soil or seedling banks in the ecosystem is a key attribute for resilience. Therefore, like soil seed banks, seedling banks survive for long periods as suppressed individuals (Antos et al. 2005) and hence may play a similar role of enhancing community resilience. As such, many long-lived perennials such as forest tree species and shrubs rely on seedling banks for recovery after disturbance.
In the context of forest regeneration, seedling banks are crucial in the Anthropocene due to the increasing levels of deforestation resulting from human disturbances such as agricultural expansion, commercial logging, industry and urbanization (Geist and Lambin 2002). When considering management options for degraded forest restoration, seedling bank features such as the density, richness, and diversity are crucial factors to consider (Bongers et al. 2009). Through seedling mortality regulation, seedling features such as density influence the direction of succession and restoration of secondary forests recovering from disturbance (Bachelot et al. 2015; Magee et al. 2021). As a result, knowledge of the seedling bank density, richness, and diversity in the context of disturbances is critical for management decisions during restoration of degraded forests.
In addition to seedling features, the length of time that has elapsed since disturbance ceased is a significant component in the recovery of degraded forests (Colon and Lugo 2006; Teegalapalli et al. 2009; Charles et al. 2018). Fewer species would colonize soon after disturbance, while more species would be present in the intermediate following disturbance, and the least number of species would be found in undisturbed or natural environments (Connell 1978). As a result, understanding seedling dynamics in relation to time elapsed since last disturbance event gives insight to the recovery process of degraded forests. However, there are few studies on forest seedling recovery that have considered a short period of time, for example <10 years after cessation of disturbance (Mwampamba and Schwartz 2011; Ssekubwa et al. 2018) despite the fact that such studies are important since seedlings determine the development of forest vegetation after disturbances (Guariguata and Ostertag 2001).
Existing studies on disturbance and species diversity have so far produced mixed results and there appears to be no consensus on the influence of disturbance on floristic metrics e.g., species richness. For example Grace and Pugesek (1997); Pollock et al. (1998); Grace (1999) have reported low species richness at high disturbance frequency whereas Grime (1973); Connell (1978) reported high species richness at intermediate levels of disturbance. In some forest ecosystems, disturbances have been reported to reduce species diversity of seedlings. For example, in the Indian tropical dry forest, Baboo et al. (2017) reported low species diversity of seedlings in highly disturbed sites. This was attributed to human disturbance in form of selective harvesting of adult trees for fuelwood and leaf fodder, which reduced seed production of adult plants and ultimately limited seed availability. Whereas there were more species in sites with gaps than non-gaps in a Neotropical forest in Panama, more than 50% of the species were absent as seedlings. This was attributed to these species’ failure to recruit despite the presence of favorable conditions for growth and survival in gaps (Hubell et al. 1999). Seedling diversity did not differ between burned and unburned stands in Southern Appalachian Oak-Hickory forests. The time lag of 15–20 years was long enough for seedling diversity in burned areas to become comparable to unburned sites, because most species censured had the ability to re-sprout (Holzmuleller et al. 2009). Other authors have reported species richness increase e.g., in sites where herbivores suppressed dominant species, allowing coexistence of more species (Tilman 1983), or decrease when disturbance is coupled with species competition for available resources such as nitrogen and destruction of plant parts through digging (Wilson and Tilman 1991).
As regards seedling density and forest disturbance, four years after gap creation in temperate forests in China resulted in high density of light demanding dominant pioneer species (Quercus momoglica and Acer mom) inside gaps and Fraxinus rhynchophylla seedling density was highest inside closed canopy (Yan et al. 2012). This suggests that, although all the three species were pioneers; the creation of gaps favored the regeneration of only two species due to their different light requirements, which affected seed germination and, eventually, seedling density. This implies that, even among species with identical regeneration requirements, disturbance might have distinct consequences. Other disturbances, like lopping, grazing, road connectivity, and distance to human population, resulted in high seedling density of native tree species such as Cedrus deodara and Pinus wallichiana at the least disturbed sites in India’s Himalayan forests (Haq et al. 2019). The most plausible explanation for these disparities was that, in comparison to the highly disturbed areas, environmental conditions such as light and moisture in the least disturbed sites remained suitable for the regeneration of these species augmented by the species’ dispersal ability.
Although the research findings above are largely from the Northern Hemisphere, there are a few and inconclusive studies on the impact of disturbance on seedling species richness, density, and diversity in tropical Africa. In Uganda, logged areas had low seedling density (Plumptre 1995) and diversity (Mwavu and Witkowski 2009) compared to unlogged forest areas in Budongo forest reserve. Ssekubwa et al. (2018) found that seedling diversity increased with restoration age in sites planted with seedlings of native tree species and sites left to regenerate naturally at Kibale tropical moist forests, implying that seedling diversity was lower at initial stages after eviction of agricultural encroachers. These studies show that results of influence of forest disturbance in form of logging on seedling species richness, diversity and density are not consistent across regions.
Theoretical Framework
Clearing forests for agriculture expansion, particularly cultivation is one of the major contributors to forest disturbances and degradation in the tropics. In Uganda, the forest cover declined from 4.9 million hectares (20% of Uganda’s land area) in 1990 to 3.6 million (14%) in 2005. This represents a 1.9% deforestation rate, which is slightly higher than in other Eastern African countries whose rate is below 1%. On private lands, nearly 1.3 million hectares were lost over the 15 years while 91,000 ha were lost in Central Forest Reserves (CFR). However, there has been renewed interest in forest restoration and conservation in the country. As a result, there are ongoing efforts by the National Forest Authority (NFA) to evacuate forest encroaches and allow regeneration. The success of these efforts largely depends on the seedling bank.
Continuous cultivation increases turnover rate of seeds from the soil seed bank and can affect seedling species richness, diversity and densities depending on disturbance history (Connell 1978). In most recently abandoned areas, seedling species richness and diversity are low because time for colonization is short. At 4–5 years since cultivation ceased, richness and diversity increases. However, richness and diversity will decline in uncultivated (intact) areas due to differences in competitive and adaptation abilities of species. Seedling banks may establish from seed banks from within the site (in situ seed bank), seeds dispersed and from re-sprouts of roots or stems (Holl 2012). Recruitment of seedlings, particularly during the initial stages of the regeneration process depends on seeds already stored in the soil. Hence, the ability to form soil seed bank is an important attribute influencing recruitment of species after disturbance events. In tropical forests, many pioneer trees form soil seed banks (Chapman and Chapman 1997; Esaete et al. 2014). Resprouting as a strategy of regeneration following disturbance also occurs in tropical forests. Although many tropical forest species are capable of resprouting, shade tolerant trees survival is higher than light demanding species (Poorter et al. 2010).
Species that are incapable of forming seed banks or resprouting can reach and colonize through the dispersal process. Wind, animals, gravity, ballistic, and water all contribute to the dispersal of forest trees (Souza et al. 2013). Dispersal increases possibilities for the species to reach favorable sites and as a consequence increases chances of seedling establishment (Babweteera and Brown 2010). Therefore a number of factors that include sources of seedlings, dispersal syndrome or mode of dispersal of seeds and the form of regeneration influence seedling regeneration after disturbance of forests. These factors are not uniform from site to site and might be the reason for the variation in regeneration success observed in previous studies.
To test the hypothesis of no relationship between seedling characteristic and time since eviction of encroachers, (i) diversity, species richness and density and (ii) dispersal and regeneration syndrome of seedlings in formerly cultivated and nature reserve areas in Mabira CFR in Uganda were assessed.
Study Area and Methods
Description of Study Site
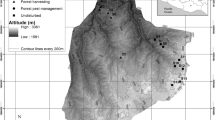
Mabira CFR is a semi-deciduous forest located in South Central Uganda between 0° 22′–0° 35′N and 32° 56′–33° 02′E (Fig. 1). Over 300 tree and shrub species have been recorded in Mabira CFR (Davenport et al. 1996). Mabira CFR is nationally important for tree species conservation (Howard et al. 2000). About 95 percent of the forest is Celtis–Chrysophyllum medium elevation semi-deciduous forest and 5 percent is Piptadeniastrum–Albizia–Celtis forest. Mabira CFR has a bimodal rainfall distribution pattern i.e., two rain seasons, the first one between March-June and the second between September and November. The amount of rainfall received ranges between 1250 and 1400 mm and temperatures range between 16 and 29 °C.
For management purposes, Mabira CFR is sub-divided into zones; Strict Nature Reserve (here in called intact), production, buffer and recreation zones (NFA 2008). According to the management plan of NFA, strict nature reserve zones are areas maintained in natural state without any form of exploitation (MWE 2017). Mabira CFR has been subjected to multiple threats within and outside the protected forest boundaries (Devine 2004). Tea and sugar cane plantations and factories, as well as enclave communities (Devine 2004) that put a lot of pressure on the forest by extracting resources surround the forest. One of the biggest problems that Mabira CFR has faced is encroachment. In the 1970s, large areas of the forest were encroached and converted to farmlands. Although the government removed encroachers in 1988–89, and Mabira was left to recover naturally, due to population pressure, encroachment has continued (Baranga 2007). In 2011 and 2013, Uganda government through NFA evicted encroachers from the Western forest block and Eastern forest blocks to allow forest regeneration. By the time of this study, the western block (WB) had been under recovery for 1–3 years and the Eastern block (EB) for 4–5 years.
Data Collection
Data for this study was collected in April 2016. Three sites were purposively selected one in the WB where people had been evicted in the previous 1–3 years, another in the EB were people had been evicted in the period 4–5 years. The last site was the strict nature reserve (intact). Following procedures by Kent (2012), three transects approximately 1 km long were laid in each site. The transects were 500 m apart oriented north to south direction. Along each transect, 10 plots (10 × 30 m) were established at WB and intact, while there were 11 in EB, at distances of 100 m apart. This gave a total of 91 plots.
The plots were sub-divided into 12 subplots of 5 × 5 m to ease location of seedling and sampling only six of the 5 × 5 m subplots, moving along the plot while switching sides. To avoid edge effects, plots were established 100 m away from the forest boundary. We recorded seedlings (plants <1.3 m tall, and girth of <20 cm). Data on dispersal syndrome and form of regeneration of the recorded species were obtained from Flora of Tropical East Africa. We also identified and enumerated trees (DBH ≥ 10 cm) in the 10 × 30 m plots.
We calculated floristic metrics of species diversity (using Shannon–Wiener index), richness (total number of species in a plot), and density of woody seedlings per plot in Statistical Package for Social Sciences (SPSS) version 23. Seedling density was computed as number of seedlings per square meter. We then evaluated if there were differences in seedling diversity, species richness and density in EB and WB and intact areas using one-way ANOVA, with time since last disturbance as fixed factor. Fisher’s least significant difference (LSD) was done for significant means. Likewise, dispersal and regeneration form of seedlings were determined and analyzed in ANOVA as described above.
We used the “vegan” package (Oksanen et al. 2020) in R version 4.1.2 (R Core Team 2021) to perform ecological analyses. To compute the contribution of individual species to the average overall Bray–Curtis dissimilarity between pairs of forest blocks, we used the simper function in the ‘vegan’ package. We also ran a non-metric multidimensional scaling ordination (NMDS, with Bray–Curtis dissimilarity distance and 999 permutations) and used “ggplot2” package (Wickham 2016) to visualize patterns of community composition in the three forest blocks. In addition, we performed a Permutational Multivariate Analysis of Variance using the adonis function in the “vegan” package to analyze and partition sums of squares using dissimilarities. We used the pairwise.adonis function for multilevel pairwise comparisons. We used the anosim function to test whether there were any statistically significant differences between the forest blocks.
Results
A total of 48 species belonging to 21 families including 37, 30 and 27 in most recently disturbed (WB) recently disturbed (EB) and intact (not disturbed) respectively were recorded. A total of 5378 individuals were assessed. A total of 44 trees (DBH ≥ 10 cm) were also recorded. The most abundant species in WB, EB and intact area was Broussonetia papyrifera (L.) L’Hér. ex Vent.occurring both as mature trees and seedlings. Celtis durandii Engl., Celtis mildbraedii Engl., Entandrophragma angolense (Welw.) C.DC., Myrianthus aboreus P.Beauv and Tabernaemontana holstii K.Schum. represented by more than ten individuals were recorded as mature trees at intact area, but were only present as seedlings in either WB and/or EB (Table 1).
Results of ANOVA revealed no significant difference (F2, 88 = 1.2420, p = 0.294) in species richness in WB (6.23 ± 2.7), EB (7.19 ± 1.8) and intact (6.9 ± 2.7) forest areas. However, there were significant differences (F2, 88 = 5.354, p = 0.006) in diversity between the three forest areas with intact having the highest diversity (1.48 ± 0.43), followed by EB (1.36 ± 0.34) and WB with the least density (1.14 ± 0.48). From the posthoc comparisons (LSD) test, the average value in WB is significantly lower than that of cultivated EB (p < 0.05) and intact (p < 0.01). Density was significantly (F2, 88 = 8.328, p < 0.001) lower in intact (58 ± 24.4), than in EB (96.8 ± 47.2) and (83.2 ± 36.8) in WB (Fig. 2). Posthoc LSD showed significant differences in density between WB and intact (p < 0.05), EB and intact (p < 0.01) and WB and EB (p < 0.01). There were more species dispersed by animals in intact than in cultivated areas. Species that regenerate by seed were represented equally in cultivated areas, while resprouting were more in EB than in cultivated WB.
Comparisons of dispersal mechanism of seedlings recorded in WB, EB and intact sites, showed no significant differences with omnibus ANOVA F-test (F2, 88 = 0.822, P > 0.05) for only wind dispersal. Animal and both animals and wind mode of dispersal were significant (p = 0.03) and (p = 0.025) respectively. Statistical comparisons using LSD posthoc test showed that there were more animal dispersed seedlings in intact than WB (p < 0.01), but not significantly between WB and EB. In relation to forms of regeneration, species that regenerate from seed were equally represented in WB and EB, while re-sprouts were more (p < 0.001) in EB compared to WB (Table 2).
Results from to dimensional NMDS (permutations = 999, stress = 0.25) show that there is no separation in species composition in WB, EB and intact, however there is more variability in intact as shown by the ellipsoid length of intact along Axis 2 (Fig. 3). This supplemented by Analysis Of Similarity (ANOSIM) showed that the WB, EB and intact were significantly dissimilar (R = 0.55, p = 0.001). Results from the Similarity Percentages (SIMPER) showed that Broussonetia papyrifera contributed to 11.98, 6.62, and 6.41% of the observed dissimilarity between Western and Intact, Western and Eastern and Intact and Eastern respectively.
Discussion
The results show a high potential for natural regeneration from seedling bank in formerly cultivated areas of Mabira CFR. Species richness was not significantly different in formerly cultivated areas and intact areas. We observed a surprisingly high number of forest tree species seedlings in an area with long history of continuous cultivation. These findings are similar to those reported by Chapman and Chapman (1997) although their study was on forest logging. These authors found no significant differences in seedling species richness between logged and unlogged forests in Kibale National Park, western Uganda. Similarly, Kirika et al. (2010) found no differences in seedling species richness of late- or early successional species in heavily and lightly logged sites in Mabira CFR. Although cultivation is expected to yield different forest recovery trajectory from logging, in this study we observed the contrary. Cultivation and logging have quite diverse effects on the forest’s floristic composition. Typically, logging eliminates mature trees of chosen species, leaving seedlings, saplings, and mature tree species that are deemed undesirable. Cultivation, on the other hand, eliminates seedlings, samples, poles, and the majority of large tree species. A few older tree species may be left in cultivated forest areas for shade or because felling equipment is difficult to come by. Fire is another type of disturbance associated with cultivation. Fire is frequently used to remove vegetation prior to cultivation. Forest recovery from these types of disturbances will almost certainly take distinct pathways.
The possible explanation for this similarity of our finding with Chapman and Chapman (1997) and Kirika et al. (2010) is intensity of disturbance. Although forest logging may appear to cause less disturbance than deforestation followed by cultivation, the process of transporting logs destroys understory vegetation, compacts the soil, and ultimately prevents seed germination and establishment and this may explain the similarity of the findings.
Alternatively, despite differences in the nature of disturbances, presence of legacy tree species was an important shared attribute. In this study, Albizia zygia (DC.), Antiaris toxicaria Lesch., Blighia unijugata Baker, Broussonetia papyrifera (L.) L’Hér. ex Vent., Celtis Africana Burm.f., Ficus exasperata Vahl, and Funtumia elastica (Preuss) Stapf were legacy trees in WB and EB. This finding collaborates earlier studies which showed that farmers around Mabira CFR deliberately plant or retain trees on farm for timber, firewood, medicine and crafts (Mulugo et al. 2020) or shade for coffee trees (Gwali et al. 2015). Furthermore, proximity of WB (l < 1 km) and EB (<20 km) to the intact block facilitated colonization of species and hence may explain the no differences in species richness in WB, EB and intact. Therefore, the areas of the forest that were not cleared provided seed sources to the formerly encroached areas.
Due to differences in time since cessation of cultivation, it would be reasonable to expect low species diversity in intact compared to formerly cultivated areas (Connell 1978). However, Mabira CFR is characterized by disturbances (Howard 1991) and illegal exploitation of resources is a common phenomenon (MWE 2017). This might explain why we found high diversity of seedlings in intact forest areas. The findings support Baboo et al. (2017) who reported high seedling diversity in less disturbed sites in an Indian tropical dry forest. The high seedling diversity in the intact zone in Mabira and Indian tropical forest areas can be explained by the similarity in the nature of the disturbances e.g., pole and fuelwood extraction which result in opening of the forest canopy. Although these disturbances opened up the forest canopy, they were not severe enough to induce drastic changes in environmental factors such as light, temperature, and moisture in the created gaps. As a result, species that take advantage of gaps to regenerate colonized in addition to species that grow in the shade and hence increasing species diversity. Low species diversity in WB compared to EB and intact can be explained in terms of time since colonization. WB had a shorter time since colonization (1–3 years after cultivation) than EB and the intact. This is consistent with Ssekubwa et al. (2018) who reported a positive effect of restoration age on woody seedling diversity.
Seedling density is a good measure of a species’ continuity in a forest when mortality is low. The findings show that seedling density was low in intact compared to cultivated areas and was lowest in WB. Low seedling density in intact areas might be explained by competition for light and shade tolerance, which allowed for more shade species where canopy is closed but not at high densities. Alternatively, conspecific negative density-dependent mortality whereby proximity to adults of the same species reduces seedling survival due to attack by host specific enemies (Janzen-Connell hypothesis) might explain low density in intact areas. Johnson et al. (2012) showed that conspecific negative density-dependent mortality is prevalent in forest species. Previous studies on seedling density in forests after disturbance report mixed results. In some studies, forest disturbance resulted in low seedling densities (Plumptre 1995, Benitez-Malvido 1998, Carlson et al. 2020) while in others, disturbance resulted in high densities (e.g., Yan et al. 2012; Pillay et al. 2018, Ligate et al. 2019). In addition to dispersal limitations (Wolfe et al. 2019), low seedling densities are linked to reduced seed supply caused by disturbances that damage mature trees and so limit seed output. Increased recruitment opportunities, particularly for species that colonize open spaces, as well as improved environmental conditions for seedling establishment and survival for such species, are the main causes of high seedling densities linked to disturbances. The low seedling density in WB can be explained by the disturbance history, in which trees were cut down to make way for cultivation and only 1–3 years had elapsed after the encroachers were evicted. This period of time may not be long enough for pioneer species to establish themselves in large numbers. In addition, the low seedling density might also be explained by how long cultivation was carried out. Studies elsewhere have shown that duration of cultivation negatively impacts number of seedlings in forests (Pérez-García and del Castillo 2017) and our study sites had been cultivated for over 40 years. However, because WB is <1 km from intact zones, seedling density is expected to increase as a result of the close proximity to seed sources.
We recorded more species dispersed by animals in intact than in previously cultivated areas of WB and EB in Mabira CFR. Animals are known to disperse many species of tropical forest trees (e.g., Howe 1996; Cordeiro and Howe 2001; Katishima et al. 2015), the site, Broussonetia papyrifera Celtis durandii, Celtis mildbraedii and Tabernaemontana holstii are all animal dispersed species (Babweteera and Brown 2010). This finding can be explained in terms presence of more wild animal species in intact areas as compared to disturbed areas. Additionally, our findings revealed that species that regenerate by seed were recorded equally in our study sites. This is not surprising since regenerating areas in forests are often colonized by pioneer tree species (Dalling and Brown 2009) and studies show that pioneer species do regenerate from seed banks (Chapman and Chapman 1997; Senbeta and Teketay 2002; Esaete et al. 2014).
We observed significant differences in species composition between WB, EB and the intact area, although there was no difference between WB and EB. This difference can be attributed to regeneration time, which is an important factor for species composition. Our findings collaborate with Mugwedi et al. (2017) who showed differences in species composition of 0–3 year and 5-year-old regenerating habitats in South Africa. This finding is also consistent with a study from Costa Rica, which showed differences in seedling species composition of 13 and 26 years (Caper et al. 2005).
Conclusions and Recommendations
Given results of this study that show that: (i) seedling species richness in cultivated and intact areas were similar, (ii) seedling density was higher in previously WB and EB compared to the intact area, we conclude that proximity to intact forest aids forest recovery, even for areas with long-term cultivation history. Our findings will inform management decisions for restoration of formerly cultivated areas of Mabira CFR and other forests in the East African region and beyond that have similar history of use. Successful forest restoration using seedling banks will result in increased carbon dioxide and other greenhouse gas absorption, mitigating global warming and improving the ecosystem for all organisms within and beyond Mabira CFR.
Further studies are necessary to examine how seedlings are linked to soil seed banks to guide management decisions on restoration approach. Also studies that monitor survival of established seedlings and their transition rates to other life stages and eventually adult tree in order to understand long-term effects of cultivation on forests. Forest regularity authorities and agencies such as NFA and Uganda Wildlife Authority should not spend resources on replanting because forests recover from the seedling banks.
References
Antos JA, Guest HJ, Parish R (2005) The tree seedling bank in an ancient Montane forest: stress tolerators in a productive environment. J Ecol 93:536–543
Baboo B, Sagar R, Bargali SS, Verma H (2017) Tree species composition, regeneration and diversity of an Indian dry tropical forest protected area. Trop Ecol 58:409–423
Babweteera F, Brown N (2010) Spatial patterns of tree recruitment in East African tropcial forests that have lost their vertebrate seed dispersers. J Trop Ecol 26:193–203
Bachelot B, Kobe RK, Vriesendorp (2015) Negative density-dependent mortality varies over time in a wet tropical forest, advantaging rare species, common species or no species. Oecologia 179:853–861
Baranga D (2007) Observations on resource use in Mabira Forest Reserve, Uganda. Afr J Ecol 45:2–6
Benitez-Malvido J (1998) Impact of forest fragmentation on seedling abundance in a tropical rain forest. Conserv Biol 12:380–389
Bongers F, Poorter L, Hawthorne WD, Sheil D (2009) The intermediate disturbance hypothesis applies to tropical forests, but disturbance contributes little to tree diversity. Ecol Lett 12:789–805
Caper RS, Chazdon RL, Brenes AR, Alvarado BV (2005) Successional dynmaics of woody seedling communities in wet tropical secondary forests. J Ecol 93:1071–1084
Carlson AR, Sibold JC, Negron JF (2020) Canopy structure and below canopy temperatures interact to shape seedling response to distrubance in a rocky mountain subalpine forest. Forest Ecol Manag 472 https://doi.org/10.1016/j.foreco.2020.118234
Chapman C, Chapman LJ (1997) Forest regeneration in logged and unlogged forests of Kibale national park, Uganda. Biotropica 29:396–412
Charles LS, Dwyer JM, Smith TJ, Connors S, Marschner P, Mayfield MM (2018) Species wood densityand the location of planted seedlings drive early-stage seedlingsurvival during tropical forest restoration. J Appl Ecol 55:1009–1018. https://doi.org/10.1111/1365-2664.1303
Colon SM, Lugo AE (2006) Recovery of a Subtropical dry forest after abandonment of different land uses. Biotropica 38:354–364
Connell JH (1978) Diversity in tropical rain forest and coral reefs. Science 199:1302–1310
Cordeiro NJ, Howe HF (2001) Low recruitment of trees dispersed by animals in African forest fragments. Conserv Biol 15:1733–1741
Dalling J, Brown J (2009) Long term persistence of pioneer species in tropical rain forest soil seed banks. Am Nat 173 https://doi.org/10.1086/597221
Davenport TRB, Howard PC, Baltzer M (1996) Mabira forest reserve: Biodiversity inventory report no. 13. Forest Department, Kampala
Devine MW (2004) Three Communities, Two Corporations, One Forest: Forest Resource Use and Conflict, Mabira forest, Uganda. Agroforestry in Landscape Mosaics Working Paper Series. World Agroforestry Centre, Yale University Tropical Resources Institute, and The University of Georgia.
Esaete J, Eycott AE, Reiniӧ J, Telford RJ, Vandvik V (2014) The Seed and Fern Spore Bank of a Recovering African Tropical Forest. Biotropica 46:677–686
Guariguata MR, Ostertag R (2001) Neotropical secondary forest succession: changes in structural and functional characteristics. For Ecol Manag 148:185–206
Geist HJ, Lambin EF (2002) Proximate causes and underlying drivers of tropical forest deforestation. Bioscience 52:143–150
Grace JB (1999) The factors controlling species density in herbaceous plant communities: as assessment. Perspect Plant Ecol EvolSyst 2:1–28
Grace JB, Pugesek BH (1997) A structural equation model of plant species richness and its application to a coastal wetland. Am Nat 149:436–460
Grime JP (1973) Control of species density in herbaceous vegetation. J Environ Manag 1:151–167
Gwali S, Agaba H, Balitta P, Hafashimana D, Nkandu J, Kuria A, Pinard F, Sinclair F (2015) Tree species diversity and abundance in coffee farms adjacent to areas of different disturbance histories in Mabira forest system, central Uganda. Int J Biodivers Sci Ecosyst Serv Manag 11:309–317
Haq SM, Rashid I, Khuroo AA, Malik ZA, Malik AH (2019) Anthropogenic disturbances alter community structure in the forest of Kashmir Himalaya. Tropical Ecol 60:6–15
Holl KD (2012) Tropical forest restoration. In: Van Andel J, Aronson J (eds) Restoration ecology. Blackwell, Malden, p 103–114
Holzmuleller EJ, Jose S, Jenkins MA (2009) The response of understory speciescomposition, diversity and seedling regeneration to repeated burning in Southern Appalachina Oak-Hickoryforests. Nat Areas J 29:255–262
Howard PC (1991) Nature conservation in Uganda’s tropical forest reserves. IUCN Tropical Forest Programme, Gland, Switzerland
Howard PC, Davenport TRB, Kigenyi FW, Viskanic P, Baltzer MC, Dickson CJ, Lwanga JL, Mathews RA, Mupada E (2000) Protected area planning in the tropics. Uganda’s Natl Syst For Nat reserves Conserv Biol 14:858–875
Howe HF (1996) Implications of seed dispersal by animals for tropical reserve management Biol Conserv 30:261–281 https://CRAN.R-project.org/package=vegan
Hu A, Zhang J, Chen XJ, Millner JP, Chang SH, Bowatte S, Hou RJ (2019) The composition, richness and evenness of seedlings from the soil seed bank of a semi-arid steppe in Northern China are affected by long term stocking rates of sheep and rainfall variation. Rangel J 41:23–32
Hubell SP, Foster RB, O’brien ST, Harms KE, Condit R, Wechster B, Wright CJ, Loo de Lao S (1999) Light-gap disturbance recruitment limitation and tree diversity in a Neotropical forest. Science 283 https://doi.org/10.1126/science.283.5401.554
Johnson DJ, Beaulieu WT, Bever JD, Clay K (2012) Conspecific negative density dependence and forest diversity. Science 336:904. https://doi.org/10.1126/science.1220269
Katishima S, Morita S, Yoshida K, Ishida A, Hayashi S, Asami T, Ito H, Miller DG, Uehara T, Hasegawa E, Matsuura K, Kasuya E, Yoshimura J (2015). The contribution of seed dispersers to tree diversity in tropical rainforests. R Soc Open Sci https://doi.org/10.1098/rsos.150330
Kent M (2012) Vegetation Description and data analysis: a practical approach, Second edition. John Wiley and Sons ltd, Uk
Kirika JM, Böhning-Gaese K, Dumbo B, Farwing N (2010) Reduced abundance of late successional trees but not of seedlings in heavily compared to lightly logged sites of three East African Tropical forests. J Trop Ecol 26:533–546
Li X, Liu W, Tang CQ (2010) The role of the seed and seedling bank in the regeneration of diverse plant communities in subtropical Ailao Mountains, Southwest China. Ecol Res 25:1171–1182
Ligate EJ, Wu C, Chen C (2019) Investigation of tropical coastal forest regeneration after farming and livestock grazing exclusion. J Res 30:1873–1884
Magee L, Wolf A, Howe R, Schubbe J, Hagenow K, Turner B (2021) Density dependence and habitat heterogeneity regulate seedling survival in a North American temperate forest. Forest Ecology and Management. https://doi.org/10.1016/j.foreco.2020.118722
Ministry of Water, Lands and Environment (2017) Forest Sector Review Report. Unpublished report, Government of Uganda.
Mugwedi LF, Rouget M, Egoh B, Sershen, Ramdhani S, Slotow R, Renteria JL (2017) An assessment of a community–based forest restoration programme in Durban (e Thekwini), South Africa. Forests 8:255. https://doi.org/10.3390/f8080255
Mulugo LW, Galabuzi C, Nabanoga GN, Turyahabwe N, Eilu G, Obua J, Kakudidi E, Sibelet N (2020) Cultural knowledge of forests and allied tree system management around Mabira Forest Reserve, Uganda. J Forest Res 31:1787–1802. https://doi.org/10.1007/s11676-019-00961-6
Mwampamba TH, Schwartz M (2011) The effects of cultivation history on forest recovery in fallows in Eastern Arc Mountain, Tanzania. For Ecol Manag 261:1042–1052
Mwavu NE, Witkowski ETF (2009) Seedling regeneration, environment and management in a semi-deciduous African tropical rain forest. J Veg Sci 20:791–804
National Forestry Authority (2008) Strategic action plan for the period 2008/9 to 2012/13 with priorities for the first five years. Government of Uganda, Kampala
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2020) vegan: Community Ecology Package. R package version 2.5-7. https://CRAN.R-project.org/package=vegan.
Pérez-García O, del Castillo RF (2017) Shifts in swidden agriculture alter the diversity of young fallows: Is the regeneration of cloud forest at stake in southern Mexico? Agric Ecosyst Environ 248:162–174
Pillay R, Hua F, Lioselle B, Benard H, Fletcher RJ (2018) Multiple stages of tree seedling recruitment are altered in tropical forests degraded by selective logging. Ecol Evol 8:8231–8242. https://doi.org/10.1002/ece3.4352
Plumptre AJ (1995) Importance of ‘seed trees’ for the natural regeneration of selectively logged forest. Common Wealth Forest Rev 74:253–258
Pollock MM, Naiman RJ, Hanley TA (1998) Plant species richness in riparian wetlands. A test of biodiversity theory. Ecology 79:94–105
Poorter L, Kitajima K, Mercado P, Chubina J, Melgar I, Prins HH (2010) Resprouting as a persistence strategy of tropical forest trees: relations with carbohydrate storage and shade tolerance. Ecology 91:2613–2627
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/
Senbeta F, Teketay D (2002) Soil seed banks in plantations and adjacent natural dry Afromontane forests of central and Southern Ethiopia. Trop Ecol 43:229–242
Souza JT, Ferraz EMN, Albuquerque UP, Araujo EL (2013) Does proximity to a mature forest contribute to the seed rain and recovery of an abandoned agricultural area in a semiarid climate? Plant Biol 16:748–756. https://doi.org/10.1111/plb.12120
Ssekubwa E, Muwanika V, Esaete J, Tabuti J, Tweheyo M (2018) Colonization of woody seedlings in the understory of actively and passively restored tropical moist forests. Restor Ecol https://doi.org/10.1111/rec.12850
Teegalapalli K, Gopi GV, Samal PK (2009) Forest recovery following shifting cultivation: an overview of existing research. Tropical Conservation. Science 2:374–387. www.tropicalconservationscience.org
Tilman D (1983) Plant succession and gopher disturbance along an experimental gradient. Oecologia 60:285–292
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York, NY ISBN 978-3-319-24277-4, https://ggplot2.tidyverse.org
Wilson SD, Tilman D (1991) Interactive effects of fertilization and disturbance on community structure and resource availability in old-field plant community. Oecologia 88:61–71
Wolfe BT, Macchiavelli R, Van Bloem SJ (2019) Seed rain along a gradient of degradation in Caribbean dry forest: Effects of dispersal limitation on the trajectory of forest recovery. Appl Veg Sci https://doi.org/10.1111/avsc.12444
Yan QL, Zhu JJ, Yu LZ (2012) Seed regeneration potential of canopy gaps at early formation stage in temperate secondary forest, Northeast China. PLoS ONE 7:e39502. https://doi.org/10.1371/journal.pone.0039502
Acknowledgements
NORAD through the Norwegian Program for Capacity Development in Higher Education and Research for Development (NORHED) project (UGA-13/0019) financed this study. Uganda national Council for Science and Technology (NS 511) provided permission for the study. We are grateful to the Ugandan National Forest Authority (NFA) for permitting access to Mabira CFR, to the staff of the Herbarium of Makerere University for specimen identification. We thank the conference organizing committee for supporting the conference participants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1
Appendix 1
Tree and seedling species abundance (1 ha) in formerly cultivated (WB Western Block and EB Eastern Block) and intact areas in Mabira Central Forest Reserve
Species | Family | Trees | Seedlings | ||||
|---|---|---|---|---|---|---|---|
WB | EB | Intact | WB | EB | Intact | ||
Acacia spp | Fabaceae | 20,800 | |||||
Acalypha fruticosa Forssk. | Euphorbiaceae | 50,000 | 74,400 | 3200 | |||
Acalypha neptunica Müll.Arg. | Euphorbiaceae | 58,000 | 36,000 | 46,800 | |||
Albizia glaberrima (Schum. and Thonn.) Benth. | Mimosaceae | 33.3 | 133.3 | 400 | 2400 | ||
Albizia grandibracteata Taub. | Mimosaceae | 400 | 800 | ||||
Albizia wightii Wight and Arn | Mimosaceae | 66.7 | 33.3 | ||||
Albizia zygia (DC.) J.F.Macbr. | Mimosaceae | 33.3 | 400 | 5600 | 170,400 | 2800 | |
Aphania spp | Sapindaceae | 33.3 | |||||
Alstonia boonei De Wild. | Apocynaceae | 33.3 | 33.3 | 133.3 | 400 | ||
Antariaris toxicaria Lesch. | Moraceae | 133.3 | 66.7 | 100 | 18,400 | 37,200 | 46,000 |
Aphania senegalensis (Poir.) Radlk. | Sapindaceae | 266.7 | 7200 | 67,200 | 17,600 | ||
Artocarpus heterophyllus Lam. | Moraceae | 66.7 | 33.3 | ||||
Blighia unijugata Baker | Sapindaceae | 33.3 | 366.7 | 46,800 | 22,800 | 24,400 | |
Broussonetia papyrifera (L.) L’Hér. ex Vent. | Moraceae | 8333.3 | 1100 | 1200 | 438,000 | 133,600 | 44,800 |
Canarium schweifurthii Engl. | Burseracece | 400 | |||||
Celtis Africana Burm.F. | Celtidaceae | 33.3 | 666.7 | 65,600 | 2400 | ||
Celtis durandii Engl. | Cananabaceae | 533.3 | 2000 | 3600 | |||
Celtis mildbraedii Engl. | Cananabaceae | 933.3 | 5200 | 10,000 | 41,600 | ||
Celtis wightii Planch. | Cananabaceae | 66.7 | |||||
Clausena anisata (Willd.) Hook.f. ex Benth | Rutaceae | 400 | |||||
Coffea ligustroides S.Moore | Rubiaceae | 13,600 | 3200 | ||||
Cola gigantea A.Chev. | Sterculiaceae | 233.3 | 66.7 | 800 | 18,800 | 38,800 | |
Cordia Africana Lam. | Boraginaceous | 4800 | |||||
Entandrophragma angolense (Welw.) C.DC. | Meliaceae | 333.3 | 2800 | 18,000 | 10,000 | ||
Ficus asperifolia Miq. | Moraceae | 11,200 | 69,600 | ||||
Ficus capensis Thunb. | Moraceae | 66.7 | 200 | 233.3 | 400 | 2400 | 8400 |
Ficus exasperata Vahl | Moraceae | 33.3 | 100 | 333.3 | 9200 | 28,400 | 10,400 |
Ficus mucuso Welw. ex Ficalho | Moraceae | 233.3 | 1200 | ||||
Ficus sur Forssk. | Moraceae | 33.3 | 1600 | ||||
Funtumia elastica (Preuss) Stapf | Apocynaceae | 166.7 | 1033.3 | 1100 | 28,400 | 23,200 | 25,600 |
Khaya senegalensis (Desv.) A.Juss. | Meliaceae | 133.3 | 1600 | 800 | |||
Kigelia africana (Lam.) Benth. | Bignoniaceae | 33.3 | 1200 | ||||
Macaranga lancifolia Pax | Euphorbiaceae | 33.3 | |||||
Margaritaria discoidea (Baill.) G.L.Webster | Euphorbiaceae | 233.3 | 4800 | 3200 | 7600 | ||
Manilkara dawei (Stapf) Chiov. | Sapotaceae | 33.3 | 5200 | 44,400 | |||
Markhamia lutea (Benth.) K.Schum. | Bignoniaceae | 33.3 | 66.7 | 39600 | 14,800 | ||
Maesopsis eminii Engl. | Rhamnaceae | 33.3 | 133.3 | ||||
Milicia excelsa (Welw.) C.C.Berg | Moraceae | 33.3 | 33.3 | 14,800 | |||
Monodora angolensis Welw. | Annonaceae | 100 | 7600 | 11200 | |||
Myrianthus arboreus P.Beauv. | Urticaceae | 333.3 | 2800 | 800 | |||
Newtonia buchananii (Baker) G.C.C.Gilbert and Boutiqu | Fabaceae | 100 | 400 | 7200 | |||
Pachystela brevipes (Baker) Baill. | Sapotaceae | 200 | 10,800 | 12000 | |||
Polyscias fulva (Hiern) Harms | Apocynaceae | 33.3 | 200 | 800 | |||
Prunus africana (Hook.f.) Kalkman | Rosaceae | 2800 | |||||
Psidium guajava L. | Myrataceae | 400 | |||||
Sapium ellipticum (Hochst.) Pax | Euphorbiaceae | 66.7 | 33.3 | 1200 | |||
Schrebera arborea A.Chev. | Oleaceae | 33.3 | |||||
Spathodea campanulata P.Beauv. | Bignoniaceae | 66.7 | 33.3 | 3200 | |||
Sterculia dawei Sprague | Sterculiaceae | 100 | 4000 | ||||
Tabernaemontana holstii K.Schum. | Apocynaceae | 600 | 10,000 | 22,000 | |||
Tamarindus indica L. | Fabaceae | 33.3 | 33.3 | 400 | 800 | ||
Teclea nobilis Delile | Rutaceae | 233.3 | 1200 | 12,000 | 26,200 | ||
Terminalia brownii Fresen. | Combretaceae | 800 | |||||
Treculia africana Decne. ex Trécul | Moraceae | 800 | |||||
Trema orientalis (L.) Blume | Cannabaceae | 33.3 | |||||
Trichilia spp | Meliaceae | 6000 | |||||
Vernonia amygdalina Delile | Asteraceae | 133.3 | |||||
Rights and permissions
About this article
Cite this article
Esaete, J., Muwanika, V.B., Musiba, R. et al. Understorey Seedling Bank in Forest Areas with a Differing Period of Recovery in Mabira Central Forest Reserve, South Central Uganda. Environmental Management 71, 159–169 (2023). https://doi.org/10.1007/s00267-022-01637-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-022-01637-4