Abstract
Purpose
To evaluate the indications, technique, results, and complications of image-guided percutaneous biopsy of the urinary bladder.
Methods
This retrospective study included 15 patients (10 male, 5 female) who underwent image-guided percutaneous biopsy of the urinary bladder between January 1999 and December 2013. The medical records, imaging studies, procedural details, and long-term follow-up of each patient were reviewed in detail to assess the feasibility of percutaneous bladder biopsy.
Results
Ten patients had focal bladder masses and 5 patients had asymmetric or diffuse bladder wall thickening. Eleven patients had either negative or unsatisfactory cystoscopies prior to the biopsy. Percutaneous biopsies were performed under computed tomography guidance in 12 patients and ultrasound in 3 patients. All procedures were technically successful and there were no procedural complications. Malignancy was confirmed in 8 patients, among whom 6 had transitional cell carcinoma, 1 cervical cancer, and 1 prostate cancer metastasis. Seven patients had a benign diagnosis, including 3 that were later confirmed by pathology following surgery and 2 patients with a false-negative result. The overall sensitivity was 80% and accuracy was 87%.
Conclusions
Image-guided percutaneous biopsy of the urinary bladder is a safe and technically feasible procedure with a high sensitivity and accuracy rate. Although image-guided bladder biopsy is an uncommon procedure, it should be considered in selected cases when more traditional methods of tissue sampling are either not possible or fail to identify abnormalities detected by cross-sectional imaging.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Bladder cancer is the second most common genitourinary malignancy in the United States. Approximately 75,000 new cases of bladder cancer were diagnosed in the United States in 2013, with an estimated 16,000 deaths [1]. Globally, bladder cancer results in approximately 150,000 annual deaths. Genetic, chemical, and environmental exposures are all risk factors associated with the development of bladder cancer [2].
The standard diagnostic evaluation of bladder tumors often begins with urine cytology, followed by cystoscopy and sometimes cross-sectional imaging [3–6]. Cystoscopy is an effective method in the work-up of patients with suspected bladder cancer as it provides direct visualization of the mass, allows for tissue sampling and detection of additional lesions that may not have been previously identified [4]. While the reported sensitivity of cystoscopy for detecting bladder tumors is up to 87% [7, 8], there are limitations including potential blind spots (i.e., bladder neck, base, and diverticula) and inability to detect serosal or submucosal lesions [9]. Inability to access the bladder in a retrograde fashion due to urethral strictures and severe patient comorbidities that preclude sedation or anesthesia are other potential factors that may hinder a safe cystoscopy.
Current imaging modalities provide the opportunity of focal lesion biopsies in various abdominal as well as deep pelvic organs to allow a diagnosis to be made in a safe and effective manner [10, 11]. In situations where bladder tumors are not readily accessible for biopsy by cystoscopy, alternative methods for acquiring tissue samples such as computed tomography (CT) or ultrasound (US)-guided percutaneous biopsy may be performed. The purpose of this study was to evaluate the indications, technique, diagnostic accuracy, and complications of image-guided percutaneous biopsy of the bladder.
Methods
A retrospective, HIPAA-compliant study was performed with institutional review board approval and waiver of informed consent. The interventional radiology electronic database was used to identify all patients who underwent image-guided percutaneous biopsy of the urinary bladder in our department from January 1999 to July 2014, which revealed 15 patients (10 male, 5 female) with a mean age of 68.7 years ± 10.9 (range, 46–85 years). The electronic medical records and the imaging studies of the patients were reviewed. The tumor location and size, the biopsy technique, results and complications, and long-term follow-up results were recorded.
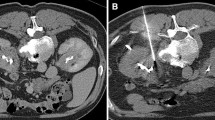
All biopsies were performed as outpatient procedures with either CT or US guidance under monitored conscious sedation. The choice between US and CT-guided biopsy was based upon operator preference as well as visibility and accessibility of the tumor by each modality. Patients were instructed to stay off anticoagulant or antiplatelet medication prior to the procedure per societal guidelines [12]. All patients had normal coagulation parameters. Patients were not administered prophylactic antibiotics prior to the procedure in accordance with societal and institutional guidelines [13]. Technical success was defined as the ability to obtain adequate tissue specimen via a percutaneous biopsy to achieve a pathological diagnosis. For each procedure, a standard departmental protocol was followed which includes placement of a 17-gage coaxial needle (Coaxial Temno EvolutionTM CareFusion, McGaw Park, IL) into the lesion through which 22-gage fine needle aspirates (FNA) (Chiba Biopsy Needle, Cook Incorporated, Bloomington, IN), and 18-gage (Coaxial Temno EvolutionTM CareFusion, McGaw Park, IL) core biopsy specimens are obtained. Passes through the coaxial needle were repeated until satisfactory amount of tissue was obtained. Both fine needle aspiration and core samples were obtained for 13 patients. The remaining two patients had core biopsy only. The specimens were then sent to pathology for analysis. An onsite pathologist was not routinely present during the biopsy. Four patients had placement of a Foley catheter at the time of the biopsy to distend the bladder and displace overlying bowel loops, thus facilitating a safe trajectory for the biopsy needle or helped localize the lesion by outlining the lesion in an otherwise collapsed bladder wall (Fig. 1).
Patient with history of bladder cancer. Axial T2-weighted MRI scan A obtained for surveillance demonstrates a polypoid lesion along the right lateral bladder wall (arrow). Cystoscopy was not possible due to severe urethral stricture in this patient. Percutaneous biopsy was performed under CT guidance (B), which showed benign stromal elements. Given history of bladder cancer and imaging findings that were discordant with the pathology results, a decision was made to proceed with radical cystectomy. Final diagnosis was transitional cell carcinoma.
For patients who did not undergo surgery, follow-up imaging and clinical notes were reviewed to confirm stability of lesions with a benign biopsy result. Pathological results in patients with subsequent surgery were reviewed and correlated with the biopsy results.
Results
Eleven patients had history of prior malignancy including bladder cancer (n = 7), prostate cancer (n = 2), cervical cancer (n = 1), and pancreatic cancer (n = 1). The remaining 4 patients did not have prior history of malignancy. Eleven patients had microscopic hematuria, and only two had urine cytology suggestive of transitional cell carcinoma (TCC) prior to the biopsy. The bladder abnormality was detected by CT in 11 patients, by magnetic resonance imaging (MRI) in 1 patient, and by both CT and MRI in 3 patients. Ten patients had a focal bladder mass of which two were in a diverticulum. Four patients had asymmetric bladder wall thickening and one patient had diffuse bladder wall thickening. The mean tumor size in patients with a focal mass was 3.3 cm ± 1.7 (range, 1.2–6 cm). The mean wall thickness in patients with wall thickening, measured perpendicular to the point of greatest thickness, was 2.6 cm ± 0.8 (range, 1.6–3.5 cm).
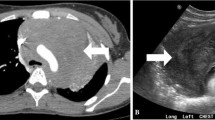
Eleven patients (73%) underwent cystoscopy prior to percutaneous biopsy, which did not reveal a lesion to target for biopsy in 5 patients (Fig. 2). Two patients had lesions within bladder diverticula that while clearly identified by CT, they were technically challenging to biopsy by cystoscopy. Three patients (20%) underwent percutaneous biopsy after cystoscopic biopsy-yielded benign results, despite clinical and imaging feature suggestive of malignancy. Four patients (27%) did not undergo cystoscopy due to the serosal appearance of the bladder abnormality by imaging and were referred directly to percutaneous biopsy. All imaging guided percutaneous biopsies were technically successful. CT (n = 12) or US (n = 3) was used for imaging guidance. The patient demographics and procedure details are summarized in Table 1.
A pathological diagnosis of malignancy was achieved in 8 patients, including TCC (n = 6), cervical cancer (n = 1) and prostate cancer metastasis (n = 1). Seven patients had a benign diagnosis. Three of these patients had pathological confirmation following the surgery. Two patients had regression of the abnormality on follow-up imaging and thus was considered true negative. The remaining two patients, however, had a false-negative result. Both patients had prior history of bladder cancer. One was admitted 11 months following the biopsy for urinary tract infection and had a repeated transurethral bladder resection, which confirmed TCC. The patient subsequently underwent a partial cystectomy. The other patient underwent radical cystectomy 1 month following the biopsy due to the high clinical suspicion and discordance of imaging and cystoscopy findings with that of the biopsy result. Suspicion for TCC was confirmed by surgical pathology. With these two false-negative results, the overall sensitivity of the study was 80% and the accuracy was 87%.
The average follow-up for the 15 patients was 24.7 months (range, 2–144 months). There were no procedure-related minor or major complications. On follow-up imaging studies, present in all of the patients, there was no evidence of tract seeding or evidence of other procedure related complications, including bladder leak.
Discussion
The majority of bladder malignancies represent TCC. Because these are mucosal-based tumors, cystoscopy is the preferred method of establishing diagnosis in patients with suspected bladder malignancy [14, 15]. Cystoscopy provides direct visualization of the lesion and allows for assessment of possible synchronous lesions. However, cystoscopy is limited in its ability to detect serosal or submucosal lesions such as infiltrative disease or drop metastases [16]. The false-negative rate of white light cystoscopy has been reported to approach 30% in some series [17]. In these situations, percutaneous biopsy may be an alternative diagnostic tool to help in establishing a diagnosis for bladder abnormalities detected by imaging. The results of our study suggest that image-guided percutaneous biopsy is safe and can provide a reasonable diagnostic alternative to cystoscopy.
Despite our small cohort, the sensitivity rate in our study is comparable to the reported rates in literature for cystoscopy. It should also be kept in mind that the majority of our patient population consisted of a selected group of challenging patients, among which a significant portion underwent prior negative work-up by cystoscopy. Our series also demonstrates how percutaneous biopsy may be helpful in targeting lesions that are located in well-recognized blind spots in cystoscopy. Five of our patients had inconclusive cystoscopies without a lesion identified to target for biopsy, and two others were in bladder diverticula both of which proved technically challenging to obtain samples by cystoscopy.
We had two false-negative results, which could either be explained by under sampling or by mis-sampling. It is worth noting that these patients had history of bladder cancer and had prior negative cystoscopies. These cases underscore the importance of close follow-up in patients with suspected bladder malignancy to ensure management of these patients be based on imaging and clinical findings and not solely based on the percutaneous biopsy results or cystoscopic examination. The imaging abnormalities should be approached even more cautiously in patients with a history of bladder cancer.
Few studies have investigated the role of image-guided percutaneous biopsy of the urinary bladder. Gani et al. included 8 patients who had US-guided bladder biopsies in a series of 159 patients undergoing percutaneous abdominal or pelvic organ biopsy [18]. However, no specific detail on bladder biopsies was given, as the main goal of the study was to use the US guidance and cost-effectiveness of abdominal tumor biopsies. The lack of literature on image-guided percutaneous biopsy can be partially explained by the widespread use of cystoscopy as the gold standard of work-up of patients with bladder lesions.
Potential complications of percutaneous bladder biopsy include hematuria, extraperitoneal or intraperitoneal hemorrhage, urinoma, and cystitis, which should be taken into account during pre-procedure planning. Tumor seeding along the biopsy tract albeit small is also a potential risk reported in a variety of malignancies. There were no minor or major procedure-related complications in this series.
This study has several limitations. It is a retrospective study that evaluates a relatively small number of patients, which prevents drawing firm conclusions with respect to the role and accuracy of image-guided percutaneous biopsy of the urinary bladder. The biopsy technique, although similar, was not entirely standard among the different operators involved. The follow-up methods and duration also differed in each patient. There was no pathological confirmation of the nature of the lesion in every patient.
The findings of this study suggest that image-guided percutaneous biopsy of the urinary bladder may have a role for selected patients with bladder abnormalities and should be considered when cystoscopic evaluation of bladder abnormalities is unsuccessful. Cystoscopy and/or biopsy remain the gold standard for the diagnosis of bladder cancer [19].
References
Siegel R, et al. (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1):9–29
Kaufman DS, Shipley WU, Feldman AS (2009) Bladder cancer. Lancet 374(9685):239–249
Grasso M, et al. (1993) Actively deflectable, flexible cystoscopes: no longer solely a diagnostic instrument. J Endourol 7(6):527–530
Burke DM, Shackley DC, O’Reilly PH (2002) The community-based morbidity of flexible cystoscopy. BJU Int 89(4):347–349
Lotan Y, Roehrborn CG (2003) Sensitivity and specificity of commonly available bladder tumor markers versus cytology: results of a comprehensive literature review and meta-analyses. Urology 61(1):109–118 (discussion 118)
Herr HW (1996) Routine CT scan in cystectomy patients: does it change management? Urology 47(3):324–325
Fielding JR, et al. (2002) Tumor detection by virtual cystoscopy with color mapping of bladder wall thickness. J Urol 167(2 Pt 1):559–562
Walker L, Liston TG, Liston TG, LLoyd-Davies RW (1993) Does flexible cystoscopy miss more tumours than rod-lens examination? Br J Urol 72(4):449–450
Tsili A, et al. (2004) Computed tomographic virtual cystoscopy for the detection of urinary bladder neoplasms. Eur Urol 46(5):579–585
Gupta S, et al. (2003) CT-guided needle biopsy of deep pelvic lesions by extraperitoneal approach through iliopsoas muscle. Cardiovasc Intervent Radiol 26(6):534–538
Gupta S, et al. (2004) Various approaches for CT-guided percutaneous biopsy of deep pelvic lesions: anatomic and technical considerations. Radiographics 24(1):175–189
Patel IJ, et al. (2012) Consensus guidelines for periprocedural management of coagulation status and hemostasis risk in percutaneous image-guided interventions. J Vasc Interv Radiol 23(6):727–736
Venkatesan AM, et al. (2010) Practice guidelines for adult antibiotic prophylaxis during vascular and interventional radiology procedures. Written by the Standards of Practice Committee for the Society of Interventional Radiology and Endorsed by the Cardiovascular Interventional Radiological Society of Europe and Canadian Interventional Radiology Association [corrected]. J Vasc Interv Radiol 21(11):1611–1630 (quiz 1631)
Grossfeld GD, et al. (2001) Evaluation of asymptomatic microscopic hematuria in adults: the American Urological Association best practice policy–part II: patient evaluation, cytology, voided markers, imaging, cystoscopy, nephrology evaluation, and follow-up. Urology 57(4):604–610
Grossfeld GD, et al. (2001) Asymptomatic microscopic hematuria in adults: summary of the AUA best practice policy recommendations. Am Fam Physician 63(6):1145–1154
Soloway MS, et al. (1978) Serial multiple-site biopsies in patients with bladder cancer. J Urol 120(1):57–59
Jocham D, Stepp H, Waidelich R (2008) Photodynamic diagnosis in urology: state-of-the-art. Eur Urol 53(6):1138–1148
Gani MS, Shafee AM, Soliman IY (2011) Ultrasound guided percutaneous fine needle aspiration biopsy/automated needle core biopsy of abdominal lesions: effect on management and cost effectiveness. Ann Afr Med 10(2):133–138
Babjuk M, et al. (2013) EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2013. Eur Urol 64(4):639–653
Conflict of interest
None of the contributing authors have any conflicts of interest, including specific financial interests or relationships and affiliations relevant to the subject matter or materials discussed in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Butros, S.R., McCarthy, C.J., Karaosmanoğlu, A.D. et al. Feasibility and effectiveness of image-guided percutaneous biopsy of the urinary bladder. Abdom Imaging 40, 1838–1842 (2015). https://doi.org/10.1007/s00261-015-0356-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-015-0356-5