Abstract
Accurate predictions of nuisance algae responses to algicide exposures are needed to guide management decisions. Copper sorption and responses of Lyngbya wollei (Farlow ex Gomont) Speziale and Dyck were measured in the laboratory and two areas in Lay Lake (AL, USA) to treatments of Captain® XTR (SePRO Corporation; chelated copper algicide) and a sequential treatment of GreenClean® Liquid (BioSafe Systems, LLC; peroxygen algicide) combined with Hydrothol® 191 (United Phosphorus, Inc.; endothall algicide) followed by Captain XTR. Measured filament viability in laboratory exposures predicted Captain XTR alone could control L. wollei in Lay Lake, with 2 mg Cu/g algae EC75. This produced a targeted field treatment of 9.7 kg Cu/ha which was divided into three applications of 0.3 mg Cu/L as Captain XTR in the treatment areas. Laboratory and field experiments indicated treatments of Captain XTR alone and the combination treatment resulted in comparable copper sorption and responses of L. wollei. Copper adsorbed greater to L. wollei in laboratory experiments than in the treated areas of Lay Lake with comparable exposures (2 mg Cu/g L. wollei). However, responses and infused copper were similar and correlated in laboratory experiments and treated areas of Lay Lake indicating infused copper is critical for governing toxicity. Laboratory exposures as mg Cu/g algae accurately predicted the necessary algicide exposure required to attain the critical burden of infused copper and elicit desired responses of L. wollei in treated areas of Lay Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Excessive growths of algae can cause significant disruption of critical water resource usages including drinking, irrigation, and recreation as well as being aesthetically displeasing (Hoagland et al. 2002; Landsberg 2002; Briand et al. 2003). Cyanobacteria toxin and taste/odor compound producers have become an increasing priority due to associated risks to humans, domestic pets, livestock, and wildlife associated with the water resource (Chorus and Bartram 1999; Falconer 1999; Carmichael et al. 2001). Lyngbya wollei is a filamentous, mat-forming cyanobacterium with documented toxin and taste/odor production that frequently causes devastating economic and ecological impacts (Paerl 1988; Speziale and Dyck 1992; Mastin et al. 2002). Efficient and effective management approaches are needed to combat this growing threat to our freshwater resources. Algicides are often the preferred management strategy due to their potential to rapidly restore water resource usages (Mastin et al. 2002). However, additional information is needed on algicide formulations and management programs to precisely select the appropriate control method to achieve the desired results while decreasing nontarget species risks. The overall goal of this research is to more accurately predict algicide efficiency and performance when targeting specific nuisance and noxious mat-forming algae infestations.
Laboratory experiments have been used to guide field algal management decisions and characterize the algicide exposure that induces a target alga to respond as desired (Bishop and Rodgers 2012). Previous studies indicate that the fundamental aspect of such laboratory experiments should base the exposure on a mass of algicide to mass of algae relationship. Copper algicide formulations can differ significantly in terms of copper partitioning and consequent algicidal efficacy (Stauber and Florence 1987; Mastin and Rodgers 2000; Murray-Gulde et al. 2002; Bishop and Rodgers 2011, 2012). Despite a large fraction of applied copper that may rapidly partition to the algae (Crist et al. 1990; Levy et al. 2007), the efficiency of a copper formulation and subsequent algal responses are primarily dictated by the amount of applied copper that transfers to internal toxic sites of action (infused; Stauber and Florence 1987; Stauber and Davies 2000; Bishop and Rodgers 2012). The efficiencies of different copper formulations need to be assessed to identify the formulation that achieves the critical burden (infused copper concentration per mass of algae required to achieve control) with the lowest amounts of applied algicide (Masuda and Boyd 1993; Mastin and Rodgers 2000; Bishop and Rodgers 2012). By measuring the critical burden of the targeted alga to a specific algicide, and quantifying algae mass present at a field site, a specific algicide formulation and corresponding amount to achieve control can be predicted. Multiple applications may be required to achieve the critical burden because algicide labels restrict the mass of copper that can be applied in a single application based on a maximum aqueous concentration. Multiple applications may be required to achieve the critical burden when there is (1) high algae biomass, (2) low water volume in treatment area, or (3) algae with low sensitivity to the algicide.
Infusion of copper algicide could hypothetically be increased following a preceding exposure to noncopper algicides such as peroxide and endothall. A peroxide and endothall treatment could remove exogenous organic matter including mucilage and periphyton. By improving copper infusion, the amount of copper used (mass Cu/g algae) to achieve the critical burden should decrease. A reduction of the amount of copper algicide required to treat algae is predicted to decrease risks associated with the copper exposure available to nontarget organisms. Despite previous field implementation at some sites (Tedrow 2007), data are still needed to assess the accuracy and refine laboratory experiments to predict the ability of combination treatments to impact copper infusion and resulting target algae response.
The goal of this research is to further correlate responses of algae to copper algicide exposures based on infusion, external adsorption, and overall affinity. By understanding the efficiencies of algicide formulations to infuse copper and resultant target algae responses, an effective algicide can be selected, and the quantity necessary to control the mass of algae present in the water can be calculated. By accurately predicting the algicide exposure needed to control algae, the likelihood of excessive or insufficient algicide exposures can be decreased. The specific objectives of this research are (1) to measure the critical burden of copper required to control L. wollei in Lay Lake, AL following exposures of Captain XTR, (2) to determine whether pretreatment with Hydrothol 191 and Green Clean Liquid followed 7 days later by Captain XTR increases copper infusion and algicidal efficacy, (3) to calculate the copper exposure and treatment program needed to control the target mass of algae in treatment areas of Lay Lake, and (4) to compare copper sorption, responses, and critical burdens of L. wollei in the laboratory and field following exposures of Captain XTR and combination treatments.
Methods
Critical Burden Measurement in the Laboratory
To measure the critical burden of copper (as Captain XTR), L. wollei and site water from field test sites in Lay Lake, AL were collected and shipped to the SePRO Research and Technology Campus (Whitakers, North Carolina, USA). Upon receipt and acclimation of samples, L. wollei was exposed to untreated control, 2, 4, 6, 8, and 10 mg Cu/g L. wollei as Captain XTR. Additional toxicity experiments were initiated at the same time exposing L. wollei to a combination of Hydrothol 191 (1.5 mg endothall/L) and GreenClean Liquid (24.8 µL hydrogen dioxide/L) followed 7 days later with Captain XTR at untreated control, 2, 4, 6, 8, and 10 mg Cu/g L. wollei. Treatments were replicated six times—three randomly selected for response measurements and three for copper analyses. Exposures were accomplished by exposing 0.1 g of L. wollei in 0.5 L of lake water (water-quality parameters were similar between sites and averaged; pH 7.1, conductivity = 111.5 μS/cm, alkalinity = 32 mg/L as CaCO3, hardness = 43 mg/L as CaCO3). Copper exposures were confirmed by measuring the acid-soluble copper concentration immediately after Captain XTR exposures were initiated. The chlorophyll a content and filament viability of L. wollei were measured 1 week after Captain XTR exposure. Filament viability was measured using the mortal stain methylene blue. As a mortal stain, cells that allow for the entry of methylene blue are considered dead (Corradi and Gorbi 1993). Staining was accomplished by immersing subsamples (~0.05 g) of L. wollei from the untreated controls and treatments in 5 mL 0.1 % methylene blue for 10 min. Filament viability was calculated as the percent of unstained filaments out of 100 randomly selected trichomes/filaments per subsample. Chlorophyll a was measured for three subsamples using fresh weight weighing from 0.05 to 0.1 g. To determine fresh weight, biomass was blotted dry with paper towels. The chlorophyll a extraction was modified from Standard Methods (APHA 2005) by freezing the sample (−12 °C) for a minimum of 24 h followed by sonication in 10 mL buffered acetone. Chlorophyll a was measured fluorometrically using a Wallac Victor2 spectrofluorometer by correlating with a matrix-matched standard calibration curve ranging from 10 to 640 μg/L (Sigma C-5753). Samples were diluted in the same matrix if needed to fall within standard curve. The EC75 was discerned using regression analysis (Microsoft 2010).
The amount of copper infused and adsorbed to L. wollei was measured from three replicates of each treatment. Copper was measured 7 days after treatment (DAT) with Captain XTR. Adsorbed copper was measured by rinsing 0.1 g ww L. wollei with 10 mL of 2 mM EDTA for 10 min to remove adhered metal ions from surface. The rinsate was filtered through a clean 0.2-µm glass fiber filter. Infused copper was measured by taking the rinsed algae, weighing, sonicating for 30 s to disperse cells, and digesting in 2 mL 70 % trace metal grade nitric acid [HNO3 (Fisher Scientific, Inc. A509)], 2 mL of 30 % H2O2, and 6 mL NanoPure™ water and heating at 180 °C until solution appeared clear (USEPA 1996; Tripathi et al. 2006). Acid-soluble copper concentrations were measured by taking 15 mL of treatment solution, then acidifying to 2 % with trace metal grade nitric acid and filtering. Copper was measured using inductively coupled plasma-optical emission spectrometry (Shimadzu ICPE 9000) using a matrix-matched calibration curve from serial dilution of a 1,000 mg/L copper standard (Fisher Scientific, Inc. SC194; APHA 2005; USEPA 1994). The limit of detection for copper was 5 µg/L. The critical burden (mg copper/g L. wollei) was identified as the lowest concentration of infused copper at which 75 % reduction in filament viability was measured.
Field Study
Three areas in Lay Lake, ~3 km apart, with dense growths of L. wollei were used in this study. One area (0.92 ha inlet; 32°59′34.90″N, 86°29′27.14″W) was randomly designated for the GreenClean and Hydrothol 191 followed by Captain XTR treatment (Combination), another area (1.38 ha inlet; 32°59′19.17″N, 86°29′56.05″W) was designated for the Captain XTR alone treatment, and the last area (0.67 ha inlet; 32°59′43.69″N, 86°29′3.53″W) was used as an untreated control. L. wollei biomass was measured from samples collected with a rake from four sampling sites per treatment area. Each area sampled per sampling site was 0.56 square meters. The exposure measured to achieve the critical burden was multiplied by the estimated L. wollei biomass in the treatment areas to calculate the amount of applied Captain XTR necessary to achieve the critical burden (e.g. [X g L. wollei per area] × [Y mg Cu per g to control L. wollei]). The calculated mass of copper required to achieve the critical burden was divided into three applications to introduce copper to lower viable mat layers.
Confirmation of copper exposures were assessed by homogenized water samples from four sampling locations from the top meter and bottom meter of the water column immediately before, 1, 4, and 24 hours after treatment (HAT). Algal samples to measure copper adsorbed onto and infused into L. wollei were collected in parallel with the water samples at the same locations to quantify mass balance of copper. Additional L. wollei samples were collected 7 DAT to compare copper sorptions in both the field and laboratory. Algal samples for measuring responses were collected immediately prior to treatment, 7, 28, and 91 DAT in untreated and treated sites. Adsorbed copper, infused copper, filament viability, and chlorophyll a content were measured in triplicate for each of the four sampling sites in the treatment areas and untreated control. In addition, biomass was estimated in treatment areas and untreated controls at the four sampling sites (0.56 m2 each) by collecting with a rake.
Results and Discussion
Critical Burden Measurement at the Bench Scale
Lyngbya wollei exposed to Captain XTR indicated that the EC75 at 7 DAT was <2 mg Cu/g algae based on percent viable filaments (Fig. 1). The mortal stain was a more sensitive indicator of algal health than chlorophyll a content (Fig. 1). We hypothesize chlorophyll a has a less sensitive response than the mortal stain due to the chlorophyll degradation rate in the L. wollei mats following treatment. However, chlorophyll a content is reported as a more sensitive measure of algicidal affects than biomass reduction (Bishop and Rodgers 2011). L. wollei cells inside the mucilaginous sheath appeared degraded in the Captain XTR treatments, and the mortal stained confirmed this observation. L. wollei appeared unaffected by the GreenClean Liquid and Hydrothol 191 treatments 7 DAT in the laboratory based on measured biomass (~50 % increase in all treatments) and visual observations in untreated controls. Copper sorption (adsorbed and infused) to L. wollei increased as the Captain XTR concentration increased (Fig. 2).
Laboratory analysis of percent viable filaments (Left) and chlorophyll a content (Right) of L. wollei 7 DAT in untreated controls, Captain XTR alone treatments (2, 4, 6, 8, and 10 mg Cu/g algae), and Hydrothol and Green Clean followed 1 week later with Captain XTR (2, 4, 6, 8, and 10 mg Cu/g algae). Error bars are ± one standard deviation
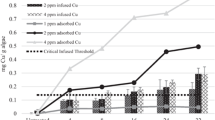
The average amount of copper adsorbed was greater than infused copper with the Captain XTR and combination treatments. The critical burdens for percent viable filaments based on infused copper were 0.0359 and 0.0378 mg Cu/g L. wollei (achieved with the 2 mg Cu/g L. wollei treatment), for the Captain XTR and combination treatments, respectively (Fig. 3). The corresponding adsorbed copper amounts were 0.04 and 0.06 mg Cu/g L. wollei (achieved with the 2 mg Cu/g L. wollei treatment), for the Captain XTR and combination treatments, respectively (Fig. 4). Despite similar trends in chlorophyll a reduction, filament viability was used to assess the critical burden due to increased sensitivity (Fig. 3). Based on measured filament viability of L. wollei to treatments in the laboratory, a 2 mg Cu/g L. wollei was a sufficient amount for copper exposure to partition and infuse enough copper to L. wollei to achieve control at two locations in Lay Lake, AL and selected for field experimentation.
Calculated Copper Exposure in the Two Treatment Areas for L. wollei Control
There was no measured difference in the L. wollei biomass between the two treated areas in Lay Lake, AL (~14,700 kg/ha) before treatment, and the calculated mass of copper per area to treat the two areas was 29.1 kg Cu/ha. Due to mat thickness and predicted copper interaction, the calculated mass of copper as Captain XTR was evenly distributed over three sequential applications each consisting of 9.7 kg Cu/ha. One application is equal to a copper concentration (as Captain XTR) of 0.3 mg Cu/L in the entire water column (~3 m water depth). However, the goal of Captain XTR application with the injection equipment (HD AQUA system designed by Aqua Services Inc.) was to target the bottom portion of the water column (1.2 m) to enhance copper delivery to where L. wollei was growing as benthic mats.
Copper Sorption to L. wollei in Lay Lake
One hour before treatment, the average acid-soluble copper concentration in the top and bottom 1.2 m of water in both treated areas ranged from <0.001 to 0.011 mg Cu/L. The averages (STD) of acid-soluble copper concentrations (1 HAT) in the bottom 1.2 m of water were 0.091 (0.126) and 0.463 (0.340) mg Cu/L for the Captain XTR alone and the combination treated areas, respectively. In the top 1.2 m of water, the averages (STD) of acid-soluble copper concentrations (1 HAT) were 0.290 (<0.01) and 0.428 (0.392) for the Captain XTR alone and combination treated areas, respectively. The measured acid-soluble copper concentrations at the top and bottom 1.2 m of water were not significantly different at either of the two treatment areas indicating movement of copper from bottom to top. Observationally, movement of copper was attributed to agitation from application equipment. Despite diffusion of some amount of acid-soluble copper, the amounts of copper adsorbed onto and infused into L. wollei increased over 24 h (Fig. 4). Based on observed trends, copper adsorption onto L. wollei occurred between 1 and 4 HAT in the area treated with Captain XTR alone and between 4 and 24 HAT in the area with the combination treatment. The copper infusion rate into L. wollei was equal to or greater than the rate of adsorption, with copper infusion occurring within 4 HAT.
Lyngbya wollei Responses Following Captain XTR Treatment in Lay Lake
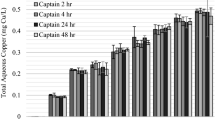
The average percentage of stained (nonviable) filaments was the greatest 7 DAT for the area treated with Captain XTR alone (Fig. 5; average = 20 %; STD = 10) and 1 DAT for the area treated with the combination (average = 34 %; STD = 10). L. wollei filament viability increased from 7 to 28 DAT for the area treated with Captain XTR alone (first application). This was likely a function of viable biomass in the lower portion of L. wollei mats, although was still decreased compared with initial levels. There was no significant difference in the percentage of unstained filaments from 7 to 28 DAT for the area with the combination treatment (α = 0.05; n = 4).
Percentage of viable filaments (top) and chlorophyll a content (bottom) of L. wollei in the Captain XTR alone treatment and the combination treatment of GreenClean and Hydrothol followed 7 days later by Captain XTR at 0 (Pretreatment), 1, 7, and 28 days after treatment. Error bars are ± one STD (n = 4)
The chlorophyll a content of L. wollei in the area treated with Captain XTR alone was approximately twice that of L. wollei treated with the combination immediately prior to Captain XTR treatment (Fig. 5). Seven DAT, the chlorophyll a content of L. wollei was significantly less than pretreatment (α = 0.05; n = 4) in both treatment areas. The chlorophyll a contents of L. wollei 7 DAT were not significantly different between the two treatment areas. However, 28 DAT the chlorophyll a content of L. wollei in the area that received the combination treatment was not significantly different than that with pretreatment (α = 0.05; n = 4). While the chlorophyll a content of L. wollei in the area with the combination treatment (28 DAT) was significantly less than the pretreatment concentrations, it was not significantly different 7 DAT (α = 0.05; n = 4). L. wollei viable biomass decreased by ~90 % in the areas treated with Captain XTR alone and the combination following the treatment program (3 treatments, 91 DAT) indicating that the exposure was effective in reducing L. wollei biomass (Fig. 6).
Comparison of Critical Burden and Responses in the Laboratory and Lay Lake
In the laboratory exposures, the quantity of copper adsorbed onto L. wollei was significantly greater than that in the treated area of Lay Lake 7 DAT (Fig. 7). However, there was no significant difference in the quantity of copper infused into L. wollei with the laboratory or field treatments 7 DAT at 2 mg Cu/g L. wollei exposure. There were no significant differences of L. wollei filament viability and chlorophyll a reduction in the laboratory and field exposures (Fig. 8).
Differences of L. wollei Responses to the Captain XTR Alone and Combination Treatments in Lay Lake
Lyngbya wollei viability was not significantly different in the treated areas, except 7 DAT where the area treated with Captain XTR had a greater percentage of mortal stained filaments than in the area that received the combination treatment (Fig. 5). Before copper algicide was applied the chlorophyll a content of L. wollei in the area treated with Captain XTR alone was significantly greater than in the area that received the combination treatment (Fig. 5). However, 7 DAT the chlorophyll a content of L. wollei exposed to the combination treatment was greater than the Captain XTR alone treated area. L. wollei biomass in either of the treated areas were not significantly different before, 7 DAT, or 28 DAT (α = 0.05; n = 4), although the average biomass of L. wollei decreased in both treated areas by 91 DAT (Fig. 6). In the untreated control site, chlorophyll a content, filament viability and biomass were not significantly different throughout the study. Average control biomass from sampling locations did decrease through the study though visual observation indicated that L. wollei mats had detached and accumulated in near shore areas.
Conclusions
The Hydrothol and GreenClean followed by Captain XTR treatment did not significantly increase either the affinity of copper to adsorb to L. wollei or the amount of infused copper within algae at lethal concentrations in laboratory or field experiments. Therefore, copper affinity and toxicity of Captain XTR were not enhanced by pretreating with Hydrothol and GreenClean. Even though copper adsorption to L. wollei was greater in the laboratory than in the field, copper infusion in L. wollei, as well as the responses of L. wollei, were similar in the laboratory and field exposures. Furthermore, copper exposures in excess of the critical burden threshold resulted in continued adsorption of copper (Tien et al. 2005), while copper infusion was not significantly different with increasing copper exposure concentrations. This suggests that when the infused critical burden threshold is attained, subsequent control is manifested independent of adsorbed copper supporting the hypothesis that infused copper is the primary fraction eliciting toxicity to L. wollei in Lay Lake. In addition, assessment of the critical burden based on infused copper accurately predicted L. wollei responses to Captain XTR exposures at the bench scale and operational treatments in Lay Lake.
Crucial aspects in designing management programs for nuisance algae infestations include critical burden, degree of algae infestation, and efficiency of the algicide. A targeted algal management approach was designed for L. wollei in Lay Lake, AL by measuring the critical burden in the laboratory with site-specific water and algae, as well as measurement of the target algal biomass in the treatment areas in Lay Lake. Management approaches using a series of algicide applications may be needed for severe nuisance algae infestations. The components of this research can be applied to a variety of field situations in order to design an efficient and effective approach for controlling nuisance algae at a specific site.
References
American Public Health Association (APHA) (2005) Standard methods for the examination of water and wastewater. 21st edn. Washington, DC
Bishop WM, Rodgers JH Jr (2011) Responses of Lyngbya magnifica Gardner to an algaecide exposure in the laboratory and field. Ecotoxicol Environ Saf 74:1832–1838
Bishop WM, Rodgers JH Jr (2012) Responses of Lyngbya wollei to exposures of copper-based algaecides: the critical burden concept. Arch Environ Contam Toxicol 62:403–410
Briand J-F, Jacquet S, Bernard C, Humbert J-F (2003) Health hazards for terrestrial vertebrates from toxic cyanobacteria in surface water ecosystems. Vet Res 34:361–377
Carmichael WW, Azevedo SMFO, An JS et al (2001) Human fatalities from cyanobacteria: chemical and biological evidence for cyanotoxins. Environ Health Perspect 109(7):663–668
Chorus I, Bartram J (1999) Toxic cyanobacteria in water: a guide to public health significance, monitoring and management. WHO, Chapman & Hall, London, p 416
Corradi MG, Gorbi G (1993) Chromium toxicity on two linked trophic levels II. Morphophysiological effects on Scenedesmus acutus. Ecotoxicol Environ Saf 25:72–78
Crist RH, Martin JR, Guptill PW, Eslinger JM, Crist DR (1990) Interaction of metals and protons with algae. 2. Ion exchange in adsorption and metal displacement by protons. Environ Sci Technol 24:337–342
Falconer IR (1999) An overview of problems caused by toxic blue-green algae (cyanobacteria) in drinking and recreational water. Environ Toxicol 14:5–12
Hoagland P, Anderson DM, Kaoru Y, White AW (2002) The economic effects of harmful algal blooms in the United States: estimates, assessment issues, and information needs. Estuaries 25(4b):819–837
Landsberg JH (2002) The effects of harmful algal blooms on aquatic creatures. Rev Fish Sci 10(2):113–390
Levy JL, Stauber JL, Jolley DF (2007) Sensitivity of marine microalgae to copper: The effect of biotic factors on copper adsorption and toxicity. Sci Total Environ 387:141–154
Mastin BJ, Rodgers JH Jr (2000) Toxicity and bioavailability of copper herbicides (Clearigate, Cutrine Plus, and Copper Sulfate) to freshwater animals. Arch Environ Contam Toxicol 39:445–451
Mastin BJ, Rodgers JH Jr, Deardorff TL (2002) Risk evaluation of cyanobacteria-dominated algal blooms in a North Louisiana reservoir. J Aquat Ecosyst Stress Recovery 9:103–114
Masuda K, Boyd CE (1993) Comparative evaluation of the solubility and algal toxicity of copper sulfate and chelated copper. Aquaculture 117:287–302
Microsoft (2010) Microsoft Excel [computer software]. Redmond, Washington
Murray-Gulde CL, Heatley JE, Schwartzman AL, Rodgers JH Jr (2002) Algicidal effectiveness of Clearigate, Cutrine-Plus, and Copper Sulfate and margins of safety associated with their use. Arch Environ Contam Toxicol 43:19–27
Paerl HW (1988) Nuisance phytoplankton blooms in coastal, estuarine, and inland waters. Limnol Oceanogr 33(4):823–847
Speziale BJ, Dyck L (1992) Comparative taxonomy of Lyngbya wollei comb. nov. (cyanobacteria). J Phycol 28:693–706
Stauber JL, Davies CM (2000) Use and limitations of microbial bioassays for assessing copper bioavailability in the aquatic environment. Environ Rev 8:255–301
Stauber JL, Florence TM (1987) Mechanism of toxicity of ionic copper and copper complexes to algae. Mar Biol 94:511–519
Tedrow OR (2007) Responses of problematic cyanobacteria to exposures of copper containing algicides. M.S. Thesis, Clemson University, 2007
Tien C-J, Sigee DC, White KN (2005) Copper adsorption kinetics of cultured algal cells and freshwater phytoplankton with emphasis on cell surface characteristics. J Appl Phycol 17:379–389
Tripathi BN, Mehta SK, Amar A, Gaur JP (2006) Oxidative stress in Scenedesmus sp. during short- and long-term exposure to Cu2+ and Zn2+. Chemosphere 62:538–544
United States Environmental Protection Agency (USEPA) (1994) Method 200.7. Determination of metals and trace elements in water and wastes by inductively coupled plasma-atomic emission spectrometry. Environmental Monitoring Systems Laboratory Office of Research and Development. Cincinnati, OH 45268
United States Environmental Protection Agency (USEPA) (1996) Microwave assisted acid digestion of siliceous and organically based matrices, method 3052. EPA SW-846, ch 3.2, USEPA. Washington, DC
Acknowledgments
This study was funded by SePRO Corporation (Carmel, IN, USA). The authors are thankful for the support provided and samples collected by Alabama Power, especially Wesley Taylor Anderson, to make this study possible. The authors are grateful for the certified professionals and advanced equipment provided by Aqua Services, Inc. to efficiently conduct the algicide applications.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bishop, W.M., Willis, B.E. & Horton, C.T. Affinity and Efficacy of Copper Following an Algicide Exposure: Application of the Critical Burden Concept for Lyngbya wollei Control in Lay Lake, AL. Environmental Management 55, 983–990 (2015). https://doi.org/10.1007/s00267-014-0433-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-014-0433-5