Abstract
Objective
Since it has been suggested that benign renal cysts can be diagnosed at unenhanced CT on the basis of homogeneity and attenuations of 20 HU or less, we determined the prevalence of renal cell carcinomas (RCCs) with these characteristics using two different methods of measuring attenuation.
Materials and methods
After IRB approval, two radiologists obtained unenhanced attenuation values of 104 RCCs (mean size 5.6 cm) using a single, large region of interest (ROI), two-thirds the size of the mass. They were then determined if the masses appeared heterogeneous. Of RCCs measuring 20 HU or less, those which appeared homogeneous were re-measured with multiple (6 or more), small (0.6 cm2 or smaller) ROIs dispersed throughout the lesion. Masses with attenuations 20 HU or less were compared to those with masses with HU greater than 20 for any differences in demographic data.
Results
Of 104 RCCS, 24 RCC had HU less than 20 using a large ROI. Of these, 21 appeared heterogeneous and 3 appeared homogeneous. Using multiple small ROIs, these three RCCs revealed maximum attenuation values above 20 HU (Range: 26–32 HU). A greater portion of RCCs measuring 20 HU or less using a large ROI were clear cell sub-type. There were no other differences.
Conclusions
Renal cell carcinoma can measure 20 HU or less at unenhanced CT when a single large ROI is used. While most appear heterogeneous, some may appear homogeneous, but will likely reveal attenuations greater than 20 HU when multiple, small ROIs are used. This knowledge may prevent some RCCs from being misdiagnosed as cysts on unenhanced CT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The diagnosis of renal masses has historically required either ultrasound, or a CT or MRI examination that is designed to evaluate a renal mass completely [1, 2]. However, renal masses are more commonly found incidentally with imaging exams that are not designed to evaluate them completely [3]. For example, unenhanced CT, used often to evaluate a variety of abdominal disorders, including flank pain and urolithiasis [4], often reveals incidental renal masses and thus radiologists are challenged with diagnosing their etiology. The primary goal is to diagnose renal cancers at an early curable stage, realizing most incidental renal masses detected at unenhanced CT are benign [1,2,3, 5].
Recent studies have suggested that benign renal cysts can be diagnosed and renal cancers excluded with confidence using unenhanced CT in masses which are homogeneous, and either measure 20 HU or less, or 70 HU or greater [3, 6,7,8]. These reports also emphasize that renal masses which are either heterogeneous, or homogeneous and contain attenuations greater than 20 HU and less than 70 HU might be cancers [9, 10]. Although most renal cell carcinomas are solid with attenuation values greater than 20 HU [10, 11], cystic renal cancers may have attenuations 20 HU or less in only a portion of the mass, and hence the number and size of the region of interest (ROI) used to measure the attenuation may be important. The literature is replete with data regarding the attenuation of renal masses and has used variable sizes and numbers of ROI’s [7, 12]. In fact, to our knowledge, a standard method of measuring attenuations of renal masses has not been described. Since it has been suggested that renal cell carcinomas (RCCs) can be diagnosed at unenhanced CT on the basis of heterogeneity and attenuations of 20 HU or more, we determined the prevalence of RCCs with these characteristics using two different methods of measuring attenuation.
Materials and methods
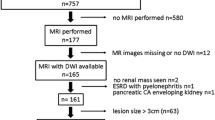
Institutional Review Board approval was obtained and informed consent waived for this retrospective HIPAA-compliant study. Of 162 pathology proven renal cell carcinomas (RCCs) between 2006 and 2014, 104 (mean size 5.64, range 1.0–15.5 cm) were examined with unenhanced abdominal CT. All CT scans were obtained in patients older than 18 years of age, within three months of establishing the diagnosis at pathology, and were performed either alone or as part of an examination that included contrast-enhanced CT images. CT scans were obtained using 16–64 channel (Light Speed, Light Speed VCT, General Electric, Milwaukee, WI) or a 64 to 128 channel multi-detector CT scanners (Definition Sensation 64, Definition AS Plus scanners, Siemens Medical Solutions, Malvern, PA). All images were acquired at 120 kVp and variable tube current. Pitch varied from 0.8 to 1.5. Images were reconstructed in the axial plane and with a section thickness ranging from 1.0 to 3.25 mm using a standard body kernel for the images acquired with General Electric CT scanners, and a B40F kernel for the images acquired with the Siemens Medical Solution scanners. Adaptive iterative reconstruction software was not used.
Renal mass analysis
Using radiology PACS work station (iSite version 3.6, Philips Healthcare), two abdominal radiologists with 34 years and 8 years of experience, respectively, independently evaluated the unenhanced CT images of each tumor. The average of the two largest perpendicular diameters was considered the diameter of each mass. Attenuation values were obtained using a large region of interest (ROI), approximately two-thirds the size of each renal mass, placed on the thinnest reconstructed image available (Fig. 1A), a method described previously by Scheida [12]. Masses with attenuation greater than 20 HU, as measured by both readers, were not analyzed further. The mean and range of the attenuation values of these masses were recorded. Masses with attenuation of 20 HU or less as measured by one or both readers were analyzed in consensus. Using window settings with a width of 400 and level of 40, masses which were not simple cyst-appearing, i.e., were either heterogeneous or had one or more septations or calcifications, were recorded and not evaluated further. The remaining homogeneous masses with an attenuation of 20 HU or less were analyzed using multiple, (>6) small (0.6 cm2 or smaller) ROIs by moving the ROI from region to region in the mass to identify regions of heterogeneity or higher attenuation (Fig. 1B). The mean attenuation value, maximum attenuation value, and range of the attenuation values of each of these masses were recorded.
Renal cell carcinoma attenuation values on unenhanced CT: diagrammatic representation of two measurement methods. A Single, large region of interest (ROI) encompassed the central, approximately two-thirds of a renal mass. B Multiple, (>6) small (0.6 cm2 or smaller) ROI attenuation measurements were positioned in multiple regions throughout each renal mass
Using data derived only from single, large ROI measurements of attenuation, masses with attenuations 20 HU or less were compared to those with attenuations greater than 20 HU for differences in patient age and gender, scanner type, mass size, and cancer sub-type. The Chi-square test was used to compare patient gender and CT scanner type. The ANOVA test was used to compare patient age and mass size. The 2-proportion z-test was used to compare cancer sub-type. All analyses were performed using the Analysis ToolPak on MS Excel (Version 2007, Redmond, WA); a p value of less than 0.05 was considered statistically significant.
Results
Of 104 RCCs, using a single, large ROI, 80 had an attenuation greater than 20 HU by both readers; 24 had an attenuation of 20 HU or less after consensus by both readers (mean, 16.5 HU, range 5.9–19.0) (Fig. 2). In our original independent review, in 22/24 there was agreement between both readers and in 2 there was disagreement, but consensus was reached as being <20 HU on these two. Of the 24 RCCs with an attenuation of 20 HU or less using a single, large ROI, 21 were not simple cyst-appearing renal masses. Of these, twelve were heterogeneous without other features (Fig. 3). Of the other nine that were heterogeneous, seven had calcifications, one had septations, and one had both septations and calcifications. There were three masses with HU less than 20, using a single large ROI. However, using multiple small ROI’s placed throughout the mass, at least one attenuation value greater than 20 HU (mean 29 HU range, 26–32 was obtained) (Figs. 4, 5, 6).
A 43-year-old male with large clear cell RCC. A Using a large ROI, the attenuation of this renal cell carcinoma was only 5.9 HU, but was considered heterogeneous in study reviewed. B Contrast enhancement shows an enhancing peripheral portion and a low density center. This tumor was a clear cell RCC at pathology
A 36-year-old male with left clear cell carcinoma. A Unenhanced CT illustrating left renal mass (arrow). B Unenhanced CT using a large ROI showed HU measurement of 16. It was considered homogeneous. C Unenhanced CT using a small ROI showed HU measurement of 25.6. D Corresponding enhanced CT demonstrating solid mass which was resected and revealed clear call RCC
A 59-year-old woman with a homogeneous-appearing RCC with HU 20 or less using a large ROI. A Unenhanced CT with 1.25 mm reconstruction showed a 4.2-cm homogenous appearing renal mass (arrow). B Unenhanced CT with 5 mm reconstruction showed a 4.2 cm homogenous appearing renal mass (arrow). C Unenhanced CT using a large ROI showed HU measurement of 18. D Unenhanced CT using a small ROI showed HU measurement of 32. E However, a follow-up CT urogram performed 11 months later for hematuria demonstrated that the mass grew to 5 cm and had a heterogeneous region (arrow) on unenhanced images. F CT urogram at 11 months shows heterogeneous enhancement. Clear cell renal cell carcinoma was found at nephrectomy
A 86-year-old male with right RCC. A Unenhanced CT using that was interpreted as a homogenous appearing renal mass (arrow). B Unenhanced CT using a large ROI showed HU measurement of 10.1. C Unenhanced CT using a small ROI showed HU measurement of 28.9. D Delayed contrast-enhanced CT through the mass. Clear cell renal cell carcinoma was found at nephrectomy
There were no differences in patient age, patient gender, mass size, and CT scanner type between the masses that measured greater than 20 HU and those which measured 20 HU or less using a single, large ROI (Table 1). However, there was a non-significant trend in the groups with only 17% in the low HU group scanned with the GE scanners compared with 44% with the GE scanner in the higher HU group (Table 1). Of the 104 RCCs, 77 (74%) were clear cell carcinomas, 20 (19%) were papillary cell carcinomas, and seven (7%) were chromophobe RCCs. Twenty of the 24 RCCs which measured 20 HU or less using a single, large ROI were clear cell sub-type. Relative to other sub-types, a significantly greater portion and a trend of masses measuring 20 HU or less were clear cell RCCs (Table 1). Clear cell carcinomas had the overall lowest mean attenuation (26.4 HU) using a single, large ROI (Table 2). All three homogeneous RCCs which measured less than 20 HU with a single large ROI were clear cell RCCs (mean diameter, 4.0 cm, range 2.8–5.1 cm) with a mean attenuation of 18 HU (range 17–19).
Discussion
It has long been known that non-calcified fat-containing renal masses can be reliably diagnosed as angiomyolipomas with unenhanced CT alone [2]. Our ability to diagnose the etiology of other renal masses with unenhanced CT alone has increased substantially in recent years [3, 6,7,8]. In large studies with unenhanced CT, simple cyst-appearing renal masses, defined as those that are homogeneous with water attenuation (−10 to +20 HU) and without septations are most always renal cysts [3, 8, 13]. Alternatively, none of 193 RCCs reported in one study were homogeneous with attenuations less than 20 HU [7].
However, Silverman et al. reported as early as 1994 that RCC can reveal attenuations 20 HU or less on unenhanced CT [11]. Birnbaum et al reported a mean unenhanced attenuation value of 19 HU among 16 RCCs; half of them measured less than 20 HU [14]. Zhang et al reported unenhanced attenuation values of 108 clear cell RCC ranging from 14 to 71 HU [15]. Ruppert-Kohlmayr et al showed that some clear cell RCCs measured less than 20 HU on unenhanced CT [16]. Although these prior reports have shown that attenuations of RCC can measure 20 HU or less, the number of masses that were also homogeneous is not known. Furthermore, the methods used to measure attenuations were either variable or not described fully [11, 12, 17,18,19,20].
It is well-established that the ability to use unenhanced CT to diagnose masses as benign cysts depends on both the measured attenuation value and the mass’s appearance [3]. Although it is recommended that a renal mass be considered homogeneous before a confident diagnosis of a cyst is made, obtaining an attenuation value of 20 HU or less might lead to misdiagnosing a renal cancer as a cyst if the visual assessment of homogeneity is not performed or is incorrect. We postulated that the prior reports of low (≤20 HU) attenuation RCCs may have been in part due to the use of large ROIs [12]. Schieda et al, in a recent study that included 55 renal cell carcinomas, used three ROI measurements (each encompassing two-thirds of each mass) and found that eight RCCs had an average attenuation 20 HU or less on unenhanced CT [12]. Therefore, we postulated that the finding of RCC with attenuations 20 HU or less may be due to both the number and size of the ROI.
Our data demonstrated that using a single, large ROI, 24 (23%) of 104 RCCs measured 20 HU or less. Although most cancers were assessed as heterogeneous, three were homogeneous and measured 20 HU or less using a single, large ROI. We would urge caution when measuring a renal mass with a large ROI. When multiple, small ROIs were used, all three revealed at least one attenuation value greater than 20 HU. Indeed, using multiple small ROIs, none of the 104 RCCs in our cohort would have been diagnosed as a cyst. All RCCs were either heterogeneous or had attenuations higher than 20 HU when evaluated with multiple, small ROIs. However, our data reveal that in addition to assessing all features of a renal mass, instead of using one or more large ROIs as has been suggested by others, it is better to use multiple, small ROIs to measure attenuation.
Our results can be explained in the following way. Measuring attenuations using large ROIs volume average heterogeneous regions of tissues that contain high and low attenuations that otherwise would be detected if multiple, small ROI were used. In other words, using multiple small ROIs adds to the visual assessment of renal masses and helps detect heterogeneity. We believe that the use of large ROIs may explain others multiple reports of low attenuation RCC [9, 11, 12, 14, 15].
Few prior studies have evaluated the effect of varying the size of the ROI used to evaluate renal masses. Rosenkrantz et al reported no significant difference between the unenhanced CT attenuation values of RCC when using a small (<0.5 cm2) ROI (mean HU of 30.5 ± 9.9) compared to attenuation values obtained with a larger ROI, equal to one half the diameter of the mass (mean HU of 28.0 ± 8.3) or for any of the ROI’s used [18]. Regarding varying the number of ROIs, Schieda et al. suggested using multiple ROIs when evaluating renal masses but their suggested size was large [12]. Our results reveal the importance of using multiple, small ROIs.
We found no statistically significant differences with respect to the scanner used to evaluate the RCCs in our cohort. Others have shown that multiple factors can affect attenuation values of renal masses, including the scanner type [21, 22]. These studies have shown differences in attenuation values between manufacturers and even differences among the same manufacturer using different reconstruction kernels [21, 22]. Although not used in our cohort, iterative reconstruction techniques may also affect attenuation values [23]. Finally, attenuation values can also be affected by patient size [24].
Our study had limitations. First, we recognize that because CT attenuation values are variable, no single attenuation value threshold is 100% sensitive and specific for the diagnosing the etiology of renal masses on unenhanced CT [23]. For example, a lower attenuation threshold might increase the sensitivity for diagnosing RCC but lower specificity, and result in misdiagnosing more cysts as cancers. Second, our study was retrospective, and did not include a cohort of renal cysts for comparison. Including such a cohort would have addressed specificity or the false-positive diagnosis of renal cell carcinoma among cysts on unenhanced CT. Thus we feel the use of a large ROI and heterogeneity should be first used to help separate solid masses from cysts on unenhanced CT. In practice, use of small ROIs would be reserved for indeterminate masses, acknowledging the fact that volume averaging could account for spurious high HU values for cysts and add additional time for image review. Further studies may be necessary to evaluate the benign end of the spectrum. However, others have limited their analyses to cancers also [7, 12]. Our purpose was to evaluate RCC only; data that include only RCCs are still sufficient to show the importance of measuring attenuation with multiple, small ROIs rather than a single large ROI. Third, we did not evaluate the effect of ROI location. The single large ROI was centered in each mass; the multiple small ROIs were positioned in multiple portions of each renal mass to maximize the chance of detecting regions greater than 20 HU. Finally, we did not evaluate the effect of varying window settings. We used a soft tissue window setting that is typically used to evaluate the abdomen to mimic clinical practice. However, narrower window settings may have improved our ability to detect heterogeneous masses or high attenuation regions [25].
In summary, 23% of RCCs had an attenuation of 20 HU or less using a single, large ROI; these included three (2.8% of total) which appeared homogeneous. Our data show that, if only a single, large ROI is used, a RCC may have attenuation 20 HU or less and be misdiagnosed as a benign renal cyst if it is not assessed for heterogeneity. Therefore, when evaluating renal masses at unenhanced CT, in addition to visually assessing for heterogeneity, we recommend using multiple, small ROIs to measure attenuation. Use of small ROIs may be beneficial in revealing otherwise non-apparent increased density within renal masses. In the future, quantitative CT textural analyses may further improve our ability to assess renal mass heterogeneity and assure that a renal cell carcinoma is not mistaken for a benign renal cyst [26].
References
Bosniak MA (1986) The current radiological approach to renal cysts. Radiology 158(1):1–10. doi:10.1148/radiology.158.1.3510019
Silverman SG, Israel GM, Herts BR, Richie JP (2008) Management of the incidental renal mass. Radiology 249(1):16–31. doi:10.1148/radiol.2491070783
Silverman SG, Israel GM, Trinh QD (2015) Incompletely characterized incidental renal masses: emerging data support conservative management. Radiology 275(1):28–42. doi:10.1148/radiol.14141144
Smith RC, Rosenfield AT, Choe KA, et al. (1995) Acute flank pain: comparison of non-contrast-enhanced CT and intravenous urography. Radiology 194(3):789–794. doi:10.1148/radiology.194.3.7862980
Berland LL, Silverman SG, Megibow AJ, Mayo-Smith WW (2014) ACR members’ response to JACR white paper on the management of incidental abdominal CT findings. J Am Coll Radiol 11(1):30–35. doi:10.1016/j.jacr.2013.06.002
Jonisch AI, Rubinowitz AN, Mutalik PG, Israel GM (2007) Can high-attenuation renal cysts be differentiated from renal cell carcinoma at unenhanced CT? Radiology 243(2):445–450. doi:10.1148/radiol.2432060559
Pooler BD, Pickhardt PJ, O’Connor SD, et al. (2012) Renal cell carcinoma: attenuation values on unenhanced CT. AJR Am J Roentgenol 198(5):1115–1120. doi:10.2214/AJR.11.7587
O’Connor SD, Silverman SG, Ip IK, Maehara CK, Khorasani R (2013) Simple cyst-appearing renal masses at unenhanced CT: can they be presumed to be benign? Radiology 269(3):793–800. doi:10.1148/radiol.13122633
Millet I, Doyon FC, Hoa D, et al. (2011) Characterization of small solid renal lesions: can benign and malignant tumors be differentiated with CT? AJR Am J Roentgenol 197(4):887–896. doi:10.2214/AJR.10.6276
Dogra V, Levine E (2003) The kidney. In: Haaga JR, Lanzieri CF, Gilkeson RC (eds) CT and MR imaging of the whole body, vol. vol 2, 4th edn. St Louis: Mosby Inc., pp 1537–1610
Silverman SG, Lee BY, Seltzer SE, et al. (1994) Small (< or = 3 cm) renal masses: correlation of spiral CT features and pathologic findings. AJR Am J Roentgenol 163(3):597–605. doi:10.2214/ajr.163.3.8079852
Schieda N, Vakili M, Dilauro M, et al. (2015) Solid renal cell carcinoma measuring water attenuation (−10 to 20 HU) on unenhanced CT. AJR Am J Roentgenol 205(6):1215–1221. doi:10.2214/AJR.15.14554
O’Connor SD, Pickhardt PJ, Kim DH, Oliva MR, Silverman SG (2011) Incidental finding of renal masses at unenhanced CT: prevalence and analysis of features for guiding management. AJR Am J Roentgenol 197(1):139–145. doi:10.2214/AJR.10.5920
Birnbaum BA, Jacobs JE, Ramchandani P (1996) Multiphasic renal CT: comparison of renal mass enhancement during the corticomedullary and nephrographic phases. Radiology 200(3):753–758. doi:10.1148/radiology.200.3.8756927
Zhang J, Lefkowitz RA, Ishill NM, et al. (2007) Solid renal cortical tumors: differentiation with CT. Radiology 244(2):494–504. doi:10.1148/radiol.2442060927
Ruppert-Kohlmayr AJ, Uggowitzer M, Meissnitzer T, Ruppert G (2004) Differentiation of renal clear cell carcinoma and renal papillary carcinoma using quantitative CT enhancement parameters. AJR Am J Roentgenol 183(5):1387–1391. doi:10.2214/ajr.183.5.1831387
Young JR, Margolis D, Sauk S, et al. (2013) Clear cell renal cell carcinoma: discrimination from other renal cell carcinoma subtypes and oncocytoma at multiphasic multidetector CT. Radiology 267(2):444–453. doi:10.1148/radiol.13112617
Rosenkrantz AB, Matza BW, Portnoy E, et al. (2014) Impact of size of region-of-interest on differentiation of renal cell carcinoma and renal cysts on multi-phase CT: preliminary findings. Eur J Radiol 83(2):239–244. doi:10.1016/j.ejrad.2013.10.020
Lee-Felker SA, Felker ER, Tan N, et al. (2014) Qualitative and quantitative MDCT features for differentiating clear cell renal cell carcinoma from other solid renal cortical masses. AJR Am J Roentgenol 203(5):W516–W524. doi:10.2214/AJR.14.12460
Bae KT, Heiken JP, Siegel CL, Bennett HF (2000) Renal cysts: is attenuation artifactually increased on contrast-enhanced CT images? Radiology 216(3):792–796. doi:10.1148/radiology.216.3.r00se14792
Birnbaum BA, Hindman N, Lee J, Babb JS (2007) Multi-detector row CT attenuation measurements: assessment of intra- and interscanner variability with an anthropomorphic body CT phantom. Radiology 242(1):109–119. doi:10.1148/radiol.2421052066
Lamba R, McGahan JP, Corwin MT, et al. (2014) CT Hounsfield numbers of soft tissues on unenhanced abdominal CT scans: variability between two different manufacturers’ MDCT scanners. AJR Am J Roentgenol 203(5):1013–1020. doi:10.2214/AJR.12.10037
Volgyes D, Pedersen M, Stray-Pedersen A, Waaler D, Martinsen AC (2016) How different iterative and filtered back projection kernels affect computed tomography numbers and low contrast detectability. J Comput Assist Tomogr. doi:10.1097/RCT.0000000000000491
Beland MD, Scappaticci AA, Machan JT, et al. (2013) Effect of patient size on mean sterile water attenuation during multiphase CT examinations. AJR Am J Roentgenol 200(5):1048–1053. doi:10.2214/AJR.12.9198
Arsava EM, Saarinen JT, Unal A, et al. (2014) Impact of window setting optimization on accuracy of computed tomography and computed tomography angiography source image-based Alberta Stroke Program Early Computed Tomography Score. J Stroke Cerebrovasc Dis 23(1):12–16. doi:10.1016/j.jstrokecerebrovasdis.2012.05.012
Raman SP, Chen Y, Schroeder JL, Huang P, Fishman EK (2014) CT texture analysis of renal masses: pilot study using random forest classification for prediction of pathology. Acad Radiol 21(12):1587–1596. doi:10.1016/j.acra.2014.07.023
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study.
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this type of study formal, consent is not required.
Informed consent
Statement of informed consent was not applicable since the manuscript does not contain any patient data.
Rights and permissions
About this article
Cite this article
McGahan, J.P., Sidhar, K., Fananapazir, G. et al. Renal cell carcinoma attenuation values on unenhanced CT: importance of multiple, small region-of-interest measurements. Abdom Radiol 42, 2325–2333 (2017). https://doi.org/10.1007/s00261-017-1131-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-017-1131-6









