Abstract
Purpose
To elucidate ultrasound features of normal placental anatomy through correlation of gray-scale and ultrasound Doppler with ferumoxytol-enhanced MRI features using US-MR image fusion.
Methods
All patients referred to MR for ultrasound findings worrisome for PAS (placenta accreta spectrum) were included in this retrospective study. MR studies included a ferumoxytol-enhanced T1-weighted MRI. Ultrasound imaging included gray-scale, color Doppler, power Doppler, and spectral Doppler techniques. After the MR, US-MRI fusion was performed by co-registering a MR acquisition to real-time US, which allowed precise, point-to-point correlation of placental features.
Results
Fourteen patients at risk for PAS were studied using the US-MR image fusion. At delivery, there were six cases without PAS (gestational age range: 24 weeks 3 days to 34 weeks 0 days), and these composed the study cohort. Placental features that were on high signal intensity on post-ferumoxytol acquisitions represent spaces with maternal blood flow and corresponded to hypoechoic areas on ultrasound created by a paucity of reflective interfaces (villi). Color and spectral Doppler allowed the separation of maternal and fetal circulations in individual perfusional domains and demonstrated spiral artery inflow, circulation around the villous tree, and return of blood flow to the basal plate. Recognizable histopathologic features by ultrasound included the central cavity, villous tree, and venous return channels.
Conclusion
Internal placental architecture can be discerned on ultrasound. This anatomy can be correlated and confirmed with ferumoxytol-MR through US-MR fusion. Understanding this structural anatomy on ultrasound could serve as a basis to identify normal and abnormal placental features.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The internal architecture of the placenta remains uncharted by ultrasound [1, 2]. Basic structural features—such as the placentone, the villous tree, and blood flow channels—are well described in the pathology literature but have not been correlated to ultrasound features in vivo.
A recent study, however, has elucidated the architecture of the fetal cotyledon – the placentone – by ferumoxytol-enhanced MR [3, 4]. The placentone is the fundamental unit of the placental perfusion. Placentone structure is symmetric around an axis drawn from a stem villous (the trunk of the villous tree) on the chorionic plate to the opposing inlet of a maternal spiral artery on the basal plate (Fig. 1). [3, 5, 6]. Over the spiral artery inlet, at the center of the placentone, there is a space or central cavity with few or no villi [3].
The placentone (fetal cotyledon) is the fundamental anatomic and perfusional unit of the placenta. Maternal arterial inflow at the basal plate is opposite a fetal stem villous at the chorionic plate. At the center of the placentone, there is around a relatively villus-free central cavity over the inlet of maternal spiral artery [3, 5, 6]. Around the central cavity is a corona of dense villi. At the periphery of the villous tree, maternal blood circulates back to the basal plate along venous outflow channels
As with all organs, the echotexture of the placenta derives from the density and orientation of reflective interfaces. In the placenta, the reflective interfaces are the numerous villi that branch from a stem villous and fill the intervillous space. A normal placenta at term has 60–70 stem villi that extend from the chorionic plate and give rise to between two to thirty branching generations of villi (average of ten generations) [3]. At the end of the finer peripheral branches are a multitude of terminal villi, tiny grape-like buds, each a small reflective interface [3]. The number and concentration of villi therefore determine the parenchymal echotexture of any placental location.
The goal of this study was to identify normal placental anatomic features evident by ultrasound using ferumoxytol-enhanced MR and ultrasound-MR fusion techniques. US-MR fusion software allows a previously acquired MR image volume to be loaded into an ultrasound system and co-registered with the real-time ultrasound to track MR and US images together. This real-time tracking provides a means to compare corresponding MR and US features precisely and unambiguously.
Methods
This retrospective study was approved by our institutional review board with waiver of informed consent. All patients referred to MR for evaluation of PAS (placenta accreta spectrum) were prospectively studied according to our standard ultrasound and MR imaging protocol which included ultrasound-MR image fusion; secondary analyses of prospectively collected data were performed in a random order.
For US-MRI image fusion, a fiducial sensor was taped securely over an iliac crest prior to MRI imaging. Co-registration of the real-time ultrasound with the MRI images was performed using the fiducial sensor initially and then refined manually. Both types of co-registration required that the patient be positioned within a weak magnetic field. Manual co-registration was performed in the sagittal plane at the internal os, or a salient feature of the placenta, such as a large vessel or lobulation.
For the ultrasound study, gray-scale, spectral and color Doppler scanning were performed. Gray-scale imaging examined for local echogenicity, vascular spaces, contours, and organization. Color Doppler was used to map both maternal and fetal flow within the placenta. Spectral Doppler tracings were acquired to evaluate flow character (arterial vs. venous) and pulse rate (maternal vs. fetal). Maternal and fetal heart rates were recorded at the beginning of the examination.
MRI imaging was performed on a clinical 1.5 T GE system (Artist, GE Healthcare) using a standard protocol that included pre-contrast SSFSE (single-shot fast spin-echo), steady-state free precession (SSFP), and diffusion weighted imaging (DWI) obtained in the sagittal, axial, and coronal planes (appendix). SSFP is a rapid method widely used in vascular imaging [7]. In 10 of the 14 cases, ferumoxytol (AMAG Pharmaceuticals, Waltham, MA, USA) was infused intravenously over 15 min at a dose of 4 mg/kg, diluted 5:1 with sterile saline, according to the recommended safety protocols [8] T1-weighted (T1w) post-contrast enhanced imaging was performed using 3D T1w spoiled gradient echo imaging with spectrally selective intermittent fat suppression (LAVA, GE Healthcare).
Ferumoxytol does not cross from the maternal circulation into the fetal circulation, therefore all areas of enhancement represent tissue perfused by maternal blood flow. Post-contrast images were compared to pre-contrast SSFSE, SSFP, DWI, and the US-MR fusion image obtained in the same plane for features of correspondence.
The ultrasound and MR images were examined for anatomic landmarks documented in the histopathologic literature including the cavity of sparse villi at the center of the villous tree, the surrounding ring of clustered villi, and the outer ring of looser, more dispersed villi corresponding to the maternal venous outflow channel (Fig. 1). The sizes of these imaging features were compared to reported sizes from histologic reports.
Results
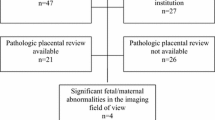
Over a three-year period, 14 patients at risk for PAS were studied using the US-MR image fusion. The indications for the MRI evaluations included one or more of the following ultrasound findings: placental hypervascularity (n = 14), myometrial thinning/ interrupted retroplacental clear space (n = 13), multiple intraplacental lakes/lacunae (n = 4), and placental heterogeneity (n = 3). All studies were performed at a single institution. Of the 14 patients, six patients had normal placentas at delivery and eight had abnormal placentas at delivery.
The six patients with normal placentas at delivery make up the study group of this report. The gestational age of these six pregnancies ranged from 24 weeks 3 days to 34 weeks 0 days (mean 30 weeks 5 days; median 30 weeks 4 days).
Post-ferumoxytol T1w images demonstrated enhancement of the maternal blood flow within the placenta, notably the intervillous space and the subplacental (myometrial) vessels (Figs. 2, 3). The signal intensities within the placentone replicate the relative contributions from enhanced maternal blood and unenhanced chorionic villi. The central cavity of the placentone is highest in signal intensity due to the sparsity of villi. The darker ring surrounding the central cavity reflects the high density of villi, and the gray outer ring reflects an intermediate density of villi produced by dispersion within the maternal venous outflow channel.
Three ring organization of the placentone. a US-MR fusion image of a normal placentone at 30 weeks 5 days. On the US image, the placentone (between arrows) is evident as a hypoechoic center (central cavity) surrounded by a thick echogenic ring (villous tree), surrounded by a hypoechoic ring (sparse villi). On the SSFP MR image, this anatomy is evident as a white center (arterial inflow area), dark ring (dense peripheral villi), and gray periphery (maternal venous outflow area). b. Enlarged SSFP MR image. c. Ferumoxytol-enhanced MR image shows the same pattern with greater clarity. The signal intensities of the placentone represent the relative admixture of enhanced maternal blood and unenhanced fetal villi
US-MR fusion image with color Doppler of a normal placenta at 30 weeks 4 days. The color Doppler images shows the blood flow in a stem villous at the center of the placentone (between arrows). Co-registered ferumoxytol-enhanced MR image shows the structure of the corresponding structure of the placentone (between arrows)
Comparison of MR images to the co-registered US images (US-MR fusion) revealed a correspondence of placental architecture (Figs. 2, 3 and 4). Placental features that were on high signal intensity (bright) on SSFP and post-contrast acquisitions represent spaces with maternal blood flow, such as the central cavity and the maternal venous return channels. Corresponding spaces on ultrasound were relatively hypoechoic due to the paucity of villi serving as reflective interfaces. Placental features that are low signal intensity (dark) on SSFSE, SSFP, and ferumoxytol-enhanced images represent unenhanced structures, such as villi. Corresponding spaces on ultrasound were relatively hyperechoic due to the abundance of villi serving as reflective interfaces.
a US-MR fusion image with color and spectral Doppler of a normal placenta at 24 weeks 3 days. Fetal stem villous in red (black arrows). Maternal venous outflow channels (white arrowheads) on either side of the villous tree. b Spectral Doppler tracing at the basal plate shows attenuated pulsatile maternal arterial flow as it enters the central cavity of the placentone. Note flow direction (colored blue). The underlying maternal heartrate is 88 bpm. c Spectral Doppler tracing shows fetal arterial flow in a stem villous as it enters the placenta from the chorionic plate (note red flow direction) diametrically opposite the maternal spiral artery on the basal plate. Fetal heart rate is 150 bpm. d Spectral Doppler tracing at the periphery of the placentone reveals maternal venous flow returning to the basal place of the placenta (note red flow direction). Peak velocity of the venous tracing is 10 cm/s
The dimensions of the anatomic features within the placentone are similar between MR, ultrasound, and histopathology (Table 1). Histopathologic diameters were obtained from the literature [4, 5].
Pulsatile maternal arterial blood was demonstrated at the basal plate by spectral and color Doppler in four of the six cases (67%). Fetal arterial blood flow directed at the central cavity was found in all six cases (100%). Venous flow directed toward the basal plate was seen at the periphery of the placentone in all six cases (100%). With gentle compression, the central cavity collapsed in all six cases (100%) (Fig. 5).
Normal placenta in a patient at 33 weeks 0 days. Two central cavities within placentones (arrows) before (a) and after (b) gentle transducer pressure. Note that the central cavities – the hypoechoic centers surrounded by rounded echogenic ring – collapse with pressure, confirming them as the low-pressure structures
Discussion
This study demonstrates a correspondence of intraplacental anatomy observed with ultrasound and MR. The use of US-MR fusion imaging allows precise point-to-point comparison of placental features as represented by these two modalities. Those areas that were higher in echogenicity on ultrasound and lower in signal intensity on SSFP and post-contrast acquisitions represent areas of abundant villi within the intervillous space. Conversely, those areas that were lower in echogenicity on ultrasound and higher in signal intensity on SSFP and post-contrast acquisitions represent areas within the maternal blood circulation containing sparse villi.
Ferumoxytol is a super-paramagnetic iron oxide (SPIO) approved for intravenous treatment of iron deficiency anemia in pregnant women and patients in renal failure [9, 10]. As an alternative to gadolinium-based agents for MR contrast, it is commonly used in pediatrics, pregnancy (e.g., for pulmonary embolus), and renal disease, among other indications [11,12,13,14,15,16]. Important for placental imaging, ferumoxytol remains within the maternal circulation and does not cross into the fetal circulation [11, 12, 17]. Therefore, ferumoxytol-enhanced MRI can be used to differentiate fetal and maternal vascular spaces.
In each placentone, maternal blood enters the intervillous space from the spiral artery, flows towards the chorionic plate, where it turns back toward the basal plate along venous outflow channels which wrap around the villous tree [5] (Fig. 1). At the center of the villous tree, there is a hollow villous-free space immediately over the inlet of the maternal spiral artery. This space or cavity is marginated by intermediate villi which tend to be morphologically immature with a paucity of terminal villi [3]. The surrounding crown of the villous tree, by contrast, is thick with terminal villi, much like abundant small leaves on a tree.
This three-ring pattern can be identified on ultrasound (Figs. 4, 6 and 7). The central cavity is an area of hypoechogenicity surround by a thick halo of echogenic villi. Around this thick halo, there is a thinner ring of intermediate echogenicity which results from dispersed villi within the venous blood flow returning toward the basal plate. Comparison between ultrasound with the corresponding MR demonstrate similar appearance and dimension of these structures. Furthermore, these dimensions are in agreement with values reported in the pathology literature [3].
Spectral and color Doppler imaging confirms this expected pattern of circulation of blood flow within the perfusion domain of the placentone. Pulsatile maternal blood flow directed into the central cavity from the spiral arteries can be found at the basal plate. Diametrically opposed to this maternal arterial inflow, fetal blood flow can be found arising from the chorionic plate, also directed toward the center of the plaecentone. At the periphery of the placentone, venous flow directed back to the basal plate along maternal venous outflow channels can be found.
With gentile compression, the central cavity collapses, confirming it to be a low-pressure vascular space. This collapse was seen in all normal placentas studied.
Similar Doppler waveforms have been described in prior reports [18]. These studies show the abundance of blood flow within the placenta but do not clearly separate the maternal and fetal circulations or contextualize blood flow within a coherent model of internal placental structure. In a mature placenta, some 40–60 stem villi give rise to a similar number of fetal cotyledons (placentones) [3]. To be clear, a placentone is not a separate anatomic compartment but rather a perfusion domain that is interconnected to other placentones within the intervillous space [17, 19].
This study has some limitations. First, pathologic correlation would have been ideal but extremely problematic as it would be difficult to restore the delivered placenta to the blood-filled and inflated state seen on US and MR. Second, the relatively small sample size prevents establishment of the range of normal placentone size and appearance over gestation. A more complete representation of the normal placental anatomy would require a larger, and ideally prospective multicenter study. The results from the current study may be useful in the design of future, comprehensive studies.
In summary, we describe the ultrasound appearance of the fundamental functional vascular exchange unit – the placentone – of the human placenta in vivo and confirm this identification with US-MR fusion imaging with ferumoxytol-enhanced MRI. This approach reveals an underlying anatomic structure to the placenta that has not been fully appreciated on ultrasound studies.
References
Fadl S, Moshiri M, Fligner CL, Katz DS, Dighe M (2017) Placental Imaging: Normal Appearance with Review of Pathologic Findings. Radiographics 37:979-998
Zaghal AA, Hussain HK, Berjawi GA (2019) MRI evaluation of the placenta from normal variants to abnormalities of implantation and malignancies. J Magn Reson Imaging 50:1702-1717
Benirschke Ka (2022) Benirschke's Pathology of the human placenta. Seventh edition. Cham : Springer, [2022] ©2022
Kliewer MA, Bockoven CG, Reeder SB, Bagley AR, Fritsch MK (2022) Ferumoxytol-Enhanced Magnetic Resonance Imaging with Volume Rendering: A New Approach for the Depiction of Internal Placental Structure In Vivo. Placenta, in press
Chernyavsky IL, Jensen OE, Leach L (2010) A mathematical model of intervillous blood flow in the human placentone. Placenta 31:44-52
Flouri D, Darby JRT, Holman SL et al (2021) Magnetic resonance imaging of placentome development in the pregnant Ewe. Placenta 105:61-69
Bhosale P, Ma J, Choi H (2009) Utility of the FIESTA pulse sequence in body oncologic imaging: review. AJR Am J Roentgenol 192:S83–93 (Quiz S94–87)
Vasanawala SS, Nguyen KL, Hope MD et al (2016) Safety and technique of ferumoxytol administration for MRI. Magn Reson Med 75:2107-2111
Gerb J, Strauss W, Derman R et al (2021) Ferumoxytol for the treatment of iron deficiency and iron-deficiency anemia of pregnancy. Ther Adv Hematol 12:20406207211018042
Huang Y, Hsu JC, Koo H, Cormode DP (2022) Repurposing ferumoxytol: Diagnostic and therapeutic applications of an FDA-approved nanoparticle. Theranostics 12:796-816
Benson DG, Schiebler ML, Nagle SK, Francois CJ (2017) Magnetic Resonance Imaging for the Evaluation of Pulmonary Embolism. Top Magn Reson Imaging 26:145-151
Benson DG, Schiebler ML, Repplinger MD et al (2017) Contrast-enhanced pulmonary MRA for the primary diagnosis of pulmonary embolism: current state of the art and future directions. Br J Radiol 90:20160901
Little JT, Bookwalter CA (2020) Magnetic Resonance Safety: Pregnancy and Lactation. Magn Reson Imaging Clin N Am 28:509-516
Starekova J NS, Schiebler ML, Reeder SB, Meduri VN. (2021) Ferumoxytol-enhanced Pulmonary MRA in Pregnancy: Evaluation of Initial Safety and Image Quality. International Society for Magnetic Resonance in Medicine Annual Meeting. ISMRM, Virtual
Aghayev A, Memon AA, Greenough PG, Nayak L, Zheng S, Siedlecki AM (2020) Alternative Diagnostic Strategy for the Assessment and Treatment of Pulmonary Embolus: A Case Series. Clin Pract Cases Emerg Med 4:308-311
Miller T, Chin MS, Gharagouzloo C et al (2021) Ferumoxytol-Enhanced Coronary Magnetic Resonance Angiography Compared to Invasive Coronary Angiography for Detection of Epicardial Coronary Artery Disease. Kidney Med 3:139-141
Nguyen SM, Wiepz GJ, Schotzko M et al (2020) Impact of ferumoxytol magnetic resonance imaging on the rhesus macaque maternal-fetal interfacedagger. Biol Reprod 102:434-444
Hernandez-Andrade E, Huntley ES, Bartal MF et al (2022) Doppler evaluation of normal and abnormal placenta. Ultrasound Obstet Gynecol 60:28-41
Frias AE, Schabel MC, Roberts VH et al (2015) Using dynamic contrast-enhanced MRI to quantitatively characterize maternal vascular organization in the primate placenta. Magn Reson Med 73:1570-1578
Acknowledgements
The authors thank Megan Vadnais, BSRT (R)(MR) and Christina K. Hendricks, RDMS for help with the studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There was no industry or grant support for the project. The authors did not receive support from any organization for the submitted work. The authors have no relevant financial or non-financial interests to disclose. The authors have no conflicts of interest.
Ethical approval
This retrospective study was approved by our institutional review board with waiver of informed consent. All patients were prospectively studied according to our standard imaging protocol.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Appendix. MR Protocol
1.5 T GE system; multichannel phased array abdominal coil; 30-35 minutes; radiologist at console to prescribe planes; Patient supine or left lateral decubitus (LLD).
-
T2 weighted SSFSE: sagittal, axial, coronal. 2D, slice/gap 3/0 mm, TE 100 ms, field of view (FOV) 30 × 30 cm, matrix 256 × 224, scan time ≈ 1.5 min
-
SSFP: sagittal, axial, coronal. 2D, slice/gap 4/0 mm, TR/TE Min/100 ms, flip angle 80°, 2 signal averages, FOV 30 × 30 cm, matrix 256 × 256, scan time ≈ 2 min
-
T1-weighted LAVA (breath hold). Prescibed plane. 3D, slice 3 mm TR/TE 4.1/Min Full, flip angle 12°, FOV 40 × 38 cm, matrix 288 × 160, scan time ≈ 20 s
-
DWI: Prescibed plane. b-value 50, 800. free breathing with respiratory triggering. 2D, slice/gap 4/0.5 mm, TE Min, FOV 38 × 40 cm, matrix 160 × 192. scan time ≈ 6 min
Ferumoxytol infusion: diluted in normal saline 5:1, slow infusion over 15 minutes. Scan after infusion is complete.
Post-contrast acquisitions (parameters as above).
High resolution non-fat saturated 3D T1w with low flip angle (prescribed plane)
Lava Flex T1 breath hold. Sagittal, axial, coronal planes.
SSFSE T2 (prescribed plane)
SSFP (prescribed plane)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kliewer, M.A., Bagley, A.R., Reeder, S.B. et al. Normal placental structural anatomy: ultrasound and doppler features elucidated with US-MR image fusion and ferumoxytol-enhanced MRI. Abdom Radiol 48, 744–751 (2023). https://doi.org/10.1007/s00261-022-03758-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03758-0