Abstract
The seemingly altruistic sentinel shifts performed alternately by individuals in group living animals have been extensively studied. However, the reactions of group members to the sentinel following its guarding shift have been almost ignored. To address this issue, we examined the reactions of groupmates to the sentinel in 20 groups of the cooperatively breeding Arabian babbler. We first induced sentineling of dominant and subordinate individuals of both sexes by feeding them ad libitum. Immediately after a sentinel bout, sentinels were not granted more copulations or more allopreening, neither did they suffer less aggression as compared to a control period not following sentinelling, even not after warning their groupmates from an approaching predator. Even more surprisingly, subordinates, who normally copulate less than dominants, copulated even less than usual after a sentinel bout. These findings support further the hypothesis that in this species, sentinelling serves first and foremost the sentinel itself, and accordingly, groupmates do not reward the sentinel.
Significance statement
Sentinel behaviour in cooperatively breeding animals gave rise to numerous debates regarding this seemingly altruistic behaviour. All the group members benefit when sentinels spot predators and pronounce loud alarm calls. Do groupmates pay back sentinels for defending them? To address this question, we examined the reactions of groupmates to the sentinel in 20 groups of the cooperatively breeding Arabian babbler. Immediately after a sentinel bout in the breeding season, sentinels did not (1) copulate more often, (2) get more allopreening, and (3) suffer less aggression, as compared to a control period, even not after warning their groupmates from an approaching predator. These findings support the hypothesis that at least in this species, sentinelling is carried out for the sake of its performer and is not an altruistic behaviour.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In many group-living animals, including mammals, birds, and even fishes, one group member positions itself at a high and prominent position from time to time, and looks around. This individual is called sentinel. If a predator approaches, the sentinel utters specific calls, termed ‘alarm calls’. Hearing these calls, the other group members frequently stop immediately whatever they are doing and escape to the closest shelter or join the sentinel in calling toward the predator. When the predator disappears, all the group members stop calling and return gradually to foraging (Bednekoff 2015). Many studies have agreed that this is a common system of anti-predator vigilance (Gaston 1977; Rasa 1986, 1989; McGowan and Woolfenden 1989; Zahavi 1990; Bednekoff 1997, 2001; Wright et al. 2001a, b; Hollen et al. 2011; Ridley et al. 2013; Santema and Clutton-Brock 2013; Dattner et al. 2015; Kern et al. 2016). There is usually one sentinel at a time (Bednekoff 2015), and the sentinel activity covers between 30 and 75% of the group’s foraging time (McGowan and Wollfenden 1989; Clutton-Brock et al. 1999; Wright et al. 2001b; Hollen et al. 2008; Walker et al. 2016). The group members, therefore, are not always protected by a sentinel when they are foraging. Previous studies found that dominants act as sentinels significantly more than subordinates in Arabian babblers, Argya squamiceps (Wright et al. 2001a, b); Florida scrub-jays, Aphelocoma coerulescens (Bednekoff and Woolfenden 2003, 2006); pied babblers, Turdoides bicolor (Hollen et al. 2011); white-browed sparrow weavers, Plocepasser mahali (Walker et al. 2016); meerkats, Suricata suricatta (Clutton-Brock et al. 1999); and dwarf mongooses, Helogale undulata (Kern et al. 2016).
Since the sentinels lose foraging time and seemingly endanger themselves (Ridley et al. 2013), several, not mutually exclusive, functions of sentinel behaviour have been suggested so far. As foragers learn from the sentinel’s alarm calls about the imminent danger and react accordingly, the sentinel benefits from the higher survivability of its group mates via group augmentation (Kokko et al. 2001; Wright et al. 2001b; Kingma et al. 2014) and kin selection (Hamilton 1964; Sherman 1977). Other hypotheses include parental care as the main motive for sentineling (Clutton-Brock et al. 2000; Sorato et al. 2012; Santema and Clutton-Brock 2013). Furthermore, reciprocal altruism had been suggested as a mechanism driving sentinel behaviour (Trivers 1971). Sentinel behaviour has also been suggested to be a handicap (because it is potentially risky and carried out on the account of foraging time) and performed in order to gain social prestige by the group members thanks to defending them, which would improve breeding probabilities in the future (Zahavi 1990; Zahavi and Zahavi 1997, pp. 133–136, 143–145; Dattner et al. 2015). An additional hypothesis suggested detecting conspecific intruders and expel them (McGowan and Woolfenden 1989; Walker et al. 2016). In contrast to most of these hypotheses (except of the last one), which assume that the sentinel endangers itself, Bednekoff (1997, 2001) suggested that a sentinel may be safer due to its ability to detect predators earlier than when it forages, and, upon detection, can either dissuade the predator from attacking by calling towards it or escape to shelter in time. Moreover, sometimes the alarm calls emitted by the sentinel may actually divert the attack away from itself towards the foragers (Kareksela et al. 2013).
Under the reciprocity (Trivers 1971) and the handicap (Zahavi 1990) hypotheses, sentinel activity should be rewarded in one way or another by group members. Under all other hypotheses, sentinel activity is independent of any behavioural response by the group members.
In this study, we tested the rewards for sentineling in the Arabian babbler, Argya squamiceps, a cooperatively breeding songbird, resident along the dry riverbeds of the Rift Valley. Arabian babblers live year-round in territorial groups of two to 20 individuals of both sexes and all ages. All group members cooperate in defending the territory against neighbouring groups and against intruding babblers who occasionally try to penetrate into the area or into a group. A linear hierarchy exists among individuals of each sex that typically corresponds to their age. Within the group, a higher rank implies both a higher priority of access to food and breeding opportunities (Zahavi 1989, 1990). However, while in certain groups the dominant pair alone can breed, in other groups, non-relative subordinates may also share breeding (Lundy et al. 1998). The Arabian babbler’s sentinel behaviour was described by Zahavi and Zahavi (1997, pp. 134–136), Naguib et al. (1999), Wright et al. (2001a, b, c), Regosin (2002), Edelaar and Wright (2006), Sommer (2011), Sommer et al. (2012), Dattner et al. (2015), Ostreiher and Heifetz (2017, 2019, 2020), and Ostreiher et al. (2021).
In the Arabian babbler, males sentinel more than females and dominants more than subordinates (Ostreiher and Heifetz 2017, 2019). During foraging, individuals sometimes behave aggressively toward others, push them away from foraging patches, exploit their work, grab from them food items, and even peck each other directly. Females suffer from aggression more than males and subordinates more than dominants. On the other hand, sometimes one babbler approaches another and starts to preen it in a friendly manner. Such allopreening may last from a few seconds up to a few minutes, and then, the individuals move away from each other and return to foraging (Zahavi 1989, 1990).
Previous feeding experiments, conducted by us and by many others in different species (Clutton-Brock et al. 1999; Wright et al. 2001c; Bednekoff and Woolfenden 2003, 2006; Ostreiher and Heifetz 2019; Ostreiher et al. 2021), showed that additional food drives the receivers to sentinel more than usual. This finding provided us the ability to compare the responses of the group members to the sentinel behaviour of one of them in three situations: after a sentinel bout in which the sentinel warned its group mates from an approaching predator, after a sentinel bout without warning, and during ordinary foraging. We hypothesized that after warning from danger, rewards are more common than without warning, and after sentinel activity in general (i.e., with or without warning), rewards are more common than during foraging. A previous study showed that group members do not increase the amount of their sentinel bouts in reaction to the increase in sentinel behaviour of one of them who received additional food (Wright et al. 2001c). Other studies found considerable differences in the amount of sentinelling between the group members (Clutton-Brock et al. 1999; Wright et al. 2001a, b; Bednekoff and Woolfenden 2003, 2006; Hollen et al. 2011; Kern et al. 2016; Walker et al. 2016). It seems, therefore, that increased sentinel activity by others is not the main factor influencing an individual’s own sentinel behaviour. However, sentinel activity could still be reciprocated by other kinds of rewards, such as reduced aggression, increasing friendly interactions like allopreening, and maybe even more copulations in the breeding season. Kern and Radford (2018) indeed found that dwarf mongooses reward sentinels by additional grooming before they go to sleep, sometimes some hours after terminating the sentinel bout. Their finding is remarkable because non-primate animals trade one type of good (defense) with another one (grooming), separate in time between the contribution and the reward, and the reward was usually given to the sentinel by one individual while the contribution was given to the whole group.
Feeding experiments revealed that additional food led to additional sentinelling (Ostreiher and Heifetz 2019; Ostreiher et al. 2021). We conducted a feeding experiment in order to test the responses of the group members to varying amounts of sentinel effort. We asked (1) Does the group members’ intensity of different social interactions vary with their peers’ sentinel activity? And (2) Does it matter whether that individual is a female or a male, and dominant or a subordinate in the social hierarchy of the group? If group members’ behaviour towards the sentinel is changes according to its sentinel effort, we expect that this change will be more pronounced for subordinates than for dominants as well as for females as compared to males since dominants and males usually sentinel more than others. We carried out the study at the beginning of the breeding season, in those days in which babblers copulate. We focused attention on three types of interactions: aggression, allopreening, and copulations. These are important measures that express agonistic (aggression) and friendly (allopreening) relationships between babblers. In addition, copulations express fitness in the short term better than any other measure. All three activities — aggression, allopreening, and copulations — are common among the group members and are easily detectable by the observers. Other social behaviours such as vocalizations, cooperation in foraging and mobbing, are less frequent, more subject to interpretation, and/or are more difficult to record accurately. For these reasons, we restricted attention in this study to the above three types of social interactions.
We hypothesized that if rewards may incentivize sentinel activity, then foragers reduce their aggression toward their peer who has just sentineled intensively, allopreen it more often, and copulate with it more, especially after being warned by it from an approaching predator. We further hypothesized that the effects of increased sentinel activity are more pronounced in subordinates and or females as these usually sentinel less frequently than dominants and males.
Methods
The study was carried out at the Shezaf Nature Reserve in the Arava Valley, in the southeastern part of Israel. The study area, the local babbler population, the Arabian babbler’s social system, and our fieldwork methods have been described by Zahavi (1989, 1990) and by Ostreiher and Heifetz (2016, 2017). At the time of the study, the study area contained about 160–260 individuals who lived in 25–32 groups. Each group was observed at least twice a week and almost every day when a group was actively nesting. The nestlings were ringed when they were 10 days old, 4 days before fledging, with four coloured rings in a unique combination. The babblers were accustomed to human presence, so we were able to stay in their vicinity as much as we wanted.
The study could have not been conducted without a feeding experiment because it would have taken many years to collect the data, and the composition of the group would have been meanwhile changed. Because dominants sentinel more than subordinates, we found it necessary to compare the reactions of the group members toward dominants versus their reactions toward subordinates. We knew in advance that following feeding, subordinates increase their sentinel activity more than dominants (Ostreiher and Heifetz 2019), and reasoned that the larger difference in their sentinel activity would potentially produce stronger reactions by their group mates, which would facilitate us to detect the reward. Moreover, we reasoned that the comparison of the reactions toward dominants versus subordinates would enable us to quantify the relationship between sentinel effort and the given rewards. Because males sentinel more than females, the same reasons caused us to compare also between the sexes.
The experiment was carried out in 20 different groups. In each group, two adult individuals, the most dominant and the most subordinate, were chosen as the focal babblers for the experiment. In ten groups, the two focal babblers were males and in ten groups females. The study was carried out in the breeding season, in the stages of nest-building and copulations, in order to find out whether additional sentinelling may lead to additional copulations (according to Zahavi 1990; Dattner et al. 2015). For the males, the groups that were chosen for the experiment were either polyandric groups (composed by one breeding female with two or more breeding males) or complex (composed of two or more breeding males and two or more breeding females). For the females, the groups that were chosen were polygynous groups (composed of one breeding male and two or more breeding females) or complex ones. In both cases, we chose groups in which the subordinates were potential breeders (not relatives of the breeders of the opposite sex) and competed for breeding.
In each group, the experiment continued 12 subsequent mornings. Every morning, the observation started at first light, when the group descended from the roost tree. Data collection started 30 min later, and continued for 3 h. In six mornings, we observed the group and no manipulation was performed. In the other six mornings, alternately, we fed one focal babbler, either the dominant or the subordinate, with mealworms (Tenebrio molitor) as much as it was willing to eat. We fed it three times: before the beginning of the 3-h observation, once again about 1 h later, and once again about 2 h later. We documented all the focal babbler’s physical interactions with other group members, including copulations, aggression, and allopreening that it received from others. Sentinel time was measured in seconds with a stopwatch. Days 1, 2, 5, 6, 9, and 10 were dedicated to one focal babbler, and days 3, 4, 7, 8, 11, and 12 were dedicated to the second one. In ten groups, we started with the dominant babbler, and in ten groups, we started with the subordinate. In each pair of days, the first day was dedicated to observation only, and no manipulation was carried out. These days were considered as control. The following day was dedicated to feeding the focal babbler of that day. This way we repeated the same treatment three times, and each focal babbler served as its own control. We compared the behaviour of the focal babbler’s group mates between days in which it gained additional food versus days which served as control. During the study, 1631 sentinel events were performed: 692 events under control conditions and 939 events following feeding. The distribution of the sentinel events between the four focal babblers is presented in Table 1, but this data set was not analysed because it is out of the focus of the current study and it repeats previous similar ones (Ostreiher and Heifetz 2019; Ostreiher et al. 2021). The experiment also enabled us to compare the reactions of the group members to the sentinel behaviour of the dominant male/female versus their reaction to the sentinel behaviour of the subordinate male/female. In each group, the 12-day series started when they started to copulate, several days after the beginning of nest building.
Out of 1631 sentinel events that were performed by 40 focal babblers, in 390 events (23.9%), an aerial raptor or a terrestrial predator approached the group. In all the cases, the sentinel was the first to notice and uttered alarm calls, and all the group members reacted to its calls. They stopped immediately what they were doing at that moment and either escaped into shelter within a bush or climbed up to the top of a tree and joined the sentinel in calling as long as the raptor or the predator was seen. None of these events was followed by a predation attempt. The raptor or the predator moved on and disappeared, and the group members returned to their routine, usually to forage. Out of these cases, for each focal babbler, we analysed three randomly chosen events in which the sentinel finished to sentinel when the predator disappeared, joined its group mates, and foraged with them at least 20 min immediately after ending the sentinel bout. We compared the focal babbler’s social interactions with its group members during a 20-min foraging across three conditions: Following a sentinel bout in which the sentinel warned its group mates from an approaching predator, following a sentinel bout without alarm calls, and during a 20-min foraging which took place on control days, did not follow a sentinel bout in the preceding hour, and started after at least 5 min intermission in foraging. We documented each of these three situations three times for each of the 40 focal babblers. Each of the nine 20-min samples (three following a sentinel bout with alarm calls, three following a sentinel bout without alarm calls, and three which did not follow a sentinel bout) was taken randomly from a different day.
For each focal babbler, we documented its mutual interactions during 9 * 20 = 180 min (3 h), totalling 7200 min (120 h) for 40 individuals. In two cases, the observations and the experiment were stopped before ending because the group started to incubate, and at least one of them, sometimes more than one, stayed in or near the nest, while other group members foraged far away. In these cases, the data were discarded, and a new set of observations, with another group, was conducted in the following year.
In general, we documented the behaviour of 126 individuals living in 20 groups, 40 of which were the focal babblers. Each group was observed 36 h (12 days * 3 h) leading to a total of 720 h (20 groups * 36 h) during 240 days (20 * 12) for the whole project. The field work started on 24.3.2009 and ended on 16.5.2015. It was not possible to record data blind because our study involved focal animals in the field.
Statistical analyses
To test our hypothesis that group mates do reward the sentinel at the end of the sentinel bout, in particular, after the sentinel uttered alarm calls towards a predator and its group mates benefited from its warning, we fitted a total of three generalized linear mixed models (Baayen 2008). These differed mainly with regard to the behaviour investigated (i.e., the response variable) but were otherwise largely identical. We fitted one model for each of the number of aggressions received (model 1), the number of instances of allopreening received (model 2), and the number of copulations (model 3). In all models, condition (factor with three levels: control period before which the focal did not sentinel; after a period of sentineling by the focal during which it did not give a warning; after a period of sentineling by the focal during which it gave a warning) was the key predictor with fixed effect. It seemed plausible that the effect of condition differs according to the rank and sex of the focal. More precisely, we reasoned that the effects of additional sentinel activity could be more pronounced for subordinates and/or females as these usually show less sentinel activity (see “Introduction”). Furthermore, subordinates and females usually receive more aggression and subordinates copulate less frequently. Hence, for such individuals, the effects of additional sentinel activity might be particularly pronounced. For these reasons, we included fixed effects for the rank and sex of the focal individual and its or their interaction(s) (see below) with condition into each model.
Since in our data dominant males never received aggressions, we excluded them from the data for model 1. For model 1, we then combined the information about the sex and rank of the focal individual in a single factor. In the other two models, we included them separately (see Table 2 for an overview of the models). Into all models, we included random intercepts for the identity of the focal and the group.
All models included random slopes for a priori reasons, though alternative analyses suggested in review show essentially the same results (see Supplementary Information).
As an overall significance test of the effects of condition and its interaction(s) with sex and rank, we compared each full model with a respective null model lacking the respective terms in the fixed effects part. When this full-null model comparison reveals significance, this indicates that condition and/or one or several of its interactions with the other predictors had a significant effect on the response. For testing the significance of individual fixed effects, we compared a model with and without the respective effect (R function drop1). All model comparisons utilized likelihood ratio tests (Dobson 2002). For more details about the statistical analysis, see the supporting information.
Results
The responses of the group members toward the sentinel
Aggression
During the data collection, we observed 167 aggressive interactions directed toward the focal babblers. None of them was directed toward dominant males. Subordinate males were subject to 31 instances of aggression, dominant females subject to 46, and subordinate females subject to 90.
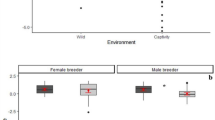
Dominant females, subordinate females, and subordinate males all received at least as much aggression following sentinel bouts as during control periods, regardless of whether they gave alarm calls during sentinel bouts (full-null model comparison χ2 = 6.809, df = 6, P = 0.339). If at all, aggression occurrences were slightly more frequent following sentinel bouts (Fig. 1a; Table SI 1), in contrast with our hypothesis.
Received number of aggressions (a), allopreenings (b), and copulations (c) during 20 min of foraging in the three conditions (after a control period without sentineling; after a sentinel bout without a warning from predator, and after a sentinel bout with a warning from a predator), separately for the different sexes and ranks. Dots show the individual observations, whereby the area of the dots is proportionate to the number of identical observations (range 1 to 27). Horizontal line segments with error bars depict the fitted model and its 95% confidence limits
Allopreening
Allopreening was a common behaviour in the groups, and 407 events were observed. Dominant males were preened 76 times, dominant females 106 times, subordinate males 109 times, and subordinate females were preened 116 times. No significant differences were found between receiving allopreening during a control period, after a sentinel bout without the sentinel warning its group mates from an approaching predator, and after a sentinel bout with such a warning (full-null model comparison χ2 = 12.99, df = 8, P = 0.112; Fig. 1b; Table SI 2). Here too, the differences were in contrast to our hypothesis, as focal individuals received slightly less allopreening after they had been sentinels (Fig. 1b).
Copulations
The study was conducted in the breeding season, and during a net time of 120 h, the 40 adult individuals (3 h per each focal babbler) were involved in 261 copulations. Dominant males copulated a total of 93 times, dominant females copulated 112 times, subordinate males 33 times, and subordinate females copulated 23 times. For dominant individuals, the numbers of copulations were very similar after control periods, after a sentinel bout without a warning given, and after sentinel bouts in which the sentinel warned its group mates from an approaching predator (Fig. 1c). In contrast with our hypothesis, however, subordinate individuals had significantly fewer copulations after a sentinel bout during which they uttered alarm calls (Fig. 1c; full-null model comparison χ2 = 16.265, df = 8, P = 0.039; see Supporting Information).
Discussion
In this study, we compared the behaviour of the group members toward the sentinel after it acted as sentinel (with or without a warning about a predator) and after a control period. The sentinel warns its group mates from approaching predators, and its absence among the foragers may reduce their competition over food. In group-living animals who develop long-term relationships, if sentinel activity is at least partially incentivized by group members, then at the end of the sentinel bout, group mates would reward the sentinel by being less aggressive towards it during foraging, maybe by preening it more frequently, and even, in the breeding season, copulating with it more often. These are common behaviours between the group members.
The Arabian babbler group members did not reward the sentinel in any noticeable way. Sentineling did not yield more tolerance in foraging as compared with control days and regardless of whether they warned or not from an approaching predator. Individuals who sentineled more than usual were not preened more and did not copulate more than usual. In fact, subordinate individuals, who normally copulate much fewer than the dominants, experienced significantly less copulations following a sentinel bout during which they uttered alarm calls towards a predator. In this sense, they were exploited rather than rewarded for the benefit they provided to the group.
In contrast to dwarf mongooses, who rewarded the sentinels by additional grooming (Kern and Radford 2018), Arabian babblers seemingly ignored the sentinels, at least within the period investigated. Moreover, subordinate babblers who finished to sentinel and started to forage suffered a little more aggressive interactions and gained significantly fewer copulations. In addition, without sentinelling all the focal individuals received slightly more allopreening (though not significantly so). We hypothesize that after ending to sentinel, the babblers were hungry and invested in foraging rather than in social activities. Moreover, joining the foragers could increase competition between foragers, maybe breach some equilibrium among them and evoke aggression. One difference between our study and Kern and Radford’s study is that we investigated immediate rewards, while they investigated delayed rewards. While we cannot rule out other rewards on other time scales, the data in hand are the opposite of predicted for immediate social rewards in Arabian babblers. Our study did not test Zahavi’s hypothesis (1990) that the sentinels gain prestige by their group members, because the term itself is vague and no testable predictions have been suggested for it so far. However, potential implications of prestige, namely tolerance in foraging, allopreening, and copulations, were not found in this study. Likewise, the lack of rewards rejects Trivers’s hypothesis of reciprocal altruism in this species.
While sentinel behaviour is often done by organisms with rich social lives, it is also carried out by Arabian babbler floaters with no group members with them (Ostreiher and Heifetz 2017, 2020). The results of this study support further the hypothesis that sentinel behaviour may be independent of rewards by others, as was suggested by Bednekoff (1997, 2001) and supported by Clutton-Brock et al. (1999) and Wright et al. (2001b), in spite of the fact that group members derive direct benefits from it. Our study opens the door for employing the same experimental procedure in other cooperatively breeding species. Cooperation and joint field work of cooperative breeder researchers are timely for understanding central questions in the behavioural ecology and sociobiology of cooperation.
Data availability
All data generated or analysed during this study are included as supplementary information in this article.
References
Baayen RH (2008) Analyzing linguistic data. Cambridge University Press, Cambridge
Bednekoff PA (1997) Mutualism among safe, selfish sentinels: a dynamic game. Am Nat 150:373–392
Bednekoff PA (2001) Coordination of safe, selfish sentinels based on mutual benefits. Ann Zool Fenn 38:5–14
Bednekoff PA (2015) Sentinel behaviour: a review and prospectus. Adv Stud Behav 47:115–146
Bednekoff PA, Woolfenden GE (2003) Florida scrub-jays (Aphelocoma coerulescens) are sentinels more when well-fed (even with no kin nearby). Ethology 109:895–904
Bednekoff PA, Woolfenden GE (2006) Florida scrub-jays compensate for the sentinel behaviour of flockmates. Ethology 112:796–800
Clutton-Brock TH, Brotherton PNM, O’Riain MJ, Griffin AS, Gaynor D, Sharpe L, Kansky R, Manser MB, McIlrath GM (2000) Individual contributions to babysitting in a cooperative mongoose, Suricata. Proc R Soc Lond B 267:301–305
Clutton-Brock TH, O’Riain MJ, Brotherton PNM, Gaynor D, Kansky R, Griffin AS, Manser M (1999) Selfish sentinels in cooperative mammals. Science 284:1640–1644
Dattner A, Zahavi A, Zahavi A (2015) Competition over guarding in the Arabian babbler (Turdoides squamiceps), a cooperative breeder. F1000Res 4:618
Dobson AJ (2002) An introduction to generalized linear models. Chapman and Hall/CRC, Boca Raton
Edelaar P, Wright J (2006) Potential prey makes excellent ornithologists: adaptive, flexible responses towards avian predation threat by arabian babblers Turdoides squamiceps living at a migratory hotspot. Ibis 148:664–671
Gaston AJ (1977) Social behaviour within groups of jungle babblers, Turdoides striatus. Anim Behav 25:828–848
Hamilton WD (1964) The genetical evolution of social behaviour. II J Theor Biol 7:17–52
Hollen LI, Bell MBV, Radford AN (2008) Cooperative sentinel calling? Foragers gain increased biomass intake. Curr Biol 18:576–579
Hollen LI, Bell MBV, Wade HM, Rose R, Russell A, Niven F, Radford AN (2011) Ecological conditions influence sentinel decisions. Anim Behav 82:1435–1441
Kareksela S, Härmä O, Lindstedt C, Siitari H, Suhonen J (2013) Effect of Willow Tit Poecile montanus alarm calls on attack rates by Pygmy Owls Glaucidium passerinum. Ibis 155:407–412
Kern JM, Radford AN (2018) Experimental evidence for delayed contingent cooperation among wild dwarf mongooses. P Natl Acad Sci USA 115:6255–6260
Kern JM, Sumner S, Radford AN (2016) Sentinel dominance status influences forager use of social information. Behav Ecol 27:1053–1060
Kingma SA, Santema P, Taborsky M, Komdeur J (2014) Group augmentation and the evolution of cooperation. Trends Ecol Evol 29:476–484
Kokko H, Johnstone RA, Clutton-Brock TH (2001) The evolution of cooperative breeding through group augmentation. Proc R Soc Lond B 268:187–196
Lundy K, Parker P, Zahavi A (1998) Reproduction by subordinates in cooperatively breeding Arabian babblers is uncommon but predictable. Behav Ecol Sociobiol 43:173–180
McGowan KJ, Woolfenden GE (1989) A sentinel system in the Florida scrub jay. Anim Behav 34:1000–1006
Naguib M, Mundry R, Ostreiher R, Hultch H, Schrader L, Todt D (1999) Cooperatively breeding Arabian babblers call differently when mobbing in different predator-induced situations. Behav Ecol 10:636–640
Ostreiher R, Heifetz A (2016) The blessing of having younger nestmates: the case of the Arabian Babbler. Behav Ecol 27:393–400
Ostreiher R, Heifetz A (2017) The sentinel behaviour of Arabian babbler floaters. R Soc Open Sci 4:160738
Ostreiher R, Heifetz A (2019) The sentineling—foraging trade-off in dominant and subordinate Arabian babblers. Ethology 125:98–105
Ostreiher R, Heifetz A (2020) The function of sentinel alarm calls in the Arabian babbler. J Avian Biol 51:e02513
Ostreiher R, Mundry R, Heifetz A (2021) On the self-regulation of sentinel activity among Arabian babbler groupmates. Anim Behav 173:81–92
Rasa OAE (1986) Coordinated vigilance in dwarf mongoose family groups: the ‘watchman song’ hypothesis and the costs of guarding. Ethology 71:340–344
Rasa OAE (1989) The costs and effectiveness of vigilance behaviour in the Dwarf Mongoose: implications for fitness and optimal group size. Ethol Ecol Evol 1:265–282
Regosin JV (2002) Alarm calling and predator discrimination in the Arabian babbler (Turdoides squamiceps). Bird Behav 15:11–19
Ridley AR, Nelson-Flower MJ, Thompson AM (2013) Is sentinel behaviour safe? An experimental investigation. Anim Behav 85:137–142
Santema P, Clutton-Brock T (2013) Meerkat helpers increase sentinel behaviour and bipedal vigilance in the presence of pups. Anim Behav 85:655–661
Sherman PW (1977) Nepotism and the evolution of alarm calls. Science 197:1246–1253
Sommer C (2011) Alarm calling and sentinel behaviour in Arabian babblers. Bioacoustics 20:357–368
Sommer C, Todt D, Ostreiher R, Mundry R (2012) Urgency-related alarm calling in Arabian babblers, Turdoides squamiceps: predator distance matters in the use of alarm call types. Behaviour 149:755–773
Sorato E, Gullett PR, Griffith SC, Russell AF (2012) Effects of predation risk on foraging behaviour and group size: adaptations in a social cooperative species. Anim Behav 84:823–834
Trivers RL (1971) The evolution of reciprocal altruism. Q Rev Biol 46:35–57
Walker LA, York JE, Young AJ (2016) Sexually selected sentinels? Evidence of a role for intrasexual competition in sentinel behaviour. Behav Ecol 27:1461–1470
Wright J, Berg E, de Kort SR, Khazin V, Maklakov AA (2001a) Cooperative sentinel behaviour in the Arabian babbler. Anim Behav 62:973–979
Wright J, Berg E, de Kort SR, Khazin V, Maklakov AA (2001b) Safe selfish sentinels in a cooperative bird. J Anim Ecol 70:1070–1079
Wright J, Maklakov AA, Khazin V (2001c) State-dependent sentinels: an experimental study in the Arabian babbler. Proc R Soc Lond B 268:821–826
Zahavi A (1989) The Arabian babbler. In: Newton I (ed) Lifetime reproduction in birds. Academic Press, London, pp 253–275
Zahavi A (1990) Arabian babblers: the quest for social status in a cooperative breeder. In: Stacey PB, Koenig WD (eds) Cooperative breeding in birds. Cambridge University Press, London, pp 103–130
Zahavi A, Zahavi A (1997) The handicap principle: a missing piece of Darwin’s puzzle. Oxford University Press, Oxford
Acknowledgements
RO is grateful to the Society for Protection of Nature in Israel (SPNI) and Hazeva Field Study Center for hosting the research project. RO is also grateful to Avner Anava, Yoel Perel, Amir Kalishow, Yuval Dagay, Arnon Datner, and Yael Alon for field assistance. We are very deeply grateful to the Associate Editor Peter Bednekoff and to two anonymous referees for their arousing and fruitful comments and suggestions which improved the manuscript very much.
Funding
Partial financial support was received from The Open University of Israel’s Research Fund (grant no. 37142).
Author information
Authors and Affiliations
Contributions
All the authors made substantial contributions to the conception and design of the work as well as the analysis and interpretation of data. All the authors drafted the work and revised it and approved the version to be published.
Corresponding author
Ethics declarations
Ethics approval
The study and the experiment meet the ASAB guidelines for ethical treatment of animals and adhered to the requirements of the Israel Nature and Parks Authority (Permit no. 30134, 31139, 32915, 41233 and 41723).
Competing interests
The authors declare no competing interests.
Additional information
Communicated by P. A. Bednekoff
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ostreiher, R., Mundry, R. & Heifetz, A. Are sentinels rewarded by their groupmates? A study in a cooperative breeder. Behav Ecol Sociobiol 77, 125 (2023). https://doi.org/10.1007/s00265-023-03403-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03403-w